Transjugular Intrahepatic Portosystemic Shunt: Devices Evolution, Technical Tips and Future Perspectives
Abstract
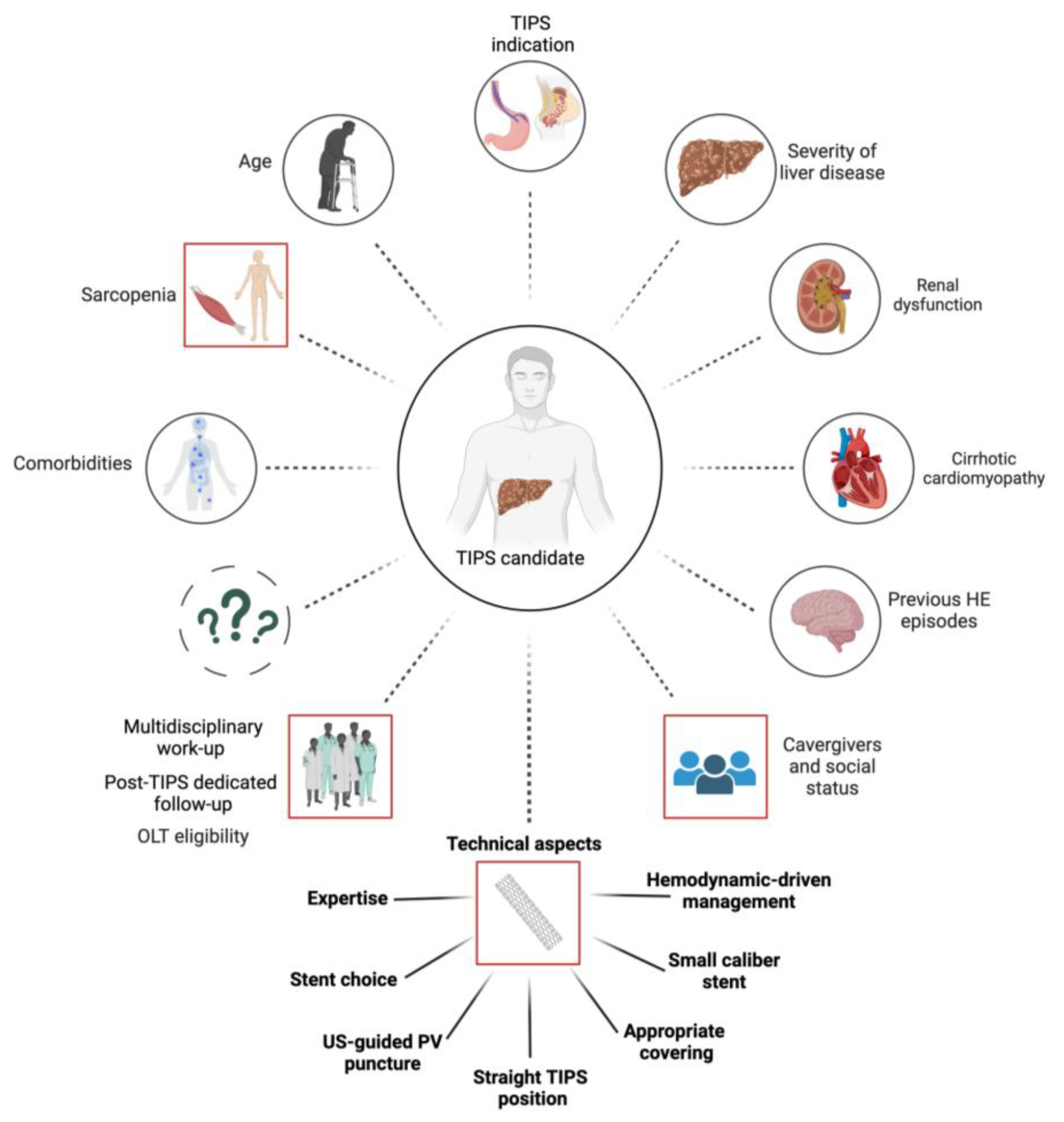
:1. Introduction
2. Device Evolution for TIPS Creation
3. Bare vs. Covered Stents: Comparative Analysis of Outcomes
4. Covered Stents: Technical Tips
5. The Technical Evolution of TIPS Does Not Stand Alone in Improving Outcomes
5.1. Hemodynamic Targets
5.2. Shunt-Related Complications
5.3. Choice of Stent Nominal Diameter
5.4. The Underdilation Strategy
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Garcia-Tsao, G.; Friedman, S.; Iredale, J.; Pinzani, M. Now There Are Many (Stages) Where before There Was One: In Search of a Pathophysiological Classification of Cirrhosis. Hepatology 2010, 51, 1445–1449. [Google Scholar] [CrossRef]
- D’Amico, G.; Garcia-Tsao, G.; Pagliaro, L. Natural History and Prognostic Indicators of Survival in Cirrhosis: A Systematic Review of 118 Studies. J. Hepatol. 2006, 44, 217–231. [Google Scholar] [CrossRef]
- Abraldes, J.G.; Tarantino, I.; Turnes, J.; Garcia-Pagan, J.C.; Rodés, J.; Bosch, J. Hemodynamic Response to Pharmacological Treatment of Portal Hypertension and Long-Term Prognosis of Cirrhosis. Hepatology 2003, 37, 902–908. [Google Scholar] [CrossRef]
- Ripoll, C.; Groszmann, R.; Garcia-Tsao, G.; Grace, N.; Burroughs, A.; Planas, R.; Escorsell, A.; Garcia-Pagan, J.C.; Makuch, R.; Patch, D.; et al. Hepatic Venous Pressure Gradient Predicts Clinical Decompensation in Patients with Compensated Cirrhosis. Gastroenterology 2007, 133, 481–488. [Google Scholar] [CrossRef]
- Vizzutti, F.; Schepis, F.; Arena, U.; Fanelli, F.; Gitto, S.; Aspite, S.; Turco, L.; Dragoni, G.; Laffi, G.; Marra, F. Transjugular Intrahepatic Portosystemic Shunt (TIPS): Current Indications and Strategies to Improve the Outcomes. Intern. Emerg. Med. 2020, 15, 37–48. [Google Scholar] [CrossRef]
- Lv, Y.; Yang, Z.; Liu, L.; Li, K.; He, C.; Wang, Z.; Bai, W.; Guo, W.; Yu, T.; Yuan, X.; et al. Early TIPS with Covered Stents versus Standard Treatment for Acute Variceal Bleeding in Patients with Advanced Cirrhosis: A Randomised Controlled Trial. Lancet Gastroenterol. Hepatol. 2019, 4, 587–598. [Google Scholar] [CrossRef]
- Hernández-Gea, V.; Procopet, B.; Giráldez, Á.; Amitrano, L.; Villanueva, C.; Thabut, D.; Ibañez-Samaniego, L.; Silva-Junior, G.; Martinez, J.; Genescà, J.; et al. Preemptive-TIPS Improves Outcome in High-Risk Variceal Bleeding: An Observational Study. Hepatology 2019, 69, 282–293. [Google Scholar] [CrossRef]
- Nicoară-Farcău, O.; Han, G.; Rudler, M.; Angrisani, D.; Monescillo, A.; Torres, F.; Casanovas, G.; Bosch, J.; Lv, Y.; Thabut, D.; et al. Effects of Early Placement of Transjugular Portosystemic Shunts in Patients with High-Risk Acute Variceal Bleeding: A Meta-Analysis of Individual Patient Data. Gastroenterology 2021, 160, 193–205.e10. [Google Scholar] [CrossRef]
- Sanyal, A.J.; Genning, C.; Reddy, K.R.; Wong, F.; Kowdley, K.V.; Benner, K.; McCashland, T.; North American Study for the Treatment of Refractory Ascites Group. The North American Study for the Treatment of Refractory Ascites. Gastroenterology 2003, 124, 634–641. [Google Scholar] [CrossRef]
- Salerno, F.; Cammà, C.; Enea, M.; Rössle, M.; Wong, F. Transjugular Intrahepatic Portosystemic Shunt for Refractory Ascites: A Meta-Analysis of Individual Patient Data. Gastroenterology 2007, 133, 825–834. [Google Scholar] [CrossRef]
- Tan, H.K.; James, P.D.; Sniderman, K.W.; Wong, F. Long-Term Clinical Outcome of Patients with Cirrhosis and Refractory Ascites Treated with Transjugular Intrahepatic Portosystemic Shunt Insertion. J. Gastroenterol. Hepatol. 2015, 30, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Bucsics, T.; Hoffman, S.; Grünberger, J.; Schoder, M.; Matzek, W.; Stadlmann, A.; Mandorfer, M.; Schwabl, P.; Ferlitsch, A.; Peck-Radosavljevic, M.; et al. ePTFE-TIPS vs. Repetitive LVP plus Albumin for the Treatment of Refractory Ascites in Patients with Cirrhosis. Liver Int. 2018, 38, 1036–1044. [Google Scholar] [CrossRef] [PubMed]
- Luca, A.; Miraglia, R.; Caruso, S.; Milazzo, M.; Sapere, C.; Maruzzelli, L.; Vizzini, G.; Tuzzolino, F.; Gridelli, B.; Bosch, J. Short- and Long-Term Effects of the Transjugular Intrahepatic Portosystemic Shunt on Portal Vein Thrombosis in Patients with Cirrhosis. Gut 2011, 60, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Pagán, J.C.; Heydtmann, M.; Raffa, S.; Plessier, A.; Murad, S.; Fabris, F.; Vizzini, G.; Gonzales Abraldes, J.; Olliff, S.; Nicolini, A.; et al. TIPS for Budd-Chiari Syndrome: Long-Term Results and Prognostics Factors in 124 Patients. Gastroenterology 2008, 135, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Fanelli, F.; Angeloni, S.; Salvatori, F.M.; Marzano, C.; Boatta, E.; Merli, M.; Rossi, P.; Attili, A.F.; Ridola, L.; Cerini, F.; et al. Transjugular Intrahepatic Portosystemic Shunt with Expanded-Polytetrafuoroethylene-Covered Stents in Non-Cirrhotic Patients with Portal Cavernoma. Dig. Liver Dis. 2011, 43, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Boike, J.R.; Thornburg, B.G.; Asrani, S.K.; Fallon, M.B.; Fortune, B.E.; Izzy, M.J.; Verna, E.C.; Abraldes, J.G.; Allegretti, A.S.; Bajaj, J.S.; et al. North American Practice-Based Recommendations for Transjugular Intrahepatic Portosystemic Shunts in Portal Hypertension. Clin. Gastroenterol. Hepatol. 2022, 20, 1636–1662.e36. [Google Scholar] [CrossRef] [PubMed]
- Rösch, J.; Hanafee, W.N.; Snow, H. Transjugular Portal Venography and Radiologic Portacaval Shunt: An Experimental Study. Radiology 1969, 92, 1112–1114. [Google Scholar] [CrossRef]
- Colapinto, R.F.; Stronell, R.D.; Birch, S.J.; Langer, B.; Blendis, L.M.; Greig, P.D.; Gilas, T. Creation of an Intrahepatic Portosystemic Shunt with a Grüntzig Balloon Catheter. Can. Med. Assoc. J. 1982, 126, 267–268. [Google Scholar]
- Hausegger, K.A.; Karnel, F.; Georgieva, B.; Tauss, J.; Portugaller, H.; Deutschmann, H.; Berghold, A. Transjugular Intrahepatic Portosystemic Shunt Creation with the Viatorr Expanded Polytetrafluoroethylene-Covered Stent-Graft. J. Vasc. Interv. Radiol. 2004, 15, 239–248. [Google Scholar] [CrossRef]
- Boyer, T.D.; Haskal, Z.J.; American Association for the Study of Liver Diseases. The Role of Transjugular Intrahepatic Portosystemic Shunt in the Management of Portal Hypertension. Hepatology 2005, 41, 386–400. [Google Scholar] [CrossRef]
- García-Pagán, J.C.; Caca, K.; Bureau, C.; Laleman, W.; Appenrodt, B.; Luca, A.; Abraldes, J.G.; Nevens, F.; Vinel, J.P.; Mössner, J.; et al. Early Use of TIPS in Patients with Cirrhosis and Variceal Bleeding. N. Engl. J. Med. 2010, 362, 2370–2379. [Google Scholar] [CrossRef]
- Bureau, C.; Thabut, D.; Oberti, F.; Dharancy, S.; Carbonell, N.; Bouvier, A.; Mathurin, P.; Otal, P.; Cabarrou, P.; Péron, J.M.; et al. Transjugular Intrahepatic Portosystemic Shunts with Covered Stents Increase Transplant-Free Survival of Patients with Cirrhosis and Recurrent Ascites. Gastroenterology 2017, 152, 157–163. [Google Scholar] [CrossRef]
- Srinivasa, R.N.; Srinivasa, R.N.; Chick, J.F.B.; Hage, A.; Saad, W.A. Transjugular Intrahepatic Portosystemic Shunt Reduction Using the GORE VIATORR Controlled Expansion Endoprosthesis: Hemodynamics of Reducing an Established 10-Mm TIPS to 8-Mm in Diameter. Cardiovasc. Interv. Radiol. 2018, 41, 518–521. [Google Scholar] [CrossRef]
- Schepis, F.; Vizzutti, F.; Garcia-Tsao, G.; Marzocchi, G.; Rega, L.; De Maria, N.; Di Maira, T.; Gitto, S.; Caporali, C.; Colopi, S.; et al. Under-Dilated TIPS Associate with Efficacy and Reduced Encephalopathy in a Prospective, Non-Randomized Study of Patients with Cirrhosis. Clin. Gastroenterol. Hepatol. 2018, 16, 1153–1162.e7. [Google Scholar] [CrossRef]
- de Franchis, R.; Bosch, J.; Garcia-Tsao, G.; Reiberger, T.; Ripoll, C.; Baveno VII Faculty. Baveno VII—Renewing Consensus in Portal Hypertension. J. Hepatol. 2022, 76, 959–974. [Google Scholar] [CrossRef] [PubMed]
- Larrue, H.; D’Amico, G.; Olivas, P.; Lv, Y.; Bucsics, T.; Rudler, M.; Sauerbruch, T.; Hernandez-Gea, V.; Han, G.; Reiberger, T.; et al. TIPS Prevents Further Decompensation and Improves Survival in Patients with Cirrhosis and Portal Hypertension in an Individual Patient Data Meta-Analysis. J. Hepatol. 2023, 79, 692–703. [Google Scholar] [CrossRef] [PubMed]
- Burgener, F.A.; Göthlin, J.H.; Gutierrez, O.H. Percutaneous Transhepatic Portography in the Dog. Investig. Radiol. 1978, 13, 26–27. [Google Scholar] [CrossRef]
- Rössle, M. TIPS: 25 Years Later. J. Hepatol. 2013, 59, 1081–1093. [Google Scholar] [CrossRef] [PubMed]
- Praveen Kumar, G.; Jafary-Zadeh, M.; Tavakoli, R.; Cui, F. Feasibility of Using Bulk Metallic Glass for Self-Expandable Stent Applications. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 1874–1882. [Google Scholar] [CrossRef]
- Jiang, W.; Zhao, W.; Zhou, T.; Wang, L.; Qiu, T. A Review on Manufacturing and Post-Processing Technology of Vascular Stents. Micromachines 2022, 13, 140. [Google Scholar] [CrossRef]
- Perarnau, J.M.; Le Gouge, A.; Nicolas, C.; d’Alteroche, L.; Borentain, P.; Saliba, F.; Minello, A.; Anty, R.; Chagneau-Derrode, C.; Bernard, P.H.; et al. Covered vs. Uncovered Stents for Transjugular Intrahepatic Portosystemic Shunt: A Randomized Controlled Trial. J. Hepatol. 2014, 60, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Bureau, C.; Pagan, J.C.G.; Layrargues, G.P.; Metivier, S.; Bellot, P.; Perreault, P.; Otal, P.; Abraldes, J.-G.; Peron, J.M.; Rousseau, H.; et al. Patency of Stents Covered with Polytetrafluoroethylene in Patients Treated by Transjugular Intrahepatic Portosystemic Shunts: Long-Term Results of a Randomized Multicentre Study. Liver Int. 2007, 27, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Narahara, Y.; Kanazawa, H.; Fukuda, T.; Matsushita, Y.; Harimoto, H.; Kidokoro, H.; Katakura, T.; Atsukawa, M.; Taki, Y.; Kimura, Y.; et al. Transjugular Intrahepatic Portosystemic Shunt versus Paracentesis plus Albumin in Patients with Refractory Ascites Who Have Good Hepatic and Renal Function: A Prospective Randomized Trial. J. Gastroenterol. 2011, 46, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Saxon, R.R.; Mendel-Hartvig, J.; Corless, C.L. Bile duct injury as a major cause of stenosis and occlusion in transjugular intrahepatic portosystemic shunts: Comparative histopathologic analysis in humans and swine. J. Vasc. Interv. Radiol. 1996, 7, 487–497. [Google Scholar] [CrossRef]
- Fanelli, F. The Evolution of Transjugular Intrahepatic Portosystemic Shunt: Tips. ISRN Hepatol. 2014, 2014, 762096. [Google Scholar] [CrossRef]
- LaBerge, J.M.; Ring, E.J.; Lake, J.R.; Ferrell, L.D.; Doherty, M.M.; Gordon, R.L.; Roberts, J.P.; Peltzer, M.Y.; Ascher, N.L. Transjugular Intrahepatic Portosystemic Shunts: Preliminary Results in 25 Patients. J. Vasc. Surg. 1992, 16, 258–267. [Google Scholar] [CrossRef]
- Sauer, P.; Theilmann, L.; Herrmann, S.; Bruckner, T.; Roeren, T.; Richter, G.; Stremmel, W.; Stiehl, A. Phenprocoumon for Prevention of Shunt Occlusion after Transjugular Intrahepatic Portosystemic Stent Shunt: A Randomized Trial. Hepatology 1996, 24, 1433–1436. [Google Scholar] [CrossRef]
- Tanihata, H.; Saxon, R.R.; Kubota, Y.; Pavcnik, D.; Uchida, B.T.; Rosch, J.; Keller, F.S.; Yamada, R.; Sato, M. Transjugular Intrahepatic Portosystemic Shunt with Silicone-Covered Wallstents: Results in a Swine Model. Radiology 1997, 205, 181–184. [Google Scholar] [CrossRef]
- Haskal, Z.J.; Brennecke, L.J. Porous and Nonporous Polycarbonate Urethane Stent-Grafts for TIPS Formation: Biologic Responses. J. Vasc. Interv. Radiol. 1999, 10, 1255–1263. [Google Scholar] [CrossRef]
- Otal, P.; Rousseau, H.; Vinel, J.P.; Ducoin, H.; Hassissene, S.; Joffre, F. High Occlusion Rate in Experimental Transjugular Intrahepatic Portosystemic Shunt Created with a Dacron-Covered Nitinol Stent. J. Vasc. Interv. Radiol. 1999, 10, 183–188. [Google Scholar] [CrossRef]
- Haskal, Z.J. Improved Patency of Transjugular Intrahepatic Portosystemic Shunts in Humans: Creation and Revision with PTFE Stent-Grafts. Radiology 1999, 213, 759–766. [Google Scholar] [CrossRef]
- Haskal, Z.J.; Davis, A.; McAllister, A.; Furth, E.E. PTFE-Encapsulated Endovascular Stent-Graft for Transjugular Intrahepatic Portosystemic Shunts: Experimental Evaluation. Radiology 1997, 205, 682–688. [Google Scholar] [CrossRef]
- Saxon, R.R.; Timmermans, H.A.; Uchida, B.T.; Petersen, B.D.; Benner, K.G.; Rabkin, J.; Keller, F.S. Stent-Grafts for Revision of TIPS Stenoses and Occlusions: A Clinical Pilot Study. J. Vasc. Interv. Radiol. 1997, 8, 539–548. [Google Scholar] [CrossRef]
- Nishimine, K.; Saxon, R.R.; Kichikawa, K.; Mendel-Hartvig, J.; Timmermans, H.A.; Shim, H.J.; Uchida, B.T.; Barton, R.E.; Keller, F.S.; Rösch, J. Improved Transjugular Intrahepatic Portosystemic Shunt Patency with PTFE-Covered Stent-Grafts: Experimental Results in Swine. Radiology 1995, 196, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.D.; Pimpalwar, S.; Jackson, R.W. A New Stent-Graft for Transjugular Intrahepatic Portosystemic Shunts. Br. J. Radiol. 2001, 74, 908–912. [Google Scholar] [CrossRef] [PubMed]
- Bureau, C.; Carlos Garcia-Pagan, J.; Otal, P.; Pomier-Layrargues, G.; Chabbert, V.; Cortez, C.; Perreault, P.; Marie Péron, J.; Abraldes, J.G.; Bouchard, L.; et al. Improved Clinical Outcome Using Polytetrafluoroethylene-Coated Stents for Tips: Results of a Randomized Study. Gastroenterology 2004, 126, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Miraglia, R.; Maruzzelli, L.; Di Piazza, A.; Mamone, G.; Caruso, S.; Gentile, G.; Tuzzolino, F.; Floridia, G.; Petridis, I.; Volpes, R.; et al. Transjugular Intrahepatic Portosystemic Shunt Using the New Gore Viatorr Controlled Expansion Endoprosthesis: Prospective, Single-Center, Preliminary Experience. Cardiovasc. Interv. Radiol. 2019, 42, 78–86. [Google Scholar] [CrossRef]
- Huang, Q.; Wu, X.; Fan, X.; Cao, J.; Han, J.; Xu, L.; Li, N. Comparison Study of Doppler Ultrasound Surveillance of Expanded Polytetrafluoroethylene-Covered Stent versus Bare Stent in Transjugular Intrahepatic Portosystemic Shunt. J. Clin. Ultrasound 2010, 38, 353–360. [Google Scholar] [CrossRef]
- Wang, L.; Xiao, Z.; Yue, Z.; Zhao, H.; Fan, Z.; Zhao, M.; He, F.; Dai, S.; Qiu, B.; Yao, J.; et al. Efficacy of Covered and Bare Stent in TIPS for Cirrhotic Portal Hypertension: A Single-Center Randomized Trial. Sci. Rep. 2016, 6, 21011. [Google Scholar] [CrossRef]
- Yang, Z.; Han, G.; Wu, Q.; Ye, X.; Jin, Z.; Yin, Z.; Qi, X.; Bai, M.; Wu, K.; Fan, D. Patency and Clinical Outcomes of Transjugular Intrahepatic Portosystemic Shunt with Polytetrafluoroethylene-Covered Stents versus Bare Stents: A Meta-Analysis: Meta-Analysis of TIPS with PTFE Stents. J. Gastroenterol. Hepatol. 2010, 25, 1718–1725. [Google Scholar] [CrossRef]
- Qi, X.; Tian, Y.; Zhang, W.; Yang, Z.; Guo, X. Covered versus Bare Stents for Transjugular Intrahepatic Portosystemic Shunt: An Updated Meta-Analysis of Randomized Controlled Trials. Ther. Adv. Gastroenterol. 2017, 10, 32–41. [Google Scholar] [CrossRef]
- Triantafyllou, T.; Aggarwal, P.; Gupta, E.; Svetanoff, W.J.; Bhirud, D.P.; Singhal, S. Polytetrafluoroethylene-Covered Stent Graft Versus Bare Stent in Transjugular Intrahepatic Portosystemic Shunt: Systematic Review and Meta-Analysis. J. Laparoendosc. Adv. Surg. Tech. 2018, 28, 867–879. [Google Scholar] [CrossRef]
- Zhu, P.; Dong, S.; Sun, P.; Belgaumkar, A.P.; Sun, Y.; Cheng, X.; Zheng, Q.; Li, T. Expanded Polytetrafluoroethylene (ePTFE)-Covered Stents versus Bare Stents for Transjugular Intrahepatic Portosystemic Shunt in People with Liver Cirrhosis. Cochrane Database Syst. Rev. 2023, 2023, CD012358. [Google Scholar] [CrossRef]
- Wang, Q.; Lv, Y.; Bai, M.; Wang, Z.; Liu, H.; He, C.; Niu, J.; Guo, W.; Luo, B.; Yin, Z.; et al. Eight Millimetre Covered TIPS Does Not Compromise Shunt Function but Reduces Hepatic Encephalopathy in Preventing Variceal Rebleeding. J. Hepatol. 2017, 67, 508–516. [Google Scholar] [CrossRef]
- Praktiknjo, M.; Abu-Omar, J.; Chang, J.; Thomas, D.; Jansen, C.; Kupczyk, P.; Schepis, F.; Garcia-Pagan, J.C.; Merli, M.; Meyer, C.; et al. Controlled Underdilation Using Novel VIATORR® Controlled Expansion Stents Improves Survival after Transjugular Intrahepatic Portosystemic Shunt Implantation. JHEP Rep. 2021, 3, 100264. [Google Scholar] [CrossRef]
- Bureau, C.; Thabut, D.; Jezequel, C.; Archambeaud, I.; D’Alteroche, L.; Dharancy, S.; Borentain, P.; Oberti, F.; Plessier, A.; De Ledinghen, V.; et al. The Use of Rifaximin in the Prevention of Overt Hepatic Encephalopathy After Transjugular Intrahepatic Portosystemic Shunt: A Randomized Controlled Trial. Ann. Intern. Med. 2021, 174, 633–640. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the Management of Patients with Decompensated Cirrhosis. J. Hepatol. 2018, 69, 406–460. [Google Scholar] [CrossRef]
- Fagiuoli, S.; Bruno, R.; Debernardi Venon, W.; Schepis, F.; Vizzutti, F.; Toniutto, P.; Senzolo, M.; Caraceni, P.; Salerno, F.; Angeli, P.; et al. Consensus Conference on TIPS Management: Techniques, Indications, Contraindications. Dig. Liver Dis. 2017, 49, 121–137. [Google Scholar] [CrossRef]
- Rossi, P.; Salvatori, F.M.; Fanelli, F.; Bezzi, M.; Rossi, M.; Marcelli, G.; Pepino, D.; Riggio, O.; Passariello, R. Polytetrafluoroethylene-Covered Nitinol Stent-Graft for Transjugular Intrahepatic Portosystemic Shunt Creation: 3-Year Experience. Radiology 2004, 231, 820–830. [Google Scholar] [CrossRef]
- Buechter, M.; Manka, P.; Gerken, G.; Canbay, A.; Blomeyer, S.; Wetter, A.; Altenbernd, J.; Kahraman, A.; Theysohn, J.M. Transjugular Intrahepatic Portosystemic Shunt in Patients with Portal Hypertension: Patency Depends on Coverage and Interventionalist’s Experience. Dig. Dis. 2018, 36, 218–227. [Google Scholar] [CrossRef]
- Sarwar, A.; Zhou, L.; Novack, V.; Tapper, E.B.; Curry, M.; Malik, R.; Ahmed, M. Hospital Volume and Mortality after Transjugular Intrahepatic Portosystemic Shunt Creation in the United States. Hepatology 2018, 67, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Lopera, J.E. A Comprehensive Review of Transjugular Intrahepatic Portosystemic Shunt-Related Complications. Semin. Interv. Radiol. 2023, 40, 55–72. [Google Scholar] [CrossRef] [PubMed]
- Steib, C.J.; Li, H.; Zhang, J.; Mayerle, J.; Ricke, J.; Gerbes, A.L.; Meyer, C.; Zipprich, A.; Trebicka, J. Transjugular Intrahepatic Portosystemic Shunt for Patients with Liver Cirrhosis: Survey Evaluating Indications, Standardization of Procedures and Anticoagulation in 43 German Hospitals. Eur. J. Gastroenterol. Hepatol. 2020, 32, 1179–1185. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Vascular Diseases of the Liver. J. Hepatol. 2016, 64, 179–202. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jiang, M.-S.; Zhang, H.-L.; Weng, N.-N.; Luo, X.-F.; Li, X.; Yang, L. Is Post-TIPS Anticoagulation Therapy Necessary in Patients with Cirrhosis and Portal Vein Thrombosis? A Randomized Controlled Trial. Radiology 2016, 279, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Bai, W.; Li, K.; Wang, Z.; Guo, W.; Luo, B.; Wang, J.; Wang, Q.; Wang, E.; Xia, D.; et al. Anticoagulation and Transjugular Intrahepatic Portosystemic Shunt for the Management of Portal Vein Thrombosis in Cirrhosis: A Prospective Observational Study. Am. J. Gastroenterol. 2021, 116, 1447–1464. [Google Scholar] [CrossRef]
- Zhang, J.-B.; Chen, J.; Zhou, J.; Wang, X.-M.; Chen, S.; Chu, J.-G.; Liu, P.; Ye, Z.-D. Systematic Review and Meta-Analysis of Trans-Jugular Intrahepatic Portosystemic Shunt for Cirrhotic Patients with Portal Vein Thrombosis. World J. Clin. Cases 2021, 9, 5179–5190. [Google Scholar] [CrossRef]
- Bosch, J. Small Diameter Shunts Should Lead to Safe Expansion of the Use of TIPS. J. Hepatol. 2021, 74, 230–234. [Google Scholar] [CrossRef]
- Deltenre, P.; Zanetto, A.; Saltini, D.; Moreno, C.; Schepis, F. The Role of Transjugular Intrahepatic Portosystemic Shunt in Patients with Cirrhosis and Ascites: Recent Evolution and Open Questions. Hepatology 2023, 77, 640–658. [Google Scholar] [CrossRef]
- Perarnau, J.M.; Noeldge, G.; Rössle, M. Intrahepatic portacaval anastomosis by the transjugular approach. Use of the Palmaz endoprosthesis. Presse Medicale 1991, 20, 1770–1772. [Google Scholar]
- Thornburg, B.; Desai, K.; Hickey, R.; Kulik, L.; Ganger, D.; Baker, T.; Abecassis, M.; Lewandowski, R.J.; Salem, R. Portal Vein Recanalization and Transjugular Intrahepatic Portosystemic Shunt Creation for Chronic Portal Vein Thrombosis: Technical Considerations. Tech. Vasc. Interv. Radiol. 2016, 19, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.P.; Sze, D.Y. Complications During Transjugular Intrahepatic Portosystemic Shunt Creation. Tech. Vasc. Interv. Radiol. 2016, 19, 61–73. [Google Scholar] [CrossRef]
- Ferral, H.; Bilbao, J.I. The Difficult Transjugular Intrahepatic Portosystemic Shunt: Alternative Techniques and “Tips” to Successful Shunt Creation. Semin. Interv. Radiol. 2005, 22, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.H.; Eche, T.; Dorczynski, C.; Otal, P.; Revel-Mouroz, P.; Zadro, C.; Partouche, E.; Fares, N.; Maulat, C.; Bureau, C.; et al. Predicting Death or Recurrence of Portal Hypertension Symptoms after TIPS Procedures. Eur. Radiol. 2022, 32, 3346–3357. [Google Scholar] [CrossRef] [PubMed]
- Gaba, R.C.; Parvinian, A.; Minocha, J.; Casadaban, L.C.; Knuttinen, M.G.; Ray, C.E.; Bui, J.T. Should Transjugular Intrahepatic Portosystemic Shunt Stent Grafts Be Underdilated? J. Vasc. Interv. Radiol. 2015, 26, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Mamone, G.; Milazzo, M.; Di Piazza, A.; Caruso, S.; Carollo, V.; Gentile, G.; Crinò, F.; Marrone, G.; Sparacia, G.; Maruzzelli, L.; et al. Transjugular Intrahepatic Portosystemic Shunt (TIPS) Complications: What Diagnostic Radiologists Should Know. Abdom. Radiol. 2022, 47, 4254–4270. [Google Scholar] [CrossRef] [PubMed]
- Vignali, C.; Bargellini, I.; Grosso, M.; Passalacqua, G.; Maglione, F.; Pedrazzini, F.; Filauri, P.; Niola, R.; Cioni, R.; Petruzzi, P. TIPS with Expanded Polytetrafluoroethylene-Covered Stent: Results of an Italian Multicenter Study. AJR Am. J. Roentgenol. 2005, 185, 472–480. [Google Scholar] [CrossRef]
- Bercu, Z.L.; Fischman, A.M.; Kim, E.; Nowakowski, F.S.; Patel, R.S.; Schiano, T.D.; Chang, C.Y.; Lookstein, R.A. TIPS for Refractory Ascites: A 6-Year Single-Center Experience with Expanded Polytetrafluoroethylene-Covered Stent-Grafts. AJR Am. J. Roentgenol. 2015, 204, 654–661. [Google Scholar] [CrossRef]
- Nardelli, S.; Gioia, S.; Pasquale, C.; Pentassuglio, I.; Farcomeni, A.; Merli, M.; Salvatori, F.M.; Nikolli, L.; Torrisi, S.; Greco, F.; et al. Cognitive Impairment Predicts the Occurrence of Hepatic Encephalopathy After Transjugular Intrahepatic Portosystemic Shunt. Am. J. Gastroenterol. 2016, 111, 523–528. [Google Scholar] [CrossRef]
- Billey, C.; Billet, S.; Robic, M.A.; Cognet, T.; Guillaume, M.; Vinel, J.P.; Péron, J.M.; Lairez, O.; Bureau, C. A Prospective Study Identifying Predictive Factors of Cardiac Decompensation After Transjugular Intrahepatic Portosystemic Shunt: The Toulouse Algorithm. Hepatology 2019, 70, 1928–1941. [Google Scholar] [CrossRef]
- Bettinger, D.; Sturm, L.; Pfaff, L.; Hahn, F.; Kloeckner, R.; Volkwein, L.; Praktiknjo, M.; Lv, Y.; Han, G.; Huber, J.P.; et al. Refining Prediction of Survival after TIPS with the Novel Freiburg Index of Post-TIPS Survival. J. Hepatol. 2021, 74, 1362–1372. [Google Scholar] [CrossRef]
- Vizzutti, F.; Celsa, C.; Calvaruso, V.; Enea, M.; Battaglia, S.; Turco, L.; Senzolo, M.; Nardelli, S.; Miraglia, R.; Roccarina, D.; et al. Mortality after Transjugular Intrahepatic Portosystemic Shunt in Older Adult Patients with Cirrhosis: A Validated Prediction Model. Hepatology 2023, 77, 476–488. [Google Scholar] [CrossRef]
- Engelmann, C.; Clària, J.; Szabo, G.; Bosch, J.; Bernardi, M. Pathophysiology of Decompensated Cirrhosis: Portal Hypertension, Circulatory Dysfunction, Inflammation, Metabolism and Mitochondrial Dysfunction. J. Hepatol. 2021, 75 (Suppl. S1), S49–S66. [Google Scholar] [CrossRef] [PubMed]
- Praktiknjo, M.; Monteiro, S.; Grandt, J.; Kimer, N.; Madsen, J.L.; Werge, M.P.; William, P.; Brol, M.J.; Turco, L.; Schierwagen, R.; et al. Cardiodynamic State Is Associated with Systemic Inflammation and Fatal Acute-on-Chronic Liver Failure. Liver Int. 2020, 40, 1457–1466. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Lai, J.C.; Boike, J.R.; German, M.; Jest, N.; Morelli, G.; Spengler, E.; Said, A.; Lee, A.; Hristov, A.; et al. Nonalcoholic Fatty Liver Disease and Diabetes Mellitus Are Associated with Post-Transjugular Intrahepatic Portosystemic Shunt Renal Dysfunction: An Advancing Liver Therapeutic Approaches Group Study. Liver Transpl. 2021, 27, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Turco, L.; Garcia-Tsao, G.; Magnani, I.; Bianchini, M.; Costetti, M.; Caporali, C.; Colopi, S.; Simonini, E.; De Maria, N.; Banchelli, F.; et al. Cardiopulmonary Hemodynamics and C-Reactive Protein as Prognostic Indicators in Compensated and Decompensated Cirrhosis. J. Hepatol. 2018, 68, 949–958. [Google Scholar] [CrossRef]
- Italian Association for the Study of the Liver (AISF) Portal Hypertension and Ascites: Patient-and Population-Centered Clinical Practice Guidelines by the Italian Association for the Study of the Liver (AISF). Dig. Liver Dis. 2021, 53, 1089–1104. [CrossRef]
- Casado, M.; Bosch, J.; García-Pagán, J.C.; Bru, C.; Bañares, R.; Bandi, J.C.; Escorsell, A.; Rodríguez-Láiz, J.M.; Gilabert, R.; Feu, F.; et al. Clinical Events after Transjugular Intrahepatic Portosystemic Shunt: Correlation with Hemodynamic Findings. Gastroenterology 1998, 114, 1296–1303. [Google Scholar] [CrossRef] [PubMed]
- Queck, A.; Schwierz, L.; Gu, W.; Ferstl, P.G.; Jansen, C.; Uschner, F.E.; Praktiknjo, M.; Chang, J.; Brol, M.J.; Schepis, F.; et al. Targeted Decrease of Portal Hepatic Pressure Gradient Improves Ascites Control after TIPS. Hepatology 2023, 77, 466–475. [Google Scholar] [CrossRef]
- Parvinian, A.; Bui, J.T.; Knuttinen, M.G.; Minocha, J.; Gaba, R.C. Transjugular Intrahepatic Portosystemic Shunt for the Treatment of Medically Refractory Ascites. Diagn. Interv. Radiol. 2014, 20, 58–64. [Google Scholar] [CrossRef]
- Nair, S.; Singh, R.; Yoselewitz, M. Correlation between Portal/Hepatic Vein Gradient and Response to Transjugular Intrahepatic Portosystemic Shunt Creation in Refractory Ascites. J. Vasc. Interv. Radiol. 2004, 15, 1431–1434. [Google Scholar] [CrossRef] [PubMed]
- Sauerbruch, T.; Mengel, M.; Dollinger, M.; Zipprich, A.; Rössle, M.; Panther, E.; Wiest, R.; Caca, K.; Hoffmeister, A.; Lutz, H.; et al. Prevention of Rebleeding from Esophageal Varices in Patients with Cirrhosis Receiving Small-Diameter Stents Versus Hemodynamically Controlled Medical Therapy. Gastroenterology 2015, 149, 660–668.e1. [Google Scholar] [CrossRef] [PubMed]
- Reverter, E.; Blasi, A.; Abraldes, J.G.; Martínez-Palli, G.; Seijo, S.; Turon, F.; Berzigotti, A.; Balust, J.; Bosch, J.; García-Pagán, J.C. Impact of Deep Sedation on the Accuracy of Hepatic and Portal Venous Pressure Measurements in Patients with Cirrhosis. Liver Int. 2014, 34, 16–25. [Google Scholar] [CrossRef]
- Walser, E.M.; Harris, V.M.; Harman, J.T.; Park, H.M.; Siddiqui, A.R. Quantification of Intrahepatic Portosystemic Shunting after Placement of a Transjugular Intrahepatic Portosystemic Shunt. J. Vasc. Interv. Radiol. 1996, 7, 263–267. [Google Scholar] [CrossRef]
- Preibsch, H.; Spira, D.; Thaiss, W.M.; Syha, R.; Nikolaou, K.; Ketelsen, D.; Lauer, U.M.; Horger, M. Impact of Transjugular Intrahepatic Portosystemic Shunt Implantation on Liver Perfusion Measured by Volume Perfusion CT. Acta Radiol. 2017, 58, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Itkin, M.; Trerotola, S.O.; Stavropoulos, S.W.; Patel, A.; Mondschein, J.I.; Soulen, M.C.; Tuite, C.M.; Shlansky-Goldberg, R.D.; Faust, T.W.; Reddy, K.R.; et al. Portal Flow and Arterioportal Shunting after Transjugular Intrahepatic Portosystemic Shunt Creation. J. Vasc. Interv. Radiol. 2006, 17, 55–62. [Google Scholar] [CrossRef]
- Gülberg, V.; Haag, K.; Rössle, M.; Gerbes, A.L. Hepatic Arterial Buffer Response in Patients with Advanced Cirrhosis. Hepatology 2002, 35, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Qi, X.; Yang, Z.; Yin, Z.; Nie, Y.; Yuan, S.; Wu, K.; Han, G.; Fan, D. Predictors of Hepatic Encephalopathy after Transjugular Intrahepatic Portosystemic Shunt in Cirrhotic Patients: A Systematic Review. J. Gastroenterol. Hepatol. 2011, 26, 943–951. [Google Scholar] [CrossRef]
- Zacharias, H.D.; Zacharias, A.P.; Gluud, L.L.; Morgan, M.Y. Pharmacotherapies That Specifically Target Ammonia for the Prevention and Treatment of Hepatic Encephalopathy in Adults with Cirrhosis. Cochrane Database Syst. Rev. 2019, 6, CD012334. [Google Scholar] [CrossRef]
- Rose, C.F.; Amodio, P.; Bajaj, J.S.; Dhiman, R.K.; Montagnese, S.; Taylor-Robinson, S.D.; Vilstrup, H.; Jalan, R. Hepatic Encephalopathy: Novel Insights into Classification, Pathophysiology and Therapy. J. Hepatol. 2020, 73, 1526–1547. [Google Scholar] [CrossRef]
- Nardelli, S.; Lattanzi, B.; Torrisi, S.; Greco, F.; Farcomeni, A.; Gioia, S.; Merli, M.; Riggio, O. Sarcopenia Is Risk Factor for Development of Hepatic Encephalopathy After Transjugular Intrahepatic Portosystemic Shunt Placement. Clin. Gastroenterol. Hepatol. 2017, 15, 934–936. [Google Scholar] [CrossRef] [PubMed]
- Praktiknjo, M.; Clees, C.; Pigliacelli, A.; Fischer, S.; Jansen, C.; Lehmann, J.; Pohlmann, A.; Lattanzi, B.; Krabbe, V.K.; Strassburg, C.P.; et al. Sarcopenia Is Associated with Development of Acute-on-Chronic Liver Failure in Decompensated Liver Cirrhosis Receiving Transjugular Intrahepatic Portosystemic Shunt. Clin. Transl. Gastroenterol. 2019, 10, e00025. [Google Scholar] [CrossRef] [PubMed]
- Riggio, O.; Angeloni, S.; Salvatori, F.M.; De Santis, A.; Cerini, F.; Farcomeni, A.; Attili, A.F.; Merli, M. Incidence, Natural History, and Risk Factors of Hepatic Encephalopathy after Transjugular Intrahepatic Portosystemic Shunt with Polytetrafluoroethylene-Covered Stent Grafts. Am. J. Gastroenterol. 2008, 103, 2738–2746. [Google Scholar] [CrossRef] [PubMed]
- Nardelli, S.; Bellafante, D.; Ridola, L.; Faccioli, J.; Riggio, O.; Gioia, S. Prevention of Post-Tips Hepatic Encephalopathy: The Search of the Ideal Candidate. Metab. Brain Dis. 2023, 38, 1729–1736. [Google Scholar] [CrossRef]
- Riggio, O.; Ridola, L.; Angeloni, S.; Cerini, F.; Pasquale, C.; Attili, A.F.; Fanelli, F.; Merli, M.; Salvatori, F.M. Clinical Efficacy of Transjugular Intrahepatic Portosystemic Shunt Created with Covered Stents with Different Diameters: Results of a Randomized Controlled Trial. J. Hepatol. 2010, 53, 267–272. [Google Scholar] [CrossRef]
- Miraglia, R.; Maruzzelli, L.; Tuzzolino, F.; Petridis, I.; D’Amico, M.; Luca, A. Transjugular Intrahepatic Portosystemic Shunts in Patients with Cirrhosis with Refractory Ascites: Comparison of Clinical Outcomes by Using 8- and 10-Mm PTFE-Covered Stents. Radiology 2017, 284, 281–288. [Google Scholar] [CrossRef]
- Luo, X.; Wang, X.; Zhu, Y.; Xi, X.; Zhao, Y.; Yang, J.; Li, X.; Yang, L. Clinical Efficacy of Transjugular Intrahepatic Portosystemic Shunt Created with Expanded Polytetrafluoroethylene-Covered Stent-Grafts: 8-Mm Versus 10-Mm. Cardiovasc. Interv. Radiol. 2019, 42, 737–743. [Google Scholar] [CrossRef]
- Trebicka, J.; Bastgen, D.; Byrtus, J.; Praktiknjo, M.; Terstiegen, S.; Meyer, C.; Thomas, D.; Fimmers, R.; Treitl, M.; Euringer, W.; et al. Smaller-Diameter Covered Transjugular Intrahepatic Portosystemic Shunt Stents Are Associated with Increased Survival. Clin. Gastroenterol. Hepatol. 2019, 17, 2793–2799.e1. [Google Scholar] [CrossRef]
- Huang, Z.; Yao, Q.; Zhu, J.; He, Y.; Chen, Y.; Wu, F.; Hua, T. Efficacy and Safety of Transjugular Intrahepatic Portosystemic Shunt (TIPS) Created Using Covered Stents of Different Diameters: A Systematic Review and Meta-Analysis. Diagn. Interv. Imaging 2021, 102, 279–285. [Google Scholar] [CrossRef]
- Gupta, A.N.; Sze, D.Y.; Rigas, D.A. Smaller Diameter and Adjustable Diameter Transjugular Intrahepatic Portosystemic Shunts. Semin. Interv. Radiol. 2023, 40, 021–026. [Google Scholar] [CrossRef]
- Jansen, C.; Möller, P.; Meyer, C.; Kolbe, C.C.; Bogs, C.; Pohlmann, A.; Schierwagen, R.; Praktiknjo, M.; Abdullah, Z.; Lehmann, J.; et al. Increase in Liver Stiffness after Transjugular Intrahepatic Portosystemic Shunt Is Associated with Inflammation and Predicts Mortality. Hepatology 2018, 67, 1472–1484. [Google Scholar] [CrossRef]
- Pieper, C.C.; Sprinkart, A.M.; Nadal, J.; Hippe, V.; Meyer, C.; Schild, H.H.; Thomas, D. Postinterventional Passive Expansion of Partially Dilated Transjugular Intrahepatic Portosystemic Shunt Stents. J. Vasc. Interv. Radiol. 2015, 26, 388–394. [Google Scholar] [CrossRef]
- Borghol, S.; Perarnau, J.M.; Pucheux, J.; D’Alteroche, L.; Ayoub, J.; Trillaud, H. Short- and Long-Term Evolution of the Endoluminal Diameter of Underdilated Stents in Transjugular Intrahepatic Portosystemic Shunt. Diagn. Interv. Imaging 2016, 97, 1103–1107. [Google Scholar] [CrossRef]
- Pieper, C.C.; Jansen, C.; Meyer, C.; Nadal, J.; Lehmann, J.; Schild, H.H.; Trebicka, J.; Thomas, D. Prospective Evaluation of Passive Expansion of Partially Dilated Transjugular Intrahepatic Portosystemic Shunt Stent Grafts-A Three-Dimensional Sonography Study. J. Vasc. Interv. Radiol. 2017, 28, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ma, J.; Zhou, C.; Yang, C.; Huang, S.; Shi, Q.; Xiong, B. Potential Benefits of Underdilation of 8-Mm Covered Stent in Transjugular Intrahepatic Portosystemic Shunt Creation. Clin. Transl. Gastroenterol. 2021, 12, e00376. [Google Scholar] [CrossRef] [PubMed]
- Saltini, D.; Caporali, C.; Indulti, F.; Bianchini, M.; Casari, F.; Prampolini, F.; Felaco, D.; Guasconi, T.; Cuffari, B.; Zanetto, A.; et al. SAT-334—TIPS under-Dilation Strategy with New Controlled Expansion Endoprosthesis: A Hemodynamic and Imaging Confirmation of Its Feasibility. J. Hepatol. 2023, 78, S287–S288. [Google Scholar] [CrossRef]



| Study | Design | Groups/Pts | Stents/Nominal Diameter (mm) | Indication/n | Etiology of Cirrhosis/n | Follow-Up (Days) | Study End-Points | Results |
|---|---|---|---|---|---|---|---|---|
| Bureau et al., 2007 [32] | Multicente, unblinded | BMS 41 | Memotherm Flexx® (BARD), Wallstent® (Boston), Luminex® (BARD), Sinus Stent® (MEDCARE)/NA # | RA, 12; AVB, 14; Prevention of rebleeding, 15. | Alcohol, 22. | 430 ± 322 | 1st—shunt dysfunction rate. 2nd—relapse of ascites (need for LVP) or gastrointestinal bleeding; number of revisions for shunt patency; rates of complication, HE and survival. | ePTFE-SG improved primary patency, reduced clinical relapses and post-TIPS HE rate. No significant difference in survival rate. |
| ePTFE-SG 39 | Viatorr® (GORE)/NA # | RA, 20; AVB, 9; Prevention of rebleeding, 10. | Alcohol, 22. | 585 ± 438 | ||||
| Huang et al., 2010 [48] | Single-center, unblinded | BMS 30 | Wallstents® (Boston)/10 | PH-related bleeding, 26; RA or hydrothorax, 4. | Viral, 29; Alcohol, 1. | 249 ± 132 | The role of Doppler US in quantitative assessment of shunt function and the usefulness of routine US follow-up of ePTFE-SGs. | Routine US surveillance may not be necessary for ePTFE-SG. ePTFE-SG improved primary patency rate. |
| ePTFE-SG 30 | Fluency® (BARD)/8 | PH-related bleeding, 25; RA or hydrothorax, 5. | Viral, 28; Alcohol, 2. | 186 ± 117 | ||||
| Perarnau et al., 2014 [31] | Multicenter, single-blind | BMS 66 | Luminexx® (BARD), Palmaz Genesis® (Cordis), Smart® (Cordis), Wallstent® (Boston), Zilver® (Cook)/NA # | Prevention of rebleeding, 22; RA, 46; Hydrothorax, 6 †. | Alcohol, 61; Viral, 8; NASH, 1; Others, 2. | 654 § (IQR 171–723) | 1st—shunt dysfunction rate. 2nd—early complications (≤1 month); symptoms recurrence; rate of HE/ disabling chronic; quality of life; early (≤1 month) and late mortality. | ePTFE-SG improved primary patency and reduced clinical relapses rate. No significant difference in post-TIPS HE and survival rate. |
| ePTFE-SG 71 | Fluency® (BARD), Fluency® + BMs, Viatorr® (GORE)/10 | Prevention of rebleeding, 20; RA, 54; Hydrothorax, 3 †. | Alcohol, 52; Viral, 10; NASH, 6; Others, 1. | 708 § (IQR 420–723) | ||||
| Wang et al., 2016 [49] | Single-center, double-blind | BMS 131 | Smart® (Cordis)/8 | PH-related bleeding, 122; RA, 22 †. | Viral, 102; Others, 25. | NA (5 year) | Restenosis/occlusion rate; recurrence of gastrointestinal bleeding, ascites/ hydrothorax; rate of secondary interventional therapy, HE and survival. | ePTFE-SGs improved both primary and secondary patency rate, and reduced clinical relapses rate.ePTFE-SG significantly improved long-term survival. No significant difference in post-TIPS HE rate. |
| ePTFE-SG 127 | Fluency® (BARD)/8 | PH-related bleeding, 123; RA, 20. | Viral, 104; Alcohol, 27. |
| Study | Design/Pts | Nominal Ø (mm) | Under-Dilation (mm) | Imaging | Sites of Measurement | Type of Measurement | Assessment of Clinical Outcomes |
|---|---|---|---|---|---|---|---|
| Gaba et al., 2015 [75] | Retro; 61 | 10 | 8 | CT | IP tract | Cross-sectional Ø from the midportion of the metal wall | No |
| Pieper et al., 2015 [112] | Retro; 29 | 8 (n = 1) 10 (n = 28) | 7 (n = 1) 8 (n = 28) | CT | PVW, IP, HVW | Cross-sectional area at each site | No |
| Borghol et al., 2016 [113] | Retro; 16 | 10 | 8 | Digital angiography | Gold ring marker-IP-HVW | Mean internal stent Ø | No |
| Pieper et al., 2017 [114] | Prosp; 20 | 10 | 8 | US | IP tract excluding PVW and HVW | Mean of 10 Ø measurements from the midportion of the wall reflex | No |
| Schepis et al., 2018 [24] | Retro; 226 Prosp: 142 | 8–10 | 6 | CT | PVW and HVW | Mean of the largest cross-sectional inner Ø | Yes |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saltini, D.; Indulti, F.; Guasconi, T.; Bianchini, M.; Cuffari, B.; Caporali, C.; Casari, F.; Prampolini, F.; Senzolo, M.; Colecchia, A.; et al. Transjugular Intrahepatic Portosystemic Shunt: Devices Evolution, Technical Tips and Future Perspectives. J. Clin. Med. 2023, 12, 6758. https://doi.org/10.3390/jcm12216758
Saltini D, Indulti F, Guasconi T, Bianchini M, Cuffari B, Caporali C, Casari F, Prampolini F, Senzolo M, Colecchia A, et al. Transjugular Intrahepatic Portosystemic Shunt: Devices Evolution, Technical Tips and Future Perspectives. Journal of Clinical Medicine. 2023; 12(21):6758. https://doi.org/10.3390/jcm12216758
Chicago/Turabian StyleSaltini, Dario, Federica Indulti, Tomas Guasconi, Marcello Bianchini, Biagio Cuffari, Cristian Caporali, Federico Casari, Francesco Prampolini, Marco Senzolo, Antonio Colecchia, and et al. 2023. "Transjugular Intrahepatic Portosystemic Shunt: Devices Evolution, Technical Tips and Future Perspectives" Journal of Clinical Medicine 12, no. 21: 6758. https://doi.org/10.3390/jcm12216758
APA StyleSaltini, D., Indulti, F., Guasconi, T., Bianchini, M., Cuffari, B., Caporali, C., Casari, F., Prampolini, F., Senzolo, M., Colecchia, A., & Schepis, F. (2023). Transjugular Intrahepatic Portosystemic Shunt: Devices Evolution, Technical Tips and Future Perspectives. Journal of Clinical Medicine, 12(21), 6758. https://doi.org/10.3390/jcm12216758








