Pancreatic T2* Magnetic Resonance Imaging for Prediction of Cardiac Arrhythmias in Transfusion-Dependent Thalassemia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Magnetic Resonance Imaging
2.3. Diagnostic Criteria and Clinical Follow-Up
2.4. Statistical Analysis
3. Results
3.1. Baseline Findings
3.2. Cardiac Complications
3.3. Characterization of Patients Who Developed Cardiac Arrhythmias
3.4. Prognostic Markers of Arrhythmias
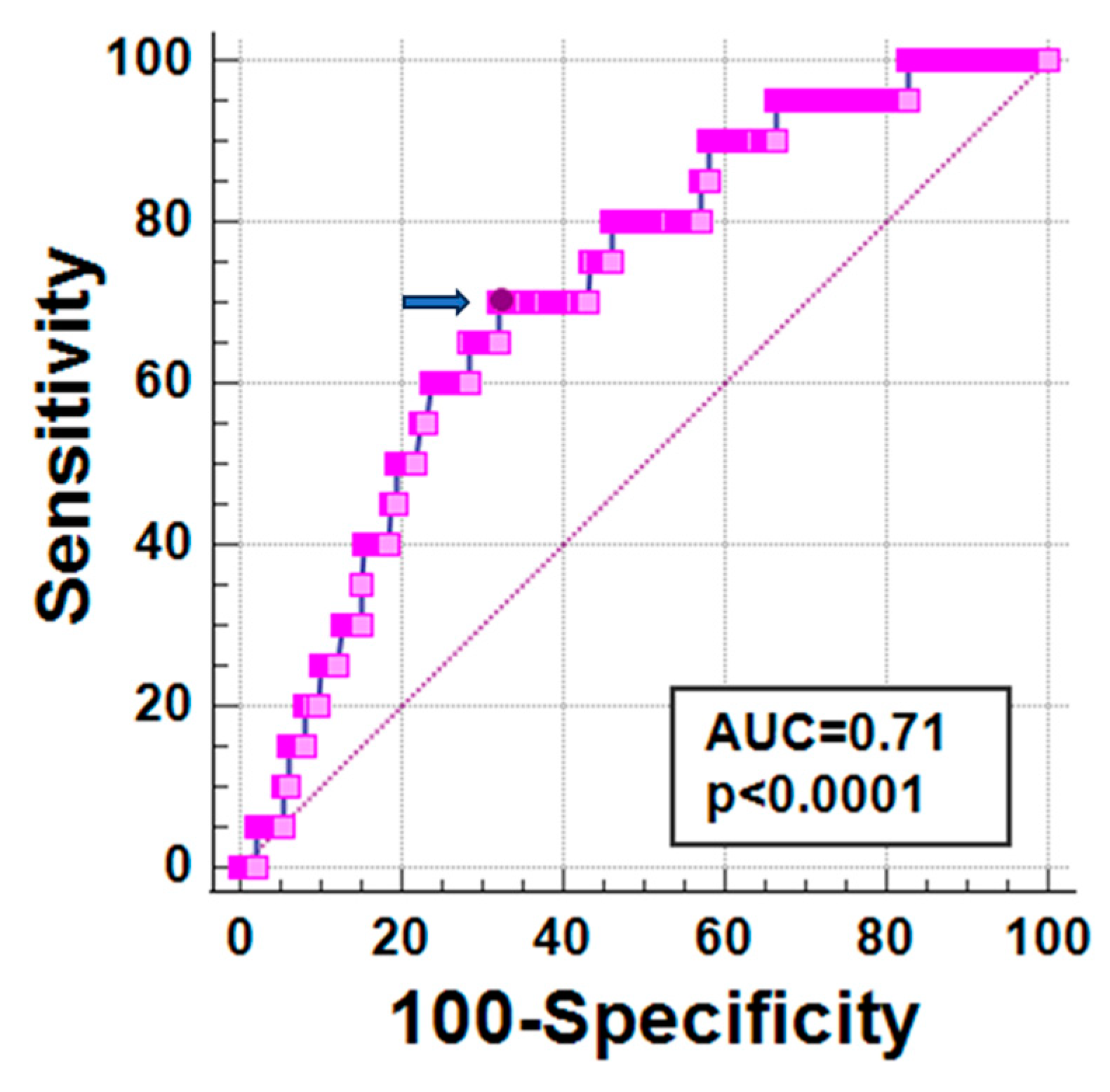
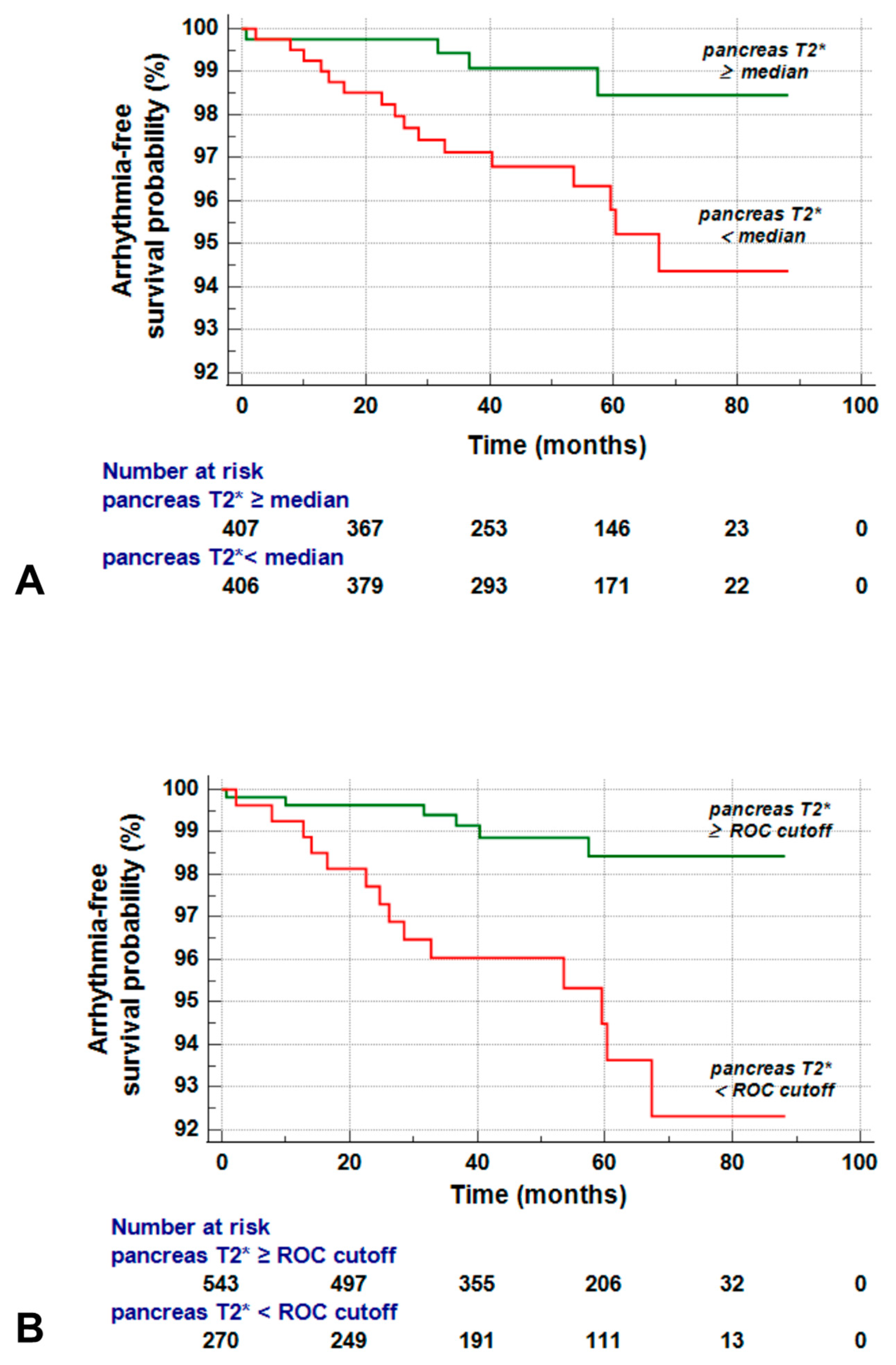
3.5. Global Pancreas T2* and Prediction of Arrhythmias
3.6. Best Cut-Off of Global Pancreas T2* for Risk Prediction
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cao, A.; Galanello, R. Beta-thalassemia. Genet. Med. 2010, 12, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Weatherall, D.J.; Clegg, J.B. The Thalassemia Syndromes; Blackwell Science: Oxford, UK, 2001. [Google Scholar]
- Musallam, K.M.; Cappellini, M.D.; Viprakasit, V.; Kattamis, A.; Rivella, S.; Taher, A.T. Revisiting the non-transfusion-dependent (NTDT) vs. transfusion-dependent (TDT) thalassemia classification 10 years later. Am. J. Hematol. 2021, 96, E54–E56. [Google Scholar] [CrossRef] [PubMed]
- Cappellini, M.D.; Cohen, A.; Porter, J.; Taher, A.; Viprakasit, V. Guidelines for the Management of Transfusion Dependent Thalassaemia (TDT), 3rd ed.; Thalassaemia International Federation: Nicosia, CY, USA, 2014. [Google Scholar]
- Farmakis, D.; Porter, J.; Taher, A.; Domenica Cappellini, M.; Angastiniotis, M.; Eleftheriou, A. 2021 Thalassaemia International Federation Guidelines for the Management of Transfusion-dependent Thalassemia. Hemasphere 2022, 6, e732. [Google Scholar] [CrossRef]
- Ozment, C.P.; Turi, J.L. Iron overload following red blood cell transfusion and its impact on disease severity. Biochim. Biophys. Acta 2009, 1790, 694–701. [Google Scholar] [PubMed]
- Wallace, D.F. The Regulation of Iron Absorption and Homeostasis. Clin. Biochem. Rev. 2016, 37, 51–62. [Google Scholar]
- Hentze, M.W.; Muckenthaler, M.U.; Andrews, N.C. Balancing acts: Molecular control of mammalian iron metabolism. Cell 2004, 117, 285–297. [Google Scholar]
- Angelucci, E.; Muretto, P.; Nicolucci, A.; Baronciani, D.; Erer, B.; Gaziev, J.; Ripalti, M.; Sodani, P.; Tomassoni, S.; Visani, G.; et al. Effects of iron overload and hepatitis C virus positivity in determining progression of liver fibrosis in thalassemia following bone marrow transplantation. Blood 2002, 100, 17–21. [Google Scholar] [CrossRef]
- Wood, J.C.; Noetzl, L.; Hyderi, A.; Joukar, M.; Coates, T.; Mittelman, S. Predicting pituitary iron and endocrine dysfunction. Ann. N. Y Acad. Sci. 2010, 1202, 123–128. [Google Scholar] [CrossRef]
- Kyriakou, A.; Skordis, N. Thalassaemia and aberrations of growth and puberty. Mediterr. J. Hematol. Infect. Dis. 2009, 1, e2009003. [Google Scholar] [CrossRef]
- Noetzli, L.J.; Mittelman, S.D.; Watanabe, R.M.; Coates, T.D.; Wood, J.C. Pancreatic iron and glucose dysregulation in thalassemia major. Am. J. Hematol. 2012, 87, 155–160. [Google Scholar]
- Noetzli, L.J.; Panigrahy, A.; Mittelman, S.D.; Hyderi, A.; Dongelyan, A.; Coates, T.D.; Wood, J.C. Pituitary iron and volume predict hypogonadism in transfusional iron overload. Am. J. Hematol. 2012, 87, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Kremastinos, D.T.; Farmakis, D. Iron overload cardiomyopathy in clinical practice. Circulation 2011, 124, 2253–2263. [Google Scholar] [CrossRef] [PubMed]
- Pepe, A.; Pistoia, L.; Gamberini, M.R.; Cuccia, L.; Peluso, A.; Messina, G.; Spasiano, A.; Allo, M.; Bisconte, M.G.; Putti, M.C.; et al. The Close Link of Pancreatic Iron With Glucose Metabolism and With Cardiac Complications in Thalassemia Major: A Large, Multicenter Observational Study. Diabetes Care 2020, 43, 2830–2839. [Google Scholar] [CrossRef] [PubMed]
- Borgna-Pignatti, C.; Rugolotto, S.; De Stefano, P.; Zhao, H.; Cappellini, M.D.; Del Vecchio, G.C.; Romeo, M.A.; Forni, G.L.; Gamberini, M.R.; Ghilardi, R.; et al. Survival and complications in patients with thalassemia major treated with transfusion and deferoxamine. Haematologica 2004, 89, 1187–1193. [Google Scholar]
- Modell, B.; Khan, M.; Darlison, M.; Westwood, M.A.; Ingram, D.; Pennell, D.J. Improved survival of thalassaemia major in the UK and relation to T2* cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 2008, 10, 42. [Google Scholar] [CrossRef]
- Meloni, A.; Pistoia, L.; Gamberini, M.R.; Cuccia, L.; Lisi, R.; Cecinati, V.; Ricchi, P.; Gerardi, C.; Restaino, G.; Righi, R.; et al. Multi-Parametric Cardiac Magnetic Resonance for Prediction of Heart Failure Death in Thalassemia Major. Diagnostics 2023, 13, 890. [Google Scholar] [CrossRef]
- Argyropoulou, M.I.; Kiortsis, D.N.; Astrakas, L.; Metafratzi, Z.; Chalissos, N.; Efremidis, S.C. Liver, bone marrow, pancreas and pituitary gland iron overload in young and adult thalassemic patients: A T2 relaxometry study. Eur. Radiol. 2007, 17, 3025–3030. [Google Scholar] [CrossRef] [PubMed]
- Berdoukas, V.; Chouliaras, G.; Moraitis, P.; Zannikos, K.; Berdoussi, E.; Ladis, V. The efficacy of iron chelator regimes in reducing cardiac and hepatic iron in patients with thalassaemia major: A clinical observational study. J. Cardiovasc. Magn. Reson. 2009, 11, 20. [Google Scholar] [CrossRef]
- Nichols-Vinueza, D.X.; White, M.T.; Powell, A.J.; Banka, P.; Neufeld, E.J. MRI guided iron assessment and oral chelator use improve iron status in thalassemia major patients. Am. J. Hematol. 2014, 89, 684–688. [Google Scholar] [CrossRef]
- Pennell, D.J.; Porter, J.B.; Cappellini, M.D.; Chan, L.L.; El-Beshlawy, A.; Aydinok, Y.; Ibrahim, H.; Li, C.K.; Viprakasit, V.; Elalfy, M.S.; et al. Deferasirox for up to 3 years leads to continued improvement of myocardial T2* in patients with beta-thalassemia major. Haematologica 2012, 97, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Chaosuwannakit, N.; Makarawate, P.; Wanitpongpun, C. The Importance of Cardiac T2* Magnetic Resonance Imaging for Monitoring Cardiac Siderosis in Thalassemia Major Patients. Tomography 2021, 7, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.T.; Porter, J.B.; Sadasivam, N.; Kaya, B.; Moon, J.C.; Velangi, M.; Ako, E.; Pancham, S. Guidelines for the monitoring and management of iron overload in patients with haemoglobinopathies and rare anaemias. Br. J. Haematol. 2022, 196, 336–350. [Google Scholar] [CrossRef] [PubMed]
- Bayraktaroglu, S.; Karadas, N.; Onen, S.; Karapinar, D.Y.; Aydinok, Y. Modern management of iron overload in thalassemia major patients guided by MRI techniques: Real-world data from a long-term cohort study. Ann. Hematol. 2022, 101, 521–529. [Google Scholar] [CrossRef]
- Chouliaras, G.; Berdoukas, V.; Ladis, V.; Kattamis, A.; Chatziliami, A.; Fragodimitri, C.; Karabatsos, F.; Youssef, J.; Karagiorga-Lagana, M. Impact of magnetic resonance imaging on cardiac mortality in thalassemia major. J. Magn. Reson. Imaging 2011, 34, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Pepe, A.; Pistoia, L.; Gamberini, M.R.; Cuccia, L.; Lisi, R.; Cecinati, V.; Maggio, A.; Sorrentino, F.; Filosa, A.; Rosso, R.; et al. National networking in rare diseases and reduction of cardiac burden in thalassemia major. Eur. Heart J. 2022, 43, 2482–2492. [Google Scholar] [CrossRef]
- Marsella, M.; Borgna-Pignatti, C.; Meloni, A.; Caldarelli, V.; Dell’Amico, M.C.; Spasiano, A.; Pitrolo, L.; Cracolici, E.; Valeri, G.; Positano, V.; et al. Cardiac iron and cardiac disease in males and females with transfusion-dependent thalassemia major: A T2* magnetic resonance imaging study. Haematologica 2011, 96, 515–520. [Google Scholar] [CrossRef]
- Kirk, P.; Roughton, M.; Porter, J.B.; Walker, J.M.; Tanner, M.A.; Patel, J.; Wu, D.; Taylor, J.; Westwood, M.A.; Anderson, L.J.; et al. Cardiac T2* magnetic resonance for prediction of cardiac complications in thalassemia major. Circulation 2009, 120, 1961–1968. [Google Scholar] [CrossRef]
- Pepe, A.; Meloni, A.; Rossi, G.; Midiri, M.; Missere, M.; Valeri, G.; Sorrentino, F.; D’Ascola, D.G.; Spasiano, A.; Filosa, A.; et al. Prediction of cardiac complications for thalassemia major in the widespread cardiac magnetic resonance era: A prospective multicentre study by a multi-parametric approach. Eur. Heart J. Cardiovasc. Imaging 2018, 19, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Cooksey, R.C.; Jouihan, H.A.; Ajioka, R.S.; Hazel, M.W.; Jones, D.L.; Kushner, J.P.; McClain, D.A. Oxidative stress, beta-cell apoptosis, and decreased insulin secretory capacity in mouse models of hemochromatosis. Endocrinology 2004, 145, 5305–5312. [Google Scholar] [CrossRef]
- Au, W.Y.; Lam, W.W.; Chu, W.; Tam, S.; Wong, W.K.; Liang, R.; Ha, S.Y. A T2* magnetic resonance imaging study of pancreatic iron overload in thalassemia major. Haematologica 2008, 93, 116–119. [Google Scholar] [CrossRef]
- Au, W.Y.; Lam, W.W.; Chu, W.W.; Yuen, H.L.; Ling, A.S.; Li, R.C.; Chan, H.M.; Lee, H.K.; Law, M.F.; Liu, H.S.; et al. A cross-sectional magnetic resonance imaging assessment of organ specific hemosiderosis in 180 thalassemia major patients in Hong Kong. Haematologica 2008, 93, 784–786. [Google Scholar] [CrossRef] [PubMed]
- Noetzli, L.J.; Papudesi, J.; Coates, T.D.; Wood, J.C. Pancreatic iron loading predicts cardiac iron loading in thalassemia major. Blood 2009, 114, 4021–4026. [Google Scholar] [CrossRef]
- Azarkeivan, A.; Hashemieh, M.; Shirkavand, A.; Sheibani, K. Correlation between Heart, Liver and Pancreas Hemosiderosis Measured by MRI T2* among Thalassemia Major Patients from Iran. Arch. Iran. Med. 2016, 19, 96–100. [Google Scholar]
- Oudit, G.Y.; Sun, H.; Trivieri, M.G.; Koch, S.E.; Dawood, F.; Ackerley, C.; Yazdanpanah, M.; Wilson, G.J.; Schwartz, A.; Liu, P.P.; et al. L-type Ca2+ channels provide a major pathway for iron entry into cardiomyocytes in iron-overload cardiomyopathy. Nat. Med. 2003, 9, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Meloni, A.; De Marchi, D.; Pistoia, L.; Grassedonio, E.; Peritore, G.; Preziosi, P.; Restaino, G.; Righi, R.; Riva, A.; Renne, S.; et al. Multicenter validation of the magnetic resonance T2* technique for quantification of pancreatic iron. Eur. Radiol. 2019, 29, 2246–2252. [Google Scholar] [CrossRef]
- Meloni, A.; De Marchi, D.; Positano, V.; Neri, M.G.; Mangione, M.; Keilberg, P.; Lendini, M.; Cirotto, C.; Pepe, A. Accurate estimate of pancreatic T2* values: How to deal with fat infiltration. Abdom. Imaging 2015, 40, 3129–3136. [Google Scholar] [CrossRef]
- Meloni, A.; Luciani, A.; Positano, V.; De Marchi, D.; Valeri, G.; Restaino, G.; Cracolici, E.; Caruso, V.; Dell’amico, M.C.; Favilli, B.; et al. Single region of interest versus multislice T2* MRI approach for the quantification of hepatic iron overload. J. Magn. Reson. Imaging 2011, 33, 348–355. [Google Scholar] [CrossRef]
- Wood, J.C.; Enriquez, C.; Ghugre, N.; Tyzka, J.M.; Carson, S.; Nelson, M.D.; Coates, T.D. MRI R2 and R2* mapping accurately estimates hepatic iron concentration in transfusion-dependent thalassemia and sickle cell disease patients. Blood 2005, 106, 1460–1465. [Google Scholar] [CrossRef]
- Cerqueira, M.D.; Weissman, N.J.; Dilsizian, V.; Jacobs, A.K.; Kaul, S.; Laskey, W.K.; Pennell, D.J.; Rumberger, J.A.; Ryan, T.; Verani, M.S. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 2002, 105, 539–542. [Google Scholar]
- Jessup, M.; Abraham, W.T.; Casey, D.E.; Feldman, A.M.; Francis, G.S.; Ganiats, T.G.; Konstam, M.A.; Mancini, D.M.; Rahko, P.S.; Silver, M.A.; et al. 2009 focused update: ACCF/AHA Guidelines for the Diagnosis and Management of Heart Failure in Adults: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: Developed in collaboration with the International Society for Heart and Lung Transplantation. Circulation 2009, 119, 1977–2016. [Google Scholar] [PubMed]
- Buxton, A.E.; Calkins, H.; Callans, D.J.; DiMarco, J.P.; Fisher, J.D.; Greene, H.L.; Haines, D.E.; Hayes, D.L.; Heidenreich, P.A.; Miller, J.M.; et al. ACC/AHA/HRS 2006 key data elements and definitions for electrophysiological studies and procedures: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Data Standards (ACC/AHA/HRS Writing Committee to Develop Data Standards on Electrophysiology). Circulation 2006, 114, 2534–2570. [Google Scholar] [PubMed]
- Davis, B.A.; Porter, J.B. Long-term outcome of continuous 24-hour deferoxamine infusion via indwelling intravenous catheters in high-risk beta-thalassemia. Blood 2000, 95, 1229–1236. [Google Scholar] [CrossRef]
- Anderson, L.J.; Westwood, M.A.; Holden, S.; Davis, B.; Prescott, E.; Wonke, B.; Porter, J.B.; Walker, J.M.; Pennell, D.J. Myocardial iron clearance during reversal of siderotic cardiomyopathy with intravenous desferrioxamine: A prospective study using T2* cardiovascular magnetic resonance. Br. J. Haematol. 2004, 127, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Tanner, M.A.; Galanello, R.; Dessi, C.; Smith, G.C.; Westwood, M.A.; Agus, A.; Pibiri, M.; Nair, S.V.; Walker, J.M.; Pennell, D.J. Combined chelation therapy in thalassemia major for the treatment of severe myocardial siderosis with left ventricular dysfunction. J. Cardiovasc. Magn. Reson. 2008, 10, 12. [Google Scholar] [CrossRef]
- Huxley, R.R.; Filion, K.B.; Konety, S.; Alonso, A. Meta-analysis of cohort and case-control studies of type 2 diabetes mellitus and risk of atrial fibrillation. Am. J. Cardiol. 2011, 108, 56–62. [Google Scholar] [CrossRef]
- Aune, D.; Feng, T.; Schlesinger, S.; Janszky, I.; Norat, T.; Riboli, E. Diabetes mellitus, blood glucose and the risk of atrial fibrillation: A systematic review and meta-analysis of cohort studies. J. Diabetes Complicat. 2018, 32, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Grisanti, L.A. Diabetes and Arrhythmias: Pathophysiology, Mechanisms and Therapeutic Outcomes. Front. Physiol. 2018, 9, 1669. [Google Scholar] [CrossRef]
- Ozturk, N.; Uslu, S.; Ozdemir, S. Diabetes-induced changes in cardiac voltage-gated ion channels. World J. Diabetes 2021, 12, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Ju, H.; Liu, T.; Yang, M.; Cheng, M.; Wu, G. Iron and atrial fibrillation: A review. Pacing Clin. Electrophysiol. 2023, 46, 312–318. [Google Scholar] [CrossRef]
- Youn, J.Y.; Zhang, J.; Zhang, Y.; Chen, H.; Liu, D.; Ping, P.; Weiss, J.N.; Cai, H. Oxidative stress in atrial fibrillation: An emerging role of NADPH oxidase. J. Mol. Cell. Cardiol. 2013, 62, 72–79. [Google Scholar] [CrossRef]
- Woo, S.-H.; Kim, J.-C.; Eslenur, N.; Trinh, T.N.; Do, L.N.H. Modulations of Cardiac Functions and Pathogenesis by Reactive Oxygen Species and Natural Antioxidants. Antioxidants 2021, 10, 760. [Google Scholar] [CrossRef]
- Vermot, A.; Petit-Härtlein, I.; Smith, S.M.E.; Fieschi, F. NADPH Oxidases (NOX): An Overview from Discovery, Molecular Mechanisms to Physiology and Pathology. Antioxidants 2021, 10, 890. [Google Scholar] [CrossRef]
- Meloni, A.; Pistoia, L.; Ricchi, P.; Allo, M.; Rosso, R.; Cuccia, L.; Casini, T.; Cecinati, V.; Serra, M.; Rossi, V.; et al. Prospective changes of pancreatic iron in patients with thalassemia major and association with chelation therapy. Blood Adv. 2023, 7, 2237–2240. [Google Scholar] [CrossRef] [PubMed]
- Ricchi, P.; Meloni, A.; Pistoia, L.; Gamberini, M.R.; Cuccia, L.; Allo, M.; Putti, M.C.; Spasiano, A.; Rosso, R.; Cecinati, V.; et al. Longitudinal prospective comparison of pancreatic iron by magnetic resonance in thalassemia patients transfusion-dependent since early childhood treated with combination deferiprone-desferrioxamine vs deferiprone or deferasirox monotherapy. Blood Transfus. 2023. ahead of print. [Google Scholar] [CrossRef]
- Ozdogan, O.; Alp, A.; Turker, M.; Atabey, B. Determination of early cardiac deterioration in beta-thalassaemia major by echocardiography. Acta Cardiol. 2013, 68, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Janse, M.J. Why does atrial fibrillation occur? Eur. Heart J. 1997, 18 (Suppl. C), C12–C18. [Google Scholar] [CrossRef] [PubMed]
- Eckstein, J.; Verheule, S.; de Groot, N.; Allessie, M.; Schotten, U. Mechanisms of perpetuation of atrial fibrillation in chronically dilated atria. Progress. Biophys. Mol. Biol. 2008, 97, 435–451. [Google Scholar] [CrossRef] [PubMed]
- Barbero, U.; Fornari, F.; Guarguagli, S.; Gaglioti, C.M.; Longo, F.; Doronzo, B.; Anselmino, M.; Piga, A. Atrial fibrillation in β-thalassemia Major Patients: Diagnosis, Management and Therapeutic Options. Hemoglobin 2018, 42, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Dai, D.F.; Chen, T.; Johnson, S.C.; Szeto, H.; Rabinovitch, P.S. Cardiac aging: From molecular mechanisms to significance in human health and disease. Antioxid. Redox Signal 2012, 16, 1492–1526. [Google Scholar] [CrossRef]
- Mirza, M.; Strunets, A.; Shen, W.K.; Jahangir, A. Mechanisms of arrhythmias and conduction disorders in older adults. Clin. Geriatr. Med. 2012, 28, 555–573. [Google Scholar] [CrossRef]
- Pepe, A.; Gamberini, M.R.; Missere, M.; Pistoia, L.; Mangione, M.; Cuccia, L.; Spasiano, A.; Maffei, S.; Cadeddu, C.; Midiri, M.; et al. Gender differences in the development of cardiac complications: A multicentre study in a large cohort of thalassaemia major patients to optimize the timing of cardiac follow-up. Br. J. Haematol. 2018, 180, 879–888. [Google Scholar] [CrossRef]
| Variable | All Patients (N = 813) | Patients without Arrhythmias (N = 793) | Patients with Arrhythmias (N = 20) | p-Value |
|---|---|---|---|---|
| Females, N (%) | 444 (54.6) | 438 (55.2) | 6 (30.0) | 0.025 |
| Age (yrs) | 36.47 ± 10.71 | 36.27 ± 10.72 | 44.47 ± 6.81 | <0.0001 |
| Splenectomy, N (%) | 449 (55.2) | 435 (54.9) | 14 (70) | 0.179 |
| Pretransfusion hemoglobin (g/dL) | 9.67 ± 0.55 | 9.67 ± 0.55 | 9.68 ± 0.54 | 0.965 |
| Serum ferritin (ng/L) | 1061.24 ± 1330.85 | 1066.71 ± 1336.84 | 860.62 ± 1099.31 | 0.059 |
| Diabetes, N (%) | 89/796 (11.2) | 83/776 (10.7) | 6/20 (30.0) | 0.007 |
| MRI LIC (mg/g/dw) | 5.88 ± 8.72 | 5.79 ± 8.52 | 9.02 ± 14.50 | 0.556 |
| Global pancreas T2* (ms) | 13.16 ± 10.49 | 13.32 ± 10.54 | 6.79 ± 5.11 | 0.001 |
| Global heart T2* (ms) | 37.32 ± 9.40 | 37.33 ± 9.38 | 36.77 ± 10.45 | 0.977 |
| N. of segments with T2* < 20 ms | 1.49 ± 4.06 | 1.49 ± 4.06 | 1.40 ± 3.89 | 0.792 |
| LV EDVI (mL/m2) | 82.15 ± 16.84 | 82.14 ± 16.67 | 82.59 ± 24.00 | 0.845 |
| LV EF (%) | 62.41 ± 6.59 | 62.38 ± 6.61 | 63.88 ± 5.43 | 0.335 |
| LV mass index (g/m2) | 54.81 ± 13.43 | 54.76 ± 13.44 | 57.18 ± 12.68 | 0.410 |
| RV EDVI (mL/m2) | 80.34 ± 17.63 | 80.34 ± 17.48 | 80.06 ± 23.97 | 0.436 |
| RV EF (%) | 60.86 ± 7.09 | 60.79 ± 7.09 | 62.59 ± 6.34 | 0.076 |
| Replacement myocardial fibrosis, N (%) | 56/224 (25.0) | 54/218 (24.8) | 2/6 (33.3) | 0.633 |
| Left atrial area (cm2/m2) | 12.89 ± 2.50 | 12.82 ± 2.43 | 15.36 ± 3.69 | 0.002 |
| Right atrial area (cm2/m2) | 11.85 ± 2.38 | 11.79 ± 2.29 | 13.75 ± 3.95 | 0.014 |
| Independent Variable | Univariate Analysis | |
|---|---|---|
| HR (95% CI) | p-Value | |
| Male gender | 2.71 (1.04–7.06) | 0.041 |
| Age | 1.11 (1.05–1.17) | <0.0001 |
| Splenectomy | 1.75 (0.67–4.54) | 0.254 |
| Pretransfusion hemoglobin | 1.12 (0.49–2.56) | 0.780 |
| Serum ferritin | 1.00 (0.99–1.00) | 0.549 |
| Diabetes | 3.15 (1.21–8.19) | 0.019 |
| MRI LIC | 1.02 (0.99–1.05) | 0.113 |
| Global pancreas T2* | 0.89 (0.81–0.98) | 0.015 |
| Global heart T2* | 0.99 (0.95–1.04) | 0.893 |
| N. of segments with T2* < 20 ms | 0.99 (0.88–1.11) | 0.831 |
| LV EDVI | 1.00 (0.97–1.03) | 0.915 |
| LV EF | 1.03 (0.96–1.11) | 0.409 |
| LV mass index | 1.01 (0.98–1.05) | 0.580 |
| RV EDVI | 0.99 (0.97–1.03) | 0.870 |
| RV EF | 1.06 (0.99–1.13) | 0.122 |
| Replacement myocardial fibrosis | 1.46 (0.27–7.99) | 0.662 |
| Left atrial area index | 1.31 (1.16–1.49) | <0.0001 |
| Right atrial area index | 1.29 (1.12–1.45) | <0.0001 |
| Multivariate Analysis with Global Pancreas T2* as Fixed Independent Variable | |||
|---|---|---|---|
| Predictor | HR (95% CI) | p-Value | |
| Model 1: crude | pancreas T2* | 0.89 (0.81–0.98) | 0.015 |
| Model 2: inclusion of age and gender | pancreas T2* | 0.88 (0.78–0.99) | 0.026 |
| age | 1.11 (1.05–1.19) | 0.001 | |
| male gender | 3.48 (1.32–9.15) | 0.012 | |
| Model 3: inclusion of diabetes | pancreas T2* | 0.89 (0.82–0.98) | 0.023 |
| diabetes | 2.37 (0.91–6.21) | 0.078 | |
| Model 3: inclusion of left atrial area index | pancreas T2* | 0.89 (0.79–0.98) | 0.024 |
| left atrial area index | 1.39 (1.20–1.62) | <0.0001 | |
| Model 4: inclusion of previous arrhythmias | pancreas T2* | 0.89 (0.82–0.98) | 0.021 |
| previous arrhythmias | 11.32 (3.27–39.19) | <0.0001 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meloni, A.; Pistoia, L.; Ricchi, P.; Positano, V.; Longo, F.; Borsellino, Z.; Cecinati, V.; Messina, G.; Corigliano, E.; Rosso, R.; et al. Pancreatic T2* Magnetic Resonance Imaging for Prediction of Cardiac Arrhythmias in Transfusion-Dependent Thalassemia. J. Clin. Med. 2023, 12, 6015. https://doi.org/10.3390/jcm12186015
Meloni A, Pistoia L, Ricchi P, Positano V, Longo F, Borsellino Z, Cecinati V, Messina G, Corigliano E, Rosso R, et al. Pancreatic T2* Magnetic Resonance Imaging for Prediction of Cardiac Arrhythmias in Transfusion-Dependent Thalassemia. Journal of Clinical Medicine. 2023; 12(18):6015. https://doi.org/10.3390/jcm12186015
Chicago/Turabian StyleMeloni, Antonella, Laura Pistoia, Paolo Ricchi, Vincenzo Positano, Filomena Longo, Zelia Borsellino, Valerio Cecinati, Giuseppe Messina, Elisabetta Corigliano, Rosamaria Rosso, and et al. 2023. "Pancreatic T2* Magnetic Resonance Imaging for Prediction of Cardiac Arrhythmias in Transfusion-Dependent Thalassemia" Journal of Clinical Medicine 12, no. 18: 6015. https://doi.org/10.3390/jcm12186015








