A Systematic Review and Meta-Analysis of the Brief Cognitive Assessment for Multiple Sclerosis (BICAMS) International Validations
Abstract
:1. Introduction
2. Methods
2.1. Search Strategy
2.2. Selection Criteria
2.3. Quality Assessment
2.4. Statistical Analysis
3. Results
3.1. Search Results
3.2. Study Characteristics and Sample Demographics
3.3. Patient Disease Information
3.4. Correlations between BICAMS and Sample Variables
3.5. Quality Ratings
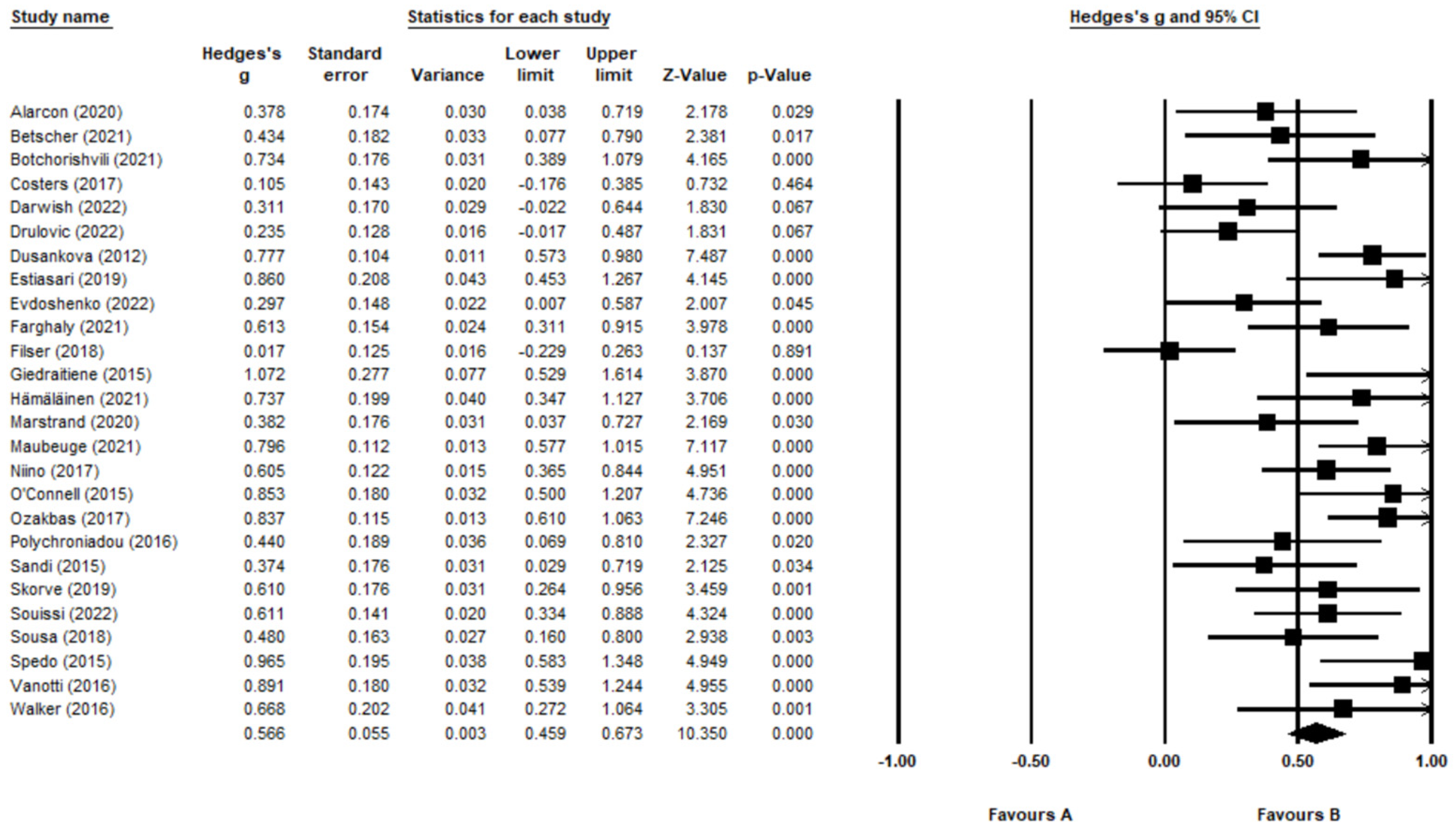
3.6. Meta-Analysis of BICAMS Validation Studies
4. Discussion
4.1. Summary of Findings
4.2. Strengths
4.3. Limitations
4.4. Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Morley, J.E.; Morris, J.C.; Berg-Weger, M.; Borson, S.; Carpenter, B.D.; del Campo, N.; Dubois, B.; Fargo, K.; Fitten, L.J.; Flaherty, J.H.; et al. Brain health: The importance of recognizing cognitive impairment: An IAGG consensus conference. J. Am. Med. Dir. Assoc. 2015, 16, 731–739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nyenhuis, D.L.; Reckow, J. Office- and bedside-based screening for cognitive impairment and the Dementias. Clin. Geriatr. Med. 2023, 39, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Franzen, S.; Watermeyer, T.J.; Pomati, S.; Papma, J.M.; Nielsen, T.R.; Narme, P.; Mukadam, N.; Lozano-Ruiz, Á.; Ibanez-Casas, I.; Goudsmit, M.; et al. Cross-cultural neuropsychological assessment in Europe: Position statement of the European consortium on Cross-Cultural Neuropsychology (eccron). Clin. Neuropsychol. 2022, 36, 546–557. [Google Scholar] [CrossRef]
- Merkley, T.L.; Esopenko, C.; Zizak, V.S.; Bilder, R.M.; Strutt, A.M.; Tate, D.F.; Irimia, A. Challenges and opportunities for harmonization of cross-cultural neuropsychological data. Neuropsychology 2022. [Google Scholar] [CrossRef]
- Nielsen, T.R. Cognitive assessment in culturally, linguistically, and educationally diverse older populations in Europe. Am. J. Alzheimer’s Dis. Other Dement. 2022, 37, 153331752211170. [Google Scholar] [CrossRef] [PubMed]
- Dobson, R.; Giovannoni, G. Multiple sclerosis—A review. Eur. J. Neurol. 2018, 26, 27–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walton, C.; King, R.; Rechtman, L.; Kaye, W.; Leray, E.; Marrie, R.; Robertson, N.; La Rocca, N.; Uitdehaag, B.; van der Mei, I.; et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult. Scler. J. 2020, 26, 1816–1821. [Google Scholar] [CrossRef]
- McGinley, M.; Goldschmidt, C.; Rae-Grant, A. Diagnosis and Treatment of Multiple Sclerosis. JAMA 2021, 325, 765. [Google Scholar] [CrossRef]
- Benedict, R.; Amato, M.; DeLuca, J.; Geurts, J. Cognitive impairment in multiple sclerosis: Clinical management, MRI, and therapeutic avenues. Lancet Neurol. 2020, 19, 860–871. [Google Scholar] [CrossRef]
- Brochet, B.; Ruet, A. Cognitive Impairment in Multiple Sclerosis With Regards to Disease Duration and Clinical Phenotypes. Front. Neurol. 2019, 10, 261. [Google Scholar] [CrossRef]
- Ruano, L.; Portaccio, E.; Goretti, B.; Niccolai, C.; Severo, M.; Patti, F.; Cilia, S.; Gallo, P.; Grossi, P.; Ghezzi, A.; et al. Age and disability drive cognitive impairment in multiple sclerosis across disease subtypes. Mult. Scler. J. 2016, 23, 1258–1267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gil-González, I.; Martín-Rodríguez, A.; Conrad, R.; Pérez-San-Gregorio, M. Quality of life in adults with multiple sclerosis: A systematic review. BMJ Open 2020, 10, e041249. [Google Scholar] [CrossRef] [PubMed]
- Kavaliunas, A.; Danylaitė Karrenbauer, V.; Binzer, S.; Hillert, J. Systematic review of the socioeconomic consequences in patients with multiple sclerosis with different levels of disability and cognitive function. Front. Neurol. 2022, 12, 2341. [Google Scholar] [CrossRef] [PubMed]
- Bruce, J.; Bruce, A.; Lynch, S.; Thelen, J.; Lim, S.; Smith, J.; Catley, D.; Reed, D.D.; Jarmolowicz, D.P. Probability discounting of treatment decisions in multiple sclerosis: Associations with disease knowledge, neuropsychiatric status, and adherence. Psychopharmacology 2018, 235, 3303–3313. [Google Scholar] [CrossRef]
- Gomes, K.; Ruiz, J.; Raskin, S.; Turner, A.; DelMastro, H.; Neto, L.; Gromisch, E. The Role of Cognitive Impairment on Physical Therapy Attendance and Outcomes in Multiple Sclerosis. J. Neurol. Phys. Ther. 2022, 46, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Drake, A.; Fuchs, T.; Dwyer, M.; Zivadinov, R.; Chapman, B.P.; Weinstock-Guttman, B.; Benedict, R.H.B. Longitudinal personality change associated with cognitive decline in multiple sclerosis. Mult. Scler. J. 2018, 24, 1909–1912. [Google Scholar] [CrossRef]
- Krasniuk, S.; Classen, S.; Morrow, S.; Alvarez, L.; He, W.; Srinivasan, S.; Monahan, M. Clinical predictors of driving simulator performance in drivers with multiple sclerosis. Mult. Scler. J. 2021, 27, 2085–2092. [Google Scholar] [CrossRef]
- Langdon, D.; Young, C. Fast Facts: Cognition in Multiple Sclerosis; S. Karger Publishers Ltd.: Oxford, UK, 2023. [Google Scholar]
- Walker, L.; Gardner, C.; Freedman, M.; MacLean, H.; Rush, C.; Bowman, M. Research-to-Practice Gaps in Multiple Sclerosis Care for Patients with Subjective Cognitive, Mental Health, and Psychosocial Concerns in a Canadian Center. Int. J. MS Care 2019, 21, 243–248. [Google Scholar] [CrossRef] [Green Version]
- Romero, K.; Shammi, P.; Feinstein, A. Neurologists’ accuracy in predicting cognitive impairment in multiple sclerosis. Mult. Scler. Relat. Disord. 2015, 4, 291–295. [Google Scholar] [CrossRef]
- Elwick, H.; Smith, L.; Mhizha-Murira, J.; Topcu, G.; Leighton, P.; Drummond, A.; Evangelou, N.; das Nair, R. Cognitive assessment in multiple sclerosis clinical care: A qualitative evaluation of stakeholder perceptions and preferences. Neuropsychol. Rehabil. 2021, 32, 1456–1474. [Google Scholar] [CrossRef]
- Kalb, R.; Beier, M.; Benedict, R.; Charvet, L.; Costello, K.; Feinstein, A.; Gingold, J.; Goverover, Y.; Halper, J.; Harris, C.; et al. Recommendations for cognitive screening and management in multiple sclerosis care. Mult. Scler. J. 2018, 24, 1665–1680. [Google Scholar] [CrossRef] [Green Version]
- Klein, O.; das Nair, R.; Ablewhite, J.; Drummond, A. Assessment and management of cognitive problems in people with multiple sclerosis: A National Survey of Clinical Practice. Int. J. Clin. Pract. 2018, 73, e13300. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence (NICE). Multiple Sclerosis in Adults: Management (NG220); NICE: London, UK, 2022. [Google Scholar]
- Aan.com. 2014. Available online: https://www.aan.com/siteassets/home-page/policy-and-guidelines/quality/quality-measures/14msmeasureset_pg.pdf (accessed on 15 March 2022).
- Meca-Lallana, V.; Gascón-Giménez, F.; Ginestal-López, R.; Higueras, Y.; Téllez-Lara, N.; Carreres-Polo, J.; Eichau-Madueño, S.; Romero-Imbroda, J.; Vidal-Jordana, Á.; Pérez-Miralles, F. Cognitive impairment in multiple sclerosis: Diagnosis and monitoring. Neurol. Sci. 2021, 42, 5183–5193. [Google Scholar] [CrossRef]
- Thrue, C.; Riemenschneider, M.; Hvid, L.; Stenager, E.; Dalgas, U. Time matters: Early-phase multiple sclerosis is accompanied by considerable impairments across multiple domains. Mult. Scler. J. 2021, 27, 1477–1485. [Google Scholar] [CrossRef]
- Langdon, D.; Amato, M.; Boringa, J.; Brochet, B.; Foley, F.; Fredrikson, S.; Hämäläinen, P.; Hartung, H.-P.; Penner, I.K.; Reder, A.T.; et al. Recommendations for a Brief International Cognitive Assessment for Multiple Sclerosis (BICAMS). Mult. Scler. J. 2012, 18, 891–898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, A. Symbol Digit Modalities Test (SDMT) Manual (revised); Western Psychological Services: Los Angeles, CA, USA, 1982. [Google Scholar]
- Portaccio, E.; Amato, M.P. Cognitive impairment in multiple sclerosis: An update on assessment and Management. NeuroSci 2022, 3, 667–676. [Google Scholar] [CrossRef]
- Delis, D.C.; Kramer, J.H.; Kaplan, E.; Ober, B.A. California Verbal Learning Test, 2nd ed.; Psychological Corporation: San Antonio, TX, USA, 2000. [Google Scholar]
- Benedict, R. The Brief Visuospatial Memory Test Revised (BVMT-R); Psychosocial Assessment Resources Inc.: Lutz, FL, USA, 1997. [Google Scholar]
- Benedict, R.; Amato, M.; Boringa, J.; Brochet, B.; Foley, F.; Fredrikson, S.; Hamalainen, P.; Hartung, H.; Krupp, L.; Penner, I.; et al. Brief International Cognitive Assessment for MS (BICAMS): International standards for validation. BMC Neurol. 2012, 12, 55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corfield, F.; Langdon, D. A Systematic Review and Meta-Analysis of the Brief Cognitive Assessment for Multiple Sclerosis (BICAMS). Neurol. Ther. 2018, 7, 287–306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freedman, M.; Devonshire, V.; Duquette, P.; Giacomini, P.; Giuliani, F.; Levin, M.; Montalban, X.; Morrow, S.A.; Oh, J.; Rotstein, D.; et al. Treatment Optimization in Multiple Sclerosis: Canadian MS Working Group Recommendations. Can. J. Neurol. Sci. J. Can. Des Sci. Neurol. 2020, 47, 437–455. [Google Scholar] [CrossRef]
- Artemiadis, A.; Bakirtzis, C.; Chatzittofis, A.; Christodoulides, C.; Nikolaou, G.; Boziki, M.; Grigoriadis, N. Brief international cognitive assessment for multiple sclerosis (BICAMS) cut-off scores for detecting cognitive impairment in multiple sclerosis. Mult. Scler. Relat. Disord. 2021, 49, 102751. [Google Scholar] [CrossRef]
- Paul, S.; Brown, A.; Hughes, A. Cognitive Assessment of Individuals with Multiple Sclerosis in the Arab World: A Systematic Review. Neuropsychol. Rev. 2019, 29, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, T.P. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, 264–269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borenstein, M.; Hedges, L.; Higgins, J.; Rothstein, H. Comprehensive Meta-Analysis Version 3; Biostat: Englewood, NJ, USA, 2005. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioural Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillside, NJ, USA, 1988. [Google Scholar]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. Fixed-effect versus random-effects models. Introduction to meta-analysis. Introd. Meta-Anal. 2009, 77, 86. [Google Scholar] [CrossRef]
- Riley, R.D.; Higgins, J.P.T.; Deeks, J.J. Interpretation of random effects meta-analyses. BMJ 2011, 342, d549. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [Green Version]
- Sterne, J.; Harbord, R. Funnel Plots in Meta-analysis. Stata J. Promot. Commun. Stat. Stata 2004, 4, 127–141. [Google Scholar] [CrossRef] [Green Version]
- Duval, S.; Tweedie, R. Trim and Fill: A Simple Funnel-Plot-Based Method of Testing and Adjusting for Publication Bias in Meta-Analysis. Biometrics 2000, 56, 455–463. [Google Scholar] [CrossRef]
- Alarcón, A.N.; Ayala, O.D.; García, J.R.; Montañés, P. Validation of the brief international cognitive assessment for multiple sclerosis (BICAMS) in a Colombian population. Mult. Scler. Relat. Disord. 2020, 42, 102072. [Google Scholar] [CrossRef]
- Betscher, E.; Guenter, W.; Langdon, D.W.; Bonek, R. Polish validation of the Brief International Cognitive Assessment For Multiple Sclerosis (BICAMS Battery): Correlation of cognitive impairment with mood disorders and fatigue. Neurol. Neurochir. Pol. 2021, 55, 59–66. [Google Scholar] [CrossRef]
- Botchorishvili, N.; Shiukashvili, N.; Mikeladze, N.; Dzagnidze, A.; Miqava, N.; Tighashvili, M.; Janelidze, M. Validity and reliability of the Georgian-language brief international cognitive assessment for multiple sclerosis (BICAMS)&NBSP. BMC Neurol. 2021, 21, 218. [Google Scholar] [CrossRef]
- Costers, L.; Gielen, J.; Eelen, P.L.; Schependom, J.V.; Laton, J.; Remoortel, A.V.; Vanzeir, E.; Wijmeersch, B.V.; Seeldrayers, P.; Haelewyck, M.-C.; et al. Does including the full CVLT-II and BVMT-R improve BICAMS? evidence from a Belgian (dutch) validation study. Mult. Scler. Relat. Disord. 2017, 18, 33–40. [Google Scholar] [CrossRef] [Green Version]
- Darwish, H.; Zeinoun, P.; Farran, N.; Ghusn, H.; Yamout, B.; Khoury, S.J. The Brief International Cognitive Assessment in multiple sclerosis (BICAMS): Validation in Arabic and Lebanese normative values. J. Int. Neuropsychol. Soc. 2022, 28, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Drulović, J.; Tončev, G.; Nadj, Č.; Obradović, D.; Eraković, J.; Mesaroš, Š.; Čukić, M.; Aleksić, D.; Andabaka, M.; Ivanović, J.; et al. Validation of the brief international cognitive assessment for multiple sclerosis (BICAMS) in a large cohort of relapsing-remitting MS patients. Acta Clin. Croat. 2022, 61, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Dusankova, J.B.; Kalincik, T.; Havrdova, E.; Benedict, R.H. Cross cultural validation of the minimal assessment of cognitive function in multiple sclerosis (MACFIMS) and the Brief International Cognitive Assessment For Multiple Sclerosis (BICAMS). Clin. Neuropsychol. 2012, 26, 1186–1200. [Google Scholar] [CrossRef] [PubMed]
- Estiasari, R.; Fajrina, Y.; Lastri, D.N.; Melani, S.; Maharani, K.; Imran, D.; Pangeran, D.; Sitorus, F. Validity and reliability of Brief International Cognitive Assessment For Multiple Sclerosis (BICAMS) in Indonesia and the correlation with quality of life. Neurol. Res. Int. 2019, 2019, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Evdoshenko, E.; Laskova, K.; Shumilina, M.; Nekrashevich, E.; Andreeva, M.; Neofidov, N.; Kalinin, I.; Nikitchenko, D.; Rogozina, A.; Kupaeva, A.; et al. Validation of the brief international cognitive assessment for multiple sclerosis (BICAMS) in the Russian population. J. Int. Neuropsychol. Soc. 2021, 28, 503–510. [Google Scholar] [CrossRef]
- Farghaly, M.; Langdon, D.W.; Shalaby, N.M.; Shehata, H.S.; Abokrysha, N.T.; Hassan, A.; Hegazy, M.I.; Elmazny, A.; Ahmed, S.; Shaheen, S.; et al. Reliability and validity of Arabic version of the Brief International Cognitive Assessment For Multiple Sclerosis: Egyptian dialect. Egypt. J. Neurol. Psychiatry Neurosurg. 2021, 57, 51. [Google Scholar] [CrossRef]
- Filser, M.; Schreiber, H.; Pöttgen, J.; Ullrich, S.; Lang, M.; Penner, I.K. The Brief International Cognitive Assessment in multiple sclerosis (BICAMS): Results from the German Validation Study. J. Neurol. 2018, 265, 2587–2593. [Google Scholar] [CrossRef]
- Giedraitienė, N.; Kizlaitienė, R.; Kaubrys, G. The bicams battery for assessment of Lithuanian-speaking multiple sclerosis patients: Relationship with age, Education, disease disability, and duration. Med. Sci. Monit. 2015, 21, 3853–3859. [Google Scholar] [CrossRef]
- Hämäläinen, P.; Leo, V.; Therman, S.; Ruutiainen, J. Validation of the Finnish version of the Brief International Cognitive Assessment For Multiple Sclerosis (BICAMS) and evaluation of the applicability of the Multiple Sclerosis Neuropsychological Questionnaire (MSNQ) and the fatigue scale for motor and cognitive functions (FSMC). Brain Behav. 2021, 11, e02087. [Google Scholar] [CrossRef]
- Marstrand, L.; Østerberg, O.; Walsted, T.; Skov, A.C.; Schreiber, K.I.; Sellebjerg, F. Brief international cognitive assessment for multiple sclerosis (BICAMS): A Danish validation study of sensitivity in early stages of ms. Mult. Scler. Relat. Disord. 2020, 37, 101458. [Google Scholar] [CrossRef]
- Maubeuge, N.; Deloire MS, A.; Brochet, B.; Ehrlé, N.; Charré-Morin, J.; Saubusse, A.; Ruet, A. French validation of the Brief International Cognitive Assessment For Multiple Sclerosis. Rev. Neurol. 2021, 177, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Niino, M.; Fukazawa, T.; Kira, J.-I.; Okuno, T.; Mori, M.; Sanjo, N.; Ohashi, T.; Fukaura, H.; Fujimori, J.; Shimizu, Y.; et al. Validation of the brief international cognitive assessment for multiple sclerosis in Japan. Mult. Scler. J.-Exp. Transl. Clin. 2017, 3, 205521731774897. [Google Scholar] [CrossRef]
- Ozakbas, S.; Yigit, P.; Cinar, B.P.; Limoncu, H.; Kahraman, T.; Kösehasanoğulları, G. The Turkish validation of the Brief International Cognitive Assessment For Multiple Sclerosis (BICAMS) battery. BMC Neurol. 2017, 17, 208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Connell, K.; Langdon, D.; Tubridy, N.; Hutchinson, M.; McGuigan, C. A preliminary validation of the Brief International Cognitive Assessment For Multiple Sclerosis (BICAMS) tool in an Irish population with multiple sclerosis (MS). Mult. Scler. Relat. Disord. 2015, 4, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Polychroniadou, E.; Bakirtzis, C.; Langdon, D.; Lagoudaki, R.; Kesidou, E.; Theotokis, P.; Tsalikakis, D.; Poulatsidou, K.; Kyriazis, O.; Boziki, M.; et al. Validation of the brief international cognitive assessment for multiple sclerosis (BICAMS) in Greek population with multiple sclerosis. Mult. Scler. Relat. Disord. 2016, 9, 68–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandi, D.; Rudisch, T.; Füvesi, J.; Fricska-Nagy, Z.; Huszka, H.; Biernacki, T.; Langdon, D.W.; Langane, É.; Vécsei, L.; Bencsik, K. The Hungarian validation of the Brief International Cognitive Assessment For Multiple Sclerosis (BICAMS) Battery and the correlation of cognitive impairment with fatigue and quality of life. Mult. Scler. Relat. Disord. 2015, 4, 499–504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skorve, E.; Lundervold, A.J.; Torkildsen, Ø.; Myhr, K.-M. The Norwegian translation of the Brief International Cognitive Assessment For Multiple Sclerosis (BICAMS). Mult. Scler. Relat. Disord. 2019, 36, 101408. [Google Scholar] [CrossRef] [PubMed]
- Souissi, A.; Mrabet, S.; Ferchichi, W.; Gharbi, A.; Nasri, A.; Djebara, M.B.; Kacem, I.; Gouider, R. Tunisian version of the Brief International Cognitive Assessment For Multiple Sclerosis: Validation and normative values. Mult. Scler. Relat. Disord. 2022, 58, 103444. [Google Scholar] [CrossRef]
- Sousa, C.; Rigueiro-Neves, M.; Miranda, T.; Alegria, P.; Vale, J.; Passos, A.M.; Langdon, D.; Sá, M. Validation of the brief international cognitive assessment for multiple sclerosis (BICAMS) in the Portuguese population with multiple sclerosis. BMC Neurol. 2018, 18, 172. [Google Scholar] [CrossRef]
- Spedo, C.T.; Frndak, S.E.; Marques, V.D.; Foss, M.P.; Pereira, D.A.; Carvalho L de Guerreiro, C.T.; Conde, R.M.; Fusco, T.; Pereira, A.J.; Gaino, S.B.; et al. Cross-cultural adaptation, reliability, and validity of the BICAMS in Brazil. Clin. Neuropsychol. 2015, 29, 836–846. [Google Scholar] [CrossRef]
- Vanotti, S.; Smerbeck, A.; Benedict, R.H.; Caceres, F. A new assessment tool for patients with multiple sclerosis from Spanish-speaking countries: Validation of the brief international cognitive assessment for MS (BICAMS) in Argentina. Clin. Neuropsychol. 2016, 30, 1023–1031. [Google Scholar] [CrossRef] [PubMed]
- Walker LA, S.; Osman, L.; Berard, J.A.; Rees, L.M.; Freedman, M.S.; MacLean, H.; Cousineau, D. Brief international cognitive assessment for multiple sclerosis (BICAMS): Canadian contribution to the International Validation Project. J. Neurol. Sci. 2016, 362, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet. Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef]
- Kurtzke, J. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444. [Google Scholar] [CrossRef] [Green Version]
- Costa, S.; Genova, H.; DeLuca, J.; Chiaravalloti, N. Information processing speed in multiple sclerosis: Past, present, and future. Mult. Scler. J. 2017, 23, 772–789. [Google Scholar] [CrossRef] [PubMed]
- Chiaravalloti, N.; DeLuca, J. Cognitive impairment in multiple sclerosis. Lancet Neurol. 2008, 7, 1139–1151. [Google Scholar] [CrossRef]
- Chiaravalloti, N.D.; DeLuca, J.; Salter, A.; Amato, M.P.; Brichetto, G.; Chataway, J.; Dalgas, U.; Farrell, R.; Feys, P.; Filippi, M.; et al. The relationship between processing speed and verbal and non-verbal new learning and memory in Progressive Multiple Sclerosis. Mult. Scler. J. 2022, 28, 1783–1792. [Google Scholar] [CrossRef]
- Sousa, V.D.; Rojjanasrirat, W. Translation, adaptation and validation of instruments or scales for use in cross-cultural health care research: A clear and user-friendly guideline. J. Eval. Clin. Pract. 2011, 17, 268–274. [Google Scholar] [CrossRef]
- Al-Jawahiri, F.; Nielsen, T.R. Effects of acculturation on the Cross-Cultural Neuropsychological Test Battery (CNTB) in a culturally and linguistically diverse population in Denmark. Arch. Clin. Neuropsychol. 2020, 36, 381–393. [Google Scholar] [CrossRef]
- Reilly, G.; Mahkawnghta, A.; Jelinek, P.; De Livera, A.; Weiland, T.; Brown, C.; Taylor, K.L.; Neate, S.L.; Jelinek, G.A.; Marck, C.H. International Differences in Multiple Sclerosis Health Outcomes and Associated Factors in a Cross-sectional Survey. Front. Neurol. 2017, 8, 229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dobson, R.; Rice, D.R.; D’hooghe, M.; Horne, R.; Learmonth, Y.; Mateen, F.J.; Marck, C.H.; Reyes, S.; Williams, M.J.; Giovannoni, G.; et al. Social Determinants of Health in multiple sclerosis. Nat. Rev. Neurol. 2022, 18, 723–734. [Google Scholar] [CrossRef] [PubMed]
- Rivera, V. Multiple Sclerosis: A Global Concern with Multiple Challenges in an Era of Advanced Therapeutic Complex Molecules and Biological Medicines. Biomedicines 2018, 6, 112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moghaddam, V.; Dickerson, A.; Bazrafshan, E.; Seyedhasani, S.; Najafi, F.; Hadei, M.; Momeni, J.; Moradi, G.; Sarmadi, M. Socioeconomic determinants of global distribution of multiple sclerosis: An ecological investigation based on Global Burden of Disease data. BMC Neurol. 2021, 21, 145. [Google Scholar] [CrossRef]






| Search Terms |
|---|
| “Multiple Sclerosis” OR “MS” OR “Clinically Isolated Syndrome” OR “CIS” AND “Brief International Cognitive Assessment for Multiple Sclerosis” OR “BICAMS” AND “Validation” OR “International Validation” OR “Validity” OR “Sensitivity” |
| Study | Country | Number of Participants | Age in Years Mean (SD), {Median}, [Range] | Gender (Female %) | Education in Years Mean (SD), {Median}, [Range] | Employment (Employed %) | MS Phenotype %(CIS/RR/SP/PP/PR) | Disease Duration in Years Mean (SD), {Median}, [Range] | EDSS Mean (SD), {Median}, [Range] |
|---|---|---|---|---|---|---|---|---|---|
| Alarcón et al. [46] | |||||||||
| MS | Columbia | 50 | 41.44 (10.99) | 64% | 14.76 (2.61) | Nr | 0/100/0/0/0 | 7.66 (5.61) | 1.33 (1.54) |
| HC | 100 | 37.75 (12.63) | 48% | 14.73 (3.57) | Nr | - | - | - | |
| Betscher et al. [47] | |||||||||
| MS | Poland | 61 | {39} | 74% | {13} | 84% | 0/74/20/6/0 | RR = {5} SP = {19.5} PP = {7.5} | RR = {3} SP = {4.75} PP = {4.5} |
| HC | 61 | {37} | 75% | {13} | 98% | - | - | - | |
| Botchorishvili et al. [48] | |||||||||
| MS | Georgia | 68 | 39.2 (9.9) | 71% | 14.3 (2.1) | 57% | 0/76/18/6/0 | 7.0 (5.7) | 3.3 (1.6) |
| HC | 68 | 38.5 (9.9) | 68% | 14.5 (1.9) | 84% | - | - | - | |
| Costers et al. [49] | |||||||||
| MS | Belgium | 97 | 45.42 (9.24) | 68% | 14.28 (1.86) | Nr | 0/84/12/4/0 | 12.97 (7.16) | 3.50 (2.50) |
| HC | 97 | 43.52 (12.69) | 75% | 14.69 (1.61) | Nr | - | - | - | |
| Darwish et al. [50] | |||||||||
| MS | Lebanon | 43 | 36.06 (12.37) | 81.4% | 14.63 (3.17) | 48.84% | 0/81/14/5/0 | 8.61 (7.36) | 1.89 (1.7) |
| HC | 180 | 45.01 (19.36) | 60% | 15.13 (3) | 56.11% | - | - | - | |
| Drulović et al. [51] | |||||||||
| MS | Serbia | 500 | 39.9 (9.4) | 70.2% | 14.0 (2.9) | Nr | 0/100/0/0/0 | 9.2 (6.7) | {2.0} |
| HC | 69 | 40.3 (11.5) | 63.77% | 14.1 (3.4) | Nr | - | - | - | |
| Dusankova et al. [52] | |||||||||
| MS | Czech Republic | 367 | 34 (10) | 68% | 14 (3) | 40% | 0/68/26/3/3 | 8 (7) | 3 (1.5) |
| HC | 134 | 34 (9) | 71% | 14 (2.5) | 73% | - | - | - | |
| Estiasari et al. [53] | |||||||||
| MS | Indonesia | 40 | {31}, [20–61] | 82.5% | >12 yrs = 75% | Nr | 0/78/22/0/0 | {4}, [0.1–15] | {3}, [1–7.5] |
| HC | 66 | {29}, [22–51] | 72.7% | >12 yrs = 89.4% | Nr | - | - | - | |
| Evdoshenko et al. [54] | |||||||||
| MS | Russia | 98 | 38.44 (11.47) | 70.4% | 15.12 (2.79) | Nr | 0/86/14/0/0 | 9.5 (7.44) | {3.0} |
| HC | 86 | 38.17 (13.29) | 63.95% | 16.26 (3.02) | Nr | - | - | - | |
| Farghaly et al. [55] | |||||||||
| MS | Egypt | 90 | 30.8 (6.7) | 77.78% | 14.5 (2.6) | Nr | 0/86/12/2/0 | 6.2 (5.8) | 2.8 (1.8) |
| HC | 85 | 30.5 (7.9) | 70.59% | 14.3 (3.3) | Nr | - | - | - | |
| Filser et al. [56] | |||||||||
| MS | Germany | 172 | 43.33 (11.64) | 68% | 10.74 (1.56) | 76.4% | 0/87/9/4/0 | Nr | Nr |
| HC | 100 | 43.04 (15.59) | 71% | 10.77 (1.58) | 92% | - | - | - | |
| Giedraitienė et al. [57] | |||||||||
| MS | Lithuania | 50 | 38.8 (10.2) | 47% | 15.9 (2.8) | 54% | 4/88/6/2/0 | 11.7 (9.2) | 3.3 (1.3) |
| HC | 20 | 36.7 (16.4) | 33% | 17.5 (3.5) | 75% | - | - | - | |
| Hämäläinen et al. [58] | |||||||||
| MS | Finland | 65 | 50.9 (8.8) | 71% | 13.8 (9.8) | 20% | 0/62/38/0/0 | 15.9 (9.8) | 4.8 (2.0) |
| HC | 45 | 49.4 (12.6) | 71% | 14.0 (2.1) | 86.7% | - | - | - | |
| Marstrand et al. [59] | |||||||||
| MS | Denmark | 65 | 37.2 (8.8) | 63% | 15.2 (2.4) | Nr | 0/100/0/0/0 | 3.9 (2.7) | 1.8 (1.2) |
| HC | 65 | 36.8 (9.6) | 63% | 15.9 (2.1) | Nr | - | - | - | |
| Maubeuge et al. [60] | |||||||||
| MS | France | 123 | 49.69 (9.41) | 63.4% | 14–16 yrs = 30.1% | 44.7% | 0/33/33/34/0 | 14.67 (9.09) | {4.0}, [0–8] |
| HC | 276 | 43.84 (12.42) | 57.3% | 14–16 yrs = 38% | Nr | - | - | - | |
| Niino et al. [61] | |||||||||
| MS | Japan | 156 | 41.4 (9.3) | 69% | 14.1 (1.9) | Nr | 0/88/11/1/0 | 10.3 (7.2) | 2.4 (2.0) |
| HC | 126 | 39.3 (11.9) | 72% | 14.3 (1.6) | Nr | - | - | - | |
| O’Connell et al. [62] | |||||||||
| MS | Ireland | 67 | 42.7 (12.8) | 68% | 14.1 (3.1) | 41.8% | 0/70/28/2/0 | 10.2 (8.4) | 1.8 (0.9) |
| HC | 66 | 43.9 (12.1) | 73% | 13.6 (2.7) | 80.3% | - | - | - | |
| Ozakbas et al. [63] | |||||||||
| MS | Turkey | 173 | 37.5 (10.7) | 71% | 13.9 (7.3) | 23.7% | 0/87/10/3/0 | 9.2 (6.1) | 2.4 (1.7) |
| HC | 153 | 36.9 (8.9) | 71% | 15.4 (8.8) | 39.1% | - | - | - | |
| Polychroniadou et al. [64] | |||||||||
| MS | Greece | 44 | 40.2 (9.9) | 61% | 13.9 (4.2) | Nr | 7/77/9/7/0 | 9.1 (4.1) | {3.5}, [1.0–6.0] |
| HC | 79 | 36.2 (10.6) | 60% | 15.6 (5.5) | Nr | - | - | - | |
| Sandi et al. [65] | |||||||||
| MS | Hungary | 65 | 41.9 (8.9) | 75% | >12 yrs = 52.3% | Nr | 0/100/0/0/0 | 11.1 (7.6) | 2.5 (1.8) |
| HC | 65 | 40.9 (11.8) | 75% | >12 yrs = 52.3% | Nr | - | - | - | |
| Skorve et al. [66] | |||||||||
| MS | Norway | 65 | 37.02 (0.40) | 64.6% | 14–16 yrs = 37% | 89.2% | 0/100/0/0/0 | 1.08 (0.74) | 1.28 (0.88) |
| HC | 68 | 38.13 (11.40) | 66.2% | 14–16 yrs = 46% | 97.0% | - | - | - | |
| Souissi et al. [67] | |||||||||
| MS | Tunisia | 104 | 33.3 (9.8) | 75% | 14–16 yrs = 14.42% | Nr | 0/88/8/4/0 | 7 (6.4) | 2.65 (2.06) |
| HC | 104 | 33.3 (9.4) | 75% | 14–16 yrs = 14.42% | Nr | - | - | - | |
| Sousa et al. [68] | |||||||||
| MS | Portugal | 105 | 38.26 (11.03) | 66.7% | 13.55 (3.71) | 58.1% | 4/92/4/0/0 | 6.52 (5.95) | {1.5}, [0–6] |
| HC | 60 | 36.17 (12.01) | 58.3% | 14.62 (3.47) | 94.9% | - | - | - | |
| Spedo et al. [69] | |||||||||
| MS | Brazil | 58 | 41.2 (12.2) | 69% | 12.7 (5.2) | Nr | 0/100/0/0/0 | 8.3 (6.6) | 4.2 (2) |
| HC | 58 | 40.3 (11.9) | 55% | 12.5 (3.6) | Nr | - | - | - | |
| Vanotti et al. [70] | |||||||||
| MS | Argentina | 50 | 43.4 (10.2) | 74% | 14.9 (2.8) | Nr | 0/78/18/4/0 | 13.1 (9.1) | 3.29 (2.55) |
| HC | 100 | 42.4 (10.1) | 75% | 14.9 (2.5) | Nr | - | - | - | |
| Walker et al. [71] | |||||||||
| MS | Canada | 57 | 45.4 (9.9) | 80% | 15.44 (2.7) | Nr | 0/77/16/7/0 | 10.11 (7.72) | 2.7 (1.85) |
| HC | 51 | 41.9 (10.8) | 86% | 16.31 (2.1) | Nr | - | - | - |
| Study | SDMT Score Mean (SD) | CVLT-II Score Mean (SD) | BVMT-R SCORE Mean (SD) | Impaired Cognition on at Least One Subtest (%) | Sensitivity (%) | Specificity (%) |
|---|---|---|---|---|---|---|
| Alarcón et al. [46] | ||||||
| MS | 46.47 (14.24) | 45.34 (10.14) a | 21.64 (6.91) | 50% | Nr | Nr |
| HC | 54.11 (12.19) | 48.78 (8.45) a | 25.67 (6.81) | - | - | - |
| Betscher et al. [47] | ||||||
| MS | 48.8 (12.1) | 51.7 (10.9) | 24 (7.7) | 34% | Nr | Nr |
| HC | 57.2 (9.7) | 56.1 (9.2) | 27.1 (5.7) | Nr | - | - |
| Botchorishvili et al. [48] | ||||||
| MS | 35.5 (12.7) | 51.0 (11.8) | 22.0 (8.0) | 43% | Nr | Nr |
| HC | 46.0 (11.8) | 58.5 (8.2) | 25.6 (6.8) | 14% | - | - |
| Costers et al. [49] | ||||||
| MS | 52.1 (13.1) | 60.1 (12.9) | 25.4 (29) | Nr | Nr | Nr |
| HC | 61 (10.2) | 61.3 (9.7) | 28.2 (5.1) | Nr | - | - |
| Darwish et al. [50] | ||||||
| MS | 47.2 (17.98) | 56.9 (10.04) b | 22 (9.79) | 61% | Nr | Nr |
| HC | 59.22 (12.27) | 54.10 (8.71) b | 24.23 (6.66) | Nr | - | - |
| Drulović et al. [51] | ||||||
| MS | 45.9 (16.7) | 50.0 (11.7) | 18.8 (7.4) | 62.9% | Nr | Nr |
| HC | 56.3 (12.9) | 52.7 (9.6) | 22.6 (5.8) | 18.6% | - | - |
| Dusankova et al. [52] | ||||||
| MS | 50 (13) | 52 (11) | 23 (7) | 58% | 94% | 86% |
| HC | 65 (9) | 60 (8) | 29 (4) | 0.7% | - | - |
| Estiasari et al. [53] | ||||||
| MS | 40.9 (14.8) | 52.0 (12.8) | 22.2 (7.7) | 40% | Nr | Nr |
| HC | 64.8 (16.2) | 61.5 (9.7) | 29.3 (5.6) | Nr | - | - |
| Evdoshenko et al. [54] | ||||||
| MS | 49.16 (13.42) | {61.5} | {26.5} | 34.69% | Nr | Nr |
| HC | 58.34 (11.52) | {65.5} | {28} | 16.28% | - | - |
| Farghaly et al. [55] | ||||||
| MS | 39.2 (13.3) | 53.7 (10.5) | 19.7 (9.2) | SDMT = 31.1% CVLT-II = 19.5% BVMT-R = 23.9% | Nr | Nr |
| HC | 50.9 (10.8) | 59.6 (8.5) | 25.4 (8.7) | SDMT = 5.8% CVLT-II = 7% BVMT-R = 8.1% | - | - |
| Filser et al. [56] | ||||||
| MS | 47.43 (11.67) | 55.35 (11.43) c | 24.44 (7.59) | 32.6% | Nr | Nr |
| HC | 56.07 (11.64) | 55.16 (10.27) c | 27.37 (5.96) | Nr | - | - |
| Giedraitienė et al. [57] | ||||||
| MS | 42.7 (13.9) | 55.9 (10) | 23.1 (7) | Nr | Nr | Nr |
| HC | 57 (11.5) | 65.7 (5.9) | 29.6 (4.1) | Nr | - | - |
| Hämäläinen et al. [58] | ||||||
| MS | 41.9 (11.8) | 43.0 (11.5) | 19.2 (8.0) | 60% | Nr | Nr |
| HC | 54.6 (8.3) | 51.3 (10.7) | 24.7 (6.8) | Nr | - | - |
| Marstrand et al. [59] | ||||||
| MS | 61.0 (10.0) | 65.4 (9.9) | 27.4 (5.8) | 32.3% | SDMT = 20.0% CVLT-II = 10.8% BVMT-R = 16.9% | SDMT = 95.4% CVLT-II = 89.2% BVMT-R = 93.8% |
| HC | 66.0 (9.6) | 68.6 (6.4) | 29.6 (3.7) | 20% | - | - |
| Maubeuge et al. [60] | ||||||
| MS | 50.31 (11.12) | 49.72 (12.77) d | 22.89 (7.26) | 50.4% | Nr | Nr |
| HC | 58.55 (8.44) | 57.78 (8.67) d | 26.73 (5.67) | 19.6% | - | - |
| Niino et al. [61] | ||||||
| MS | 47.9 (14) | 48.6 (12.6) | 23.5 (8.4) | Nr | Nr | Nr |
| HC | 61 (9.5) | 55.7 (10.5) | 28.3 (5.4) | Nr | - | - |
| O’Connell et al. [62] | ||||||
| MS | 46.0 (12.9) | 45.3 (10.2) | 17.9 (7.1) | 57% | Nr | Nr |
| HC | 56.1 (10.6) | 53.6 (9.1) | 20.9 (6.5) | 17% | - | - |
| Ozakbas et al. [63] | ||||||
| MS | 43.2 (12.5) | 45.7 (11.3) | 16.9 (8.5) | 45.1% | Nr | Nr |
| HC | 53.5 (9.5) | 53.9 (7.7) | 22.5 (9.2) | Nr | - | - |
| Polychroniadou et al. [64] | ||||||
| MS | 45.0 (17.2) | 55.5 (12.3) e | 18.5 (8.3) | 47% | Nr | Nr |
| HC | 61.4 (13.1) | 60.5 (10.7) e | 22.1 (6.5) | Nr | - | - |
| Sandi et al. [65] | ||||||
| MS | 55.6 (15.5) | 55.4 (10.7) | 22.5 (8.5) | 52.3% | Nr | Nr |
| HC | 66.2 (12.4) | 59.0 (8.3) | 26.7 (5.6) | Nr | - | - |
| Skorve et al. [66] | ||||||
| MS | 54.65 (10.79) | 54.55 (10.86) | 26.55 (5.76) | 46.2% | Nr | Nr |
| HC | 58.52 (10.53) | 60.32 (7.75) | 29.03 (4.01) | Nr | - | - |
| Souissi et al. [67] | ||||||
| MS | 36 (13) | 42 (7) f | 23 (9) | 73.1% | SDMT = 74% TVLT = 76% f BVMT-R = 75% | SDMT = 56% TVLT = 55% f BVMT-R = 53.5% |
| HC | 47 (15) | 46 (6) f | 27 (7) | Nr | - | - |
| Sousa et al. [68] | ||||||
| MS | 51.77 (11.20) | 55.05 (11.84) | 21.72 (7.27) | 24.8% | Nr | Nr |
| HC | 58.68 (10.02) | 60.47 (10.12) | 24.68 (5.52) | Nr | - | - |
| Spedo et al. [69] | ||||||
| MS | 35.9 (16.1) | 42.1 (12.4) | 19.9 (8.6) | Nr | Nr | Nr |
| HC | 47.5 (13) | 53.4 (10.8) | 23.8 (7.7) | Nr | - | - |
| Vanotti et al. [70] | ||||||
| MS | 45.1 (16.1) | 50.9 (12.4) | 20.7 (7.74) | Nr | Nr | Nr |
| HC | 56.7 (10.9) | 60.9 (10.5) | 23.4 (5.8) | Nr | - | - |
| Walker et al. [71] | ||||||
| MS | 49.7 (10.8) | 51.6 (10.1) | 24.6 (6.5) | 57.9% | SDMT = 97.5% CVLT-II = 82.5% BVMT-R = 77.5% | SDMT = 88.2% CVLT-II = 70.6% BVMT-R = 82.4% |
| HC | 59.1 (8.5) | 57.7 (7.9) | 29.8 (3.6) | Nr | - | - |
| Study | BICAMS Scores and Sample Variables | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age (r) | Disease Duration (r) | EDSS (r) | Education Years (r) | |||||||||
| SDMT | CVLT-II | BVMT-R | SDMT | CVLT-II | BVMT-R | SDMT | CVLT-II | BVMT-R | SDMT | CVLT-II | BVMT-R | |
| Alarcón et al. [46] | ||||||||||||
| MS | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr |
| HC | Nr | - | Nr | - | - | - | - | - | - | Nr | - | Nr |
| Betscher et al. [47] | ||||||||||||
| MS | −0.28 * | Nr | −0.26 * | Nr | Nr | Nr | −0.58 *** | −0.31 * | −0.27 * | 0.36 * | 0.42 *** | 0.5 *** |
| HC | −0.35 * | Nr | Nr | - | - | - | - | - | - | 0.44 *** | 0.47 *** | 0.27 * |
| Botchorishvili et al. [48] | ||||||||||||
| MS | −0.400* | −0.112 | −0.192 | −0.177 | −0.106 | 0.125 | −0.582 *** | −0.403 *** | −0.342 *** | 0.243 * | 0.207 | 0.297 * |
| HC | −0.457 *** | −0.368 *** | −0.506 *** | - | - | - | - | - | - | 0.523 *** | 0.439 * | 0.348 * |
| Costers et al. [49] | ||||||||||||
| MS | −0.34 *** | −0.10 | −0.29 ** | Nr | Nr | Nr | −0.44 *** | −0.35 *** | −0.43 *** | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Darwish et al. [50] | ||||||||||||
| MS | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr |
| HC | Nr | - | Nr | - | - | - | - | - | - | Nr | - | Nr |
| Drulović et al. [51] | ||||||||||||
| MS | −0.225 * | −0.232 * | −0.271 * | −0.109 * | −0.880 | −0.207 * | −0.466 * | −0.320 * | −0.360 * | 0.339 * | 0.298 * | 0.190 * |
| HC | −0.605 * | −0.430 * | −0.374 * | - | - | - | - | - | - | 0.521 * | 0.552 * | 0.394 * |
| Dusankova et al. [52] | ||||||||||||
| MS | Nr | Nr | Nr | 0.44 * | 0.39 * | 0.41 * | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Estiasari et al. [53] | ||||||||||||
| MS | −0.004 | −0.11 | 0.02 | −0.23 | −0.19 | −0.18 | −0.5 *** | −0.46 * | −0.49 * | {47}, [15–69] | {54}, [23–72] | {24.5}, [4–32] |
| HC | −0.27 * | −0.11 | −0.28 * | - | - | - | - | - | - | {63}, [42–110] * | {63}, [36–77] | {31}, [14–36] |
| Evdoshenko et al. [54] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Farghaly et al. [55] | ||||||||||||
| MS | −0.26 a,* | −0.17 a | −0.26 a,* | −0.41 a,*** | −0.18 a | −0.27 a,* | −0.37 a,*** | −0.31 a,* | −0.19 a | 0.36 a,*** | 0.27 a,* | 0.25 a,* |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Filser et al. [56] | ||||||||||||
| MS | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr |
| HC | Nr | - | Nr | - | - | - | - | - | - | Nr | - | Nr |
| Giedraitienė et al. [57] | ||||||||||||
| MS | Nr | Nr | Nr | −0.3 a | −0.2 a | −0.2 a | −5.9 a,*** | −3.7 a,*** | −2.3 a,*** | 2.4 a,* | 2.4 a,* | 1.0 a,* |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | 2.0 a,* | 1.2 a,* | 0.9 a,* |
| Hämäläinen et al. [58] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Marstrand et al. [59] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Maubeuge et al. [60] | ||||||||||||
| MS | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr |
| HC | Nr | - | Nr | - | - | - | - | - | - | Nr | - | Nr |
| Niino et al. [61] | ||||||||||||
| MS | –0.37 *** | –0.25 * | –0.30 *** | –0.30 *** | –0.12 | –0.27 *** | –0.56 *** | –0.29 *** | −0.46 *** | 0.07 | 0.13 | 0.001 |
| HC | –0.44 *** | –0.23 * | –0.25 * | - | - | - | - | - | - | 0.24 * | 0.25 * | 0.05 |
| O’Connell et al. [62] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Ozakbas et al. [63] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | −0.46 * | −0.40 * | −0.24 | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Polychroniadou et al. [64] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Sandi et al. [65] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | Nr | Nr | Nr | Nr |
| Skorve et al. [66] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Souissi et al. [67] | ||||||||||||
| MS | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr | Nr | - | Nr |
| HC | Nr | - | Nr | - | - | - | - | - | - | Nr | - | Nr |
| Sousa et al. [68] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | −0.497 *** | −0.334 *** | −0.275 * | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Spedo et al. [69] | ||||||||||||
| MS | −0.30 * | −0.30 * | −0.29 * | Nr | Nr | Nr | Nr | Nr | Nr | 0.29 * | 0.18 * | 0.27 * |
| HC | −0.49 * | - | −0.34 * | - | - | - | - | - | - | 0.49 * | 0.37 * | - |
| Vanotti et al. [70] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
| Walker et al. [71] | ||||||||||||
| MS | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | Nr | - | 0.20 * | - |
| HC | Nr | Nr | Nr | - | - | - | - | - | - | Nr | Nr | Nr |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Potticary, H.; Langdon, D. A Systematic Review and Meta-Analysis of the Brief Cognitive Assessment for Multiple Sclerosis (BICAMS) International Validations. J. Clin. Med. 2023, 12, 703. https://doi.org/10.3390/jcm12020703
Potticary H, Langdon D. A Systematic Review and Meta-Analysis of the Brief Cognitive Assessment for Multiple Sclerosis (BICAMS) International Validations. Journal of Clinical Medicine. 2023; 12(2):703. https://doi.org/10.3390/jcm12020703
Chicago/Turabian StylePotticary, Hannah, and Dawn Langdon. 2023. "A Systematic Review and Meta-Analysis of the Brief Cognitive Assessment for Multiple Sclerosis (BICAMS) International Validations" Journal of Clinical Medicine 12, no. 2: 703. https://doi.org/10.3390/jcm12020703
APA StylePotticary, H., & Langdon, D. (2023). A Systematic Review and Meta-Analysis of the Brief Cognitive Assessment for Multiple Sclerosis (BICAMS) International Validations. Journal of Clinical Medicine, 12(2), 703. https://doi.org/10.3390/jcm12020703





