Abstract
The limitation of traditional delivery methods for fecal microbiota transplantation (FMT) gave birth to colonic transendoscopic enteral tubing (TET) to address the requirement of frequent FMTs. Colonic TET as a novel endoscopic intervention has received increasing attention in practice since 2015 in China. Emerging studies from multiple centers indicate that colonic TET is a promising, safe, and practical delivery method for microbial therapy and administering medication with high patient satisfaction. Intriguingly, colonic TET has been used to rescue endoscopy-related perforations by draining colonic air and fluid through the TET tube. Recent research based on collecting ileocecal samples through a TET tube has contributed to demonstrating community dynamics in the intestine, and it is expected to be a novel delivery of proof-of-concept in host–microbiota interactions and pharmacological research. The present article aims to review the concept and techniques of TET and to explore microbial therapy, colonic drainage, and microbial research based on colonic TET.
1. Introduction
The relationship between the intestinal microbiome and diseases has been studied and documented through developments in the field of microbiology and metabolomics. The essence of microbial therapy is to reconstruct the patient’s gut microbiota, and fecal microbiota transplantation (FMT) is the most common method. Recently, increasing evidence has demonstrated the therapeutic potential of FMT in many diseases including recurrent Clostridioides difficile infection (rCDI) [1], inflammatory bowel disease (IBD) [2,3], refractory irritable bowel syndrome [4], autism [5], diabetes mellitus [6], serious antibiotics-associated diarrhea [7], radiation enteritis [8], non-erosive reflux disease [9], and other microbiota-related diseases.
FMT-related delivery methods are traditionally divided into the upper gut, the mid-gut, and the lower gut [10,11,12]. Oral capsule is a delivery method via the upper gut [13]. The mid-gut routes for FMT include the gastroscopy, nasojejunal tube, percutaneous endoscopic gastro-jejunostomy, and mid-gut transendoscopic enteral tubing [14]. The microbiota suspension can be infused into the lower gut through enema, colonoscopy, distal ileum stoma, colostomy, or colonic transendoscopic tubing (TET) [12]. Colonic TET, a novel delivery pathway, was initially designed for multiple FMT and colon administration [12]. Recently, a systematic review reported that patients who underwent colonic TET had the lowest incidence of delivery-related adverse events (AEs) compared with patients using other delivery routes such as capsule, gastroscopy, colonoscopy, and mid-gut tube, etc. [15]. In addition, Allegretti et al. also stated that TET was a promising approach for FMT due to its considerable improvement in safety [16].
Endoscopic placement of an intestinal decompression tube is a practical technique for the treatment of acute intestinal dilation. However, patients with decompression tubes face many difficulties in conducting their daily tasks. Colonic TET, as a delivery method for microbial therapy, could be used to solve the problem of the development of colonic perforation due to IBD or endoscopic-associated injury to avoid surgery [17]. There is a clinical necessity of using a maintainable colonic tube by combining the use of decompression and medication delivery.
Based on the novel implanting method of colonic TET, which directly connects the deep intestine to the exterior, its application is already beyond microbial therapy. The innovation of colonic TET has been proven to be a new non-invasive method of sampling fecal suspension from the cecum for microbiomics and metabolomics research [18]. In addition, colonic TET could represent a novel delivery method for proof-of-concept in pharmacological research. Considering the multiple applications of colonic TET, this review aims to present up-to-date evidence of colonic TET in microbial therapy, colonic drainage, and host–microbiota interaction.
2. The Concept and Technique of Colonic TET
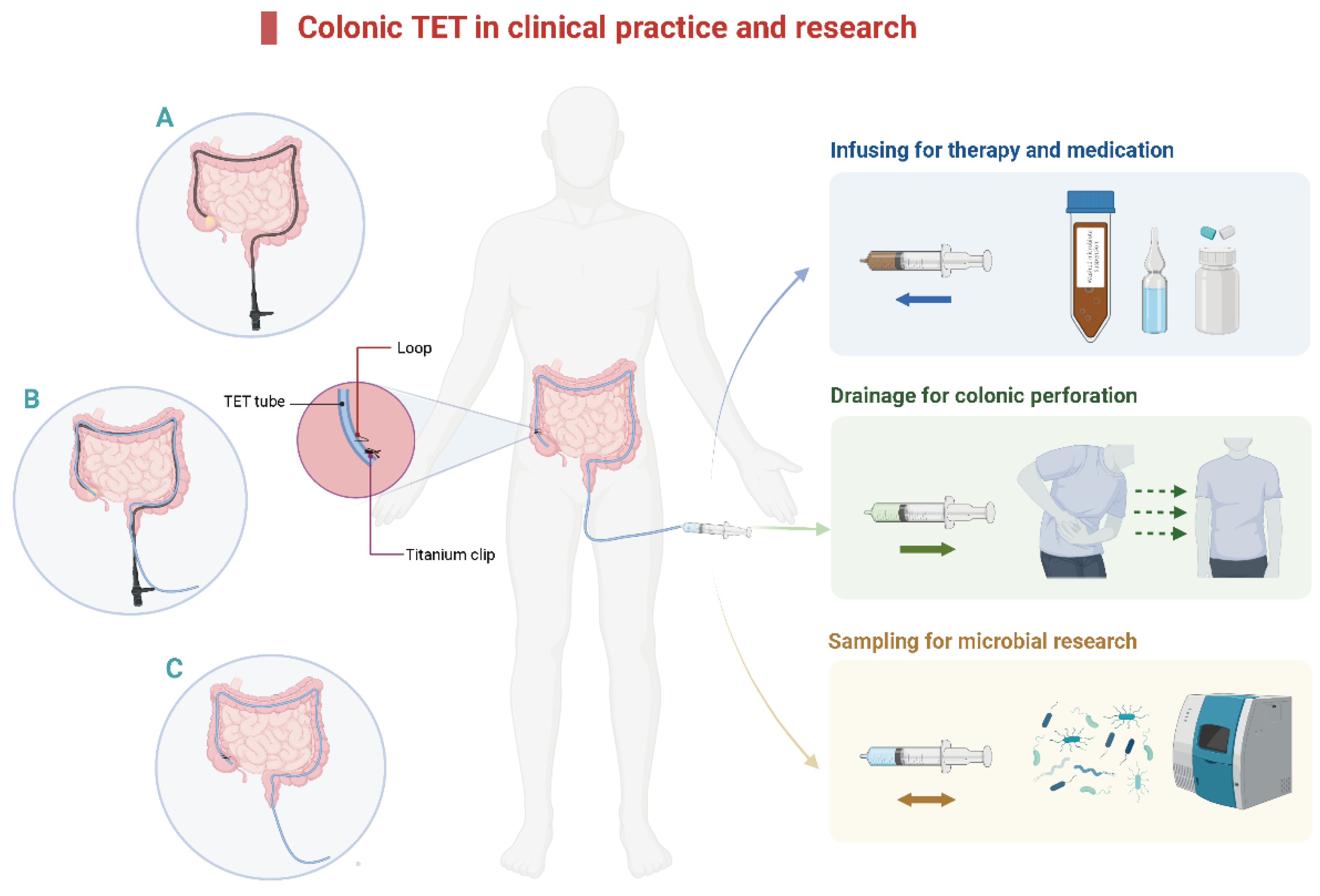
The concept of TET was first reported in 2015 [12]. The main clinical applications of colonic TET are shown in Figure 1. A tiny and soft TET tube should be inserted into the deep colon with endoscopic guidance. After that, the endoscope is removed from the colon while the TET tube is maintained at the target location. Then, the endoscope is re-inserted to fix the TET tube in place [12,19]. The colonic TET tube (FMT medical, Nanjing, China) has three separate loops attached to the tube: the first, second, and third site/station. The first loop is fixed to the proximal end of the colon, 10 cm away from the subsequent loop [19]. Each line-loop on the tube is used to fix the tube to the intestinal wall with one or two endoscopic clips (e.g., ROOC-D-26-195-C, ≥10 mm, Nanjing Microtech Co.; HX-610-135 L, 135°, Olympus). The location and number of clips are based on the mucosal folds, disease severity, and duration for which the tube needs to be retained. Generally, 1–2 clips at the first site and 0–2 clips at the second and/or third site (as required) are recommended [12,19,20]. A previous study indicated that using more than four clips had no extra benefit in prolonging the maintenance time of the TET tube [19]. There is a guide wire within the tube for colonic TET, which is removed from the distal TET tube when the colonoscope is withdrawn from the intestine. Subsequently, the TET tube outside the anus is fixed to the hip lubricated with paraffin.
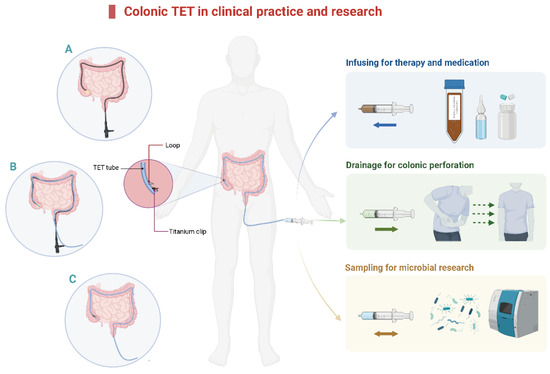
Figure 1.
The diagram of colonic TET in clinical practice and research. (A). Insert the colonic TET into the endoscopic channel when the endoscope reaches the target location, then remove the endoscope. (B). Re-insert the endoscope and hold the TET tube in place. (C). Insert the titanium clip to fix the loop of the colonic TET tube onto the intestinal wall. TET: transendoscopic enteral tubing.
After the endoscope arrives at the target location (e.g., the cecum, ascending colon, transverse colon, and descending colon), the TET tube lubricated with paraffin oil is inserted through the endoscopic channel (diameter >3.2 mm is recommended). A recent study demonstrated that the implantation of a colonic TET tube was quick and safe, although it required double cecal intubation. Compared with regular colonoscopy, the whole cecal intubation time was decreased with the help of cap-assisted colonoscopy, especially the second cecal intubation time (2.8 min vs. 2.2 min, p < 0.001) [21].
3. The Applications of Colonic TET
3.1. Colonic TET for Microbial Therapy
FMT via colonoscopy is impractical for patients who require multiple FMTs to undergo repeated colonoscopy in a short period of time. As for pediatric or older patients who cannot care for themselves, there is a high risk of choking into the airway accidentally when swallowing the oral capsules. Furthermore, it is also not recommended for patients who are unconscious. The microbiota suspension delivered by enema can only reach the rectal and sigmoid colon, making it difficult for patients to retain it in the gut for sufficient time. Therefore, colonic TET was developed to meet the needs of multiple FMTs. In addition, whole or local colonic administration of medications is possible through colonic TET including mesalazine and corticosteroids [12,19]. A study by Oancea et al. demonstrated that the local administration of thioguanine in the rectum might be an effective treatment for colitis [22]. They highlighted the advantages of local drug administration, which reduces the risk of serious side effects associated with systemic delivery. Moreover, colonic TET works for patients who can endure regular enemas. The differences among the common delivery methods are shown in Table 1. The improved methodology of FMT was termed “washed microbiota transplantation” (WMT), which is based on an automatic filtration and washing process and the related delivery [23]. During the washing process, more types, quantities of viruses, and pro-inflammatory mediators are washed out to improve the safety of WMT. Recently, Lu et al. found that colonic TET for delivering WMT was the predominant method used in ulcerative colitis (UC, 67.2%) [24].

Table 1.
Comparison of the different delivery methods.
Colonic TET was recommended by the most recent consensus from the FMT-standardization study group in Asia in 2019 [25] and an international FMT expert group in 2020 [26] due to its convenience and safety for WMT in clinical applications (Figure 2). A questionnaire analysis from Liang’s group indicated that an increasing proportion of the public were aware of FMT and had positive outlooks toward the use of FMT in the treatment of IBD and other diseases in recent years [27]. However, another questionnaire study about the recognition and attitudes of FMT through TET in patients with IBD revealed that a large proportion of participants were unaware of the concept of TET, suggesting that it is necessary to increase public attention and promote the medical application of colonic TET [28].
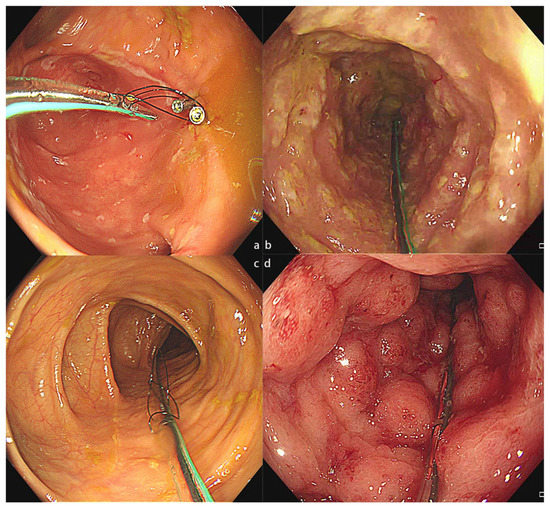
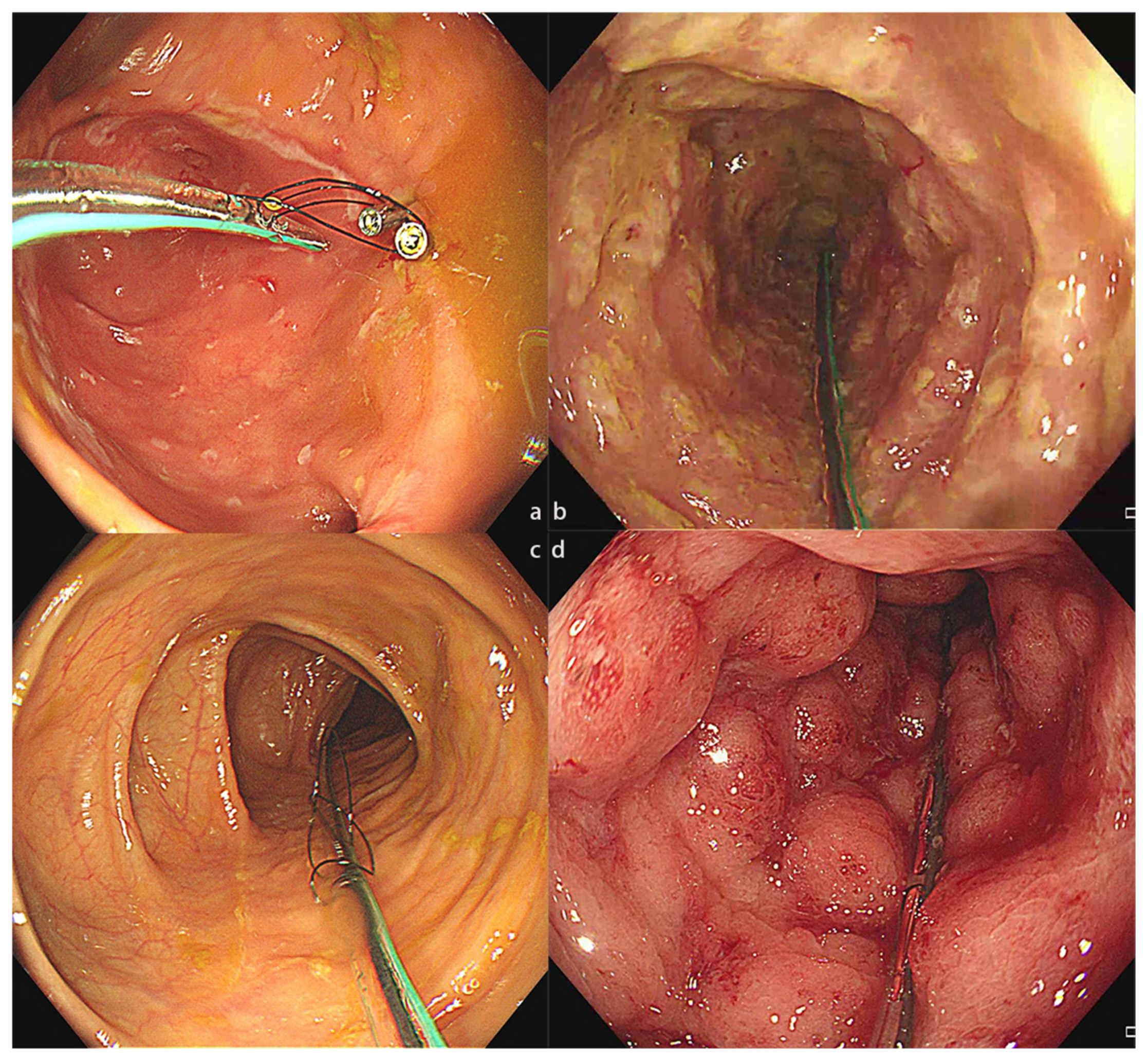
Figure 2.
The colonic TET tube used in different disease conditions. (a) The colonic TET tube in the intestine of a patient with CD. (b). The colonic TET tube in the intestine of a patient with rCDI. (c) The colonic TET tube in the intestine of a patient with IBS. (d) The colonic TET tube in the intestine of a patient with UC. CD, Crohn’s disease; rCDI, recurrent Clostridioides difficile infection; IBS, irritable bowel syndrome; UC, ulcerative colitis; TET, transendoscopic enteral tubing.
Generally, colonic TET is removed actively or falls out spontaneously after microbial therapy or medication treatment; the latter outcome is preferred in clinical practice. Due to the difference in the sample size in studies, the median retention time of colonic TET has been reported to be 12.4 days [12] and 8.6 days [19] in adults and 6 days in children [20]; the difference between adults and children can be attributed to the number and type of endoscopic clip. All of the existing studies indicate that endoscopic clips are an independent factor affecting the retention time. The reported success rate of performing colonic TET was 100% in both adult [19] and pediatric patients [20]. Moreover, physician–patient satisfaction [29] for colonic TET in adults was 97.8% [19], and the reasons for dissatisfaction were not mentioned in the relevant study. In children, the satisfaction rate was 100% [20].
Recently, Philip et al. reported on a patient with fulminant CDI requiring surgical loop ileostomy. The patient underwent rescue FMT, which was safely delivered by a Foley catheter through the ileostomy. The case highlighted the positive contribution of the Foley catheter in multiple FMTs, avoiding re-operation and unnecessary colonoscopy [30]. The role of the Foley catheter in this case was similar to colonic TET; however, it is not as stable as colonic TET because it is not fixed to the intestinal wall.
Several studies regarding FMT delivery via colonic TET demonstrated a high efficacy [31]. Zhou’s group indicated that in 47 patients with UC who underwent FMT treatment via colonic TET, the rate of steroid-free clinical response was 84.1% and steroid-free clinical remission was 70.5% at one month post-FMT [32]. Ding et al. demonstrated that the clinical response of UC patients one month post-FMT via colonic TET was 83.3% [3]. Moreover, Chen’s group reported that in 30 patients with active UC who underwent FMT via colonic TET and enema, the clinical response rate was 59.3% and the clinical remission rate was 40.7% [33]. Remarkably, there was no difference in efficacy between patients who underwent FMT via colonic TET or via other delivery routes (gastroscopy and nasojejunal TET) in different studies [3,34,35,36]. Nie’s group further demonstrated that regardless of whether it was gastroscopy or colonic TET, the delivery route might not affect fecal IgA-bacteria interactions after FMT [37].
Recently, it has been reported that two patients with IgA nephropathy who received FMT through colonic TET for 6–7 months both achieved partial clinical remission [38]. Furthermore, WMT via colonic TET was shown by He’s group to reduce the serum uric acid in patients with hyperuricemia and acute gout [39,40]. Details regarding the application of colonic TET in published articles are shown in Table 2. Of note, colonic TET is not recommended for traditional manual preparation of FMT because tube obstruction was reported while delivering the manual microbiota suspension in another study [41]. Recent reports have highlighted that WMT as a new methodology of FMT contributes to the decreased incidence of AEs compared with manual FMT [23]. Evidence suggests that colonic TET is an efficient, safe, and satisfying delivery route for FMT.

Table 2.
The reported indications, clinical success rates, satisfaction rates, and adverse events of colonic TET.
3.2. Colonic TET for Drainage and Decompression from Deep Colon
Stricture formation and intestinal perforation are common complications of Crohn’s disease (CD) and results from the disease process, surgery, or drugs. The related practical recommendations about dealing with IBD-related strictures and perforation have been released by the International Interventional IBD Group [46,47]. Subsequently, the correct and prudent application of endoscopic stricturotomy and ileo-colonic resection have been emphasized by expert opinions [48,49]. Hence, the endoscopic procedure in the treatment of IBD-associated complications is still a challenge. Although endoscopic decompression of acute intestinal distension can reduce mortality in critically-ill patients [50], the trans-anal decompression tube should be applied discreetly as it may cause intestinal perforation [51]. Unlike traditional trans-anal depression tube placement in the left colon [52,53], colonic TET is a novel interventional procedure that will be promising for bringing benefits to patients under endoscopy and avoiding surgery.
Endoscopic perforations are usually observed in patients with CD-associated strictures [54]. The European Society of Gastrointestinal Endoscopy (ESGE) [55] and the International Intervention IBD Group [46] recommended that endoscopic intervention should be considered depending on the type and size of iatrogenic perforation. Moreover, diversion of the digestive luminal contents and decompression of tension pneumoperitoneum should be performed [55], and colonic TET could play a significant role in this situation. Endoscopy is the correct option if the perforation is found immediately; however, endoscopic reintervention can entail new risks and it may be difficult to locate the site of perforation. Furthermore, prompt diagnosis will affect patient outcomes, and if the endoscopic perforation is found too late, there is a risk of infection. Surgery must be performed once the patient shows symptoms of generalized peritonitis or sepsis. However, if a patient with acute colonic obstruction is in poor condition, emergency surgery compared with elective colon cancer resection has a higher mortality rate [56]. Therefore, placing a colonic TET as palliative treatment and awaiting a better time for surgery is necessary in critical illness.
Zhang et al. reported two cases of stricturing CD in the transverse colon in the same patient who underwent endoscopic balloon dilation and one case with UC and spreading mild dysplasia in the sigmoid colon, both patients suffered perforation after therapeutic endoscopy [17]. A colonic TET with loops was fixed to the ascending or descending colon wall with the intention of WMT and frequently delivering medications. However, perforation was identified by X-ray several days after endoscopy, and the colonic TET was immediately used for draining the air and fluid in the colon with syringe suction. Eventually, all patients recovered rapidly via colonic TET and were free from surgery [17]. In cases of intestinal pressure exceeding atmospheric pressure, regardless of whether surgery is necessary for the patient, colonic TET could be used to drain intestinal fluid and reduce the tension by opening the cap of the distal tube outside the intestine. In addition, colonic TET can be used to deliver antibiotics to prevent or treat infection. Of note, regular endoscopic procedures such as endoscopic mucosal resection within the colon and cap-assistant endoscopic sclerotherapy for hemorrhoids and prolapse have no effect on colonic TET [57].
4. Host-Microbiota Interaction Based on Sampling via Colonic TET
4.1. Discovery in Host–Microbiota Interaction
Microorganisms play critical roles in various physiological functions of the host. Exploring the human microbiota–host interaction could reflect the connection between health and disease [58,59]. Principally, all gut microbiota-derived metabolites are produced in one of three ways: directly from ingested compounds, from host-derived substrates, or de novo from primary metabolites [59]. Therefore, finding a more effective method to sample metabolites is vital. In most studies, fecal samples were used to study the gut microbiome. Although they can be acquired easily, continual fecal samples are rarely taken within short intervals [60]. Some studies found that the ileocecal microbiome, localized in the middle part of the gastrointestinal tract, had relatively higher diversity than the fecal microbiome [61]. Moreover, studies on sampling the intestinal lavage fluid (IVF) microbiome found that pathogenic microbiota was more abundant in the IVF than in feces, and the microbiome in the IVF may be a better indicator for evaluating the risk of developing colorectal cancer compared with fecal samples [62].
Microbial circadian rhythmicity is a feature of mammalian metabolism that might be a significant factor in the development of metabolic disease [63]. Lora V’s group found that the intestinal microbiota in the mouse small intestine programs diurnal metabolic rhythms through histone deacetylase 3 [64]. To investigate community dynamics in the intestine with better resolution, Wang et al. applied colonic TET to extract cecum fluid samples from healthy volunteers twice daily (10 a.m. and 10 p.m.) via syringe, from which metagenomic, metatranscriptomic, metabolomic, and virome analyses were conducted [65,66]. The results revealed the individuality of reconstruction in the microbiome composition, functions, and shared characteristics of the internal resilience of the gut microbiome. Sampling the ileocecal microbiota in situ provides unique insights into the diurnal patterns or circadian rhythms of the human gut microbiome for the first time. Based on samples from a healthy human cecum, Liu et al. further identified that gut microbial methionine impacts circadian clock gene expression and the reactive oxygen species level in the host gastrointestinal tract [18]. Moreover, Fawad et al. reported that gut microbe-generated short-chain fatty acids entrained intestinal epithelial circadian rhythms by inhibiting histone deacetylase [67]. Collection of intestinal fluid with a sterile syringe is recommended. Colonic TET is currently the best non-invasive tool for collecting microbial samples from the deep colon in humans.
4.2. Precision Delivery of Potential Microbiota and Its Metabolites
The interplay between the metabolic activities of the intestinal microbiome and its host forms a significant component of health [68]. The basis of this interaction is mediated by the release of microbially-derived metabolites that interact with the immune or metabolic systems of the host [69]. To date, accumulating evidence focusing on host microbiota has shown that some potential gut microbiota and derived metabolites can be used to reflect and treat different diseases [70]. For example, Meng’s group found that colitis could be treated with indole-3-propionic acid [70], which was also recently demonstrated by Serger et al. to promote nerve regeneration and repair in mice [71]. A 9-amino-acid peptide called D3 was designed by Zhao’s group, which could largely increase the abundance of Akkermansia muciniphila and downregulate CD36 to improve obesity [72]. Moreover, a study by Zhao’s group demonstrated that supplementation with Bacteroides uniformis improved autism spectrum disorder-like behaviors in a mouse model, which added new evidence for the host–microbiota interaction [73]. Table 3 presents some potential substances for the prevention or treatment of disease. In these studies, oral administration was the primary delivery. However, one possibility is that the concentration and properties of these substances had changed when they arrived at their destination in the intestine, which was not mentioned in the articles. Therefore, how to effectively transfer potential metabolites for treatment to the intestine has triggered further investigation.

Table 3.
The potential substances for the prevention or treatment of disease.
To slow down the drug release in the acidic upper gastrointestinal tract after oral administration, novel prebiotic and postbiotic synergistic delivery microcapsules were proposed by Zhao’s team [82]. In addition, Zhong et al. proposed an orally deliverable strategy based on microalgae, which leveraged the biological properties of microalgal carriers to improve the bioavailability of loaded drugs for the treatment of colon cancer and colitis [85]. Similarly, Kaur et al. designed an orally administrable cargo transport device named bacterioboat, which consists of surface-encapsulated mesoporous nanoparticles on metabolically active Lactobacillus reuteri as a drug carrier that is suitable for oral administration [86]. In vivo studies have shown that the oral delivery of 5-fluorouracil via bacterioboat led to increased potency, resulting in improved shrinkage of solid tumors, enhanced life expectancy, and reduced side effects. Decorated bacteria for drug delivery in intestinal disease treatment and cancer therapy will be an innovative strategy in the future if we ignore the cost of development [87].
4.3. The Proof-of-Concept on Translational Microbial Research
A number of the exact mechanisms have not been verified in clinical research, and studies focusing on the interaction between the host or pharmacy and microbiota remain in the proof-of-concept stage [88]. Compared with the attempts of the precision delivery of potential metabolites and drugs, the advantage and feasibility of colonic TET as a novel delivery of the proof-of-concept in host–microbiota interactions and pharmacological research could be highlighted. Polyphenols mainly exist in plant-based food and are known to be beneficial in IBD alleviation. Singh et al. found that a combination of polyphenols from green tea extract and a prebiotic improved the beneficial gut microbiota (Lactobacillus, Bifidobacteria, Akkermansia, Roseburia spp.) abundance, restored Firmicutes/Bacteriodetes, and improved the Prevotella/Bacteroides proportions to effectively reduce the level of inflammation [89]. Enlightened by these studies, combined latent beneficial substances or FMT and probiotics via colonic TET might be a potential method to further explore the relationship between disease and gut microbiota [90].
5. Conclusions
Strategically choosing a new pathway like colonic TET might be more effective than traditional delivery methods in future research. Increasing studies have demonstrated that colonic TET is a promising, safe, and practical delivery method. The present review demonstrates the benefits of this novel technique in providing new options for the improvement of microbial therapy, rescue therapy for patients with endoscopic-related perforation, and research on dynamic host–microbiota interactions. There is no doubt that this review will improve the understanding of colonic TET for researchers, physicians, and patients in clinical practice and basic studies.
Author Contributions
W.W. and G.L. contributed equally to this work. They conducted information collection and wrote the manuscript. X.W. and Q.W. revised the manuscript. F.Z. designed the research, revised the manuscript, and provided valuable suggestions for this study. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Key Research and Development Program of China (2021YFA0717004) and the Nanjing Medical University Fan Daiming Research Funds for Holistic Integrative Medicine.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
Faming Zhang conceived the concept of GenFMTer and transendoscopic enteral tubing and related devices. Other authors declare that they have no competing interests.
References
- Surawicz, C.M.; Brandt, L.J.; Binion, D.G.; Ananthakrishnan, A.N.; Curry, S.R.; Gilligan, P.H.; McFarland, L.V.; Mellow, M.; Zuckerbraun, B.S. Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am. J. Gastroenterol. 2013, 108, 478–498; quiz 499. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Ding, X.; Li, Q.; Wu, X.; Dai, M.; Long, C.; He, Z.; Cui, B.; Zhang, F. Efficacy of faecal microbiota transplantation in Crohn’s disease: A new target treatment? Microb. Biotechnol. 2020, 13, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Li, Q.; Li, P.; Zhang, T.; Cui, B.; Ji, G.; Lu, X.; Zhang, F. Long-Term Safety and Efficacy of Fecal Microbiota Transplant in Active Ulcerative Colitis. Drug Saf. 2019, 42, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Chen, H.; Luo, Q.; Xu, H.; He, J.; Li, Y.; Zhou, Y.; Yao, F.; Nie, Y.; Zhou, Y. Relief of irritable bowel syndrome by fecal microbiota transplantation is associated with changes in diversity and composition of the gut microbiota. J. Dig. Dis. 2019, 20, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Zhong, H.; Huang, D.; Wu, L.; He, X. Beneficial Effects of Repeated Washed Microbiota Transplantation in Children with Autism. Front. Pediatr. 2022, 10, 928785. [Google Scholar] [CrossRef]
- Meijnikman, A.S.; Gerdes, V.E.; Nieuwdorp, M.; Herrema, H. Evaluating Causality of Gut Microbiota in Obesity and Diabetes in Humans. Endocr. Rev. 2018, 39, 133–153. [Google Scholar] [CrossRef]
- Dai, M.; Liu, Y.; Chen, W.; Buch, H.; Shan, Y.; Chang, L.; Bai, Y.; Shen, C.; Zhang, X.; Huo, Y.; et al. Rescue fecal microbiota transplantation for antibiotic-associated diarrhea in critically ill patients. Crit. Care 2019, 23, 324. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Li, Q.; Li, P.; Chen, X.; Xiang, L.; Bi, L.; Zhu, J.; Huang, X.; Cui, B.; Zhang, F. Fecal microbiota transplantation: A promising treatment for radiation enteritis? Radiother. Oncol. 2020, 143, 12–18. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, X.; Cai, J.; Yuan, Y.; Xie, W.; Xu, J.; Xia, H.H.; Zhang, M.; He, X.; Wu, L. Washed microbiota transplantation reduces proton pump inhibitor dependency in nonerosive reflux disease. World J. Gastroenterol. 2021, 27, 513–522. [Google Scholar] [CrossRef]
- Gulati, M.; Singh, S.K.; Corrie, L.; Kaur, I.P.; Chandwani, L. Delivery routes for faecal microbiota transplants: Available, anticipated and aspired. Pharmacol. Res. 2020, 159, 104954. [Google Scholar] [CrossRef]
- Zhang, F.; Cui, B.; He, X.; Nie, Y.; Wu, K.; Fan, D.; FMT-Standardization Study Group. Microbiota transplantation: Concept, methodology and strategy for its modernization. Protein Cell 2018, 9, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Xiang, J.; He, Z.; Zhang, T.; Xu, L.; Cui, B.; Li, P.; Huang, G.; Ji, G.; Nie, Y.; et al. Colonic transendoscopic enteral tubing: A novel way of transplanting fecal microbiota. Endosc. Int. Open 2016, 4, E610–E613. [Google Scholar] [CrossRef] [PubMed]
- Youngster, I.; Russell, G.H.; Pindar, C.; Ziv-Baran, T.; Sauk, J.; Hohmann, E.L. Oral, capsulized, frozen fecal microbiota transplantation for relapsing Clostridium difficile infection. JAMA 2014, 312, 1772–1778. [Google Scholar] [CrossRef]
- Long, C.; Yu, Y.; Cui, B.; Jagessar, S.A.R.; Zhang, J.; Ji, G.; Huang, G.; Zhang, F. A novel quick transendoscopic enteral tubing in mid-gut: Technique and training with video. BMC Gastroenterol. 2018, 18, 37. [Google Scholar] [CrossRef] [PubMed]
- Marcella, C.; Cui, B.; Kelly, C.R.; Ianiro, G.; Cammarota, G.; Zhang, F. Systematic review: The global incidence of faecal microbiota transplantation-related adverse events from 2000 to 2020. Aliment. Pharmacol. Ther. 2020, 53, 33–42. [Google Scholar] [PubMed]
- Allegretti, J.R.; Mullish, B.H.; Kelly, C.; Fischer, M. The evolution of the use of faecal microbiota transplantation and emerging therapeutic indications. Lancet 2019, 394, 420–431. [Google Scholar] [CrossRef]
- Zhang, F.; Wen, Q.; Cui, B. Drainage via colonic transendoscopic enteral tubing increases our confidence in rescuing endoscopy-associated perforation. Endoscopy 2021, 54, E201–E202. [Google Scholar] [CrossRef]
- Liu, X.; Dai, M.; Ma, Y.; Zhao, N.; Wang, Z.; Yu, Y.; Xu, Y.; Zhang, H.; Xiang, L.; Tian, H.; et al. Reconstruction and Dynamics of the Human Intestinal Microbiome Observed In Situ. Engineering 2021, 15, 89–101. [Google Scholar] [CrossRef]
- Zhang, T.; Long, C.; Cui, B.; Buch, H.; Wen, Q.; Li, Q.; Ding, X.; Ji, G.; Zhang, F. Colonic transendoscopic tube-delivered enteral therapy (with video): A prospective study. BMC Gastroenterol. 2020, 20, 135. [Google Scholar] [CrossRef]
- Zhong, M.; Buch, H.; Wen, Q.; Long, C.; Cui, B.; Zhang, F. Colonic Transendoscopic Enteral Tubing: Route for a Novel, Safe, and Convenient Delivery of Washed Microbiota Transplantation in Children. Gastroenterol. Res. Pract. 2021, 2021, 6676962. [Google Scholar] [CrossRef]
- Wen, Q.; Liu, K.-J.; Cui, B.-T.; Li, P.; Wu, X.; Zhong, M.; Wei, L.; Tu, H.; Yuan, Y.; Lin, D.; et al. Impact of cap-assisted colonoscopy during transendoscopic enteral tubing: A randomized controlled trial. World J. Gastroenterol. 2020, 26, 6098–6110. [Google Scholar] [CrossRef] [PubMed]
- Oancea, I.; Movva, R.; Das, I.; de Cárcer, D.A.; Schreiber, V.; Yang, Y.; Purdon, A.; Harrington, B.; Proctor, M.; Wang, R.; et al. Colonic microbiota can promote rapid local improvement of murine colitis by thioguanine independently of T lymphocytes and host metabolism. Gut 2016, 66, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Lu, G.; Zhao, Z.; Liu, Y.; Shen, Q.; Li, P.; Chen, Y.; Yin, H.; Wang, H.; Marcella, C.; et al. Washed microbiota transplantation vs. manual fecal microbiota transplantation: Clinical findings, animal studies and in vitro screening. Protein Cell 2020, 11, 251–266. [Google Scholar] [CrossRef]
- Lu, G.; Wang, W.; Li, P.; Wen, Q.; Cui, B.; Zhang, F. Washed preparation of faecal microbiota changes the transplantation related safety, quantitative method and delivery. Microb. Biotechnol. 2022, 15, 2439–2449. [Google Scholar] [CrossRef] [PubMed]
- Fecal Microbiota Transplantation-Standardization Study Group. Nanjing consensus on methodology of washed microbiota transplantation. Chin. Med. J. 2020, 133, 2330–2332. [Google Scholar] [CrossRef] [PubMed]
- Ianiro, G.; Mullish, B.H.; Kelly, C.R.; Kassam, Z.; Kuijper, E.J.; Ng, S.C.; Iqbal, T.H.; Allegretti, J.R.; Bibbo, S.; Sokol, H.; et al. Reorganisation of faecal microbiota transplant services during the COVID-19 pandemic. Gut 2020, 69, 1555–1563. [Google Scholar] [CrossRef]
- Zhang, Y.; Xue, X.; Su, S.; Zhou, H.; Jin, Y.; Shi, Y.; Lin, J.; Wang, J.; Li, X.; Yang, G.; et al. Patients and physicians’ attitudes change on fecal microbiota transplantation for inflammatory bowel disease over the past 3 years. Ann. Transl. Med. 2021, 9, 1619. [Google Scholar] [CrossRef]
- Zhong, M.; Sun, Y.; Wang, H.-G.; Marcella, C.; Cui, B.-T.; Miao, Y.-L.; Zhang, F.-M. Awareness and attitude of fecal microbiota transplantation through transendoscopic enteral tubing among inflammatory bowel disease patients. World J. Clin. Cases 2020, 8, 3786–3796. [Google Scholar] [CrossRef]
- Dai, M.; Zhang, T.; Li, Q.; Cui, B.; Xiang, L.; Ding, X.; Rong, R.; Bai, J.; Zhu, J.; Zhang, F. The bowel preparation for magnetic resonance enterography in patients with Crohn’s disease: Study protocol for a randomized controlled trial. Trials 2019, 20, 1. [Google Scholar] [CrossRef]
- Philip, S.; Tageldin, O.; Mansoor, M.S.; Richter, S. Successful Fecal Microbiota Transplant Delivered by Foley Catheter Through a Loop Ileostomy in a Patient with Severe Complicated Clostridioides difficile Infection. ACG Case Rep. J. 2022, 9, e00801. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, S.; Borody, T.J.; Zhang, F.M. Encyclopedia of fecal microbiota transplantation: A review of effectiveness in the treatment of 85 diseases. Chin. Med. J. 2022, 135, 1927–1939. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Jin, Z.; Yang, Z.; Zhang, J.; Ma, X.; Guan, J.; Sun, B.; Chen, X. Fecal Microbiota Transplantation Ameliorates Active Ulcerative Colitis by Downregulating Pro-inflammatory Cytokines in Mucosa and Serum. Front. Microbiol. 2022, 13, 818111. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Huang, H.; Xu, H.; Luo, Q.; He, J.; Li, Y.; Zhou, Y.; Nie, Y.; Zhou, Y. Fecal microbiota transplantation ameliorates active ulcerative colitis. Exp. Ther. Med. 2020, 19, 2650–2660. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Zhang, T.; Xiao, Y.; Tian, L.; Cui, B.; Ji, G.; Liu, Y.-Y.; Zhang, F. Timing for the second fecal microbiota transplantation to maintain the long-term benefit from the first treatment for Crohn’s disease. Appl. Microbiol. Biotechnol. 2018, 103, 349–360. [Google Scholar] [CrossRef]
- Li, Q.; Ding, X.; Liu, Y.; Marcella, C.; Dai, M.; Zhang, T.; Bai, J.; Xiang, L.; Wen, Q.; Cui, B.; et al. Fecal Microbiota Transplantation is a Promising Switch Therapy for Patients with Prior Failure of Infliximab in Crohn’s Disease. Front. Pharmacol. 2021, 12, 658087. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Liu, X.L.; Zhang, Y.J.; Nie, Y.Z.; Wu, K.C.; Shi, Y.Q. Efficacy and safety of fecal microbiota transplantation by washed preparation in patients with moderate to severely active ulcerative colitis. J. Dig. Dis. 2020, 21, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.-Q.; Huang, H.-L.; Peng, W.; Liu, Y.-D.; Zhou, Y.-L.; Xu, H.-M.; Zhang, L.-J.; Zhao, C.; Nie, Y.-Q. Altered Pattern of Immunoglobulin A-Targeted Microbiota in Inflammatory Bowel Disease After Fecal Transplantation. Front. Microbiol. 2022, 13, 873018. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Bai, M.; Yang, X.; Wang, Y.; Li, R.; Sun, S. Alleviation of refractory IgA nephropathy by intensive fecal microbiota transplantation: The first case reports. Ren. Fail. 2021, 43, 928–933. [Google Scholar] [CrossRef]
- Cai, J.; Chen, X.; He, Y.; Wu, B.; Zhang, M.; Wu, L. Washed microbiota transplantation reduces serum uric acid levels in patients with hyperuricaemia. World J. Clin. Cases 2022, 10, 3401–3413. [Google Scholar] [CrossRef]
- Xie, W.R.; Yang, X.Y.; Deng, Z.H.; Zheng, Y.M.; Zhang, R.; Wu, L.H.; Cai, J.Y.; Kong, L.P.; Xia, H.H.; He, X.X. Effects of washed microbiota transplantation on serum uric acid levels, symptoms and intestinal barrier function in patients with acute and recurrent gout: A pilot study. Dig. Dis. 2022, 40, 684–690. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Zhang, F.; Su, Y.; Wang, J.; Wu, D.; Hsu, W. Initial experience of fecal microbiota transplantation in gastrointestinal disease: A case series. Kaohsiung J. Med. Sci. 2019, 35, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Sun, X.; Liu, Z. Application of colonic transendoscopic enteral tubing in the treatment of extensive moderate ulcerative colitis. Chin. J. Digest. Med. Imageol. 2021, 11, 106–110. [Google Scholar] [CrossRef]
- Long, C.; He, Z.; Cui, B.; Zhang, T.; Wen, Q.; Li, Q.; Zhang, J.; Ji, G.; Zhang, F. Methodology, safety and applications of colonic transendoscopic enteral tubing (with video). Chin. J. Dig. Endosc. 2020, 37, 28–32. [Google Scholar] [CrossRef]
- Luo, W.; Xie, W.; Chen, Q.; Xue, L.; He, X. Evaluation of the efficacy and safety of fecal microbiota transplantation in different ways on ulcerative colitis. Chin. J. Gastroenterol. Hepatol. 2020, 29, 1008–1011. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, J.; Wang, S.; Xu, W. Application of transendoscopic enteral tubing in the treatment of extensive colonic ulcerative colitis. J. Pract. Med. 2019, 35, 2824–2827. [Google Scholar] [CrossRef]
- Shen, B.; Kochhar, G.S.; Navaneethan, U.; Cross, R.K.; Farraye, F.A.; Iacucci, M.; Schwartz, D.A.; Gonzalez-Lama, Y.; Schairer, J.; Kiran, R.P.; et al. Endoscopic evaluation of surgically altered bowel in inflammatory bowel disease: A consensus guideline from the Global Interventional Inflammatory Bowel Disease Group. Lancet Gastroenterol. Hepatol. 2021, 6, 482–497. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Kochhar, G.; Navaneethan, U.; Farraye, F.A.; Schwartz, D.A.; Iacucci, M.; Bernstein, C.N.; Dryden, G.; Cross, R.; Bruining, D.H.; et al. Practical guidelines on endoscopic treatment for Crohn’s disease strictures: A consensus statement from the Global Interventional Inflammatory Bowel Disease Group. Lancet Gastroenterol. Hepatol. 2020, 5, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Lan, N.; Hull, T.L.; Shen, B. Endoscopic stricturotomy and ileo-colonic resection in patients with primary Crohn’s disease-related distal ileum strictures. Gastroenterol. Rep. 2020, 8, 312–318. [Google Scholar] [CrossRef]
- Zhang, L.; Lan, N.; Wu, X.; Shen, B. Endoscopic stricturotomy in the treatment of anastomotic strictures in inflammatory bowel disease (IBD) and non-IBD patients. Gastroenterol. Rep. 2019, 8, 143–150. [Google Scholar] [CrossRef]
- Book, T.; Kirstein, M.M.; Schneider, A.; Manns, M.P.; Voigtländer, T. Endoscopic decompression of acute intestinal distension is associated with reduced mortality in critically ill patients. BMC Gastroenterol. 2020, 20, 87. [Google Scholar] [CrossRef]
- Hiraki, M.; Tanaka, T.; Okuyama, K.; Kubo, H.; Ikeda, O.; Kitahara, K. Colon perforation caused by transanal decompression tube after laparoscopic low anterior resection: A case report. Int. J. Surg. Case Rep. 2021, 80, 105640. [Google Scholar] [CrossRef] [PubMed]
- Endo, S.; Kumamoto, K.; Enomoto, T.; Koizumi, K.; Kato, H.; Saida, Y. Comparison of survival and perioperative outcome of the colonic stent and the transanal decompression tube placement and emergency surgery for left-sided obstructive colorectal cancer: A retrospective multi-center observational study “The CODOMO study”. Int. J. Color. Dis. 2020, 36, 987–998. [Google Scholar] [CrossRef] [PubMed]
- An, Y.; Wang, N.; Yang, Z.; Li, Y.; Xu, B.; Guo, G.; Sun, M. Efficacy of transanal drainage tube and self-expanding metallic stent in acute left malignant colorectal obstruction. Ann. Palliat. Med. 2020, 9, 1614–1621. [Google Scholar] [CrossRef] [PubMed]
- Endo, K.; Takahashi, S.; Shiga, H.; Kakuta, Y.; Kinouchi, Y.; Shimosegawa, T. Short and long-term outcomes of endoscopic balloon dilatation for Crohn’s disease strictures. World J. Gastroenterol. 2013, 19, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Paspatis, G.A.; Arvanitakis, M.; Dumonceau, J.-M.; Barthet, M.; Saunders, B.; Turino, S.Y.; Dhillon, A.; Fragaki, M.; Gonzalez, J.-M.; Repici, A.; et al. Diagnosis and management of iatrogenic endoscopic perforations: European Society of Gastrointestinal Endoscopy (ESGE) Position Statement—Update 2020. Endoscopy 2020, 52, 792–810. [Google Scholar] [CrossRef]
- Belle, S. Endoscopic Decompression in Colonic Distension. Visc. Med. 2021, 37, 142–148. [Google Scholar] [CrossRef]
- Zhang, T.; Xu, L.J.; Xiang, J.; He, Z.; Peng, Z.Y.; Huang, G.M.; Ji, G.Z.; Zhang, F.M. Cap-assisted endoscopic sclerotherapy for hemorrhoids: Methods, feasibility and efficacy. World J. Gastrointest. Endosc. 2015, 7, 1334–1340. [Google Scholar] [CrossRef]
- Dorrestein, P.C.; Mazmanian, S.K.; Knight, R. Finding the missing links among metabolites, microbes, and the host. Immunity 2014, 40, 824–832. [Google Scholar] [CrossRef]
- Silpe, J.E.; Balskus, E.P. Deciphering Human Microbiota–Host Chemical Interactions. ACS Central Sci. 2020, 7, 20–29. [Google Scholar] [CrossRef]
- Tang, Q.; Jin, G.; Wang, G.; Liu, T.; Liu, X.; Wang, B.; Cao, H. Current Sampling Methods for Gut Microbiota: A Call for More Precise Devices. Front. Cell. Infect. Microbiol. 2020, 10, 151. [Google Scholar] [CrossRef]
- Stearns, J.C.; Lynch, M.D.J.; Senadheera, D.B.; Tenenbaum, H.C.; Goldberg, M.B.; Cvitkovitch, D.G.; Croitoru, K.; Moreno-Hagelsieb, G.; Neufeld, J.D. Bacterial biogeography of the human digestive tract. Sci. Rep. 2011, 1, 170. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Sun, J.; Yao, F.; Lin, K.; Yuan, Y.; Chen, Y.; Han, H.; Li, Z.; Zou, J.; Jiao, X. Microbiome in Intestinal Lavage Fluid May Be A Better Indicator in Evaluating the Risk of Developing Colorectal Cancer Compared with Fecal Samples. Transl. Oncol. 2020, 13, 100772. [Google Scholar] [CrossRef] [PubMed]
- Reitmeier, S.; Kiessling, S.; Clavel, T.; List, M.; Almeida, E.L.; Ghosh, T.S.; Neuhaus, K.; Grallert, H.; Linseisen, J.; Skurk, T.; et al. Arrhythmic Gut Microbiome Signatures Predict Risk of Type 2 Diabetes. Cell Host Microbe 2020, 28, 258–272.e6. [Google Scholar] [CrossRef] [PubMed]
- Kuang, Z.; Wang, Y.; Li, Y.; Ye, C.; Ruhn, K.A.; Behrendt, C.L.; Olson, E.N.; Hooper, L.V. The intestinal microbiota programs diurnal rhythms in host metabolism through histone deacetylase 3. Science 2019, 365, 1428–1434. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Zhang, Y.; Dai, M.; Xu, J.; Chen, L.; Zhang, F.; Zhao, N.; Wang, J. Profiling of Human Gut Virome with Oxford Nanopore Technology. Med. Microecol. 2020, 4, 100012. [Google Scholar] [CrossRef]
- Liu, X.; Ma, Y.; Yu, Y.; Zhang, W.; Shi, J.; Zhang, X.; Dai, M.; Wang, Y.; Zhang, H.; Zhang, J.; et al. Gut microbial methionine impacts circadian clock gene expression and reactive oxygen species level in host gastrointestinal tract. Protein Cell 2022, pwac021. [Google Scholar] [CrossRef]
- Fawad, J.A.; Luzader, D.H.; Hanson, G.F.; Moutinho, T.J.; McKinney, C.A.; Mitchell, P.G.; Brown-Steinke, K.; Kumar, A.; Park, M.; Lee, S.; et al. Histone deacetylase inhibition by gut microbe-generated short chain fatty acids entrains intestinal epithelial circadian rhythms. Gastroenterology 2022, 163, 1377–1390.e11. [Google Scholar] [CrossRef]
- He, R.; Li, P.; Wang, J.; Cui, B.; Zhang, F.; Zhao, F. The interplay of gut microbiota between donors and recipients determines the efficacy of fecal microbiota transplantation. Gut Microbes 2022, 14, 2100197. [Google Scholar] [CrossRef]
- Cox, T.O.; Lundgren, P.; Nath, K.; Thaiss, C.A. Metabolic control by the microbiome. Genome. Med. 2022, 14, 80. [Google Scholar] [CrossRef]
- Zhao, X.; Jiang, L.; Fang, X.; Guo, Z.; Wang, X.; Shi, B.; Meng, Q. Host-microbiota interaction-mediated resistance to inflammatory bowel disease in pigs. Microbiome 2022, 10, 115. [Google Scholar] [CrossRef]
- Serger, E.; Luengo-Gutierrez, L.; Chadwick, J.S.; Kong, G.; Zhou, L.; Crawford, G.; Danzi, M.C.; Myridakis, A.; Brandis, A.; Bello, A.T.; et al. The gut metabolite indole-3 propionate promotes nerve regeneration and repair. Nature 2022, 607, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, B.; Wang, N.; Zuo, Z.; Wei, H.; Zhao, F. A novel peptide protects against diet-induced obesity by suppressing appetite and modulating the gut microbiota. Gut 2022, gutjnl-2022-328035. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zhang, B.; Ji, P.; Zuo, Z.; Huang, Y.; Wang, N.; Liu, C.; Liu, S.J.; Zhao, F. Changes to gut amino acid transporters and microbiome associated with increased E/I ratio in Chd8(+/−) mouse model of ASD-like behavior. Nat. Commun. 2022, 13, 1151. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Song, H.; Luo, Z.; Wu, H.; Chen, L.; Wang, Y.; Cui, H.; Zhang, Y.; Wang, B.; Li, W.; et al. Acetylcholine ameliorates colitis by promoting IL-10 secretion of monocytic myeloid-derived suppressor cells through the nAChR/ERK pathway. Proc. Natl. Acad. Sci. USA 2021, 118, e2017762118. [Google Scholar] [CrossRef]
- Messaoudene, M.; Pidgeon, R.; Richard, C.; Ponce, M.; Diop, K.; Benlaifaoui, M.; Nolin-Lapalme, A.; Cauchois, F.; Malo, J.; Belkaid, W.; et al. A Natural Polyphenol Exerts Antitumor Activity and Circumvents Anti–PD-1 Resistance through Effects on the Gut Microbiota. Cancer Discov. 2022, 12, 1070–1087. [Google Scholar] [CrossRef]
- Bell, K.J.; Saad, S.; Tillett, B.J.; McGuire, H.M.; Bordbar, S.; Yap, Y.A.; Nguyen, L.T.; Wilkins, M.R.; Corley, S.; Brodie, S.; et al. Metabolite-based dietary supplementation in human type 1 diabetes is associated with microbiota and immune modulation. Microbiome 2022, 10, 9. [Google Scholar] [CrossRef]
- Shin, J.; Noh, J.R.; Choe, D.; Lee, N.; Song, Y.; Cho, S.; Kang, E.J.; Go, M.J.; Ha, S.K.; Chang, D.H.; et al. Ageing and rejuvenation models reveal changes in key microbial communities associated with healthy ageing. Microbiome 2021, 9, 240. [Google Scholar] [CrossRef]
- Gu, Y.; Wang, C.; Qin, X.; Zhou, B.; Liu, X.; Liu, T.; Xie, R.; Liu, J.; Wang, B.; Cao, H. Saccharomyces boulardii, a yeast probiotic, inhibits gut motility through upregulating intestinal serotonin transporter and modulating gut microbiota. Pharmacol. Res. 2022, 181, 106291. [Google Scholar] [CrossRef]
- Griffin, M.E.; Espinosa, J.; Becker, J.L.; Luo, J.-D.; Carroll, T.S.; Jha, J.K.; Fanger, G.R.; Hang, H.C. Enterococcus peptidoglycan remodeling promotes checkpoint inhibitor cancer immunotherapy. Science 2021, 373, 1040–1046. [Google Scholar] [CrossRef]
- Lu, H.; Xu, X.; Fu, D.; Gu, Y.; Fan, R.; Yi, H.; He, X.; Wang, C.; Ouyang, B.; Zhao, P.; et al. Butyrate-producing Eubacterium rectale suppresses lymphomagenesis by alleviating the TNF-induced TLR4/MyD88/NF-κB axis. Cell Host Microbe 2022, 30, 1139–1150.e1137. [Google Scholar] [CrossRef]
- Erny, D.; Dokalis, N.; Mezö, C.; Castoldi, A.; Mossad, O.; Staszewski, O.; Frosch, M.; Villa, M.; Fuchs, V.; Mayer, A.; et al. Microbiota-derived acetate enables the metabolic fitness of the brain innate immune system during health and disease. Cell Metab. 2021, 33, 2260–2276.e7. [Google Scholar] [CrossRef]
- Yang, K.; Wang, X.; Huang, R.; Wang, H.; Lan, P.; Zhao, Y. Prebiotics and Postbiotics Synergistic Delivery Microcapsules from Microfluidics for Treating Colitis. Adv. Sci. 2022, 9, e2104089. [Google Scholar] [CrossRef]
- Federici, S.; Kredo-Russo, S.; Valdés-Mas, R.; Kviatcovsky, D.; Weinstock, E.; Matiuhin, Y.; Silberberg, Y.; Atarashi, K.; Furuichi, M.; Oka, A.; et al. Targeted suppression of human IBD-associated gut microbiota commensals by phage consortia for treatment of intestinal inflammation. Cell 2022, 185, 2879–2898.e24. [Google Scholar] [CrossRef] [PubMed]
- Fluckiger, A.; Daillère, R.; Sassi, M.; Sixt, B.S.; Liu, P.; Loos, F.; Richard, C.; Rabu, C.; Alou, M.T.; Goubet, A.-G.; et al. Cross-reactivity between tumor MHC class I–restricted antigens and an enterococcal bacteriophage. Science 2020, 369, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Zhong, D.; Zhang, D.; Chen, W.; He, J.; Ren, C.; Zhang, X.; Kong, N.; Tao, W.; Zhou, M. Orally deliverable strategy based on microalgal biomass for intestinal disease treatment. Sci. Adv. 2021, 7, abi9265. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Ghosh, S.; Bhowmick, A.; Gadhave, K.; Datta, S.; Ghosh, A.; Garg, N.; Mahajan, R.L.; Basu, B.; Choudhury, D. Bacterioboat—A novel tool to increase the half-life period of the orally administered drug. Sci. Adv. 2022, 8, eabh1419. [Google Scholar] [CrossRef]
- Wu, F.; Liu, J. Decorated bacteria and the application in drug delivery. Adv. Drug. Deliv. Rev. 2022, 188, 114443. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, X.; Wang, J. Small molecules in the big picture of gut microbiome-host cross-talk. Ebiomedicine 2022, 81, 104085. [Google Scholar] [CrossRef]
- Singh, D.P.; Singh, J.; Boparai, R.K.; Zhu, J.; Mantri, S.; Khare, P.; Khardori, R.; Kondepudi, K.K.; Chopra, K.; Bishnoi, M. Isomalto-oligosaccharides, a prebiotic, functionally augment green tea effects against high fat diet-induced metabolic alterations via preventing gut dysbacteriosis in mice. Pharmacol. Res. 2017, 123, 103–113. [Google Scholar] [CrossRef]
- Elkrief, A.; Routy, B. First clinical proof-of-concept that FMT can overcome resistance to ICIs. Nat. Rev. Clin. Oncol. 2021, 18, 325–326. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).