The SARS-COV-2 Seroprevalence among Oncology Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. The Characterisation and Location of the Study
2.2. Patient Population
2.3. Protocol for IgG/IgM Antibody Detection
2.4. Statistical Analysis
3. Results
3.1. General Characteristics
3.2. Seroprevalence of COVID-19
3.3. Vaccination in the Population
3.4. The Regression Model
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ciotti, M.; Ciccozzi, M.; Terrinoni, A.; Jiang, W.; Wang, C.; Bernardini, S. The COVID-19 pandemic. Crit. Rev. Clin. Lab. Sci. 2020, 57, 365–388. [Google Scholar] [CrossRef] [PubMed]
- Junejo, Y.; Ozaslan, M.; Safdar, M.; Khailany, R.A.; Rehman, S.; Yousaf, W.; Khan, M.A. Novel SARS-CoV-2/COVID-19: Origin, pathogenesis, genes and genetic variations, immune responses and phylogenetic analysis. Gene Rep. 2020, 20, 100752. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Hong, N.; Zhou, X.; He, J.; Ma, Y.; Jiang, H.; Han, L.; Chang, F.; Shan, G.; Zhu, W. Evaluation of the secondary transmission pattern and epidemic prediction of COVID-19 in the four metropolitan areas of China. Front. Med. 2020, 7, 171. [Google Scholar] [CrossRef] [PubMed]
- Cucinotta, D.; Vanelli, M. WHO Declares COVID-19 a Pandemic. Acta Biomed. 2020, 91, 157–160. [Google Scholar]
- Sinha, S.; Kundu, C.N. Cancer and COVID-19: Why are cancer patients more susceptible to COVID-19? Med. Oncol. 2021, 38, 101. [Google Scholar] [CrossRef]
- Dai, M.; Liu, D.; Liu, M.; Zhou, F.; Li, G.; Chen, Z.; Zhang, Z.; You, H.; Wu, M.; Zheng, Q. Patients with Cancer Appear More Vulnerable to SARS-CoV-2: A Multicenter Study during the COVID-19 OutbreakPatients with Cancer in SARS-COV-2 Infection. Cancer Discov. 2020, 10, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Shaman, J. COVID-19 pandemic dynamics in India, the SARS-CoV-2 Delta variant and implications for vaccination. J. R. Soc. Interface 2022, 19, 20210900. [Google Scholar] [CrossRef]
- Made, F.; Wilson, K.; Jina, R.; Tlotleng, N.; Jack, S.; Ntlebi, V.; Kootbodien, T. Distribution of cancer mortality rates by province in South Africa. Cancer Epidemiol. 2017, 51, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Sacoronavirus Latest Vaccine Statistics. SA Corona Virus Online Portal. Available online: https://sacoronavirus.co.za/latest-vaccine-statistics/ (accessed on 5 August 2022).
- Maslo, C.; Friedland, R.; Toubkin, M.; Laubscher, A.; Akaloo, T.; Kama, B. Characteristics and outcomes of hospitalized patients in South Africa during the COVID-19 Omicron wave compared with previous waves. JAMA 2022, 327, 583–584. [Google Scholar] [CrossRef]
- Somdyala, N.I.; Bradshaw, D.; Gelderblom, W.C.; Parkin, D.M. Cancer incidence in a rural population of South Africa, 1998–2002. Int. J. Cancer 2010, 127, 2420–2429. [Google Scholar] [CrossRef]
- Moodley, J.; Constant, D.; Mwaka, A.D.; Scott, S.E.; Walter, F.M. Mapping awareness of breast and cervical cancer risk factors, symptoms and lay beliefs in Uganda and South Africa. PLoS ONE 2020, 15, e0240788. [Google Scholar] [CrossRef]
- Chai, P.; Yu, J.; Ge, S.; Jia, R.; Fan, X. Genetic alteration, RNA expression, and DNA methylation profiling of coronavirus disease 2019 (COVID-19) receptor ACE2 in malignancies: A pan-cancer analysis. J. Hematol. Oncol. 2020, 13, 43. [Google Scholar] [CrossRef]
- Yu, J.; Chai, P.; Ge, S.; Fan, X. Recent Understandings Toward Coronavirus Disease 2019 (COVID-19): From Bench to Bedside. Front. Cell. Dev. Biol. 2020, 8, 476. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, H.; Mikami, T.; Chopra, N.; Yamada, T.; Chernyavsky, S.; Rizk, D.; Cruz, C. Do patients with cancer have a poorer prognosis of COVID-19? An experience in New York City. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2020, 31, 1088–1089. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.C.; Sausville, E.L.; Girish, V.; Yuan, M.L.; Vasudevan, A.; John, K.M.; Sheltzer, J.M. Cigarette Smoke Exposure and Inflammatory Signaling Increase the Expression of the SARS-CoV-2 Receptor ACE2 in the Respiratory Tract. Dev. Cell. 2020, 53, 514–529.e3. [Google Scholar] [CrossRef]
- Liang, W.; Guan, W.; Chen, R.; Wang, W.; Li, J.; Xu, K.; Li, C.; Ai, Q.; Lu, W.; Liang, H.; et al. Cancer patients in SARS-CoV-2 infection: A nationwide analysis in China. Lancet Oncol. 2020, 21, 335–337. [Google Scholar] [CrossRef]
- Zhang, L.; Zhu, F.; Xie, L.; Wang, C.; Wang, J.; Chen, R.; Jia, P.; Guan, H.Q.; Peng, L.; Chen, Y.; et al. Clinical characteristics of COVID-19-infected cancer patients: A retrospective case study in three hospitals within Wuhan, China. Ann. Oncol. 2020, 31, 894–901. [Google Scholar] [CrossRef] [PubMed]
- SA’s COVID-19 Vaccination Rollout Kicks Off in Khayelitsha—SA Corona Virus Online Portal. Available online: https://sacoronavirus.co.za/2021/02/18/sas-covid-19-vaccination-rollout-kicks-off-in-khayelitsha/ (accessed on 8 August 2022).
- Burger, R.; Köhler, T.; Golos, A.M.; Buttenheim, A.M.; English, R.; Tameris, M.; Maughan-Brown, B. Longitudinal changes in COVID-19 vaccination intent among South African adults: Evidence from the NIDS-CRAM panel survey, February to May 2021. BMC Public Health 2022, 22, 422. [Google Scholar] [CrossRef]
- Ostergaard, S.D.; Schmidt, M.; Horvath-Puho, E.; Thomsen, R.W.; Sorensen, H.T. Thromboembolism and the Oxford-AstraZeneca COVID-19 vaccine: Side-effect or coincidence? Lancet 2021, 397, 1441–1443. [Google Scholar] [CrossRef]
- Wise, J. Covid-19: European countries suspend use of Oxford-AstraZeneca vaccine after reports of blood clots. BMJ 2021, 372, 699. [Google Scholar] [CrossRef]
- Chen, N.; Jotwani, A.; Li, A. Care Delivery in Cancer Patients with Asymptomatic COVID-19 Infection in a Tertiary, Safety-Net Hospital in Houston, Texas. Am. J. Clin. Oncol. 2021, 44, 409–412. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Natarajan, V.; Murthy, P.; Meghal, T.; Xu, Y.; Wiesel, O. The prevalence of asymptomatic COVID-19 infection in cancer patients. A cross-sectional study at a tertiary cancer center in New York City. Cancer Treat Res. Commun. 2021, 27, 100346. [Google Scholar] [CrossRef] [PubMed]
- Piciu, A.; Manole, S.; Piciu, D.; Dreve, T.; Roman, A. Asymptomatic COVID-19 cancer patients incidentally discovered during F18-FDG PET/CT monitoring. Med. Pharm. Rep. 2021, 94, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Lidenge, S.J.; Ngowi, J.R.; Peña, P.B.; Clegg, A.A.; Ngalamika, O.; Mwita, C.J.; Mwaiselage, J.; Wood, C. Lower SARS-CoV-2 Seroprevalence among Cancer Patients in Sub-Saharan Africa. J. Clin. Med. 2022, 11, 4428. [Google Scholar]
- Ofori-Asenso, R.; Ogundipe, O.; Agyeman, A.A.; Chin, K.L.; Mazidi, M.; Ademi, Z.; De Bruin, M.L.; Liew, D. Cancer is associated with severe disease in COVID-19 patients: A systematic review and meta-analysis. Ecancermedicalscience 2020, 14, 1047. [Google Scholar] [CrossRef]
- Tian, Y.; Qiu, X.; Wang, C.; Zhao, J.; Jiang, X.; Niu, W.; Huang, J.; Zhang, F. Cancer associates with risk and severe events of COVID-19: A systematic review and meta-analysis. Int. J. Cancer 2021, 148, 363–374. [Google Scholar] [CrossRef]
- Robilotti, E.V.; Babady, N.E.; Mead, P.A.; Rolling, T.; Perez-Johnston, R.; Bernardes, M.; Bogler, Y.; Caldararo, M.; Figueroa, C.J.; Glickman, M.S. Determinants of COVID-19 disease severity in patients with cancer. Nat. Med. 2020, 26, 1218–1223. [Google Scholar] [CrossRef]
- El-Sharkawi, D.; Iyengar, S. Haematological cancers and the risk of severe COVID-19: Exploration and critical evaluation of the evidence to date. Br. J. Haematol. 2020, 190, 336–345. [Google Scholar] [CrossRef]
- Ng, K.W.; Faulkner, N.; Cornish, G.H.; Rosa, A.; Harvey, R.; Hussain, S.; Ulferts, R.; Earl, C.; Wrobel, A.G.; Benton, D.J. Preexisting and de novo humoral immunity to SARS-CoV-2 in humans. Science 2020, 370, 1339–1343. [Google Scholar] [CrossRef]
- Ng, K.W.; Faulkner, N.; Wrobel, A.G.; Gamblin, S.J.; Kassiotis, G. Heterologous humoral immunity to human and zoonotic coronaviruses: Aiming for the achilles heel. Semin. Immunol. 2021, 55, 101507. [Google Scholar] [CrossRef]

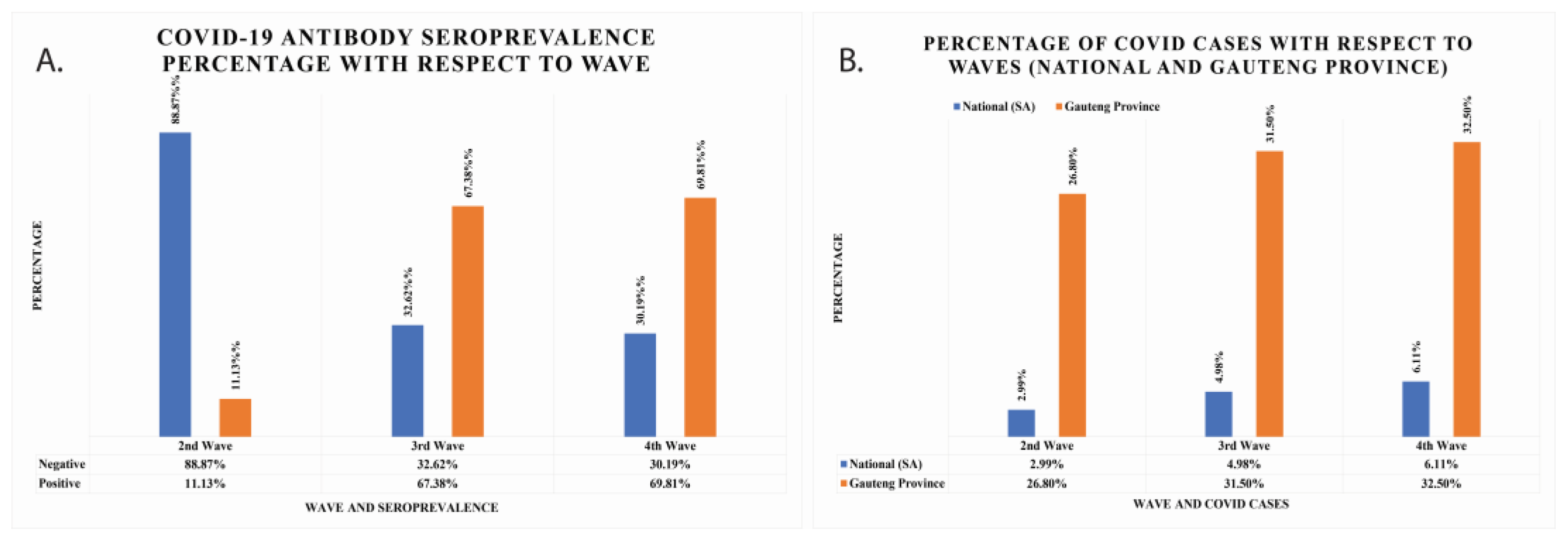

| Count of Antibody Test Status | Percentage % | Overall Seropositivity % | |
|---|---|---|---|
| IgG and IgM positive | 95 | 12.50 | Positive= 33.16 |
| IgG positive | 153 | 20.13 | |
| IgM positive | 4 | 0.53 | |
| Negative | 508 | 66.84 | Negative= 66.84 |
| Total | 760 |
| SARS-CoV-2 Seropositive | SARS-CoV-2 Seronegative | p-Value | |||
|---|---|---|---|---|---|
| n (252) | % | n (508) | % | ||
| Age (years) | 0.27 | ||||
| <40 | 37 | 14.70 | 82 | 16.14 | |
| 40–69 | 163 | 64.70 | 345 | 67.91 | |
| ≥70 | 52 | 20.60 | 81 | 15.94 | |
| Gender | 0.023 | ||||
| Male | 106 | 42.06 | 171 | 33.66 | |
| Female | 146 | 57.94 | 337 | 66.34 | |
| Smoker | 0.009 | ||||
| Yes | 32 | 12.70 | 71 | 13.98 | |
| No | 208 | 82.54 | 431 | 84.84 | |
| Ex-smoker | 12 | 4.76 | 6 | 1.18 | |
| Co-morbidities | |||||
| HIV | 0.14 | ||||
| Yes | 57 | 22.62 | 92 | 18.11 | |
| No | 195 | 77.38 | 416 | 81.89 | |
| Hypertension | <0.001 | ||||
| Yes | 89 | 35.32 | 66 | 12.99 | |
| No | 163 | 64.68 | 442 | 87.01 | |
| Diabetes | 0.941 | ||||
| Yes | 52 | 20.63 | 106 | 20.87 | |
| No | 200 | 79.37 | 402 | 79.13 | |
| Cancer type | <0.001 | ||||
| Breast Cancer | 39 | 15.48 | 158 | 31.10 | |
| Gynaecologic Cancer | 79 | 31.35 | 83 | 16.34 | |
| Colon and Rectal | 8 | 3.17 | 32 | 6.30 | |
| Prostate | 81 | 32.14 | 82 | 16.14 | |
| Others | 45 | 17.86 | 153 | 30.12 | |
| Symptoms | 0.009 | ||||
| Yes | 9 | 3.57 | 44 | 8.66 | |
| No | 243 | 96.43 | 464 | 91.34 | |
| SARS-CoV-2 Seropositive | SARS-CoV-2 Seronegative | p-Value | |||
|---|---|---|---|---|---|
| n (252) | % | n (508) | % | ||
| Treatment Intent | <0.001 | ||||
| Radical | 191 | 75.79 | 356 | 70.08 | |
| Palliative | 10 | 3.97 | 123 | 24.21 | |
| Others a | 51 | 20.24 | 29 | 5.71 | |
| Previous Cancer Treatment | |||||
| Chemotherapy | <0.001 | ||||
| Yes | 16 | 6.35 | 85 | 16.73 | |
| No | 236 | 93.65 | 423 | 83.27 | |
| Radiotherapy | 0.397 | ||||
| Yes | 32 | 12.70 | 54 | 10.63 | |
| No | 220 | 87.30 | 454 | 89.37 | |
| Zoladex | 0.531 | ||||
| Yes | 1 | 0.40 | 4 | 0.79 | |
| No | 251 | 99.60 | 504 | 99.21 | |
| Surgery | <0.001 | ||||
| Yes | 40 | 15.87 | 178 | 35.04 | |
| No | 212 | 84.13 | 330 | 64.96 | |
| Othersb | <0.001 | ||||
| Yes | 174 | 69.05 | 265 | 52.17 | |
| No | 78 | 30.95 | 243 | 47.83 | |
| Current Cancer Treatment | |||||
| Chemotherapy | <0.001 | ||||
| Yes | 34 | 13.49 | 231 | 45.47 | |
| No | 218 | 86.51 | 277 | 54.53 | |
| Radiotherapy | <0.001 | ||||
| Yes | 100 | 39.68 | 119 | 23.43 | |
| No | 152 | 60.32 | 389 | 76.57 | |
| Zoladex | 0.469 | ||||
| Yes | 4 | 1.59 | 5 | 0.98 | |
| No | 248 | 98.41 | 503 | 99.02 | |
| Surgery | 0.612 | ||||
| Yes | 1 | 0.40 | 1 | 0.20 | |
| No | 251 | 99.60 | 507 | 99.80 | |
| Othersb | <0.001 | ||||
| Yes | 122 | 48.41 | 177 | 34.84 | |
| No | 130 | 51.59 | 331 | 65.16 | |
| Odds Ratio (OR) | 95% CI | ||
|---|---|---|---|
| Lower | Upper | ||
| Age Group | |||
| <40 | Ref | ||
| 40–69 | 0.706 | 0.419 | 1.189 |
| ≥70 | 0.728 | 0.365 | 1.452 |
| Gender | |||
| Male | Ref | ||
| Female | 0.773 | 0.404 | 1.479 |
| Smoker | |||
| No | Ref | ||
| Yes | 0.987 | 0.574 | 1.699 |
| Ex-smoker | 2.154 | 0.704 | 6.591 |
| Co-morbidities | |||
| HIV | |||
| No | Ref | ||
| Yes | 1.251 | 0.778 | 2.011 |
| Hypertension | |||
| No | Ref | ||
| Yes | 1.820 | 1.181 | 2.804 |
| Diabetes | |||
| No | Ref | ||
| Yes | 1.373 | 0.856 | 2.203 |
| Cancer type | |||
| Others | Ref | ||
| Breast Cancer | 1.908 | 0.988 | 3.683 |
| Colon and Rectal Cancer | 1.532 | 0.596 | 3.939 |
| Gynaecologic Cancer | 2.224 | 1.187 | 4.166 |
| Prostate Cancer | 2.500 | 1.290 | 4.844 |
| Symptoms | |||
| No | Ref | ||
| Yes | 0.661 | 0.265 | 1.407 |
| Treatment Intent | |||
| Others/None | Ref | ||
| Radical | 0.081 | 0.031 | 0.210 |
| Palliative | 0.303 | 0.161 | 0.571 |
| Previous Cancer Treatment | |||
| Chemotherapy | |||
| No | Ref | ||
| Yes | 0.471 | 0.220 | 1.010 |
| Radiotherapy | |||
| No | Ref | ||
| Yes | 0.752 | 0.316 | 1.786 |
| Zoladex | |||
| No | Ref | ||
| Yes | 0.144 | 0.013 | 1.556 |
| Surgery | |||
| No | Ref | ||
| Yes | 0.411 | 0.179 | 0.943 |
| Others/Completed/None | |||
| No | Ref | ||
| Yes | 0.668 | 0.284 | 1.570 |
| Current Cancer Treatment | |||
| Chemotherapy | |||
| No | Ref | ||
| Yes | 0.435 | 0.180 | 1.051 |
| Radiotherapy | |||
| No | Ref | ||
| Yes | 1.237 | 0.457 | 3.351 |
| Zoladex | |||
| No | Ref | ||
| Yes | 0.853 | 0.167 | 4.353 |
| Surgery | |||
| No | Ref | ||
| Yes | 2.522 | 0.123 | 51.689 |
| Others/Completed/None | |||
| No | Ref | ||
| Yes | 0.956 | 0.325 | 2.814 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kgatle, M.; Das, R.; Lawal, I.; Boshomane, T.; Mokoala, K.; Gaspar, C.; Mbokazi, L.; Nkambule, N.; Gow, V.; Ndlovu, H.; et al. The SARS-COV-2 Seroprevalence among Oncology Patients. J. Clin. Med. 2023, 12, 529. https://doi.org/10.3390/jcm12020529
Kgatle M, Das R, Lawal I, Boshomane T, Mokoala K, Gaspar C, Mbokazi L, Nkambule N, Gow V, Ndlovu H, et al. The SARS-COV-2 Seroprevalence among Oncology Patients. Journal of Clinical Medicine. 2023; 12(2):529. https://doi.org/10.3390/jcm12020529
Chicago/Turabian StyleKgatle, Mankgopo, Rajesh Das, Ismaheel Lawal, Tebatso Boshomane, Kgomotso Mokoala, Cattleya Gaspar, Lydia Mbokazi, Nonhlanhla Nkambule, Veronique Gow, Honest Ndlovu, and et al. 2023. "The SARS-COV-2 Seroprevalence among Oncology Patients" Journal of Clinical Medicine 12, no. 2: 529. https://doi.org/10.3390/jcm12020529










