Challenges for Optimization of Reverse Shoulder Arthroplasty Part II: Subacromial Space, Scapular Posture, Moment Arms and Muscle Tensioning
Abstract
:1. Introduction
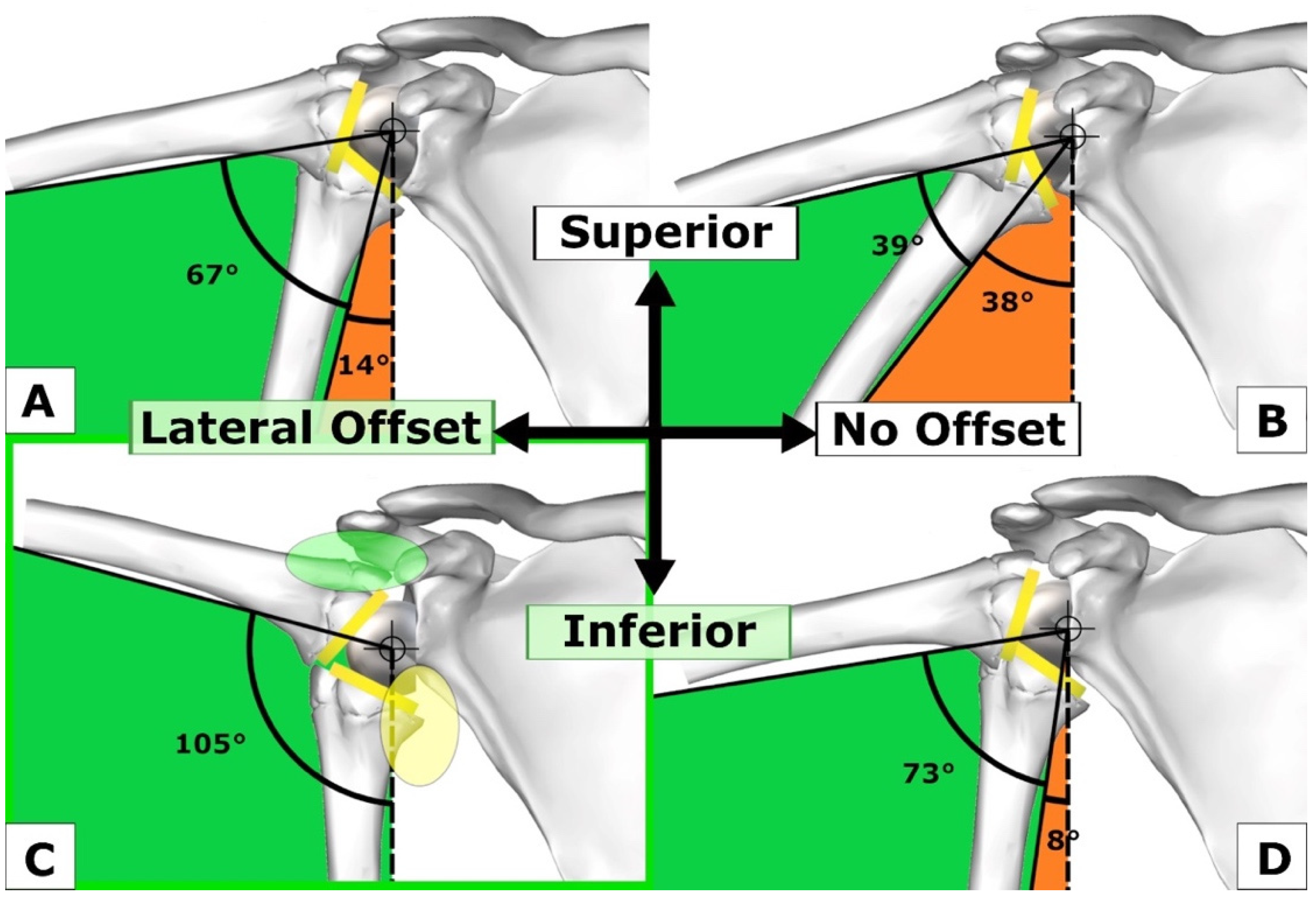
2. Conservation of Sufficient Subacromial and Coracohumeral Space
- (1)
- Lateralization of COR of GS;
- (2)
- GS distalization (BP low, BP small, GS eccentric);
- (3)
- GS inferior tilt;
- (4)
- Increasing NSA from 130° to 150°.
3. Scapular Posture
4. Moment Arms and Muscle Tensioning
4.1. Biomechanics: Moment Arms and Muscle Tensioning
4.2. Arm Length and Prevention of Nerve Injuries
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Walch, G.; Vezeridis, P.S.; Boileau, P.; Deransart, P.; Chaoui, J. Three-dimensional planning and use of patient-specific guides improve glenoid component position: An in vitro study. J. Shoulder Elb. Surg. 2015, 24, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Moroder, P.; Akgün, D.; Plachel, F.; Baur, A.D.J.; Siegert, P. The influence of posture and scapulothoracic orientation on the choice of humeral component retrotorsion in reverse total shoulder arthroplasty. J. Shoulder Elb. Surg. 2020, 29, 1992–2001. [Google Scholar] [CrossRef] [PubMed]
- Werthel, J.-D.; Walch, G.; Vegehan, E.; Deransart, P.; Sanchez-Sotelo, J.; Valenti, P. Lateralization in reverse shoulder arthroplasty: A descriptive analysis of different implants in current practice. Int. Orthop. 2019, 43, 2349–2360. [Google Scholar] [CrossRef] [PubMed]
- Bauer, S.; Corbaz, J.; Athwal, G.S.; Walch, G.; Blakeney, W.G. Lateralization in Reverse Shoulder Arthroplasty. J. Clin. Med. 2021, 10, 5380. [Google Scholar] [CrossRef]
- Giles, J.W.; Langohr, D.G.; Johnson, J.A.; Athwal, G.S. Implant Design Variations in Reverse Total Shoulder Arthroplasty Influence the Required Deltoid Force and Resultant Joint Load. Clin. Orthop. Relat. Res. 2015, 473, 3615–3626. [Google Scholar] [CrossRef]
- Langohr, G.D.G.; Giles, J.W.; Athwal, G.S.; Johnson, J.A. The effect of glenosphere diameter in reverse shoulder arthroplasty on muscle force, joint load, and range of motion. J. Shoulder Elb. Surg. 2015, 24, 972–979. [Google Scholar] [CrossRef]
- Giles, J.W.; Langohr, G.D.G.; Johnson, J.A.; Athwal, G.S. The rotator cuff muscles are antagonists after reverse total shoulder arthroplasty. J. Shoulder Elb. Surg. 2016, 25, 1592–1600. [Google Scholar] [CrossRef]
- Lädermann, A.; Edwards, T.B.; Walch, G. Arm lengthening after reverse shoulder arthroplasty: A review. Int. Orthop. 2013, 38, 991–1000. [Google Scholar] [CrossRef]
- Hamilton, M.A.; Diep, P.; Roche, C.; Flurin, P.H.; Wright, T.W.; Zuckerman, J.D.; Routman, H. Effect of reverse shoulder design philosophy on muscle moment arms. J. Orthop. Res. 2015, 33, 605–613. [Google Scholar] [CrossRef]
- Peljovich, A.; Ratner, J.A.; Marino, J. Update of the Physiology and Biomechanics of Tendon Transfer Surgery. J. Hand Surg. 2010, 35, 1365–1369. [Google Scholar] [CrossRef]
- Marion, B.; Leclère, F.M.; Casoli, V.; Paganini, F.; Unglaub, F.; Spies, C.; Valenti, P. Potential axillary nerve stretching during RSA implantation: An anatomical study. Anat. Sci. Int. 2014, 89, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Neeley, R.; Simon, P.; Christmas, K.N.; Gorman, R.A., II; Amador, I.E.; Frankle, M.A.; Mighell, M.A. Radiographic outcomes of patients undergoing reverse shoulder arthroplasty using inlay versus onlay components: Is there really a difference? Semin. Arthroplast. JSES 2021, 31, 620–628. [Google Scholar] [CrossRef]
- Lädermann, A.; Tay, E.; Collin, P.; Piotton, S.; Chiu, C.-H.; Michelet, A.; Charbonnier, C. Effect of critical shoulder angle, glenoid lateralization, and humeral inclination on range of movement in reverse shoulder arthroplasty. Bone Jt. Res. 2019, 8, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Seebauer, L.; Walter, W.; Keyl, W. Reverse Total Shoulder Arthroplasty for the Treatment of Defect Arthropathy. Eur. J. Trauma 2005, 31, 508–520. [Google Scholar] [CrossRef]
- Nyffeler, R.W.; Werner, C.M.; Gerber, C. Biomechanical relevance of glenoid component positioning in the reverse Delta III total shoulder prosthesis. J. Shoulder Elb. Surg. 2005, 14, 524–528. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Levy, J.C.; E Lee, W.; Keller, T.S.; E Maitland, M. Center of Rotation Affects Abduction Range of Motion of Reverse Shoulder Arthroplasty. Clin. Orthop. Relat. Res. 2007, 458, 78–82. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Levy, J.C.; Frankle, M.A.; Cuff, D.; Keller, T.S.; Pupello, D.R.; Lee, W.E. Evaluation of abduction range of motion and avoidance of inferior scapular impingement in a reverse shoulder model. J. Shoulder Elb. Surg. 2008, 17, 608–615. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Iv, C.A.C.; Luo, Z.-P.; Pupello, D.R.; Frankle, M.A. Range of impingement-free abduction and adduction deficit after reverse shoulder arthroplasty. Hierarchy of surgical and implant-design-related factors. J. Bone Jt. Surg. 2008, 90, 2606–2615. [Google Scholar] [CrossRef]
- Boileau, P.; Morin-Salvo, N.; Gauci, M.-O.; Seeto, B.L.; Chalmers, P.N.; Holzer, N.; Walch, G. Angled BIO-RSA (bony-increased offset–reverse shoulder arthroplasty): A solution for the management of glenoid bone loss and erosion. J. Shoulder Elb. Surg. 2017, 26, 2133–2142. [Google Scholar] [CrossRef]
- Lädermann, A.; Denard, P.J.; Boileau, P.; Farron, A.; Deransart, P.; Walch, G. What is the best glenoid configuration in onlay reverse shoulder arthroplasty? Int. Orthop. 2018, 42, 1339–1346. [Google Scholar] [CrossRef]
- Bauer, S.; Blakeney, W.G.; Goyal, N.; Flayac, H.; Wang, A.; Corbaz, J. Posteroinferior relevant scapular neck offset in reverse shoulder arthroplasty: Key player for motion and friction-type impingement in a computer model. J. Shoulder Elb. Surg. 2022, 31, 2638–2646. [Google Scholar] [CrossRef] [PubMed]
- Ben Kibler, W. The Role of the Scapula in Athletic Shoulder Function. Am. J. Sports Med. 1998, 26, 325–337. [Google Scholar] [CrossRef]
- Culham, E.; Peat, M. Functional Anatomy of the Shoulder Complex. J. Orthop. Sports Phys. Ther. 1993, 18, 342–350. [Google Scholar] [CrossRef]
- Kebaetse, M.; McClure, P.; Pratt, N.A. Thoracic position effect on shoulder range of motion, strength, and three-dimensional scapular kinematics. Arch. Phys. Med. Rehabil. 1999, 80, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Ludewig, P.M.; Reynolds, J.F. The Association of Scapular Kinematics and Glenohumeral Joint Pathologies. J. Orthop. Sports Phys. Ther. 2009, 39, 90–104. [Google Scholar] [CrossRef] [PubMed]
- Ailon, T.; Shaffrey, C.I.; Lenke, L.G.; Harrop, J.S.; Smith, J.S. Progressive Spinal Kyphosis in the Aging Population. Neurosurgery 2015, 77, S164–S172. [Google Scholar] [CrossRef] [PubMed]
- Finley, M.A.; Lee, R.Y. Effect of sitting posture on 3-dimensional scapular kinematics measured by skin-mounted electromagnetic tracking sensors. Arch. Phys. Med. Rehabil. 2003, 84, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Roghani, T.; Zavieh, M.K.; Manshadi, F.D.; King, N.; Katzman, W. Age-related hyperkyphosis: Update of its potential causes and clinical impacts—Narrative review. Aging Clin. Exp. Res. 2016, 29, 567–577. [Google Scholar] [CrossRef]
- Braun, B.L.; Amundson, L.R. Quantitative assessment of head and shoulder posture. Arch. Phys. Med. Rehabil. 1989, 70, 322–329. [Google Scholar]
- Reintgen, C.; Armington, S.; Vigan, M.; Werthel, J.-D.; Patrick, M.; King, J.; Wright, T.; Schoch, B. Influence of Thoracic Kyphosis on Reverse Total Shoulder Arthroplasty Outcomes. J. Am. Acad. Orthop. Surg. 2021, 29, 840–847. [Google Scholar] [CrossRef]
- Moroder, P.; Urvoy, M.M.; Raiss, P.; Werthel, J.-D.; Akgün, D.; Chaoui, J.; Siegert, P. Patient Posture Affects Simulated ROM in Reverse Total Shoulder Arthroplasty: A Modeling Study Using Preoperative Planning Software. Clin. Orthop. Relat. Res. 2021, 480, 619–631. [Google Scholar] [CrossRef]
- Boileau, P.; Moineau, G.; Roussanne, Y.; O’Shea, K. Bony increased-offset reversed shoulder arthroplasty: Minimizing scapular impingement while maximizing glenoid fixation. Clin. Orthop. 2011, 469, 2558–2567. [Google Scholar] [CrossRef]
- Katz, D.; Valenti, P.; Kany, J.; Elkholti, K.; Werthel, J.-D. Does lateralisation of the centre of rotation in reverse shoulder arthroplasty avoid scapular notching? Clinical and radiological review of one hundred and forty cases with forty five months of follow-up. Int. Orthop. 2015, 40, 99–108. [Google Scholar] [CrossRef]
- Wong, M.T.; Langohr, G.D.G.; Athwal, G.S.; Johnson, J.A. Implant positioning in reverse shoulder arthroplasty has an impact on acromial stresses. J. Shoulder Elb. Surg. 2016, 25, 1889–1895. [Google Scholar] [CrossRef]
- Hess, F.; Zettl, R.; Smolen, D.; Knoth, C. Anatomical reconstruction to treat acromion fractures following reverse shoulder arthroplasty. Int. Orthop. 2018, 42, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Bauer, S.; Traverso, A.; Walch, G. Locked 90°-double plating of scapular spine fracture after reverse shoulder arthroplasty with union and good outcome despite plate adjacent acromion fracture. BMJ Case Rep. 2020, 13, e234727. [Google Scholar] [CrossRef] [PubMed]
- Harman, M.; Frankle, M.; Vasey, M.; Banks, S. Initial glenoid component fixation in “reverse” total shoulder arthroplasty: A biomechanical evaluation. J. Shoulder Elb. Surg. 2005, 14 (Suppl. S1), 162S–167S. [Google Scholar] [CrossRef] [PubMed]
- Franceschetti, E.; de Sanctis, E.G.; Ranieri, R.; Palumbo, A.; Paciotti, M.; Franceschi, F. The role of the subscapularis tendon in a lateralized reverse total shoulder arthroplasty: Repair versus nonrepair. Int. Orthop. 2019, 43, 2579–2586. [Google Scholar] [CrossRef]
- Routman, H.D.; Flurin, P.-H.; Wright, T.W.; Zuckerman, J.D.; A Hamilton, M.; Roche, C.P. Reverse Shoulder Arthroplasty Prosthesis Design Classification System. Bull. Hosp. Jt. Dis. 2015, 73 (Suppl. S1), S5–S14. [Google Scholar]
- Kadum, B.; Wahlström, P.; Khoschnau, S.; Sjödén, G.; Sayed-Noor, A. Association of lateral humeral offset with functional outcome and geometric restoration in stemless total shoulder arthroplasty. J. Shoulder Elb. Surg. 2016, 25, e285–e294. [Google Scholar] [CrossRef]
- Vourazeris, J.D.; Wright, T.W.; Struk, A.M.; King, J.J.; Farmer, K.W. Primary reverse total shoulder arthroplasty outcomes in patients with subscapularis repair versus tenotomy. J. Shoulder Elb. Surg. 2017, 26, 450–457. [Google Scholar] [CrossRef]
- Boileau, P.; Walch, G. The three-dimensional geometry of the proximal humerus. Implications for surgical technique and prosthetic design. J. Bone Joint Surg. Br. 1997, 79, 857–865. [Google Scholar] [CrossRef]
- Boutsiadis, A.; Lenoir, H.; Denard, P.J.; Panisset, J.-C.; Brossard, P.; Delsol, P.; Guichard, F.; Barth, J. The lateralization and distalization shoulder angles are important determinants of clinical outcomes in reverse shoulder arthroplasty. J. Shoulder Elb. Surg. 2018, 27, 1226–1234. [Google Scholar] [CrossRef] [PubMed]
- Mahendraraj, K.A.; Colliton, E.; Muniz, A.; Menendez, M.E.; Jawa, A. Assessing the validity of the distalization and lateralization shoulder angles following reverse total shoulder arthroplasty. Semin. Arthroplast. JSES 2020, 30, 291–296. [Google Scholar] [CrossRef]
- Di Giacomo, G. The Effect of Reverse Shoulder Arthroplasty Design Parameters on Rotator Cuff and, Deltoid Muscle Torques: A Computational Biomechanical Analysis. In Proceedings of the Advanced Shoulder Arthroplasty Meeting, Snowbird, UT, USA, 21 January 2022. [Google Scholar]
- Lädermann, A.; Williams, M.D.; Melis, B.; Hoffmeyer, P.; Walch, G. Objective evaluation of lengthening in reverse shoulder arthroplasty. J. Shoulder Elb. Surg. 2009, 18, 588–595. [Google Scholar] [CrossRef]
- LiBrizzi, C.L.; Rojas, J.; Joseph, J.; Bitzer, A.; McFarland, E.G. Incidence of clinically evident isolated axil-lary nerve injury in 869 primary anatomic and reverse total shoulder arthroplasties without routine identification of the axillary nerve. JSES Open Access 2019, 3, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Schmalzl, J.; Fenwick, A.; Reichel, T.; Schmitz, B.; Jordan, M.; Meffert, R.; Plumhoff, P.; Boehm, D.; Gilbert, F. Anterior deltoid muscle tension quantified with shear wave ultrasound elastography correlates with pain level after reverse shoulder arthroplasty. Eur. J. Orthop. Surg. Traumatol. 2022, 32, 333–339. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bauer, S.; Blakeney, W.G.; Wang, A.W.; Ernstbrunner, L.; Corbaz, J.; Werthel, J.-D. Challenges for Optimization of Reverse Shoulder Arthroplasty Part II: Subacromial Space, Scapular Posture, Moment Arms and Muscle Tensioning. J. Clin. Med. 2023, 12, 1616. https://doi.org/10.3390/jcm12041616
Bauer S, Blakeney WG, Wang AW, Ernstbrunner L, Corbaz J, Werthel J-D. Challenges for Optimization of Reverse Shoulder Arthroplasty Part II: Subacromial Space, Scapular Posture, Moment Arms and Muscle Tensioning. Journal of Clinical Medicine. 2023; 12(4):1616. https://doi.org/10.3390/jcm12041616
Chicago/Turabian StyleBauer, Stefan, William G. Blakeney, Allan W. Wang, Lukas Ernstbrunner, Jocelyn Corbaz, and Jean-David Werthel. 2023. "Challenges for Optimization of Reverse Shoulder Arthroplasty Part II: Subacromial Space, Scapular Posture, Moment Arms and Muscle Tensioning" Journal of Clinical Medicine 12, no. 4: 1616. https://doi.org/10.3390/jcm12041616
APA StyleBauer, S., Blakeney, W. G., Wang, A. W., Ernstbrunner, L., Corbaz, J., & Werthel, J.-D. (2023). Challenges for Optimization of Reverse Shoulder Arthroplasty Part II: Subacromial Space, Scapular Posture, Moment Arms and Muscle Tensioning. Journal of Clinical Medicine, 12(4), 1616. https://doi.org/10.3390/jcm12041616