Harnessing the Power of Precision Medicine and Novel Biomarkers to Treat Crohn’s Disease
Abstract
:1. Introduction
2. Current Diagnostic Approaches and Limitations
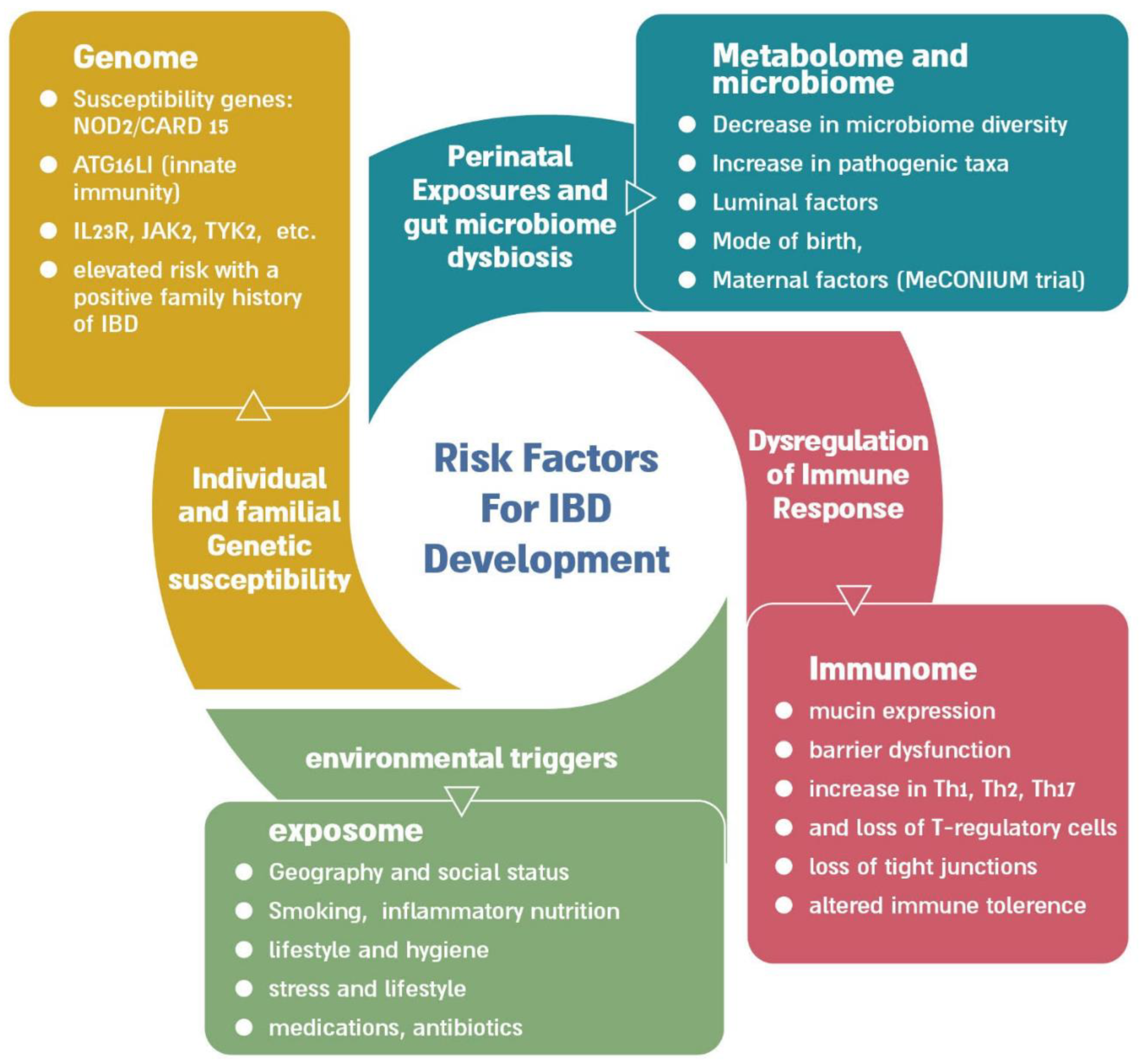
3. IBD Has a Complex and Multifactorial Pathogenesis Which Leads to Heterogeneous Disease Phenotypes
3.1. Genomic and Epigenomic Alterations
3.2. IBD Interactome and Exposome
3.3. The IBD Gut Microbiome
3.3.1. The Chronic Dysbiosis Theory
3.3.2. Unique Characteristics of the Gut Microbiota in IBD Patients Can Predict Disease Phenotypes and Responses to Treatment
4. Exploring New Frontiers in IBD Management
4.1. The Rise of Personalized Medicine: A New Era of Healthcare
Personalized vs. Precision Medicine
4.2. The Era of Disease-Modifying Endpoints
The Search for Novel Biomarkers
5. The Implementation of IBD-Associated Biomarkers Can Be Considered across Various Stages of the Disease
5.1. Diagnostic Biomarkers
5.2. Stratification and Prognostication Biomarkers
5.3. ‘Response to Treatment’ Biomarkers
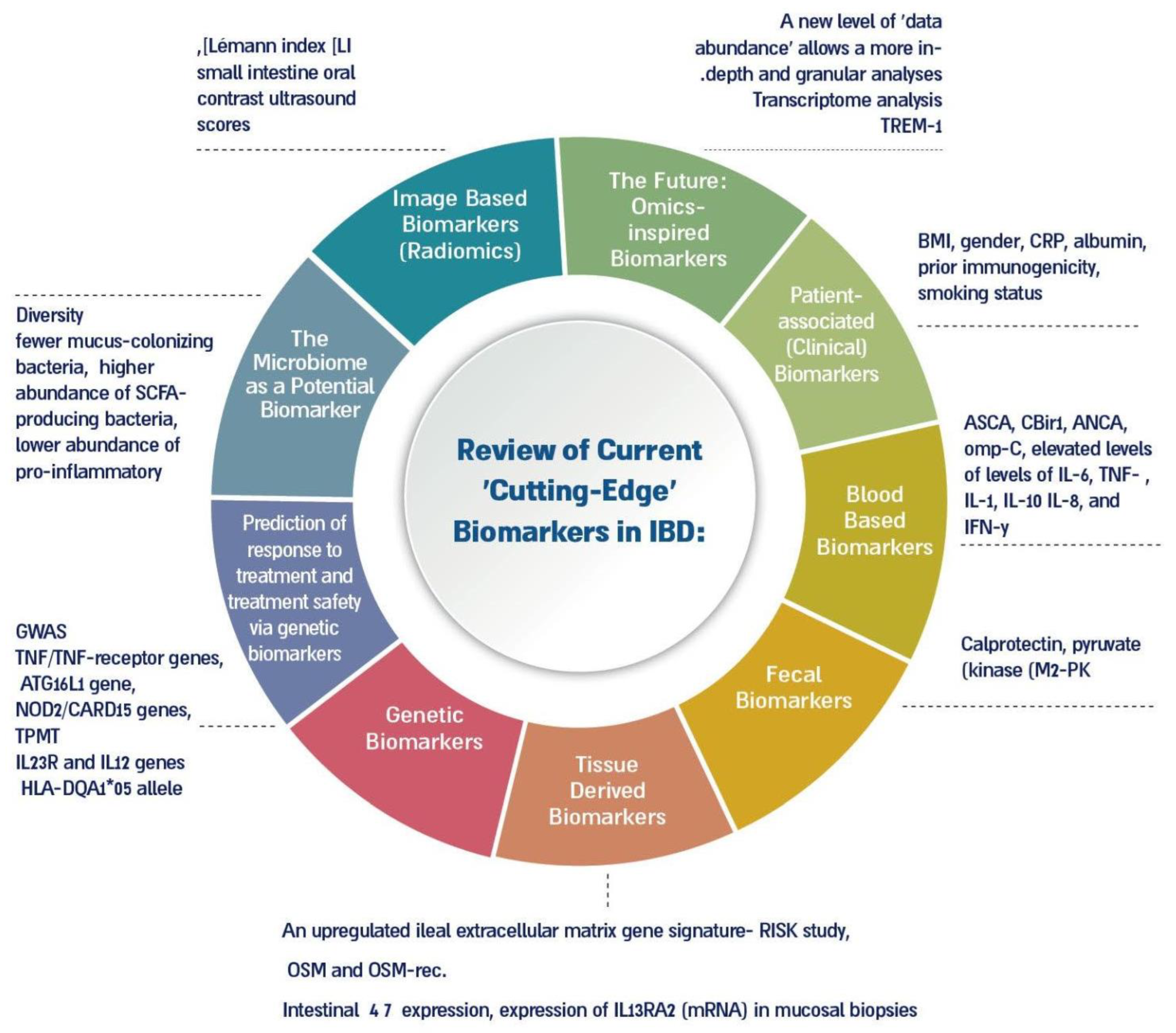
6. Review of Current ‘Cutting-Edge’ Biomarkers in IBD, Consistent with the Origin of the Biomarker
6.1. Patient-Associated (Clinical) Biomarkers
Blood Based Biomarkers
7. Serum Biomarkers Predicting Response to Treatment
7.1. Fecal Biomarkers
7.2. Tissue-Derived Biomarkers
7.2.1. Oncostatin M
7.2.2. Expression of IL13RA2 (mRNA) in Mucosal Biopsies
7.3. Genetic Biomarkers
7.3.1. Genetic Profiling of Biomarkers
7.3.2. HLA-DQA1*05 as a Genetic Predictor of Immunogenicity
7.3.3. Prediction of Safety via Genetic Biomarkers
7.3.4. Prediction of Response to Treatment via Genetic Biomarkers
7.4. The Microbiome as a Potential Biomarker
7.5. Image-Based Biomarkers (Radiomics)
7.6. The Future: Omics-Inspired Biomarkers
Transcriptional Profiling
8. Integrating Biomarkers in Everyday Clinical Practice—Reality or Wishful Thinking
9. Putting the Pieces Together
9.1. Applying Precision Medicine in IBD Care—Uncovering the How, When, and Who
9.2. Summary: Precision Medicine in IBD: The Quest Continues
Funding
Informed Consent Statement
Conflicts of Interest
References
- Xavier, R.J.; Podolsky, D.K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 2007, 448, 427–434. [Google Scholar] [CrossRef]
- Peyrin-Biroulet, L.; Loftus, E.; Colombel, J.-F.; Sandborn, W.J. The natural history of adult Crohn’s disease in population-based cohorts. Am. J. Gastroenterol. 2010, 105, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Cosnes, J.; Cattan, S.; Blain, A.; Beaugerie, L.; Carbonnel, F.; Parc, R.; Gendre, J.-P. Long-term evolution of disease behavior of Crohn’s disease. Inflamm. Bowel Dis. 2002, 8, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Ray, K. IBD: Genotypes and phenotypes of IBD. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 672. [Google Scholar] [CrossRef] [PubMed]
- Denson, L.A.; Curran, M.; McGovern, D.P.B.; Koltun, W.A.; Duerr, R.H.; Kim, S.C.; Sartor, R.B.; Sylvester, F.A.; Abraham, C.; De Zoeten, E.F.; et al. Challenges in IBD Research: Precision Medicine. Inflamm. Bowel Dis. 2019, 25 (Suppl. S2), S31–S39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borg-Bartolo, S.P.; Boyapati, R.K.; Satsangi, J.; Kalla, R. Precision medicine in inflammatory bowel disease: Concept, progress and challenges. F1000Research 2020, 9, 54. [Google Scholar] [CrossRef] [Green Version]
- GBD 2017 Inflammatory Bowel Disease Collaborators. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020, 5, 17–30. [Google Scholar] [CrossRef] [Green Version]
- Marinelli, C.; Savarino, E.; Inferrera, M.; Lorenzon, G.; Rigo, A.; Ghisa, M.; Facchin, S.; D’Incà, R.; Zingone, F. Factors Influencing Disability and Quality of Life during Treatment: A Cross-Sectional Study on IBD Patients. Gastroenterol. Res. Pract. 2019, 2019, 5354320. [Google Scholar] [CrossRef]
- Dulai, P.S.; Peyrin-Biroulet, L.; Danese, S.; Sands, B.E.; Dignass, A.; Turner, D.; Mantzaris, G.; Schölmerich, J.; Mary, J.-Y.; Reinisch, W.; et al. Approaches to Integrating Biomarkers Into Clinical Trials and Care Pathways as Targets for the Treatment of Inflammatory Bowel Diseases. Gastroenterology 2019, 157, 1032–1043.e1. [Google Scholar] [CrossRef] [PubMed]
- De Souza, H.S.; Fiocchi, C. Immunopathogenesis of IBD: Current state of the art. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N.; Bernstein, C.N.; Iliopoulos, D.; MacPherson, A.; Neurath, M.F.; Ali, R.A.R.; Vavricka, S.R.; Fiocchi, C. Environmental triggers in IBD: A review of progress and evidence. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 39–49. [Google Scholar] [CrossRef]
- Peplow, M. The 100,000 Genomes Project. BMJ 2016, 353, i1757. [Google Scholar] [CrossRef] [PubMed]
- Zhernakova, A.; Van Diemen, C.C.; Wijmenga, C. Detecting shared pathogenesis from the shared genetics of immune-related diseases. Nat. Rev. Genet. 2009, 10, 43–55. [Google Scholar] [CrossRef]
- De Souza, H.S.P.; Fiocchi, C.; Iliopoulos, D. The IBD interactome: An integrated view of aetiology, pathogenesis and therapy. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 739–749. [Google Scholar] [CrossRef]
- Zeng, Z.; Mukherjee, A.; Zhang, H. From Genetics to Epigenetics, Roles of Epigenetics in Inflammatory Bowel Disease. Front. Genet. 2019, 10, 1017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cho, J.H.; Brant, S.R. Recent insights into the genetics of inflammatory bowel disease. Gastroenterology 2011, 140, 1704–1712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Honda, K.; Littman, D.R. The microbiome in infectious disease and inflammation. Annu. Rev. Immunol. 2012, 30, 759–795. [Google Scholar] [CrossRef] [Green Version]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomaa, E.Z. Human gut microbiota/microbiome in health and diseases: A review. Antonie Van Leeuwenhoek 2020, 113, 2019–2040. [Google Scholar] [CrossRef]
- Grondin, J.A.; Kwon, Y.H.; Far, P.M.; Haq, S.; Khan, W.I. Mucins in Intestinal Mucosal Defense and Inflammation: Learning From Clinical and Experimental Studies. Front. Immunol. 2020, 11, 2054. [Google Scholar] [CrossRef]
- Michielan, A.; D’incà, R. Intestinal Permeability in Inflammatory Bowel Disease: Pathogenesis, Clinical Evaluation, and Therapy of Leaky Gut. Mediat. Inflamm. 2015, 2015, 628157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lloyd-Price, J.; Arze, C.; Ananthakrishnan, A.N.; Schirmer, M.; Avila-Pacheco, J.; Poon, T.W.; Andrews, E.; Ajami, N.J.; Bonham, K.S.; Brislawn, C.J.; et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature 2019, 569, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Yan, P.G.; Li, J.N. Advances in the understanding of the intestinal micro-environment and inflammatory bowel disease. Chin. Med. J. 2020, 133, 834–841. [Google Scholar] [CrossRef]
- Rutgeerts, P.; Peeters, M.; Hiele, M.; Vantrappen, G.; Pennincx, F.; Aerts, R.; Kerremans, R.; Goboes, K. Effect of faecal stream diversion on recurrence of Crohn’s disease in the neoterminal ileum. Lancet 1991, 338, 771–774. [Google Scholar] [CrossRef]
- Eindor-Abarbanel, A.; Healey, G.R.; Jacobson, K. Therapeutic Advances in Gut Microbiome Modulation in Patients with Inflammatory Bowel Disease from Pediatrics to Adulthood. Int. J. Mol. Sci. 2021, 22, 12506. [Google Scholar] [CrossRef] [PubMed]
- Moayyedi, P.; Surette, M.G.; Kim, P.T.; Libertucci, J.; Wolfe, M.; Onischi, C.; Armstrong, D.; Marshall, J.K.; Kassam, Z.; Reinisch, W.; et al. Fecal Microbiota Transplantation Induces Remission in Patients With Active Ulcerative Colitis in a Randomized Controlled Trial. Gastroenterology 2015, 149, 102–109.e6. [Google Scholar] [CrossRef] [Green Version]
- Paramsothy, S.; Paramsothy, R.; Rubin, D.T.; Kamm, M.A.; Kaakoush, N.O.; Mitchell, H.M.; Castaño-Rodríguez, N. Faecal Microbiota Transplantation for Inflammatory Bowel Disease: A Systematic Review and Meta-analysis. J. Crohn’s Colitis 2017, 11, 1180–1199. [Google Scholar] [CrossRef] [Green Version]
- Doherty, M.K.; Ding, T.; Koumpouras, C.; Telesco, S.E.; Monast, C.; Das, A.; Brodmerkel, C.; Schloss, P.D. Fecal Microbiota Signatures Are Associated with Response to Ustekinumab Therapy among Crohn’s Disease Patients. mBio 2018, 9, e02120-17. [Google Scholar] [CrossRef] [Green Version]
- Dovrolis, N.; Drygiannakis, I.; Filidou, E.; Kandilogiannakis, L.; Arvanitidis, K.; Tentes, I.; Kolios, G.; Valatas, V. Gut Microbial Signatures Underline Complicated Crohn’s Disease but Vary Between Cohorts; An In Silico Approach. Inflamm. Bowel Dis. 2019, 25, 217–225. [Google Scholar] [CrossRef]
- Kostic, A.D. The dynamics of the human infant gut microbiome in development and in progression toward type 1 diabetes. Cell Host Microbe 2015, 17, 260–273. [Google Scholar] [CrossRef] [Green Version]
- Danilova, N.A. Markers of dysbiosis in patients with ulcerative colitis and Crohn’s disease. Ter. Arkhive 2019, 91, 17–24. [Google Scholar] [CrossRef]
- Belizário, J.E.; Faintuch, J. Microbiome and Gut Dysbiosis. Exp. Suppl. 2018, 109, 459–476. [Google Scholar] [CrossRef]
- Collins, F.S.; Varmus, H. A new initiative on precision medicine. N. Engl. J. Med. 2015, 372, 793–795. [Google Scholar] [CrossRef] [Green Version]
- Burstein, H.J. The distinctive nature of HER2-positive breast cancers. N. Engl. J. Med. 2005, 353, 1652–1654. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ginsburg, G.S.; Phillips, K.A. Precision Medicine: From Science To Value. Health Aff. 2018, 37, 694–701. [Google Scholar] [CrossRef]
- Suran, M. Researchers Fill Gaps in Human Genome Sequence. JAMA 2022, 327, 1949. [Google Scholar] [CrossRef] [PubMed]
- Peyrin-Biroulet, L.; Sandborn, W.; Sands, B.E.; Reinisch, W.; Bemelman, W.; Bryant, R.V.; D’Haens, G.; Dotan, I.; Dubinsky, M.; Feagan, B.; et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): Determining Therapeutic Goals for Treat-to-Target. Am. J. Gastroenterol. 2015, 110, 1324–1338. [Google Scholar] [CrossRef]
- Elhag, D.A.; Kumar, M.; Saadaoui, M.; Akobeng, A.K.; Al-Mudahka, F.; Elawad, M.; Al Khodor, S. Inflammatory Bowel Disease Treatments and Predictive Biomarkers of Therapeutic Response. Int. J. Mol. Sci. 2022, 23, 6966. [Google Scholar] [CrossRef]
- Hart, A.L.; Rubin, D.T. Entering the Era of Disease Modification in Inflammatory Bowel Disease. Gastroenterology 2022, 162, 1367–1369. [Google Scholar] [CrossRef]
- Nell, V.P.K.; Machold, K.P.; Eberl, G.; Stamm, T.A.; Uffmann, M.; Smolen, J.S. Benefit of very early referral and very early therapy with disease-modifying anti-rheumatic drugs in patients with early rheumatoid arthritis. Rheumatology 2004, 43, 906–914. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomollón, F.; Dignass, A.; Annese, V.; Tilg, H.; Van Assche, G.; Lindsay, J.O.; Peyrin-Biroulet, L.; Cullen, G.J.; Daperno, M.; Kucharzik, T.; et al. 3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn’s Disease 2016: Part 1: Diagnosis and Medical Management. J. Crohn’s Colitis 2017, 11, 3–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiorino, G.; Danese, S. Diagnostic Delay in Crohn’s Disease: Time for Red Flags. Dig. Dis. Sci. 2016, 61, 3097–3098. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.; Hu, J.; Seki, A.; Eisele, C.; Nair, N.; Huang, R.; Tarassishin, L.; Jharap, B.; Cote-Daigneault, J.; Mao, Q.; et al. Infants born to mothers with IBD present with altered gut microbiome that transfers abnormalities of the adaptive immune system to germ-free mice. Gut 2020, 69, 42–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verstockt, B.; Noor, N.M.; Marigorta, U.M.; Pavlidis, P.; Deepak, P.; Ungaro, R.C.; Fiocchi, C.; Torres, J.; Scharl, M.; Scientific Workshop Steering Committee. Results of the Seventh Scientific Workshop of ECCO: Precision Medicine in IBD-Disease Outcome and Response to Therapy. J. Crohn’s Colitis 2021, 15, 1431–1442. [Google Scholar] [CrossRef] [PubMed]
- Noor, N.M.; Sousa, P.; Paul, S.; Roblin, X. Early Diagnosis, Early Stratification, and Early Intervention to Deliver Precision Medicine in IBD. Inflamm. Bowel Dis. 2022, 28, 1254–1264. [Google Scholar] [CrossRef]
- Lee, S.; Kuenzig, M.E.; Ricciuto, A.; Zhang, Z.; Shim, H.H.; Panaccione, R.; Kaplan, G.G.; Seow, C.H. Smoking May Reduce the Effectiveness of Anti-TNF Therapies to Induce Clinical Response and Remission in Crohn’s Disease: A Systematic Review and Meta-analysis. J. Crohn’s Colitis 2021, 15, 74–87. [Google Scholar] [CrossRef] [PubMed]
- Biasci, D.; Lee, J.C.; Noor, N.M.; Pombal, D.R.; Hou, M.; Lewis, N.; Ahmad, T.; Hart, A.; Parkes, M.; McKinney, E.F.; et al. A blood-based prognostic biomarker in IBD. Gut 2019, 68, 1386–1395. [Google Scholar] [CrossRef] [Green Version]
- Sandborn, W.J. Serologic markers in inflammatory bowel disease: State of the art. Rev. Gastroenterol. Disord. 2004, 4, 167–174. Available online: https://www.ncbi.nlm.nih.gov/pubmed/15580151 (accessed on 20 March 2023).
- Dubinsky, M.C. Emerging Strategies in the Use of IBD-related Serologic Markers. Gastroenterol. Hepatol. 2008, 4, 557–559. Available online: https://www.ncbi.nlm.nih.gov/pubmed/21960935 (accessed on 20 March 2023).
- Billiet, T.; Cleynen, I.; Ballet, V.; Claes, K.; Princen, F.; Singh, S.; Ferrante, M.; Van Assche, G.; Gils, A.; Vermeire, S. Evolution of cytokines and inflammatory biomarkers during infliximab induction therapy and the impact of inflammatory burden on primary response in patients with Crohn’s disease. Scand. J. Gastroenterol. 2017, 52, 1086–1092. [Google Scholar] [CrossRef]
- Parkes, M.; Noor, N.M.; Dowling, F.; Leung, H.; Bond, S.; Whitehead, L.; Upponi, S.; Kinnon, P.; Sandham, A.P.; Lyons, P.A.; et al. Predicting Outcomes For Crohn’s dIsease using a moLecular biomarkEr (PROFILE): Protocol for a multicentre, randomised, biomarker-stratified trial. BMJ Open 2018, 8, e026767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boyapati, R.; Kalla, R.; Satsangi, J.; Ho, G.-T. Biomarkers in Search of Precision Medicine in IBD. Am. J. Gastroenterol. 2016, 111, 1682–1690. [Google Scholar] [CrossRef] [Green Version]
- Sands, B.E. Leukocyte Anti-Trafficking Strategies: Current Status and Future Directions. Dig. Dis. 2017, 35, 13–20. [Google Scholar] [CrossRef]
- Pathirana, W.G.W.; Chubb, S.P.; Gillett, M.J.; Vasikaran, S.D. Faecal Calprotectin. Clin. Biochem. Rev. 2018, 39, 77–90. Available online: https://www.ncbi.nlm.nih.gov/pubmed/30828114 (accessed on 20 March 2023).
- Ho, G.T.; Lee, H.M.; Brydon, G.; Ting, T.; Hare, N.; Drummond, H.; Shand, A.G.; Bartolo, D.C.; Wilson, R.G.; Dunlop, M.G.; et al. Fecal calprotectin predicts the clinical course of acute severe ulcerative colitis. Am. J. Gastroenterol. 2009, 104, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Van Rheenen, P.F.; Van De Vijver, E.; Fidler, V. Faecal calprotectin for screening of patients with suspected inflammatory bowel disease: Diagnostic meta-analysis. BMJ 2010, 341, c3369. [Google Scholar] [CrossRef] [Green Version]
- Turner, D.; Ricciuto, A.; Lewis, A.; D’Amico, F.; Dhaliwal, J.; Griffiths, A.M.; Bettenworth, D.; Sandborn, W.J.; Sands, B.E.; Reinisch, W.; et al. STRIDE-II: An Update on the Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) Initiative of the International Organization for the Study of IBD (IOIBD): Determining Therapeutic Goals for Treat-to-Target strategies in IBD. Gastroenterology 2021, 160, 1570–1583. [Google Scholar] [CrossRef] [PubMed]
- Frin, A.-C.; Filippi, J.; Boschetti, G.; Flourie, B.; Drai, J.; Ferrari, P.; Hebuterne, X.; Nancey, S. Accuracies of fecal calprotectin, lactoferrin, M2-pyruvate kinase, neopterin and zonulin to predict the response to infliximab in ulcerative colitis. Dig. Liver Dis. 2017, 49, 11–16. [Google Scholar] [CrossRef]
- Kugathasan, S.; A Denson, L.; Walters, T.D.; Kim, M.-O.; Marigorta, U.M.; Schirmer, M.; Mondal, K.; Liu, C.; Griffiths, A.; Noe, J.D.; et al. Prediction of complicated disease course for children newly diagnosed with Crohn’s disease: A multicentre inception cohort study. Lancet 2017, 389, 1710–1718. [Google Scholar] [CrossRef] [Green Version]
- West, N.R.; Hegazy, A.N.; Owens, B.M.J.; Bullers, S.J.; Linggi, B.; Buonocore, S.; Coccia, M.; Görtz, D.; This, S.; Stockenhuber, K.; et al. Oncostatin M drives intestinal inflammation and predicts response to tumor necrosis factor-neutralizing therapy in patients with inflammatory bowel disease. Nat. Med. 2017, 23, 579–589. [Google Scholar] [CrossRef] [Green Version]
- Verstockt, S.; Verstockt, B.; Machiels, K.; Vancamelbeke, M.; Ferrante, M.; Cleynen, I.; De Hertogh, G.; Vermeire, S. Oncostatin M Is a Biomarker of Diagnosis, Worse Disease Prognosis, and Therapeutic Nonresponse in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2021, 27, 1564–1575. [Google Scholar] [CrossRef]
- Rath, T.; Billmeier, U.; Ferrazzi, F.; Vieth, M.; Ekici, A.; Neurath, M.F.; Atreya, R. Effects of Anti-Integrin Treatment with Vedolizumab on Immune Pathways and Cytokines in Inflammatory Bowel Diseases. Front. Immunol. 2018, 9, 1700. [Google Scholar] [CrossRef] [Green Version]
- Arijs, I.; Li, K.; Toedter, G.; Quintens, R.; Van Lommel, L.; Van Steen, K.; Leemans, P.; De Hertogh, G.; Lemaire, K.; Ferrante, M.; et al. Mucosal gene signatures to predict response to infliximab in patients with ulcerative colitis. Gut 2009, 58, 1612–1619. [Google Scholar] [CrossRef] [Green Version]
- Verstockt, B.; Verstockt, S.; Creyns, B.; Tops, S.; Van Assche, G.; Gils, A.; Ceuppens, J.L.; Vermeire, S.; Ferrante, M.; Breynaert, C. Mucosal IL13RA2 expression predicts nonresponse to anti-TNF therapy in Crohn’s disease. Aliment. Pharm. 2019, 49, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Cleynen, I. Molecular Profiling of Inflammatory Bowel Disease: Is It Ready for Use in Clinical Decision-Making? Cells 2019, 8, 535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koder, S.; Repnik, K.; Ferkolj, I.; Pernat, C.; Skok, P.; Weersma, R.K.; Potočnik, U. Genetic polymorphism in ATG16L1 gene influences the response to adalimumab in Crohn’s disease patients. Pharmacogenomics 2015, 16, 191–204. [Google Scholar] [CrossRef]
- Garand, M.; Kumar, M.; Huang, S.S.Y.; Al Khodor, S. A literature-based approach for curating gene signatures in multifaceted diseases. J. Transl. Med. 2020, 18, 279. [Google Scholar] [CrossRef]
- Adler, J.; Rangwalla, S.C.; A Dwamena, B.; Higgins, P.D. The prognostic power of the NOD2 genotype for complicated Crohn’s disease: A meta-analysis. Am. J. Gastroenterol. 2011, 106, 699–712. [Google Scholar] [CrossRef]
- Khera, A.V.; Chaffin, M.; Aragam, K.G.; Haas, M.E.; Roselli, C.; Choi, S.H.; Natarajan, P.; Lander, E.S.; Lubitz, S.A.; Ellinor, P.T.; et al. Genome-wide polygenic scores for common diseases identify individuals with risk equivalent to monogenic mutations. Nat. Genet. 2018, 50, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Sazonovs, A.; Kennedy, N.A.; Moutsianas, L.; Heap, G.A.; Rice, D.L.; Reppell, M.; Bewshea, C.M.; Chanchlani, N.; Walker, G.J.; Perry, M.H.; et al. HLA-DQA1*05 Carriage Associated with Development of Anti-Drug Antibodies to Infliximab and Adalimumab in Patients With Crohn’s Disease. Gastroenterology 2020, 158, 189–199. [Google Scholar] [CrossRef] [Green Version]
- A Heap, G.; International Serious Adverse Events Consortium; Weedon, M.N.; Bewshea, C.M.; Singh, A.; Chen, M.; Satchwell, J.B.; Vivian, J.P.; So, K.; Dubois, P.C.; et al. HLA-DQA1–HLA-DRB1 variants confer susceptibility to pancreatitis induced by thiopurine immunosuppressants. Nat. Genet. 2014, 46, 1131–1134. [Google Scholar] [CrossRef]
- Heap, G.; So, K.; Weedon, M.; Edney, N.; Bewshea, C.; Singh, A.; Annese, V.; Beckly, J.; Buurman, D.; Chaudhary, R.; et al. Clinical Features and HLA Association of 5-Aminosalicylate (5-ASA)-induced Nephrotoxicity in Inflammatory Bowel Disease. J. Crohn’s Colitis 2016, 10, 149–158. [Google Scholar] [CrossRef] [Green Version]
- Arijs, I.; Quintens, R.; Van Lommel, L.; Van Steen, K.; De Hertogh, G.; Lemaire, K.; Schraenen, A.; Perrier, C.; Van Assche, G.; Vermeire, S.; et al. Predictive value of epithelial gene expression profiles for response to infliximab in Crohn’s disease. Inflamm. Bowel Dis. 2010, 16, 2090–2098. [Google Scholar] [CrossRef]
- Verstockt, B.; Verstockt, S.; Veny, M.; Dehairs, J.; Arnauts, K.; Van Assche, G.; De Hertogh, G.; Vermeire, S.; Salas, A.; Ferrante, M. Expression Levels of 4 Genes in Colon Tissue Might Be Used to Predict Which Patients Will Enter Endoscopic Remission After Vedolizumab Therapy for Inflammatory Bowel Diseases. Clin. Gastroenterol. Hepatol. 2020, 18, 1142–1151.e10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ananthakrishnan, A.N.; Luo, C.; Yajnik, V.; Khalili, H.; Garber, J.J.; Stevens, B.W.; Cleland, T.; Xavier, R.J. Gut Microbiome Function Predicts Response to Anti-integrin Biologic Therapy in Inflammatory Bowel Diseases. Cell Host Microbe 2017, 21, 603–610.e3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caenepeel, C.; Tabib, N.S.S.; Vieira-Silva, S.; Vermeire, S. Review article: How the intestinal microbiota may reflect disease activity and influence therapeutic outcome in inflammatory bowel disease. Aliment. Pharm. 2020, 52, 1453–1468. [Google Scholar] [CrossRef]
- Pariente, B.; Mary, J.-Y.; Danese, S.; Chowers, Y.; De Cruz, P.; D’Haens, G.; Loftus, E.; Louis, E.; Panes, J.; Schölmerich, J.; et al. Development of the Lémann index to assess digestive tract damage in patients with Crohn’s disease. Gastroenterology 2015, 148, 52–63.e3. [Google Scholar] [CrossRef]
- Calabrese, E.; Zorzi, F.; Zuzzi, S.; Ooka, S.; Onali, S.; Petruzziello, C.; Lasinio, G.J.; Biancone, L.; Rossi, C.; Pallone, F. Development of a numerical index quantitating small bowel damage as detected by ultrasonography in Crohn’s disease. J. Crohn’s Colitis 2012, 6, 852–860. [Google Scholar] [CrossRef]
- Ashton, J.J.; Mossotto, E.; Ennis, S.; Beattie, R.M. Personalising medicine in inflammatory bowel disease-current and future perspectives. Transl. Pediatr. 2019, 8, 56–69. [Google Scholar] [CrossRef]
- Marigorta, U.M.; Denson, L.A.; Hyams, J.S.; Mondal, K.; Prince, J.; Walters, T.D.; Griffiths, A.; Noe, J.D.; Crandall, W.V.; Rosh, J.R.; et al. Transcriptional risk scores link GWAS to eQTLs and predict complications in Crohn’s disease. Nat. Genet. 2017, 49, 1517–1521. [Google Scholar] [CrossRef] [PubMed]
- Verstockt, B.; Verstockt, S.; Blevi, H.; Cleynen, I.; de Bruyn, M.; Van Assche, G.; Vermeire, S.; Ferrante, M. TREM-1, the ideal predictive biomarker for endoscopic healing in anti-TNF-treated Crohn’s disease patients? Gut 2019, 68, 1531–1533. [Google Scholar] [CrossRef]
- Rifai, N.; Gerszten, R.E. Biomarker discovery and validation. Clin. Chem. 2006, 52, 1635–1637. [Google Scholar] [CrossRef]
- Sparrow, M.P.; Papamichael, K.; Ward, M.G.; Riviere, P.; Laharie, D.; Paul, S.; Roblin, X. Therapeutic Drug Monitoring of Biologics During Induction to Prevent Primary Non-Response. J. Crohn’s Colitis 2020, 14, 542–556. [Google Scholar] [CrossRef] [PubMed]
- Colombel, J.F.; Panaccione, R.; Bossuyt, P.; Lukas, M.; Baert, F.; Vaňásek, T.; Danalioglu, A.; Novacek, G.; Armuzzi, A.; Hébuterne, X.; et al. Effect of tight control management on Crohn’s disease (CALM): A multicentre, randomized, controlled phase 3 trial. Lancet 2017, 390, 2779–2789. [Google Scholar] [CrossRef] [PubMed]
- Gaujoux, R.; Starosvetsky, E.; Maimon, N.; Vallania, F.; Bar-Yoseph, H.; Pressman, S.; Weisshof, R.; Goren, I.; Rabinowitz, K.; Waterman, M.; et al. Cell-centred meta-analysis reveals baseline predictors of anti-TNFα non-response in biopsy and blood of patients with IBD. Gut 2019, 68, 604–614. [Google Scholar] [CrossRef] [PubMed]
| Dysbiosis | Reduced microbial diversity and increased abundance of certain bacterial species | [30] |
| Firmicutes/Bacteroidetes ratio | Increased Firmicutes/Bacteroidetes ratio in IBD patients | [31] |
| Bacterial species | Increased abundance of bacterial species, including Escherichiacoli, Enterobacteriaceae, and Fusobacterium, in IBD patients | [31] |
| Viruses | Increased abundance of viruses, including bacteriophages, in IBD patients | [32] |
| Fungi | Increased abundance of fungi, including Candida, in IBD patients | [32] |
| Metabolites | Altered production of metabolites, such as short-chain fatty acids and mucin, in IBD patients | [31] |
| Biomarkers | Description | Exemplars | Leading Studies | References |
|---|---|---|---|---|
| Diagnostic biomarkers | Biomarkers aid in the process of identifying patients at high risk to develop IBD or in early diagnosis of ibd | Genetic mutations, Perinatal exposures, Environmental Exposures Early life microbiome | Meconium Study Gem Project | [10,45,80] |
| Stratification and Prognostication Biomarkers | Separates patients into discrete groups early in the course of the disease in an attempt to support treatment decisions. | Oncostatin M (OSM) and OSM receptor NOD-2 OMP-C ASCA\ANCA | The Genome-wide polygenic risk score | [44,49,51,59,60] |
| A prognostic blood test derived from a gene-expression signature of CD8 + T cells | PROFILE trial | |||
| An ECM-derived signature derived from intestinal biopsies of newly diagnosed pediatric CD patients | The RISK study | |||
| ‘Predicting response to treatment’ biomarkers | Biomarkers predicting response to treatment, preferably specific to the mode of action, aiding in treatment decisions | OSM and OSM receptors serum concentrations of IL22 Smoking status BMI TREM-I | [60,62,73,74,81] | |
| Treatment safety biomarkers | Biomarkers aimed to identify patients at higher risk to develop side effects | TPMT HLA-DQA1*05 | PANTS study | [48,70] |
| Treatment response biomarkers | Noninvasive biomarkers aid in evaluating treatment response, thus reducing the need for invasive assessments | Serum CRP Fecal calprotectin Therapeutic drug monitoring Multi-omics data | CALM study | [37,53,75,82,83,84] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kriger-Sharabi, O.A.; Kopylov, U. Harnessing the Power of Precision Medicine and Novel Biomarkers to Treat Crohn’s Disease. J. Clin. Med. 2023, 12, 2696. https://doi.org/10.3390/jcm12072696
Kriger-Sharabi OA, Kopylov U. Harnessing the Power of Precision Medicine and Novel Biomarkers to Treat Crohn’s Disease. Journal of Clinical Medicine. 2023; 12(7):2696. https://doi.org/10.3390/jcm12072696
Chicago/Turabian StyleKriger-Sharabi, Ofra Aviva, and Uri Kopylov. 2023. "Harnessing the Power of Precision Medicine and Novel Biomarkers to Treat Crohn’s Disease" Journal of Clinical Medicine 12, no. 7: 2696. https://doi.org/10.3390/jcm12072696




