Ustekinumab Promotes Radiological Fistula Healing in Perianal Fistulizing Crohn’s Disease: A Retrospective Real-World Analysis
Abstract
:1. Introduction
2. Methods
2.1. Study Design
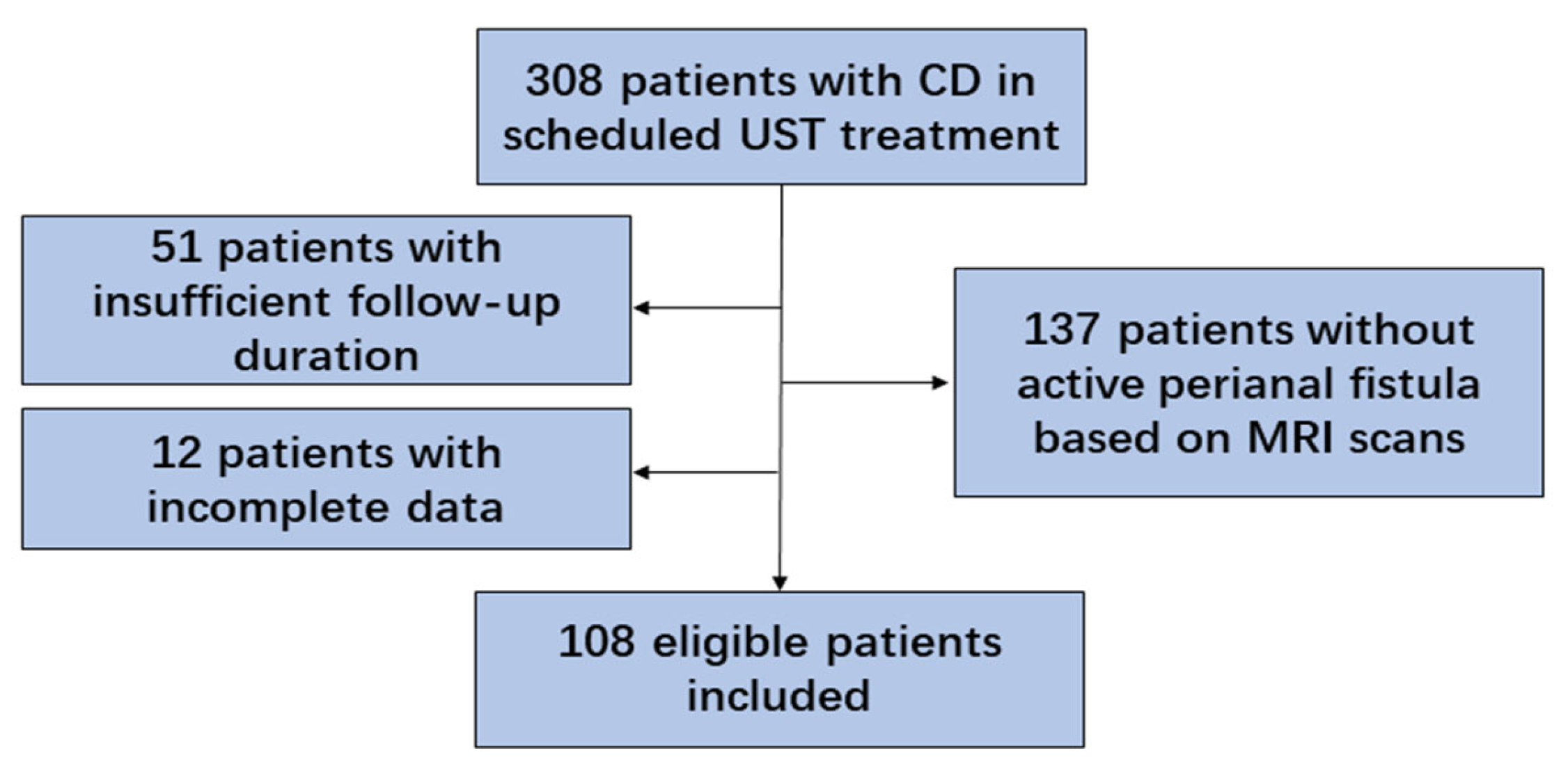
2.2. Patients
2.3. Definition
2.4. Statistical Analysis
3. Results
3.1. Patients’ Characteristics
3.2. Efficacy of UST on CD
3.3. Efficacy of UST on Perianal Fistulas
3.4. Efficacy of UST on Anti-TNF Naïve and Exposure Patients
3.5. Relationship of CD Clinical Remission and Clinical Fistula Response
3.6. Exposure–Response Effect of UST on Perianal Fistulizing CD
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Caron, B.; D’Amico, F.; Danese, S.; Peyrin-Biroulet, L. Endpoints for Perianal Crohn’s Disease Trials: Past, Present and Future. J. Crohns Colitis 2021, 15, 1387–1398. [Google Scholar] [CrossRef]
- Shmidt, E.; Ho, E.Y.; Feuerstein, J.D.; Singh, S.; Terdiman, J.P. Spotlight: Medical Management of Moderate to Severe Luminal and Perianal Fistulizing Crohn’s Disease. Gastroenterology 2021, 160, 2511. [Google Scholar] [CrossRef]
- Barreiro Dominguez, E.M.; Vazquez-Garcia, I.; Perez-Corbal, L.; Ballinas Miranda, J.R.; Antelo, J.S.; Parajo Calvo, A. Mesenchymal stem cells for the treatment of perianal fistulizing Crohn’s disease-A video vignette. Colorectal Dis. 2022, 24, 1441–1442. [Google Scholar] [CrossRef]
- Wiseman, J.; Chawla, T.; Morin, F.; de Buck van Overstraeten, A.; Weizman, A.V. A Multi-Disciplinary Approach to Perianal Fistulizing Crohn’s Disease. Clin. Colon. Rectal Surg. 2022, 35, 51–57. [Google Scholar] [CrossRef]
- Gecse, K.B.; Bemelman, W.; Kamm, M.A.; Stoker, J.; Khanna, R.; Ng, S.C.; Panes, J.; van Assche, G.; Liu, Z.; Hart, A.; et al. A global consensus on the classification, diagnosis and multidisciplinary treatment of perianal fistulising Crohn’s disease. Gut 2014, 63, 1381–1392. [Google Scholar] [CrossRef] [Green Version]
- Sands, B.E.; Anderson, F.H.; Bernstein, C.N.; Chey, W.Y.; Feagan, B.G.; Fedorak, R.N.; Kamm, M.A.; Korzenik, J.R.; Lashner, B.A.; Onken, J.E.; et al. Infliximab maintenance therapy for fistulizing Crohn’s disease. N. Engl. J. Med. 2004, 350, 876–885. [Google Scholar] [CrossRef]
- Colombel, J.F.; Sandborn, W.J.; Rutgeerts, P.; Enns, R.; Hanauer, S.B.; Panaccione, R.; Schreiber, S.; Byczkowski, D.; Li, J.; Kent, J.D.; et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn’s disease: The CHARM trial. Gastroenterology 2007, 132, 52–65. [Google Scholar] [CrossRef] [Green Version]
- Adedokun, O.J.; Xu, Z.; Gasink, C.; Jacobstein, D.; Szapary, P.; Johanns, J.; Gao, L.L.; Davis, H.M.; Hanauer, S.B.; Feagan, B.G.; et al. Pharmacokinetics and Exposure Response Relationships of Ustekinumab in Patients with Crohn’s Disease. Gastroenterology 2018, 154, 1660–1671. [Google Scholar] [CrossRef] [Green Version]
- Feagan, B.G.; Sandborn, W.J.; Gasink, C.; Jacobstein, D.; Lang, Y.; Friedman, J.R.; Blank, M.A.; Johanns, J.; Gao, L.L.; Miao, Y.; et al. Ustekinumab as Induction and Maintenance Therapy for Crohn’s Disease. N. Engl. J. Med. 2016, 375, 1946–1960. [Google Scholar] [CrossRef]
- Yao, J.Y.; Zhang, M.; Wang, W.; Peng, X.; Zhao, J.Z.; Liu, T.; Li, Z.W.; Sun, H.T.; Hu, P.; Zhi, M. Ustekinumab trough concentration affects clinical and endoscopic outcomes in patients with refractory Crohn’s disease: A Chinese real-world study. BMC Gastroenterol. 2021, 21, 380. [Google Scholar] [CrossRef]
- Rubin de Celix, C.; Chaparro, M.; Gisbert, J.P. Real-World Evidence of the Effectiveness and Safety of Ustekinumab for the Treatment of Crohn’s Disease: Systematic Review and Meta-Analysis of Observational Studies. J. Clin. Med. 2022, 11, 4202. [Google Scholar] [CrossRef]
- Honap, S.; Meade, S.; Ibraheim, H.; Irving, P.M.; Jones, M.P.; Samaan, M.A. Effectiveness and Safety of Ustekinumab in Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Dig. Dis. Sci. 2022, 67, 1018–1035. [Google Scholar] [CrossRef]
- Gomollon, F.; Dignass, A.; Annese, V.; Tilg, H.; Van Assche, G.; Lindsay, J.O.; Peyrin-Biroulet, L.; Cullen, G.J.; Daperno, M.; Kucharzik, T.; et al. 3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn’s Disease 2016: Part 1: Diagnosis and Medical Management. J. Crohns Colitis 2017, 11, 3–25. [Google Scholar] [CrossRef] [Green Version]
- Inflammatory Bowel Disease Group; Chinese Society of Gastroenterology; Chinese Medical Association. Chinese consensus on diagnosis and treatment in inflammatory bowel disease (2018, Beijing). J. Dig. Dis. 2021, 22, 298–317. [Google Scholar] [CrossRef]
- Kotze, P.G.; Ma, C.; Almutairdi, A.; Panaccione, R. Clinical utility of ustekinumab in Crohn’s disease. J. Inflamm. Res. 2018, 11, 35–47. [Google Scholar] [CrossRef] [Green Version]
- Satsangi, J.; Silverberg, M.S.; Vermeire, S.; Colombel, J.F. The Montreal classification of inflammatory bowel disease: Controversies, consensus, and implications. Gut 2006, 55, 749–753. [Google Scholar] [CrossRef] [Green Version]
- Thia, K.; Faubion, W.A., Jr.; Loftus, E.V., Jr.; Persson, T.; Persson, A.; Sandborn, W.J. Short CDAI: Development and validation of a shortened and simplified Crohn’s disease activity index. Inflamm. Bowel Dis. 2011, 17, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Irvine, E.J. Usual therapy improves perianal Crohn’s disease as measured by a new disease activity index. McMaster IBD Study Group. J. Clin. Gastroenterol. 1995, 20, 27–32. [Google Scholar]
- Adegbola, S.O.; Dibley, L.; Sahnan, K.; Wade, T.; Verjee, A.; Sawyer, R.; Mannick, S.; McCluskey, D.; Bassett, P.; Yassin, N.; et al. Development and initial psychometric validation of a patient-reported outcome measure for Crohn’s perianal fistula: The Crohn’s Anal Fistula Quality of Life (CAF-QoL) scale. Gut 2021, 70, 1649–1656. [Google Scholar] [CrossRef]
- Rutgeerts, P.; Geboes, K.; Vantrappen, G.; Beyls, J.; Kerremans, R.; Hiele, M. Predictability of the postoperative course of Crohn’s disease. Gastroenterology 1990, 99, 956–963. [Google Scholar] [CrossRef]
- Daperno, M.; D’Haens, G.; Van Assche, G.; Baert, F.; Bulois, P.; Maunoury, V.; Sostegni, R.; Rocca, R.; Pera, A.; Gevers, A.; et al. Development and validation of a new, simplified endoscopic activity score for Crohn’s disease: The SES-CD. Gastrointest. Endosc. 2004, 60, 505–512. [Google Scholar] [CrossRef]
- Ng, S.C.; Plamondon, S.; Gupta, A.; Burling, D.; Swatton, A.; Vaizey, C.J.; Kamm, M.A. Prospective evaluation of anti-tumor necrosis factor therapy guided by magnetic resonance imaging for Crohn’s perineal fistulas. Am. J. Gastroenterol. 2009, 104, 2973–2986. [Google Scholar] [CrossRef]
- Van Assche, G.; Vanbeckevoort, D.; Bielen, D.; Coremans, G.; Aerden, I.; Noman, M.; D’Hoore, A.; Penninckx, F.; Marchal, G.; Cornillie, F.; et al. Magnetic resonance imaging of the effects of infliximab on perianal fistulizing Crohn’s disease. Am. J. Gastroenterol. 2003, 98, 332–339. [Google Scholar] [CrossRef]
- Gu, B.; Venkatesh, K.; Williams, A.J.; Ng, W.; Corte, C.; Gholamrezaei, A.; Ghaly, S.; Xuan, W.; Paramsothy, S.; Connor, S. Higher infliximab and adalimumab trough levels are associated with fistula healing in patients with fistulising perianal Crohn’s disease. World J. Gastroenterol. 2022, 28, 2597–2608. [Google Scholar] [CrossRef]
- Dunleavy, K.A.; Pardi, D.S. Biologics: How far can they go in Crohn’s disease? Gastroenterol. Rep. 2022, 10, goac049. [Google Scholar] [CrossRef]
- Yang, B.L.; Chen, Y.G.; Gu, Y.F.; Chen, H.J.; Sun, G.D.; Zhu, P.; Shao, W.J. Long-term outcome of infliximab combined with surgery for perianal fistulizing Crohn’s disease. World J. Gastroenterol. 2015, 21, 2475–2482. [Google Scholar] [CrossRef]
- Papamichael, K.; Vande Casteele, N.; Jeyarajah, J.; Jairath, V.; Osterman, M.T.; Cheifetz, A.S. Higher Postinduction Infliximab Concentrations Are Associated with Improved Clinical Outcomes in Fistulizing Crohn’s Disease: An ACCENT-II Post Hoc Analysis. Am. J. Gastroenterol. 2021, 116, 1007–1014. [Google Scholar] [CrossRef]
- Sands, B.E.; Gasink, C.; Jacobstein, D.; Gao, L.L.; Johanns, J.; Colombel, J.F.; de Villiers, W.J.; Sandborn, W.J. Fistula healing in prvotal studies of Ustekinumab in Crohn’s disease. Gastroenterology 2017, 152, S185. [Google Scholar] [CrossRef]
- Chapuis-Biron, C.; Kirchgesner, J.; Pariente, B.; Bouhnik, Y.; Amiot, A.; Viennot, S.; Serrero, M.; Fumery, M.; Allez, M.; Siproudhis, L.; et al. Ustekinumab for Perianal Crohn’s Disease: The BioLAP Multicenter Study from the GETAID. Am. J. Gastroenterol. 2020, 115, 1812–1820. [Google Scholar] [CrossRef]
- Biemans, V.B.C.; van der Meulen-de Jong, A.E.; van der Woude, C.J.; Lowenberg, M.; Dijkstra, G.; Oldenburg, B.; de Boer, N.K.H.; van der Marel, S.; Bodelier, A.G.L.; Jansen, J.M.; et al. Ustekinumab for Crohn’s Disease: Results of the ICC Registry, a Nationwide Prospective Observational Cohort Study. J. Crohns Colitis 2020, 14, 33–45. [Google Scholar] [CrossRef]
- Attauabi, M.; Burisch, J.; Seidelin, J.B. Efficacy of ustekinumab for active perianal fistulizing Crohn’s disease: A systematic review and meta-analysis of the current literature. Scand. J. Gastroenterol. 2021, 56, 53–58. [Google Scholar] [CrossRef]
- Johnson, A.M.; Barsky, M.; Ahmed, W.; Zullow, S.; Galati, J.; Jairath, V.; Narula, N.; Peerani, F.; Click, B.H.; Coburn, E.S.; et al. The Real-World Effectiveness and Safety of Ustekinumab in the Treatment of Crohn’s Disease: Results from the SUCCESS Consortium. Am. J. Gastroenterol. 2022, in press. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Li, H.; Xu, H.; Tang, W.; Fan, G.; Yang, X. Can the simplified magnetic resonance index of activity be used to evaluate the degree of activity in Crohn’s disease? BMC Gastroenterol. 2021, 21, 409. [Google Scholar] [CrossRef] [PubMed]
- Karmiris, K.; Bielen, D.; Vanbeckevoort, D.; Vermeire, S.; Coremans, G.; Rutgeerts, P.; Van Assche, G. Long-term monitoring of infliximab therapy for perianal fistulizing Crohn’s disease by using magnetic resonance imaging. Clin. Gastroenterol. Hepatol. 2011, 9, 130–136. [Google Scholar] [CrossRef]
- Panes, J.; Rimola, J. Perianal fistulizing Crohn’s disease: Pathogenesis, diagnosis and therapy. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 652–664. [Google Scholar] [CrossRef] [PubMed]
- Hindryckx, P.; Jairath, V.; Zou, G.; Feagan, B.G.; Sandborn, W.J.; Stoker, J.; Khanna, R.; Stitt, L.; van Viegen, T.; Shackelton, L.M.; et al. Development and Validation of a Magnetic Resonance Index for Assessing Fistulas in Patients with Crohn’s Disease. Gastroenterology 2019, 157, 1233–1244.e35. [Google Scholar] [CrossRef] [Green Version]
- El-Matary, W.; Walters, T.D.; Huynh, H.Q.; deBruyn, J.; Mack, D.R.; Jacobson, K.; Sherlock, M.E.; Church, P.; Wine, E.; Carroll, M.W.; et al. Higher Postinduction Infliximab Serum trough Levels Are Associated with Healing of Fistulizing Perianal Crohn’s Disease in Children. Inflamm. Bowel Dis. 2019, 25, 150–155. [Google Scholar] [CrossRef]
- Grossberg, L.B.; Cheifetz, A.S.; Papamichael, K. Therapeutic Drug Monitoring of Biologics in Crohn’s Disease. Gastroenterol. Clin. N. Am. 2022, 51, 299–317. [Google Scholar] [CrossRef]
- Sands, B.E.; Kramer, B.C.; Gasink, C.; Jacobstein, D.; Gao, L.L.; Ma, T.; Adedokun, O.J.; Colombel, J.F.; Schwartz, D.A. Association of Ustekinumab Serum Concentrations and Perianal Fistula Resolution inthe Crohn’s Disease Uniti Program. Gastroenterology 2019, 156, S1099–S1100. [Google Scholar] [CrossRef]
- Glass, J.; Alsamman, Y.; Chittajallu, P.; Ahmed, T.; Fudman, D. Ustekinumab Dose Escalation Effective in Real-World Use for Luminal and Perianal Crohn’s Disease. Inflamm. Bowel Dis. 2020, 26, S76. [Google Scholar] [CrossRef]




| Variables | Total Patients (n = 108) |
|---|---|
| Male, n (%) | 80 (74.1) |
| Age at diagnosis, [years, mean ± S.D.E] | 29.2 ± 1.0 |
| Disease duration, [years, mean ± S.D.E] | 4.3 ± 0.4 |
| Montreal classification | |
| Age, n (%) | |
| A1 (≤16 years) | 8 (7.4) |
| A2 (17–40 years) | 87 (80.6) |
| A3 (>40 years) | 13 (12.0) |
| Disease behavior, n (%) | |
| B1 (non-stricturing, non-penetrating) | 66 (61.1) |
| B2 (stricturing) | 13 (12.0) |
| B3 (penetrating) | 29 (26.9) |
| Disease location, n (%) | |
| L1 (ileal) | 20 (18.5) |
| L2 (colonic) | 11 (10.2) |
| L3 (ileocolonic) | 77 (71.3) |
| L4 (upper GI) | 21 (19.4) |
| Fistula type, n (%) | |
| Simple | 61 (56.5) |
| Complex | 47 (43.5) |
| Fistula location, n (%) | |
| Superficial | 20 (18.5) |
| Inter-sphincteric | 69 (63.9) |
| Trans-sphincteric | 17 (15.7) |
| Supra-sphincteric | 2 (1.9) |
| Extra-sphincteric | 0 (0) |
| Van Assche at baseline, median (IQR) | 9.0 (7.0,14.0) |
| Proctitis, n (%) | 62 (57.4) |
| Perianal abscess, n (%) | 32 (29.6) |
| Previous medication, n (%) | |
| Steroids | 50 (46.3) |
| Immunosuppressants 1 | 76 (70.4) |
| Anti-TNF agents 2 | 70 (64.8) |
| Previous intestinal surgery, n (%) | 27 (25.0) |
| Extraintestinal manifestation, n (%) | 8 (7.4) |
| Variables | Baseline | Week 16/20 | p Value |
|---|---|---|---|
| Inflammatory burden (mean ± S.D.E) | |||
| CRP (mg/L) | 24.0 ± 3.2 | 14.6 ± 2.4 | 0.002 |
| ESR (mm/h) | 22.8 ± 2.3 | 18.2 ± 1.6 | 0.051 |
| Platelet (×109/L) | 311.9 ± 9.4 | 296.4 ± 9.1 | 0.090 |
| Nutritional state (mean ± S.D.E) | |||
| Hemoglobin (g/L) | 119.0 ± 2.1 | 129.8 ± 2.1 | <0.001 |
| Alb (g/L) | 36.9 ± 5.1 | 40.2 ± 5.7 | <0.001 |
| BMI | 19.0 ± 2.9 | 19.3 ± 3.3 | 0.247 |
| Intestinal clinical evaluation (IQR) | |||
| CDAI | 179.5 (117.6, 258.2) | 112.2 (71.9, 171.8) | <0.001 |
| Fistula clinical evaluation (IQR) | |||
| PDAI | 7.5 (5.0, 10.0) | 5.0 (3.0, 8.0) | <0.001 |
| CAF-QoL | 49.0 (32.3, 60.0) | 23.5 (9.3, 38.8) | <0.001 |
| Variables | Anti-TNF Naïve 1 | Anti-TNF Exposure | p Value |
|---|---|---|---|
| Intestinal clinical remission, n/n (%) (n = 108) | 30/38 (78.9) | 41/70 (58.6) | 0.033 |
| Intestinal clinical response, n/n (%) (n = 108) | 30/38 (78.9) | 47/70 (67.1) | 0.195 |
| Fistula clinical remission, n/n (%) (n = 108) | 20/38 (52.6) | 24/70 (34.3) | 0.064 |
| Fistula clinical response, n/n (%) (n = 108) | 25/38 (65.8) | 42/70 (60.0) | 0.554 |
| Endoscopic remission, n/n (%) (n = 99) | 14/35 (40.0) | 17/64 (26.6) | 0.381 |
| Endoscopic response, n/n (%) (n = 99) | 20/35 (57.1) | 25/64 (39.1) | 0.260 |
| Radiological remission, n/n (%) (n = 67) | 14/25 (56.0) | 16/42 (38.1) | 0.154 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, J.; Zhang, H.; Su, T.; Peng, X.; Zhao, J.; Liu, T.; Wang, W.; Hu, P.; Zhi, M.; Zhang, M. Ustekinumab Promotes Radiological Fistula Healing in Perianal Fistulizing Crohn’s Disease: A Retrospective Real-World Analysis. J. Clin. Med. 2023, 12, 939. https://doi.org/10.3390/jcm12030939
Yao J, Zhang H, Su T, Peng X, Zhao J, Liu T, Wang W, Hu P, Zhi M, Zhang M. Ustekinumab Promotes Radiological Fistula Healing in Perianal Fistulizing Crohn’s Disease: A Retrospective Real-World Analysis. Journal of Clinical Medicine. 2023; 12(3):939. https://doi.org/10.3390/jcm12030939
Chicago/Turabian StyleYao, Jiayin, Heng Zhang, Tao Su, Xiang Peng, Junzhang Zhao, Tao Liu, Wei Wang, Pinjin Hu, Min Zhi, and Min Zhang. 2023. "Ustekinumab Promotes Radiological Fistula Healing in Perianal Fistulizing Crohn’s Disease: A Retrospective Real-World Analysis" Journal of Clinical Medicine 12, no. 3: 939. https://doi.org/10.3390/jcm12030939






