A Real-World Study of Achievement Rate and Predictive Factors of Clinical and Deep Remission to Biologics in Patients with Severe Asthma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Assessments
2.3. Definitions of Clinical and Deep Remissions
2.4. Statistical Analysis
3. Results
3.1. Patient Characteristics at Baseline and after 1-Year Follow-Up of Biologics
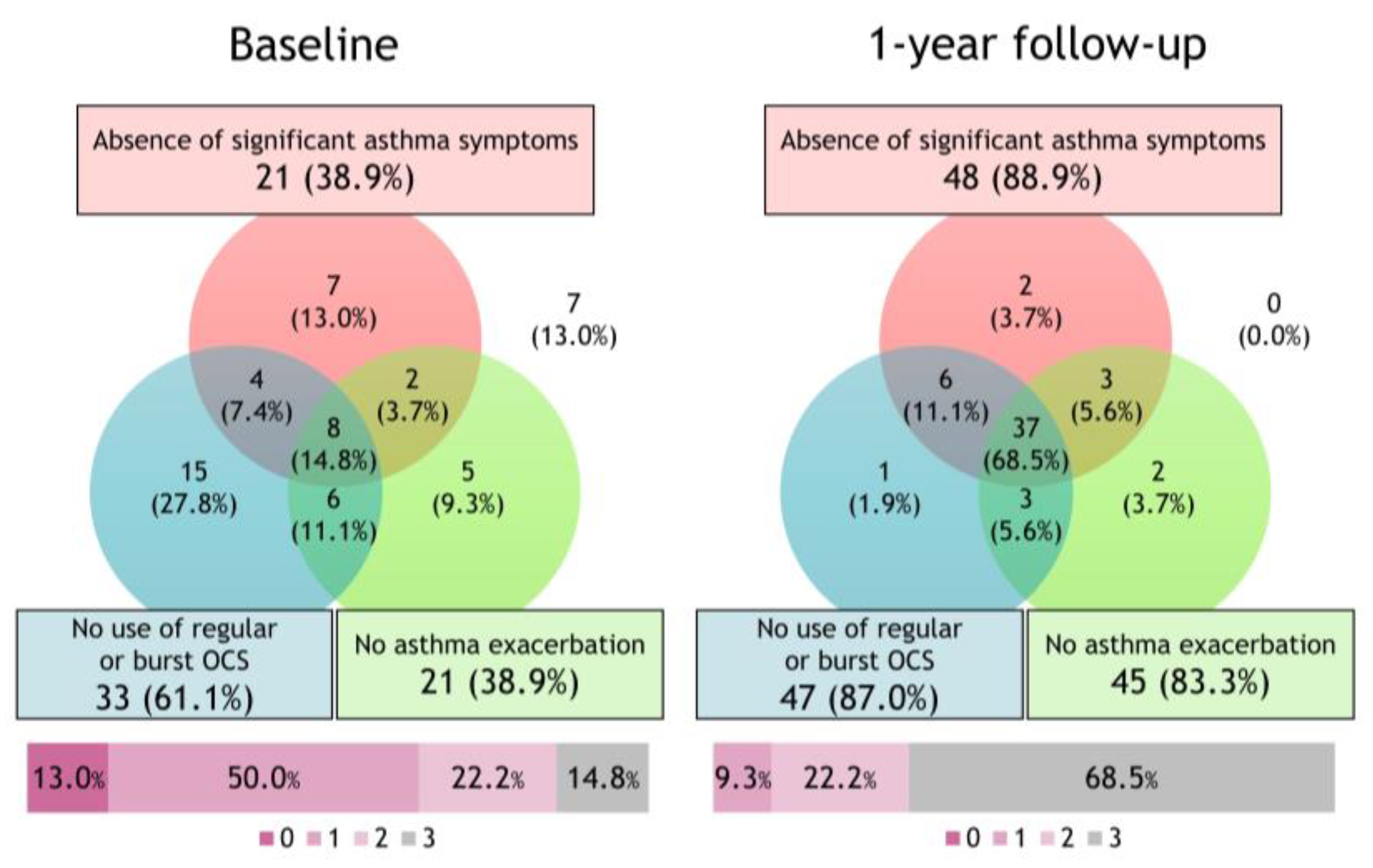
3.2. Changes in the Proportion of Achievement of the Deep Remission Component after Initiating Biologics
3.3. Comparison of Patient Characteristics between the Clinical Remission and Non-Clinical Remission Groups
3.4. Comparison of Patient Characteristics between the Deep Remission and Non-Deep Remission Groups
3.5. Predictors of Clinical and Deep Remission to Biologics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Global Initiative for Asthma. 2022 GINA Report, Global Strategy for Asthma Management and Prevention, Updated 2020. Available online: http://ginasthma.org (accessed on 2 April 2023).
- Chung, K.F.; Wenzel, S.E.; Brozek, J.L.; Bush, A.; Castro, M.; Sterk, P.J.; Adcock, I.M.; Bateman, E.D.; Bel, E.H.; Bleecker, E.R.; et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur. Respir. J. 2014, 43, 343–373. [Google Scholar] [CrossRef] [PubMed]
- Nagase, H.; Adachi, M.; Matsunaga, K.; Yoshida, A.; Okoba, T.; Hayashi, N.; Emoto, K.; Tohda, Y. Prevalence, disease burden, and treatment reality of patients with severe, uncontrolled asthma in Japan. Allergol. Int. 2020, 69, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.J.; Busby, J.; Pfeffer, P.E.; Menzies-Gow, A.; Brown, T.; Gore, R.; Doherty, M.; Mansur, A.H.; Message, S.; Niven, R.; et al. Characterisation of patients with severe asthma in the UK Severe Asthma Registry in the biologic era. Thorax 2021, 76, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, K.; Adachi, M.; Nagase, H.; Okoba, T.; Hayashi, N.; Tohda, Y. Association of low-dosage systemic corticosteroid use with disease burden in asthma. NPJ Prim. Care Respir. Med. 2020, 30, 35. [Google Scholar] [CrossRef] [PubMed]
- Skov, I.R.; Madsen, H.; Henriksen, D.P.; Andersen, J.H.; Pottegård, A.; Davidsen, J.R. Low-dose oral corticosteroids in asthma associates with increased morbidity and mortality. Eur. Respir. J. 2022, 60, 2103054. [Google Scholar] [CrossRef]
- Frøssing, L.; Silberbrandt, A.; Von Bülow, A.; Backer, V.; Porsbjerg, C. The Prevalence of Subtypes of Type 2 Inflammation in an Unselected Population of Patients with Severe Asthma. J. Allergy Clin. Immunol. Pract. 2021, 9, 1267–1275. [Google Scholar] [CrossRef]
- Oishi, K.; Hirano, T.; Matsuda, K.; Hamada, K.; Uehara, S.; Suetake, R.; Yamaji, Y.; Asami-Noyama, M.; Edakuni, N.; Matsunaga, K. The prevalence and clinical characteristics of T2-high and T2-low endotype of severe uncontrolled asthma distinguished by combining popular type-2 biomarkers. Eur. Respir. J. 2019, 54, PA2749. [Google Scholar] [CrossRef]
- Lommatzsch, M.; Brusselle, G.G.; Canonica, G.W.; Jackson, D.J.; Nair, P.; Buhl, R.; Virchow, J.C. Disease-modifying anti-asthmatic drugs. Lancet 2022, 399, 1664–1668. [Google Scholar] [CrossRef]
- Nagase, H.; Suzukawa, M.; Oishi, K.; Matsunaga, K. Biologics for severe asthma: The real-world evidence, effectiveness of switching, and prediction factors for the efficacy. Allergol. Int. 2023, 72, 11–23. [Google Scholar] [CrossRef]
- Schett, G.; Emery, P.; Tanaka, Y.; Burmester, G.; Pisetsky, D.S.; Naredo, E.; Fautrel, B.; van Vollenhoven, R. Tapering biologic and conventional DMARD therapy in rheumatoid arthritis: Current evidence and future directions. Ann. Rheum. Dis. 2016, 75, 1428–1437. [Google Scholar] [CrossRef]
- Upham, J.W.; Le Lievre, C.; Jackson, D.J.; Masoli, M.; Wechsler, M.E.; Price, D.B.; Mansur, A.; Detoraki, A.; Altraja, A.; James, A.; et al. Defining a Severe Asthma Super-Responder: Findings from a Delphi Process. J. Allergy Clin. Immunol. Pract. 2021, 9, 3997–4004. [Google Scholar] [CrossRef] [PubMed]
- Menzies-Gow, A.; Bafadhel, M.; Busse, W.W.; Casale, T.B.; Kocks, J.W.H.; Pavord, I.D.; Szefler, S.J.; Woodruff, P.G.; de Giorgio-Miller, A.; Trudo, F.; et al. An expert consensus framework for asthma remission as a treatment goal. J. Allergy Clin. Immunol. 2020, 145, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Hamada, K.; Oishi, K.; Murata, Y.; Hirano, T.; Matsunaga, K. Feasibility of Discontinuing Biologics in Severe Asthma: An Algorithmic Approach. J. Asthma Allergy 2021, 14, 1463–1471. [Google Scholar] [CrossRef] [PubMed]
- Niimi, A.; Fukunaga, K.; Taniguchi, M.; Nakamura, Y.; Tagaya, E.; Horiguchi, T.; Yokoyama, A.; Yamaguchi, M.; Nagata, M. Executive summary: Japanese guidelines for adult asthma (JGL) 2021. Allergol. Int. 2023, 72, 207–226. [Google Scholar] [CrossRef]
- Ministry of Heath, Labour and Welfare. Ethical Guidelines for Medical and Health Research Involving Human Subjects. Available online: https://www.mhlw.go.jp/file/06-Seisakujouhou-10600000-Daijinkanboukouseikagakuka/0000153339.pdf (accessed on 1 July 2020).
- Matsunaga, K.; Kuwahira, I.; Hanaoka, M.; Saito, J.; Tsuburai, T.; Fukunaga, K.; Matsumoto, H.; Sugiura, H.; Ichinose, M. An official JRS statement: The principles of fractional exhaled nitric oxide (FeNO) measurement and interpretation of the results in clinical practice. Respir. Investig. 2021, 59, 34–52. [Google Scholar] [CrossRef]
- Yuji, M. The Examination Committee of Criteria for ‘Obesity Disease’ in Japan, Japan Society for the Study of Obesity. New Criteria for ‘Obesity Disease’ in Japan. Circ. J. 2002, 66, 987–992. [Google Scholar] [CrossRef]
- Juniper, E.F.; O’Byrne, P.M.; Guyatt, G.H.; Ferrie, P.J.; King, D.R. Development and validation of a questionnaire to measure asthma control. Eur. Respir. J. 1999, 14, 902–907. [Google Scholar] [CrossRef]
- Reddel, H.K.; Taylor, D.R.; Bateman, E.D.; Boulet, L.P.; Boushey, H.A.; Busse, W.W.; Casale, T.B.; Chanez, P.; Enright, P.L.; Gibson, P.G.; et al. An official American Thoracic Society/European Respiratory Society statement: Asthma control and exacerbations: Standardizing endpoints for clinical asthma trials and clinical practice. Am. J. Respir. Crit. Care Med. 2009, 180, 59–99. [Google Scholar] [CrossRef]
- Pavord, I.; Busse, W.; Israel, E.; Szefler, S.; Chen, Z.; Daizadeh, N.; Lederer, D.; Mannent, L.; Amin, N.; Laws, E.; et al. Dupilumab Treatment Leads to Clinical Asthma Remission in Patients With Uncontrolled Moderate-to-Severe Asthma With Type 2 Inflammation. J. Allergy Clin. Immunol. 2021, 147, AB4. [Google Scholar] [CrossRef]
- Menzies-Gow, A.; Hoyte, F.L.; Price, D.B.; Cohen, D.; Barker, P.; Kreindler, J.; Jison, M.; Brooks, C.L.; Papeleu, P.; Katial, R. Clinical Remission in Severe Asthma: A Pooled Post Hoc Analysis of the Patient Journey with Benralizumab. Adv. Ther. 2022, 39, 2065–2084. [Google Scholar] [CrossRef]
- Domingo Ribas, C.; Pavord, I.; Price, R.; Howarth, P.; Oppenheimer, J.; Heaney, L.; Nagase, H.; Pizzichini, E.; Banas Conejero, D.; Gardiner, F. Mepolizumab Treatment Leads to Clinical Remission in Patients with Severe Eosinophilic Asthma: Results from the Real-World REDES Study. In C101. Expanding the Use of Asthma Biologics and Other Novel Therapeutics to Additional Patient Populations; American Thoracic Society: New York, NY, USA, 2022; p. A4834. [Google Scholar]
- Eger, K.; Kroes, J.A.; Brinke, A.T.; Bel, E.H. Long-Term Therapy Response to Anti-IL-5 Biologics in Severe Asthma-A Real-Life Evaluation. J. Allergy Clin. Immunol. Pract. 2021, 9, 1194–1200. [Google Scholar] [CrossRef]
- Shimoda, T.; Odajima, H.; Okamasa, A.; Kawase, M.; Komatsubara, M.; Mayer, B.; Yancey, S.; Ortega, H. Efficacy and safety of mepolizumab in Japanese patients with severe eosinophilic asthma. Allergol. Int. 2017, 66, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Ohta, K.; Adachi, M.; Tohda, Y.; Kamei, T.; Kato, M.; Fitzgerald, J.M.; Takanuma, M.; Kakuno, T.; Imai, N.; Wu, Y.; et al. Efficacy and safety of benralizumab in Japanese patients with severe, uncontrolled eosinophilic asthma. Allergol. Int. 2018, 67, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.; McDonald, V.M.; Pavord, I.D.; Gibson, P.G. Asthma remission- what is it and how can it be achieved? Eur. Respir. J. 2022, 60, 2102583. [Google Scholar] [CrossRef] [PubMed]
- Ajeganova, S.; Huizinga, T. Sustained remission in rheumatoid arthritis: Latest evidence and clinical considerations. Ther. Adv. Musculoskelet. Dis. 2017, 9, 249–262. [Google Scholar] [CrossRef]
- Hirano, T.; Matsunaga, K. Late-onset asthma: Current perspectives. J. Asthma Allergy 2018, 11, 19–27. [Google Scholar] [CrossRef]
- McDonald, V.M.; Fingleton, J.; Agusti, A.; Hiles, S.A.; Clark, V.L.; Holland, A.E.; Marks, G.B.; Bardin, P.P.; Beasley, R.; Pavord, I.D.; et al. Treatable traits: A new paradigm for 21st century management of chronic airway diseases: Treatable Traits Down Under International Workshop report. Eur. Respir. J. 2019, 53, 1802058. [Google Scholar] [CrossRef]
- Melhorn, J.; Howell, I.; Pavord, I.D. Should we apply a treatable traits approach to asthma care? Ann. Allergy Asthma Immunol. 2022, 128, 390–397. [Google Scholar] [CrossRef]
- Buhl, R.; Bel, E.; Bourdin, A.; Dávila, I.; Douglass, J.A.; FitzGerald, J.M.; Jackson, D.J.; Lugogo, N.L.; Matucci, A.; Pavord, I.D.; et al. Effective Management of Severe Asthma with Biologic Medications in Adult Patients: A Literature Review and International Expert Opinion. J. Allergy Clin. Immunol. Pract. 2022, 10, 422–432. [Google Scholar] [CrossRef]
- Menzies-Gow, A.; Corren, J.; Bourdin, A.; Chupp, G.; Israel, E.; Wechsler, M.E.; Brightling, C.E.; Griffiths, J.M.; Hellqvist, Å.; Bowen, K.; et al. Tezepelumab in Adults and Adolescents with Severe, Uncontrolled Asthma. N. Engl. J. Med. 2021, 384, 1800–1809. [Google Scholar] [CrossRef]
- Chikumoto, A.; Oishi, K.; Hamada, K.; Hirano, T.; Kakugawa, T.; Kanesada, K.; Matsunaga, K. Sequential Biotherapy Targeting IL-5 and IL-4/13 in Patients with Eosinophilic Asthma with Sinusitis and Otitis Media. Biomolecules 2022, 12, 522. [Google Scholar] [CrossRef] [PubMed]


| Characteristics | All Patients (n = 54) | |
|---|---|---|
| Baseline | One-Year Follow-Up | |
| Clinical Characteristics | ||
| Age (years) | 61 (48–73) | - |
| Duration of asthma (years) | 18 (4–31) | - |
| Asthma onset ≥ 18 years, n (%) | 42 (77.8) | - |
| Sex: male, n (%) | 28 (51.9) | - |
| Body mass index (kg/m2) | 23.6 (21.2–26.8) | - |
| Obesity, n (%) (BMI ≥ 30 kg/m2) | 20 (37.0) | |
| Former smoker, n (%) | 22 (40.7) | - |
| Type 2 biomarker | ||
| Blood eosinophils (/μL) | 501 (320–938) | 61 (0–219) * |
| FeNO, (ppb) | 64 (37–123) | 32 (20–60) * |
| Atopy | 37 (68.5) | - |
| Total serum IgE (IU/mL) | 256 (76–661) | - |
| Comorbidity | ||
| Allergic rhinitis, n (%) | 34 (63.0) | - |
| Chronic rhinosinusitis, n (%) | 37 (68.5) | - |
| Uncontrolled features | ||
| Exacerbation in the previous year, 0/1/≥2, n (%) | 22/11/21(40.7/20.4/38.9) | 45/7/2 (83.3/13.0/3.7) * |
| ACQ | 1.6 (1.0–2.4) | 0.7 (0.4–1.0) * |
| FEV1 % predicted (%) | 77.0 (51.8–91.5) | 90.8 (73.3–99.0) * |
| Treatment | ||
| Use of ICS/LABA, n (%) | 54 (100.0) | - |
| Use of LAMA, n (%) | 33 (61.1) | - |
| Use of LTRA, n (%) | 29 (53.7) | - |
| Use of maintenance OCS, n (%) | 20 (37.0) | 7 (13.0) * |
| Maintenance OCS dose † (mg/day) | 6.0 (5.0–10.0) | 0.0 (0.0–2.9) * |
| Type of biologics, Oma/Mep/Ben/Dup/Switch, n (%) | 6/4/18/13/13 (11.1/7.4/33.3/24.1/24.1) | - |
| Number of each remission criterion achieved, 0/1/2/3/4/5, n (%) | 4/14/23/11/2/0 (7.4/25.9/42.6/20.4/3.7/0.0) | 0/7/10/3/17/17 (0.0/13.0/18.5/5.6/31.5/31.5) * |
| Characteristics | Clinical Remission (n = 37) | Non-Clinical Remission (n = 17) | p-Value |
|---|---|---|---|
| Clinical characteristics | |||
| Age (years) | 64 (54–73) | 55 (47–73) | 0.333 |
| Duration of asthma (years) | 13 (4–28) | 27 (16–40) | 0.006 |
| Asthma onset ≥ 18 years, n (%) | 32 (86.5) | 10 (58.8) | 0.035 |
| Sex: male, n (%) | 17 (46.0) | 11 (64.7) | 0.249 |
| Body mass index (kg/m2) | 23.6 (20.5–26.1) | 25.0 (20.5–26.1) | 0.208 |
| Obesity | 11 (29.7) | 9 (52.9) | 0.134 |
| Former smoker, n (%) | 10 (27.0) | 12 (70.6) | 0.004 |
| Type 2 biomarker | |||
| Blood eosinophils (/μL) | 503 (372–909) | 478 (216–1153) | 0.635 |
| FeNO (ppb) | 67 (33–133) | 56 (42–101) | 0.802 |
| Atopy | 27 (73.0) | 10 (58.8) | 0.353 |
| Total serum IgE (IU/mL) | 347 (81–662) | 199 (70–675) | 0.635 |
| Comorbidity | |||
| Allergic rhinitis, n (%) | 24 (64.9) | 10 (58.8) | 0.765 |
| Chronic sinusitis, n (%) | 25 (67.6) | 12 (70.6) | 1.000 |
| Uncontrolled features | |||
| Exacerbation in the previous year, 0/1/≥2, n (%) | 17/8/12 (46.0/21.6/32.4) | 5/3/9 (29.4/17.7/52.9) | 0.140 |
| ACQ | 1.6 (1.0–2.2) | 2.4 (1.2–2.4) | 0.011 |
| FEV1 % predicted (%) | 91.5 (78.0–96.4) | 71.5 (49.4–84.2) | 0.0005 |
| Treatment | |||
| Use of ICS/LABA, n (%) | 17 (100.0) | 37 (100.0) | |
| Use of LAMA, n (%) | 21 (56.8) | 12 (70.6) | 0.383 |
| Use of maintenance OCS, n (%) | 10 (27.0) | 10 (58.8) | 0.035 |
| Maintenance OCS dose † (mg/day) | 8.0 (5.0–11.3) | 5.5 (5.0–10.0) | 0.501 |
| Type of biologics, Oma/Mep/Ben/Dup/Switch, n (%) | 4/1/14/11/7 (10.8/2.7/37.8/29.7/18.9) | 6/4/12/6/9 (16.2/10.8/32.4/16.2/24.3) | 0.131 |
| Characteristics | Deep Remission (n = 17) | Non-Deep Remission (n = 37) | p-Value |
|---|---|---|---|
| Clinical characteristics | |||
| Age (years) | 62 (52–72) | 61 (48–73) | 0.926 |
| Duration of asthma (years) | 5 (2–23) | 19 (10–36) | 0.006 |
| Asthma onset ≥ 18 years, n (%) | 16 (94.1) | 26 (70.3) | 0.078 |
| Sex: male, n (%) | 6 (35.3) | 22 (59.5) | 0.144 |
| Body mass index (kg/m2) | 23.6 (20.0–25.5) | 23.6 (21.4–28.0) | 0.412 |
| Obesity | 4 (23.5) | 16 (43.2) | 0.229 |
| Former smoker, n (%) | 5 (29.4) | 17 (46.0) | 0.372 |
| Type 2 biomarker | |||
| Blood eosinophils (/μL) | 580 (430–893) | 478 (314–953) | 0.301 |
| FeNO (ppb) | 43 (19–96) | 67 (44–143) | 0.053 |
| Atopy | 9 (52.9) | 28 (75.7) | 0.121 |
| Total serum IgE (IU/mL) | 219 (74–659) | 283 (75–675) | 0.752 |
| Comorbidity | |||
| Allergic rhinitis, n (%) | 11 (64.7) | 23 (62.2) | 1.000 |
| Chronic sinusitis, n (%) | 12 (70.6) | 25 (67.6) | 1.000 |
| Uncontrolled features | |||
| Exacerbation in the previous year, 0/1/≥2, n (%) | 7/4/6 (41.2/23.5/35.3) | 15/7/15 (40.5/18.9/40.5) | 0.791 |
| ACQ | 1.6 (1.1–2.4) | 1.6 (1.0–2.4) | 0.501 |
| FEV1 % predicted (%) | 91.5 (78.0–96.4) | 71.5 (49.4–84.2) | 0.0005 |
| Treatment | |||
| Use of ICS/LABA, n (%) | 17 (100.0) | 37 (100.0) | |
| Use of LAMA, n (%) | 7 (41.2) | 26 (70.3) | 0.070 |
| Use of maintenance OCS, n (%) | 5 (29.4) | 15 (40.5) | 0.549 |
| Maintenance OCS dose † (mg/day) | 10.0 (5.5–12.5) | 5.0 (5.0–10.0) | 0.217 |
| Type of biologics, Oma/Mep/Ben/Dup/Switch, n (%) | 0/0/6/7/4 (0.0/0.0/35.3/41.2/23.5) | 6/4/12/6/9 (16.2/10.8/32.4/16.2/24.3) | 0.106 |
| Number of each remission criterion achieved | 2 (1.5–3) | 2 (1–2) | 0.293 |
| Predictor | OR (95% CI) | p-Value |
|---|---|---|
| Asthma duration ≤ 5 years | 10.91 (1.30–91.27) | 0.028 |
| FEV1 ≥ 75% predicted | 3.38 (1.017–11.26) | 0.047 |
| Predictor | OR (95% CI) | p-Value |
|---|---|---|
| Asthma duration ≤ 5 years | 7.38 (2.01–27.16) | 0.0026 |
| FEV1 ≥ 75% predicted | 26.29 (3.13–220.47) | 0.0026 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oishi, K.; Hamada, K.; Murata, Y.; Matsuda, K.; Ohata, S.; Yamaji, Y.; Asami-Noyama, M.; Edakuni, N.; Kakugawa, T.; Hirano, T.; et al. A Real-World Study of Achievement Rate and Predictive Factors of Clinical and Deep Remission to Biologics in Patients with Severe Asthma. J. Clin. Med. 2023, 12, 2900. https://doi.org/10.3390/jcm12082900
Oishi K, Hamada K, Murata Y, Matsuda K, Ohata S, Yamaji Y, Asami-Noyama M, Edakuni N, Kakugawa T, Hirano T, et al. A Real-World Study of Achievement Rate and Predictive Factors of Clinical and Deep Remission to Biologics in Patients with Severe Asthma. Journal of Clinical Medicine. 2023; 12(8):2900. https://doi.org/10.3390/jcm12082900
Chicago/Turabian StyleOishi, Keiji, Kazuki Hamada, Yoriyuki Murata, Kazuki Matsuda, Syuichiro Ohata, Yoshikazu Yamaji, Maki Asami-Noyama, Nobutaka Edakuni, Tomoyuki Kakugawa, Tsunahiko Hirano, and et al. 2023. "A Real-World Study of Achievement Rate and Predictive Factors of Clinical and Deep Remission to Biologics in Patients with Severe Asthma" Journal of Clinical Medicine 12, no. 8: 2900. https://doi.org/10.3390/jcm12082900





