Abstract
Background: Regarding weight loss outcomes, the results published after laparoscopic sleeve gastrectomy (LSG) and laparoscopic Roux-en-Y (LRYGB) are conflicting. At this time, no clear evidence exists that outcomes from LSG are similar to those for LRYGB. The main objective of this study was to compare the percent of total weight loss (%TWL) between LRYGB and LSG over the first 2 years using a multilevel mixed-effects linear regression. Methods: Data were collected from a prospectively maintained database of patients who underwent primary laparoscopic bariatric surgery from January 2016 to December 2017 at a French accredited bariatric center. The medical records of 435 consecutive patients were analyzed. %TWL was calculated at each follow-up surgical consultation and used as a repeated outcome variable in our models to assess the long-term %TWL. Due to this hierarchical structure of the data (%TWL at each visit = level 1) within patients (level 2), a multilevel linear regression adjusted for age, sex, preoperative BMI and comorbidities was used. Results: Among the medical records of 435 consecutive patients included, 266 patients underwent LRYGB and 169 underwent LSG. The average %TWL at 2 years was 31.7% for the LRYGB group and 25.8% for the LSG group. The final multivariate model showed that, compared with LRYGB, LSG was associated with a decreased %TWL at over 2 years of follow-up (β: −4.01; CI95%: −5.47 à −2.54; p ≤ 0.001). Conclusion: This observational study suggests that compared with LRYGB, LSG was associated with a decreased %TWL at 2 years using a multilevel model. Further studies are required to confirm the results observed with this statistical model.
1. Introduction
The last 30 years have brought a dramatic increase in the worldwide prevalence of obesity, causing considerable social and economic burden [1,2,3]. There is strong evidence that bariatric surgery, compared with medical treatment alone, results in substantial and long-term weight loss in morbidly obese patients [4,5,6]. Laparoscopic sleeve gastrectomy (LSG) and laparoscopic Roux-en-Y gastric bypass (LRYGB) are currently the two most frequently performed procedures in bariatric surgery, and weight loss is considered to be the primary indicator of its success [7,8]. At least 70 randomized controlled trials (RCTs) or observational studies (OS) have directly compared LRYGB and LSG [9,10,11,12,13,14,15,16]. While the majority of RCTs have shown that patients undergoing LRYGB and LSG experience similar weight loss [13,14,16,17], OS have shown that LRYGB patients achieve greater weight loss than LSG patients [9,11]. Furthermore, in daily practice, the choice of procedure is based on a shared decision-making process [18] (i.e., patients’ own preferences and values, comorbidities, intraoperative findings, and surgeons’ expertise and habits) that may not be measured in RCTs; hence, there is interest in complementary observational studies. Finally, traditional approaches to assess long-term weight loss in bariatric surgery have relied on a single time-point analysis, either through bivariate or regression models. However, this analysis requires, from a statistical point of view, to take into account the presence of repeated weight measurements for the same individual over time. To preserve this hierarchical structure, multilevel mixed-effects linear models should be used to assess weight loss over time [19].
To our knowledge, no superiority in terms of long-term weight loss efficacy has been demonstrated between LSG and LRYGB, so there is no consensus.
The main objective was to compare the %TWL between LRYGB and LSG over the first 2 years using a multilevel mixed-effects linear regression.
2. Materials and Methods
2.1. Study Design
The medical records of 435 consecutive patients who underwent laparoscopic bariatric surgery between January 2016 to December 2017 were analyzed. The data were recorded prospectively. The inclusion criteria for this survey specified patients older than 18 years who underwent, at our specialized and accredited bariatric center, either LRYGB or LSG as a primary operation and had one or more weight measurements obtained during postoperative visits. All indications for bariatric surgery were assessed according to the International Federation for the Surgery of Obesity and Metabolic Disorders criteria [7,8] and endorsed in an interdisciplinary consensus meeting.
2.2. Surgical Technique
All procedures were standardized to be performed in the same way at our center. “The decision to perform LSG or RYGB took into account both the patient’s choice, maximum preoperative BMI and related comorbidities as well as the proposal of the multidisciplinary consultation meeting”. The surgical techniques used for this study were as previously described in the literature [20,21,22].
2.3. Data Collection
All relevant data included patient features (sex, age and comorbidities), pre- (surgery weight) and postoperative biometric values [weight, height, body mass index (BMI) and percentage of total weight loss (%TWL)]. The %TWL was calculated according to the following formula: [(surgery weight-follow-up weight)/surgery weight] × 100. %TWL was calculated at each follow-up surgical consultation and used as a repeated outcome variable in our models to assess the long-term %TWL.
All patients were assessed as part of a routine surgical follow-up program in the outpatient clinic and were seen on a regular schedule at approximately 1, 3, 6, 12, 18 and 24 months postoperatively. The exact day of each consultation was retrospectively collected for all patients. Thus, a delay corresponding to the difference between the day of the surgery and the day of the consultation was calculated.
2.4. Statistical Analyses
Chi-square and Fisher’s exact tests were used to identify statistically significant differences for descriptive comparisons between both groups. p < 0.05 was defined as statistically significant.
Efficacy in terms of weight loss was assessed using %TWL. The %TWL readings at each postoperative follow-up consultation for the same patient corresponded to the use of repeated data. Due to this hierarchical structure of the data (%TWL at each visit = level 1) within patients (level 2, n = 435), a multilevel mixed-effects linear regression adjusted for age, sex, preoperative BMI and comorbidities was used.
Subsequently, the continuous variable %TWL was introduced into the model as the most flexible form of a three-node restricted cubic spline (Mkspline in STATA).
p = 0.05 was considered significant in the final model. All statistical analyses were performed with Stata/SE version 13 (StataCorp, College Station, TX, USA).
This study was approved by the local medical ethics committee and was declared to the CNIL (2204611v0). The requirement for patient consent was waived owing to the retrospective nature of the study.
3. Results
3.1. Demographic and Clinical Characteristics
Between January 2016 and December 2017, 266 patients underwent LRYGB and 169 underwent LSG. Both groups were similar regarding age and obesity-related comorbidities (Table 1). The LRYGB group had a significantly higher prevalence of females. Conversely, the LSG group was significantly associated with both higher preoperative biometric values and ASA score. Both groups were similar regarding age and obesity-related comorbidities.

Table 1.
Characteristics of patients (n = 435) who underwent bariatric surgery (LRYGB or LSG) at the University Hospital of Caen, 2016–2017.
3.2. Weight Loss
The average %TWL at 2 years was 31.7% for the LRYGB group and 25.8% for the LSG group.
Negative β coefficients indicate lower long-term %TWL. Schematically, a significant variable with a negative β means that the patient lost less weight.
3.2.1. Two Years %TWL
The final multivariate model shows that compared with LRYGB, LSG was associated with a decreased %TWL at over 2 years of follow-up (β: −4.01; IC 95%: −5.47 à −2.54; p ≤ 0.001) (Table 2).

Table 2.
Linear mixed model of %TWL from 1 month to over 2 years of follow-up after bariatric surgery at the University Hospital of Caen, 2016–2017.
3.2.2. Weight Loss Curve
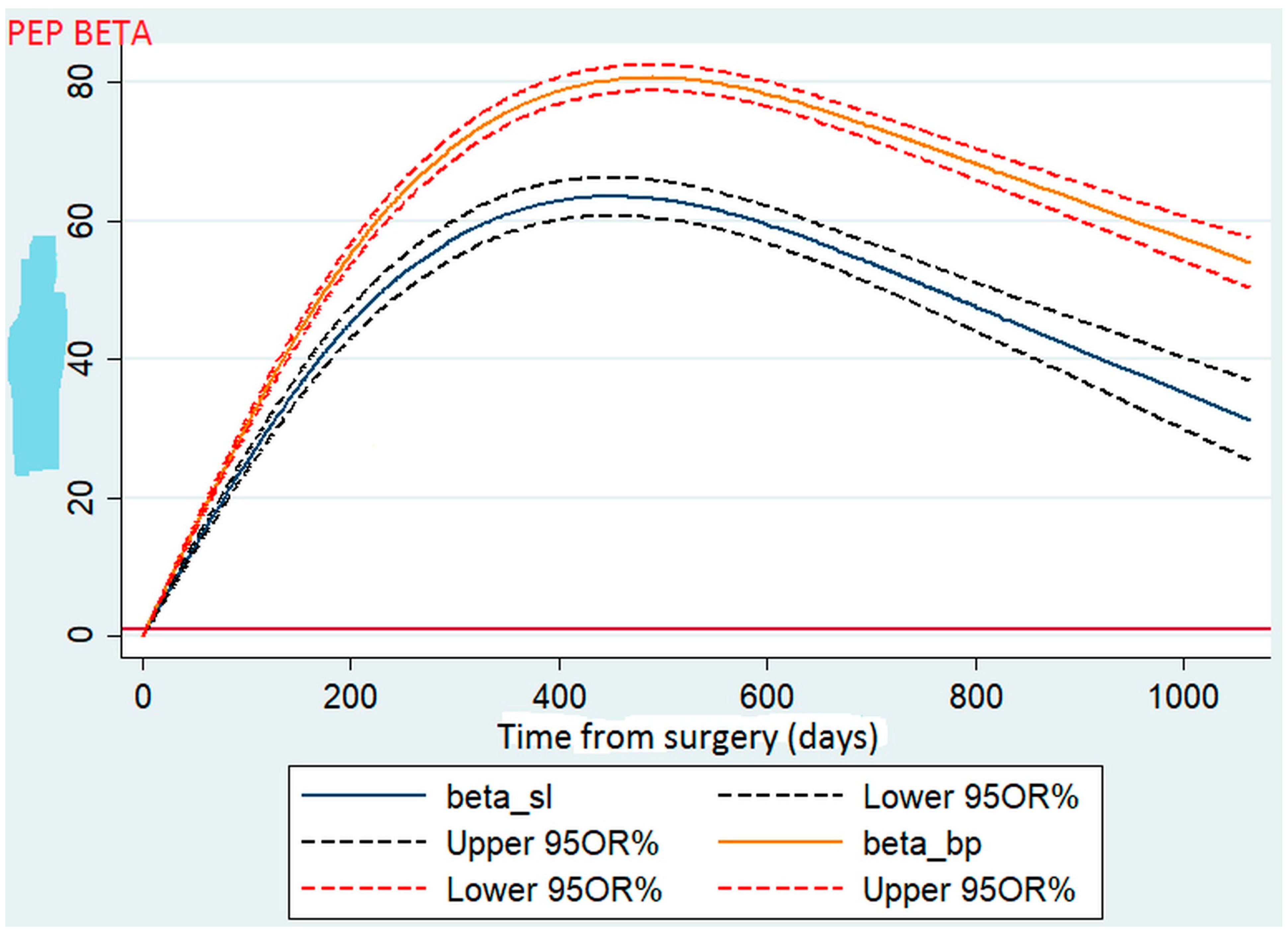
To test the hypothesis of linearity due to the inclusion of the %TWL in a continuous form, we used a four-node cubic spline model. Using a spline (Figure 1), we found that after the two procedures, the patients routinely experienced weight loss in a gradual fashion during the first 2 years. For each procedure, the mean %TWL peaked during the first year (approximately 16 months) after surgery and then fell within the normal distribution, similar to a bell curve.
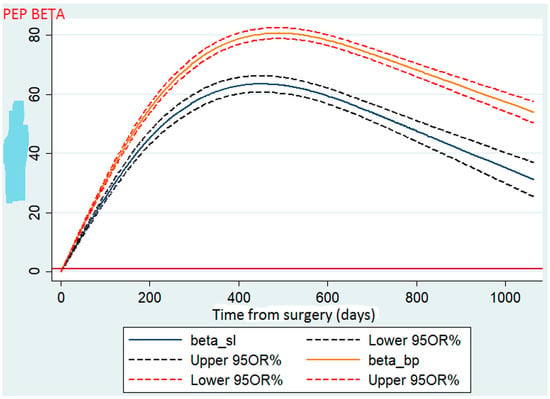
Figure 1.
Comparison of total weight loss between sleeve gastrectomy and gastric bypass.
4. Discussion
Using a multivariate linear regression in our study sample, we found that LSG compared with LRYGB was associated with a decrease in %TWL at 2 years of follow-up. These findings are compelling because the majority of current studies (including RCTs) have reported little or no difference in short-term weight loss between LRYGB and LSG [13,14,15,23,24,25,26,27,28,29]. A subset of other studies have found that LRYGB results in greater weight loss than LSG at 1 to 4 years of follow-up [9,10,12,30,31,32,33].
Patients of our survey with LSG were characterized by higher preoperative BMI as compared to those with RYGB, while baseline obesity-related comorbidities were similar. Importantly, even after adjusting for age, sex and preoperative biometric values, LSG remained associated with lower %TWL than LRYGB.
A number of RCTs have been devoted to compare weight loss outcomes between LSG and LRYGB, most of which have shown similar weight loss between the two techniques [13,14,34]. In a recent meta-analysis, including nine RCTs, Han and al. reported no significant difference in terms of %PEEP between the two procedures [−0.16 (95% CI: −0.52 to 0.19; p = 0.36)]. This conclusion is supported by two previous RCTs, with a follow-up over 5 years greater than 80% [13,14]. Even though they are randomized trials, these results should be interpreted with caution due to the limited number of patients included in each arm (between 20 and 100 patients) and the short follow-up time that has been traditionally reported [34,35,36].
Conversely, observational studies generally show greater weight loss in patients who received RYGB [9,11,12,37]. Thus, the multicenter study, PCORnet Cohort Study, compared the weight results in 32,208 patients who received RYGB against 29,693 patients who received SG [11]. The results show an average total weight loss at 5 years of 25.5% (95% CI, 25.1–25.9%) for RYGB versus 18.8% (95% CI, 18.0–19.6%) for LSG. That is a difference of 6.7% in terms of %PPT (95% CI, 5.8 to 7.7; p < 0.001) in favor of RYGB. Overall, the results of observational studies suggest that the difference observed between the two techniques is greater in a non-randomized setting and may be the consequence of unmeasured differences. Indeed, in daily practice, the choice of procedure is based on a process of shared decisions taking into account preoperatively the preferences of the patient, his comorbidities, the habits and the expertise of the surgeon and/or his center but also intraoperative findings [18]. It is therefore a set of essential factors that cannot be taken into account during an RCT; hence, the interest of complementary observational studies.
Today, one of the difficulties in synthesizing the literature on the subject is based on the lack of uniformity in the analysis tools used. Most experts agree that weight loss and gain should be expressed as a percentage of preoperative weight (which also has the advantage of being easy to use in clinical practice) [38,39]. However, primary studies have evaluated weight loss in other forms, in particular using the %PEP, the average loss of BMI, the weight lost or the percentage loss of excess BMI (%PE-BMI) [16].
Finally, traditional approaches to assess long-term weight loss have relied on a single time-point analysis, either through bivariate models or through regression models that do not respect the fundamental assumption of independence of observations [40]. Very few studies have applied statistical methods for the analysis of repeated measures [11,13,14]. The use of models that did not take this statistical specificity into account may have led to erroneous results.
Furthermore, several studies gave results about excess weight loss after bariatric surgery without multilevel analyses [13,14,41,42]. Thus, statistical analyses were performed with logistic regression model or variance analysis.
Although the data recording is done prospectively, it is a single-center retrospective study with all its inherent limitations. It is also an observational study, which risks unobserved confounding. Finally, the resolution of comorbidities could not be assessed due to a lack of available data, even though this is a parameter for determining the effectiveness of bariatric surgery. Our results must also be interpreted with caution due to the lack of data on long-term complications and quality of life, which are fundamental parameters to be taken into consideration. Similarly, the problem of loss to follow-up patients cannot be excluded, as reported in a recent review of the literature [43,44,45].
However, the study has also several strengths. Our cohort benefited from a relatively high number of patients (n = 435) and a low rate of loss to follow-up 2 years after surgery associated with a satisfactory 2-year follow-up rate (approximately 70%) compared to literature data [13,14,37,43]. This can be explained in particular by standardized patient monitoring within our team for several years; this is done both on a regular basis and over the long term. Indeed, the follow-up of patients has been standardized within our team for several years [20,21,22].
Finally, we applied a multilevel mixed-effects linear model. To our knowledge, only three studies previously considered recorded weights as repeated measures by applying an adapted statistical method with conflicting results. The SLEEVEPASS study was considered to have failed to meet the equivalence criteria set out in the study design, whereas LRYGB was significantly associated with greater excess weight loss at 5 years [13]. The SM-BOSS found no differences in the percentage of body mass index loss, and slight differences in the 5-year body mass index (approximately 1 kg/m2) were found between studies [14]. Conversely, the PCORnet Cohort Study, a large multicenter observational study, found that patients lost more weight with LRYGB than with LSG at 1, 3 and 5 years [36].
The main strengths of our study are based, in our opinion, on the application of a statistical model taking into account the presence of longitudinal data, on the use of recommended monitoring tools (%PPT in particular) [38,39] and on a comprehensive analysis of the literature.
5. Conclusions
In this observational study reflecting daily practice, we found that the use of LSG compared with the use of LRYGB was associated with a decreased %TWL at 2 years using a multivariate linear regression. Taken together, the findings from observational studies suggest that the differences in weight loss outcomes between LRYGB and LSG are slightly larger in nonrandomized settings and may be due to unmeasured differences.
Further studies, either RCTs or OS, are needed to confirm the results observed with this statistical model, also taking into account long-term outcomes (i.e., the resolution of co-morbidities, the prevalence of long-term complications and patients’ quality of life).
Author Contributions
Methodology: C.P., O.D., B.M., A.A. and V.B.; Conceptualization: O.D., A.A.; Methodology: C.P., O.D., B.M.; Software: A.L.B., V.S.; Validation: A.A., G.L.; Formal: C.P., O.D., B.M., A.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent Statement
The requirement for patient consent was waived owing to the retrospective nature of the study. This study was declared to the French Data Protection Authority, CNIL (2204611v0).
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kim, D.D.; Basu, A. Estimating the Medical Care Costs of Obesity in the United States: Systematic Review, Meta-Analysis, and Empirical Analysis. Value Health 2016, 19, 602–613. [Google Scholar] [CrossRef] [PubMed]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Finucane, M.M.; Stevens, G.A.; Cowan, M.J.; Danaei, G.; Lin, J.K.; Paciorek, C.J.; Singh, G.M.; Gutierrez, H.R.; Lu, Y.; Bahalim, A.N.; et al. National, regional, and global trends in body-mass index since 1980: Systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet 2011, 377, 557–567. [Google Scholar] [CrossRef]
- Angrisani, L.; Santonicola, A.; Iovino, P.; Formisano, G.; Buchwald, H.; Scopinaro, N. Bariatric Surgery Worldwide 2013. Obes. Surg. 2015, 25, 1822–1832. [Google Scholar] [CrossRef] [PubMed]
- Mingrone, G.; Panunzi, S.; De Gaetano, A.; Guidone, C.; Iaconelli, A.; Nanni, G.; Castagneto, M.; Bornstein, S.; Rubino, F. Bariatric–metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet 2015, 386, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Schauer, P.R.; Bhatt, D.L.; Kirwan, J.P.; Wolski, K.; Brethauer, S.A.; Navaneethan, S.D.; Aminian, A.; Pothier, C.E.; Kim, E.S.; Nissen, S.E.; et al. Bariatric Surgery versus Intensive Medical Therapy for Diabetes—3-Year Outcomes. N. Engl. J. Med. 2014, 370, 2002–2013. [Google Scholar] [CrossRef] [PubMed]
- Melissas, J. IFSO Guidelines for Safety, Quality, and Excellence in Bariatric Surgery. Obes. Surg. 2008, 18, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Fried, M.; Yumuk, V.; Oppert, J.-M.; Scopinaro, N.; Torres, A.J.; Weiner, R.; Yashkov, Y.; Frühbeck, G. Interdisciplinary European Guidelines on Metabolic and Bariatric Surgery. Obes. Facts 2013, 6, 449–468. [Google Scholar] [CrossRef]
- Maciejewski, M.L.; Arterburn, D.E.; Van Scoyoc, L.; Smith, V.A.; Yancy, W.S., Jr.; Weidenbacher, H.J.; Livingston, E.H.; Olsen, M.K. Bariatric Surgery and Long-term Durability of Weight Loss. JAMA Surg. 2016, 151, 1046–1055. [Google Scholar] [CrossRef]
- Li, J.; Lai, D.; Wu, D. Laparoscopic Roux-en-Y Gastric Bypass Versus Laparoscopic Sleeve Gastrectomy to Treat Morbid Obesity-Related Comorbidities: A Systematic Review and Meta-analysis. Obes. Surg. 2015, 26, 429–442. [Google Scholar] [CrossRef]
- Arterburn, D.; Wellman, R.; Emiliano, A.; Smith, S.R.; Odegaard, A.O.; Murali, S.; Williams, N.; Coleman, K.J.; Courcoulas, A.; Coley, R.Y.; et al. Comparative Effectiveness and Safety of Bariatric Procedures for Weight Loss: A PCORnet cohort study. Ann. Intern. Med. 2018, 169, 741–750. [Google Scholar] [CrossRef] [PubMed]
- Sudan, R.; Maciejewski, M.L.; Wilk, A.R.; Nguyen, N.T.; Ponce, J.; Morton, J.M. Comparative effectiveness of primary bariatric operations in the United States. Surg. Obes. Relat. Dis. 2017, 13, 826–834. [Google Scholar] [CrossRef]
- Peterli, R.; Wölnerhanssen, B.; Peters, T.; Vetter, D.; Kröll, D.; Borbély, Y.; Schultes, B.; Beglinger, C.; Drewe, J.; Schiesser, M.; et al. Effect of Laparoscopic Sleeve Gastrectomy vs Laparoscopic Roux-en-Y Gastric Bypass on Weight Loss in Patients with Morbid Obesity: The SM-BOSS randomized clinical trial. JAMA 2018, 319, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Salminen, P.; Helmiö, M.; Ovaska, J.; Juuti, A.; Leivonen, M.; Peromaa-Haavisto, P.; Hurme, S.; Soinio, M.; Nuutila, P.; Victorzon, M. Effect of Laparoscopic Sleeve Gastrectomy vs Laparoscopic Roux-en-Y Gastric Bypass on Weight Loss at 5 Years Among Patients with Morbid Obesity: The SLEEVEPASS randomized clinical trial. JAMA 2018, 319, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Helmiö, M.; Victorzon, M.; Ovaska, J.; Leivonen, M.; Juuti, A.; Jaser, N.; Peromaa, P.; Tolonen, P.; Hurme, S.; Salminen, P. SLEEVEPASS: A randomized prospective multicenter study comparing laparoscopic sleeve gastrectomy and gastric bypass in the treatment of morbid obesity: Preliminary results. Surg. Endosc. 2012, 26, 2521–2526. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.H.; Le, Q.A. Effectiveness of bariatric surgical procedures: A systematic review and network meta-analysis of randomized controlled trials. Medicine 2017, 96, e8632. [Google Scholar] [CrossRef]
- Colquitt, J.L.; Pickett, K.; Loveman, E.; Frampton, G.K. Surgery for weight loss in adults. Cochrane Database Syst. Rev. 2014, 2014, CD003641. [Google Scholar] [CrossRef]
- Weinstein, A.L.; Marascalchi, B.J.; Spiegel, M.A.; Saunders, J.K.; Fagerlin, A.; Parikh, M. Patient Preferences and Bariatric Surgery Procedure Selection; the Need for Shared Decision-Making. Obes. Surg. 2014, 24, 1933–1939. [Google Scholar] [CrossRef]
- Laird, N.M.; Ware, J.H. Random-Effects Models for Longitudinal Data. Biometrics 1982, 38, 963. [Google Scholar] [CrossRef]
- Contival, N.; Menahem, B.; Gautier, T.; Le Roux, Y.; Alves, A. Guiding the non-bariatric surgeon through complications of bariatric surgery. J. Visc. Surg. 2018, 155, 27–40. [Google Scholar] [CrossRef]
- Meunier, H.; Le Roux, Y.; Fiant, A.-L.; Marion, Y.; Bion, A.L.; Gautier, T.; Contival, N.; Lubrano, J.; Fobe, F.; Zamparini, M.; et al. Does the Implementation of Enhanced Recovery After Surgery (ERAS) Guidelines Improve Outcomes of Bariatric Surgery? A Propensity Score Analysis in 464 Patients. Obes. Surg. 2019, 29, 2843–2853. [Google Scholar] [CrossRef] [PubMed]
- Vallois, A.; Menahem, B.; Le Roux, Y.; Bion, A.L.; Meunier, H.; Gautier, T.; Contival, N.; Mulliri, A.; Lubrano, J.; Parienti, J.-J.; et al. Revisional Roux-en-Y Gastric Bypass: A Safe Surgical Opportunity? Results of a Case-Matched Study. Obes. Surg. 2018, 29, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Barzin, M.; Khalaj, A.; Motamedi, M.A.; Shapoori, P.; Azizi, F.; Hosseinpanah, F. Safety and effectiveness of sleeve gastrectomy versus gastric bypass: One-year results of Tehran Obesity Treatment Study (TOTS). Gastroenterol. Hepatol. Bed Bench 2016, 9, S62–S69. [Google Scholar] [PubMed]
- Chang, W.W.; Hawkins, D.N.; Brockmeyer, J.R.; Faler, B.J.; Hoppe, S.W.; Prasad, B.M. Factors influencing long-term weight loss after bariatric surgery. Surg. Obes. Relat. Dis. 2019, 15, 456–461. [Google Scholar] [CrossRef]
- Kehagias, I.; Karamanakos, S.N.; Argentou, M.; Kalfarentzos, F. Randomized Clinical Trial of Laparoscopic Roux-en-Y Gastric Bypass Versus Laparoscopic Sleeve Gastrectomy for the Management of Patients with BMI < 50 kg/m2. Obes. Surg. 2011, 21, 1650–1656. [Google Scholar] [CrossRef]
- Ignat, M.; Vix, M.; Imad, I.; D’Urso, A.; Perretta, S.; Marescaux, J.; Mutter, D. Randomized trial of Roux-en-Y gastric bypass versus sleeve gastrectomy in achieving excess weight loss. Br. J. Surg. 2016, 104, 248–256. [Google Scholar] [CrossRef]
- Dogan, K.; Gadiot, R.P.M.; Aarts, E.O.; Betzel, B.; van Laarhoven, C.J.H.M.; Biter, L.U.; Mannaerts, G.H.H.; Aufenacker, T.J.; Janssen, I.M.C.; Berends, F.J. Effectiveness and Safety of Sleeve Gastrectomy, Gastric Bypass, and Adjustable Gastric Banding in Morbidly Obese Patients: A Multicenter, Retrospective, Matched Cohort Study. Obes. Surg. 2014, 25, 1110–1118. [Google Scholar] [CrossRef]
- Shoar, S.; Saber, A.A. Long-term and midterm outcomes of laparoscopic sleeve gastrectomy versus Roux-en-Y gastric bypass: A systematic review and meta-analysis of comparative studies. Surg. Obes. Relat. Dis. 2016, 13, 170–180. [Google Scholar] [CrossRef]
- Leyba, J.L.; Llopis, S.N.; Aulestia, S.N. Laparoscopic Roux-en-Y Gastric Bypass Versus Laparoscopic Sleeve Gastrectomy for the Treatment of Morbid Obesity. A Prospective Study with 5 Years of Follow-Up. Obes. Surg. 2014, 24, 2094–2098. [Google Scholar] [CrossRef]
- Jammu, G.S.; Sharma, R. A 7-Year Clinical Audit of 1107 Cases Comparing Sleeve Gastrectomy, Roux-En-Y Gastric Bypass, and Mini-Gastric Bypass, to Determine an Effective and Safe Bariatric and Metabolic Procedure. Obes. Surg. 2015, 26, 926–932. [Google Scholar] [CrossRef]
- Jalilvand, A.; Blaszczak, A.; Dewire, J.; Detty, A.; Needleman, B.; Noria, S. Laparoscopic sleeve gastrectomy is an independent predictor of poor follow-up and reaching ≤40% excess body weight loss at 1, 2, and 3 years after bariatric surgery. Surg. Endosc. 2019, 34, 2572–2584. [Google Scholar] [CrossRef] [PubMed]
- Praveenraj, P.; Gomes, R.M.; Kumar, S.; Perumal, S.; Senthilnathan, P.; Parthasarathi, R.; Rajapandian, S.; Palanivelu, C. Comparison of weight loss outcomes 1 year after sleeve gastrectomy and Roux-en-Y gastric bypass in patients aged above 50 years. J. Minimal Access Surg. 2016, 12, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, H.; Cao, Z.; Sun, X.; Zhang, C.; Cai, W.; Liu, R.; Hu, S.; Qin, M. A Randomized Clinical Trial of Laparoscopic Roux-en-Y Gastric Bypass and Sleeve Gastrectomy for the Treatment of Morbid Obesity in China: A 5-Year Outcome. Obes. Surg. 2014, 24, 1617–1624. [Google Scholar] [CrossRef] [PubMed]
- Grönroos, S.; Helmiö, M.; Juuti, A.; Tiusanen, R.; Hurme, S.; Löyttyniemi, E.; Ovaska, J.; Leivonen, M.; Peromaa-Haavisto, P.; Mäklin, S.; et al. Effect of Laparoscopic Sleeve Gastrectomy vs Roux-en-Y Gastric Bypass on Weight Loss and Quality of Life at 7 Years in Patients with Morbid Obesity: The SLEEVEPASS randomized clinical tria. JAMA Surg. 2021, 156, 137. [Google Scholar] [CrossRef] [PubMed]
- Peterli, R.; Wölnerhanssen, B.; Vetter, D.; Nett, P.; Gass, M.; Borbély, Y.; Peters, T.; Schiesser, M.; Schultes, B.; Beglinger, C.; et al. Laparoscopic Sleeve Gastrectomy Versus Roux-Y-Gastric Bypass for Morbid Obesity—3-Year Outcomes of the Prospective Randomized Swiss Multicenter Bypass or Sleeve Study (SM-BOSS). Ann. Surg. 2017, 265, 466–473. [Google Scholar] [CrossRef]
- Wölnerhanssen, B.K.; Peterli, R.; Hurme, S.; Bueter, M.; Helmiö, M.; Juuti, A.; Meyer-Gerspach, A.C.; Slawik, M.; Peromaa-Haavisto, P.; Nuutila, P.; et al. Laparoscopic Roux-en-Y gastric bypass versus laparoscopic sleeve gastrectomy: 5-year outcomes of merged data from two randomized clinical trials (SLEEVEPASS and SM-BOSS). Br. J. Surg. 2020, 108, 49–57. [Google Scholar] [CrossRef]
- Ahmed, B.; King, W.C.; Gourash, W.; Belle, S.H.; Hinerman, A.; Pomp, A.; Dakin, G.; Courcoulas, A.P. Long-term weight change and health outcomes for sleeve gastrectomy (SG) and matched Roux-en-Y gastric bypass (RYGB) participants in the Longitudinal Assessment of Bariatric Surgery (LABS) study. Surgery 2018, 164, 774–783. [Google Scholar] [CrossRef]
- van Rijswijk, A.-S.; van Olst, N.; Schats, W.; van der Peet, D.L.; van de Laar, A.W. What Is Weight Loss After Bariatric Surgery Expressed in Percentage Total Weight Loss (%TWL)? A Systematic Review. Obes Surg. Août 2021, 31, 3833–3847. [Google Scholar] [CrossRef]
- Grover, B.T.; Morell, M.C.; Kothari, S.N.; Borgert, A.J.; Kallies, K.J.; Baker, M.T. Defining Weight Loss After Bariatric Surgery: A Call for Standardization. Obes. Surg. 2019, 29, 3493–3499. [Google Scholar] [CrossRef]
- Applied Longitudinal Data Analysis: Modeling Change and Event Occurrence—Oxford Scholarship [Internet]. [cité 19 août 2021]. Available online: https://oxford.universitypressscholarship.com/view/10.1093/acprof:oso/9780195152968.001.0001/acprof-9780195152968 (accessed on 29 December 2022).
- Hu, Z.; Sun, J.; Li, R.; Wang, Z.; Ding, H.; Zhu, T.; Wang, G. A Comprehensive Comparison of LRYGB and LSG in Obese Patients Including the Effects on QoL, Comorbidities, Weight Loss, and Complications: A Systematic Review and Meta-Analysis. Obes. Surg. 2019, 30, 819–827. [Google Scholar] [CrossRef]
- Han, Y.; Jia, Y.; Wang, H.; Cao, L.; Zhao, Y. Comparative analysis of weight loss and resolution of comorbidities between laparoscopic sleeve gastrectomy and Roux-en-Y gastric bypass: A systematic review and meta-analysis based on 18 studies. Int. J. Surg. 2020, 76, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Toolabi, K.; Sarkardeh, M.; Vasigh, M.; Golzarand, M.; Vezvaei, P.; Kooshki, J. Comparison of Laparoscopic Roux-en-Y Gastric Bypass and Laparoscopic Sleeve Gastrectomy on Weight Loss, Weight Regain, and Remission of Comorbidities: A 5 Years of Follow-up Study. Obes. Surg. 2019, 30, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Auge, M.; Dejardin, O.; Menahem, B.; Bion, A.L.; Savey, V.; Launoy, G.; Bouvier, V.; Alves, A. Analysis of the Lack of Follow-Up of Bariatric Surgery Patients: Experience of a Reference Center. J. Clin. Med. 2022, 11, 6310. [Google Scholar] [CrossRef] [PubMed]
- Auge, M.; Menahem, B.; Savey, V.; Bion, A.L.; Alves, A. Long-term complications after gastric bypass and sleeve gastrectomy: What information to give to patients and practitioners, and why? J. Visc. Surg. 2022, 159, 298–308. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).