Combination of Supramicrosurgical Lymphatico-Venular Anastomosis (sLVA) and Lymph-Sparing Liposuction in Treating Cancer-Related Lymphedema: Rationale for a Regional One-Stage Approach
Abstract
:1. Introduction
2. Materials and Methods
2.1. Treatment
2.2. Follow-Up
2.3. Data Analysis
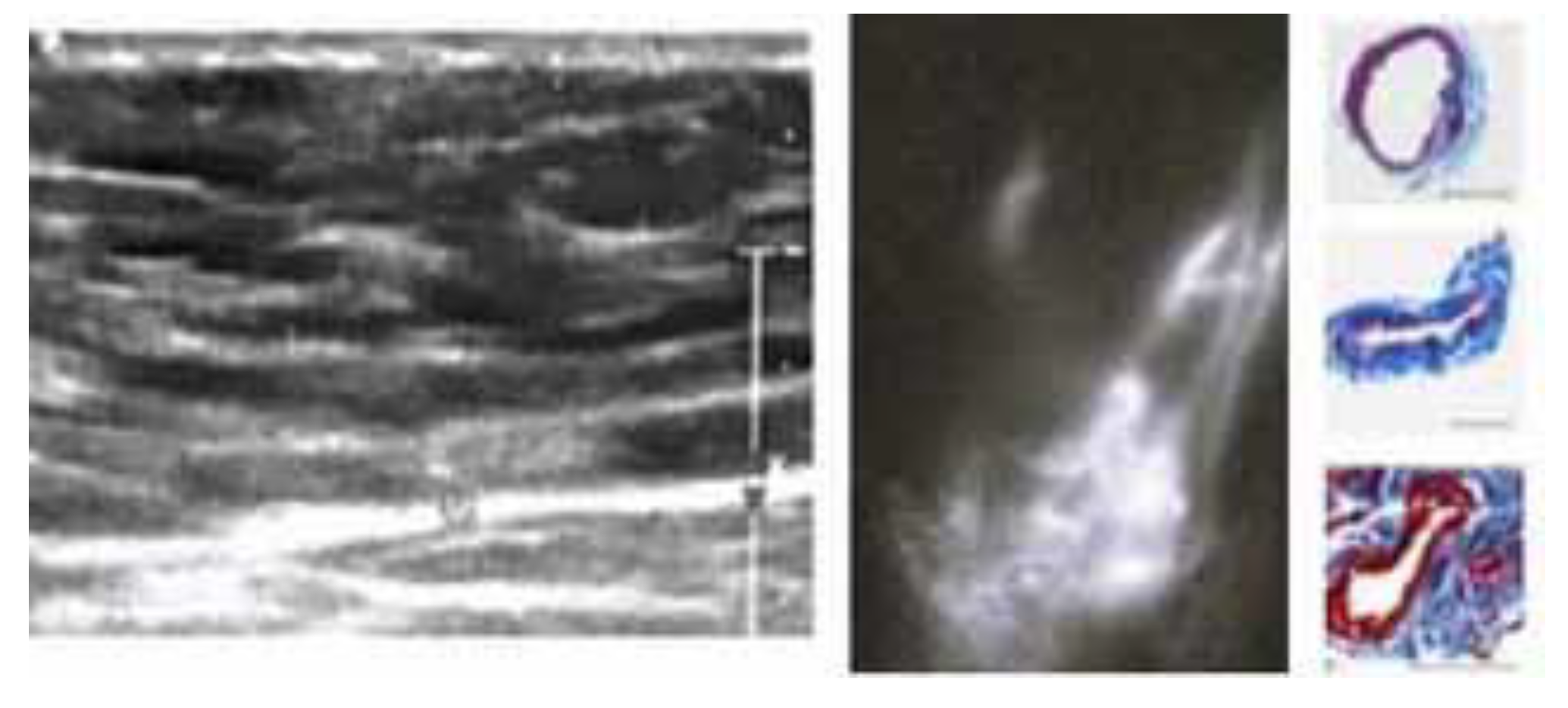
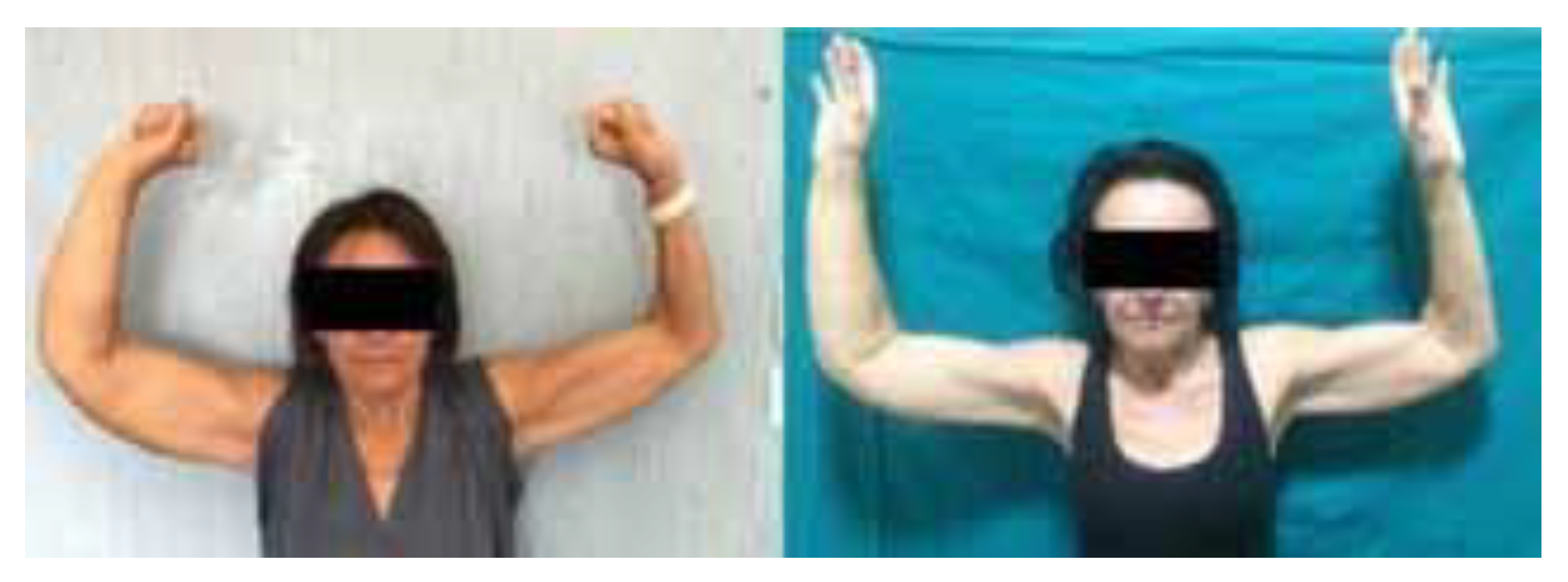
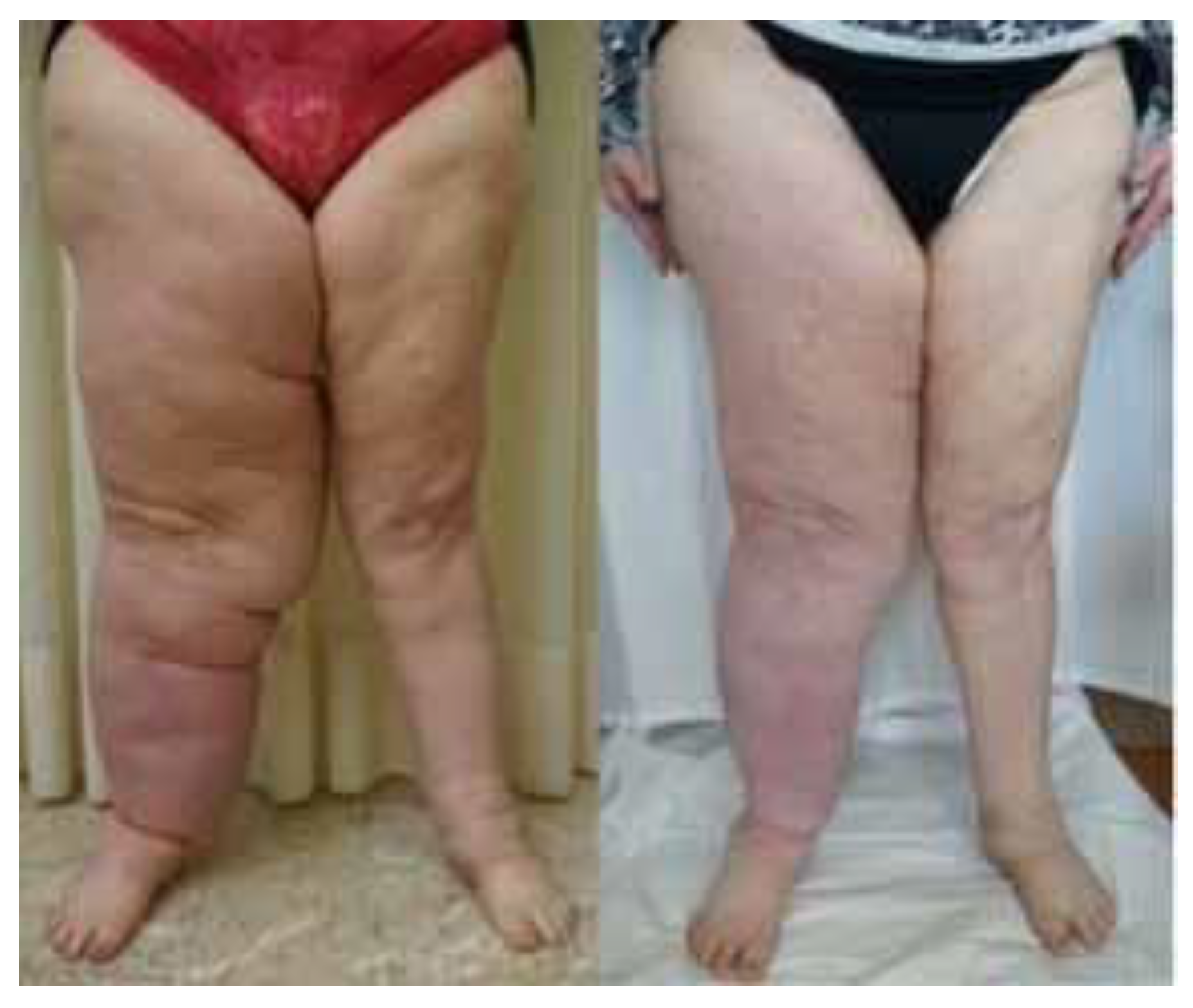
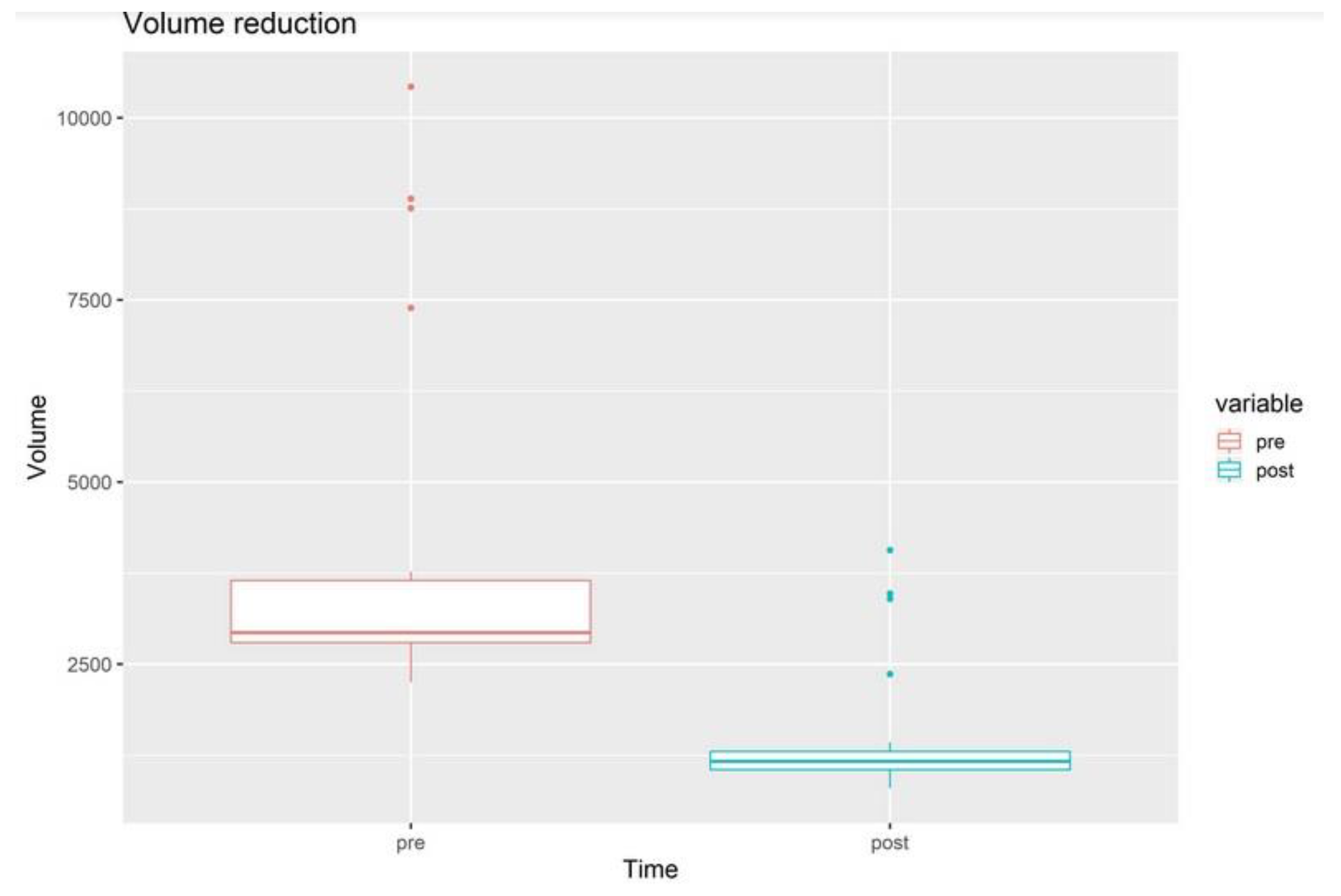
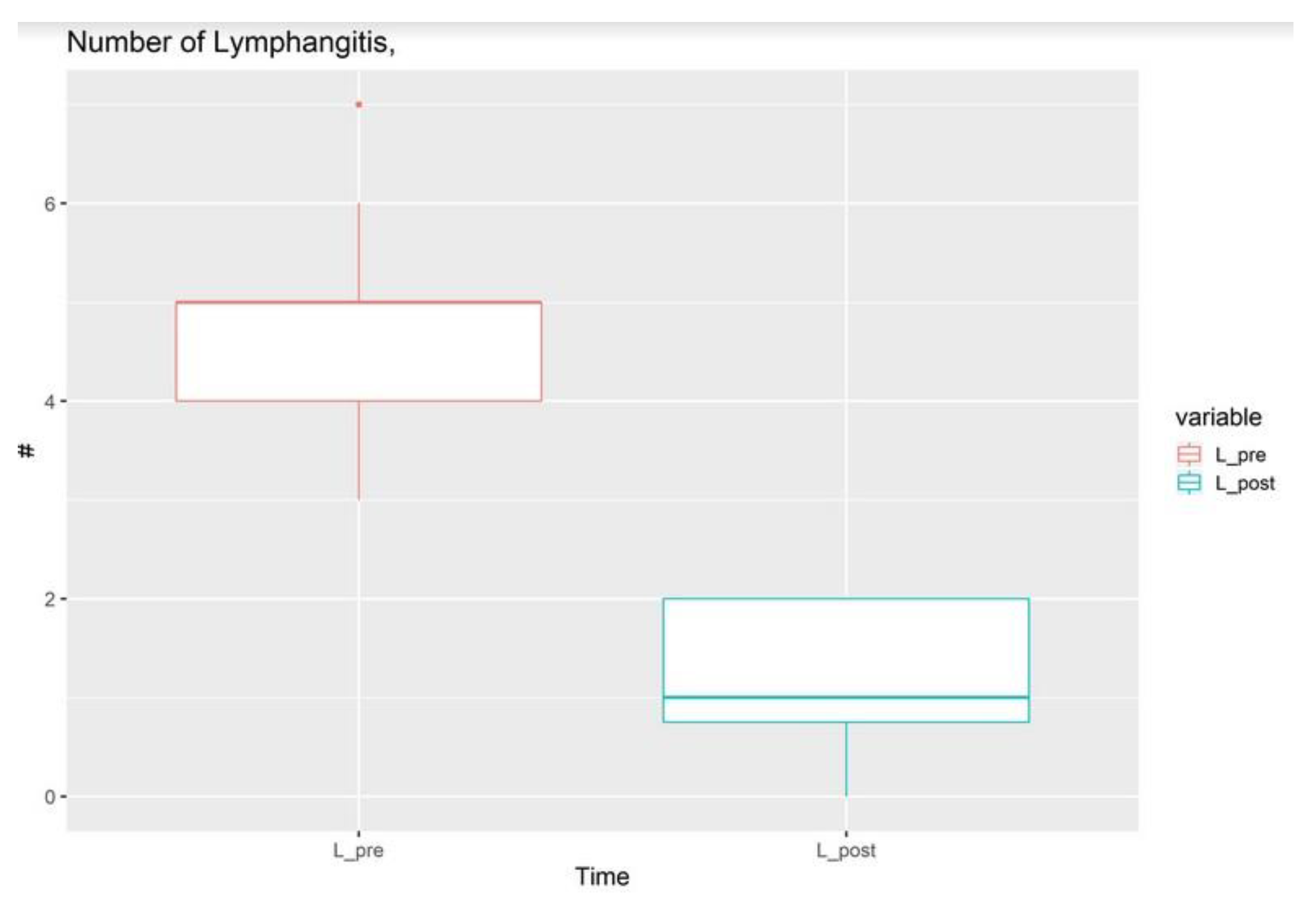
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: 2013 Consensus Document of the International Society of Lymphology. Lymphology 2013, 46, 1–11. [Google Scholar] [PubMed]
- Huang, T.W.; Kuo, K.N.; Chen, K.H.; Chen, C.; Hou, W.H.; Lee, W.H.; Chao, T.Y.; Tsai, J.T.; Su, C.M.; Huang, M.T.; et al. Recommendation for axillary lymph node dissection in women with early breast cancer and sentinel node metastasis: A systematic review and meta-analysis of randomized controlled trials using the GRADE system. Int. J. Surg. 2016, 34, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Alagkiozidis, I.; Weedon, J.; Grossman, A.; Wang, P.; Mize, B.; Wilson, K.; Shah, T.; Economos, K.; Salame, G.; Abulafia, O.; et al. Extent of lymph node dissection and overall survival in patients with uterine carcinosarcoma, papillary serous and endometrioid adenocarcinoma: A retrospective cohort study. Int. J. Surg. 2015, 24 Pt A, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Weber, E.; Aglianò, M.; Bertelli, E.; Gabriele, G.; Gennaro, P.; Barone, V. Lymphatic Collecting Vessels in Health and Disease: A Review of Histopathological Modifications in Lymphedema. Lymphat. Res. Biol. 2022, 20, 468–477. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yamamoto, T.; Yoshimatsu, H.; Yamamoto, N. Complete lymph flow reconstruction: A free vascularized lymph node true perforator flap transfer with efferent lymphaticolymphatic anastomosis. J. Plast. Reconstr. Aesthet. Surg. 2016, 69, 1227–1233. [Google Scholar] [CrossRef] [PubMed]
- Park, J.K.; Seo, J.; Yang, E.J.; Kang, Y.; Heo, C.Y.; Myung, Y. Association of lymphatic flow velocity with surgical outcomes in patients undergoing lymphovenous anastomosis for breast cancer-related lymphedema. Breast Cancer 2022, 29, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Hoskin, T.L.; Habermann, E.B.; Cheville, A.L.; Boughey, J.C. Breast Cancer-Related Lymphedema Risk is Related to Multidisciplinary Treatment and Not Surgery Alone: Results from a Large Cohort Study. Ann. Surg. Oncol. 2017, 24, 2972–2980. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Schaverien, M.V.; Badash, I.; Patel, K.M.; Selber, J.C.; Cheng, M.H. Vascularized Lymph Node Transfer for Lymphedema. Semin. Plast. Surg. 2018, 32, 28–35. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, W.F.; Zeng, W.F.; Hawkes, P.J.; Man, J.; Bowen, M. Lymphedema Liposuction with Immediate Limb Contouring. Plast. Reconstr. Surg. Glob. Open 2019, 12, e2513. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Brorson, H.; Ohlin, K.; Olsson, G.; Långström, G.; Wiklund, I.; Svensson, H. Quality of life following liposuction and conservative treatment of arm lymphedema. Lymphology 2006, 39, 8–25. [Google Scholar] [PubMed]
- Mihara, M.; Hara, H.; Hayashi, Y.; Narushima, M.; Yamamoto, T.; Todokoro, T.; Iida, T.; Sawamoto, N.; Araki, J.; Kikuchi, K.; et al. Pathological steps of cancer-related lymphedema: Histological changes in the collecting lymphatic vessels after lymphadenectomy. PLoS ONE 2012, 7, e41126, Erratum in PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ciudad, P.; Manrique, O.J.; Bustos, S.S.; Agko, M.; Huang, T.C.; Vizcarra, L.; Nuñez, M.L.; Lo Torto, F.; Forte, A.J. Single-stage VASER-assisted liposuction and lymphatico-venous anastomoses for the treatment of extremity lymphedema: A case series and systematic review of the literature. Gland Surg. 2020, 9, 545–557. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Brazio, P.S.; Nguyen, D.H. Combined Liposuction and Physiologic Treatment Achieves Durable Limb Volume Normalization in Class II-III Lymphedema: A Treatment Algorithm to Optimize Outcomes. Ann. Plast. Surg. 2021, 86 (Suppl. S3), S384–S389. [Google Scholar] [CrossRef] [PubMed]
- Chromy, A.; Zalud, L.; Dobsak, P.; Suskevic, I.; Mrkvicova, V. Limb volume measurements: Comparison of accuracy and decisive parameters of the most used present methods. Springerplus 2015, 4, 707. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yamamoto, T.; Matsuda, N.; Doi, K.; Oshima, A.; Yoshimatsu, H.; Todokoro, T.; Ogata, F.; Mihara, M.; Narushima, M.; Iida, T.; et al. The earliest finding of indocyanine green lymphography in asymptomatic limbs of lower extremity lymphedema patients secondary to cancer treatment: The modified dermal backflow stage and concept of subclinical lymphedema. Plast. Reconstr. Surg. 2011, 128, 314e–321e. [Google Scholar] [CrossRef] [PubMed]
- Deptula, P.; Zhou, A.; Posternak, V.; He, H.; Nguyen, D. Multimodality Approach to Lymphedema Surgery Achieves and Maintains Normal Limb Volumes: A Treatment Algorithm to Optimize Outcomes. J. Clin. Med. 2022, 11, 598. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lasso, J.M.; Alonso-Farto, J.C. Indocyanine green-guided liposuction for patients presenting with residual nonpitting edema after lymphovenous anastomosis. J. Plast. Reconstr. Aesthet. Surg. 2022, 75, 2482–2492. [Google Scholar] [CrossRef] [PubMed]
- Agha, R.; Abdall-Razak, A.; Crossley, E.; Dowlut, N.; Iosifidis, C.; Mathew, G.; STROCSS Group. STROCSS 2019 Guideline: Strengthening the reporting of cohort studies in surgery. Int. J. Surg. 2019, 72, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, M.G.; Toyserkani, N.M.; Hansen, F.C.G.; Thomsen, J.B.; Sørensen, J.A. Prospective Validation of Indocyanine Green Lymphangiography Staging of Breast Cancer-Related Lymphedema. Cancers 2021, 13, 1540. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Weiss, J.; Daniel, T. Validation of the lymphedema life impact scale (llis): A condition-specific measurement tool for persons with lymphedema. Lymphology 2015, 48, 128–138. [Google Scholar] [PubMed]
- Gennaro, P.; Gabriele, G.; Mihara, M.; Kikuchi, K.; Salini, C.; Aboh, I.; Cascino, F.; Chisci, G.; Ungari, C. Supramicrosurgical lymphatico-venular anastomosis (LVA) in treating lymphoedema: 36-months preliminary report. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 4642–4653. [Google Scholar] [PubMed]
- Kwon, J.G.; Kim, Y.; Jang, M.Y.; Suh, H.P.; Pak, C.J.; Keeley, V.; Jeon, J.Y.; Hong, J.P. The Quality of Life after Lymphaticovenous Anastomosis in 118 Lower Limb Lymphedema Patients. Arch. Plast. Surg. 2023, 50, 514–522. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Borman, P.; Yaman, A.; Denizli, M.; Karahan, S.; Özdemir, O. The reliability and validity of Lymphedema Quality of Life Questionnaire-Arm in Turkish patients with upper limb lymphedema related with breast cancer. Turk. J. Phys. Med. Rehabil. 2018, 64, 205–212. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, W.F.; Yamamoto, T.; Fisher, M.; Liao, J.; Carr, J. The “Octopus” Lymphaticovenular Anastomosis: Evolving Beyond the Standard Supermicrosurgical Technique. J. Reconstr. Microsurg. 2015, 31, 450–457. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, S.A.; Staley, A.C.; Vicini, F.; Thiruchelvam, P.; Hutchison, N.A.; Mendez, J.; MacNeill, F.; Rockson, S.G.; DeSnyder, S.M.; Klimberg, S.; et al. Considerations for Clinicians in the Diagnosis, Prevention, and Treatment of Breast Cancer-Related Lymphedema: Recommendations from a Multidisciplinary Expert ASBrS Panel: Part 1: Definitions, Assessments, Education, and Future Directions. Ann. Surg. Oncol. 2017, 24, 2818–2826. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, S.A.; DeSnyder, S.M.; Klimberg, S.; Alatriste, M.; Boccardo, F.; Smith, M.L.; Staley, A.C.; Thiruchelvam, P.T.R.; Hutchison, N.A.; Mendez, J.; et al. Considerations for Clinicians in the Diagnosis, Prevention, and Treatment of Breast Cancer-Related Lymphedema, Recommendations from an Expert Panel: Part 2: Preventive and Therapeutic Options. Ann. Surg. Oncol. 2017, 24, 2827–2835. [Google Scholar] [CrossRef] [PubMed]
- Sleigh, B.C.; Manna, B. Lymphedema; Stat Pearls Publishing: St. Petersburg, FL, USA, 2020. [Google Scholar]
- Cheng, H.; Luan, J.; Mu, D.; Wang, Q.; Qi, J.; Li, Z.; Fu, S. M1/M2 Macrophages Play Different Roles in Adipogenic Differentiation of PDGFRα+ Preadipocytes In Vitro. Aesthetic. Plast. Surg. 2019, 43, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Kataru, R.P.; Mehrara, B.J. Histopathologic Features of Lymphedema: A Molecular Review. Int. J. Mol. Sci. 2020, 21, 2546. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gennaro, P.; Chisci, G.; Mazzei, F.; Gabriele, G. Magnetic resonance lymphangiography: How to prove it? J. Magn. Reson. Imaging 2016, 44, 509–510. [Google Scholar] [CrossRef] [PubMed]
- Barone, V.; Borghini, A.; Tedone Clemente, E.; Aglianò, M.; Gabriele, G.; Gennaro, P.; Weber, E. New Insights into the Pathophysiology of Primary and Secondary Lymphedema: Histopathological Studies on Human Lymphatic Collecting Vessels. Lymphat. Res. Biol. 2020, 18, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Brorson, H. Liposuction in Lymphedema Treatment. J. Reconstr. Microsurg. 2016, 32, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Gennaro, P.; Gabriele, G.; Salini, C.; Chisci, G.; Cascino, F.; Xu, J.F.; Ungari, C. Our supramicrosurgical experience of lymphaticovenular anastomosis in lymphoedema patients to prevent cellulitis. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 674–679. [Google Scholar] [PubMed]
- Sood, J.; Jayaraman, L.; Sethi, N. Liposuction: Anaesthesia challenges. Indian J. Anaesth. 2011, 55, 220–227. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Park, K.E.; Allam, O.; Chandler, L.; Mozzafari, M.A.; Ly, C.; Lu, X.; Persing, J.A. Surgical management of lymphedema: A review of current literature. Gland Surg. 2020, 9, 503–511. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Campisi, C.C.; Ryan, M.; Boccardo, F.; Campisi, C. Fibro-Lipo-Lymph-Aspiration with a Lymph Vessel Sparing Procedure to Treat Advanced Lymphedema after Multiple Lymphatic-Venous Anastomoses: The Complete Treatment Protocol. Ann. Plast. Surg. 2017, 78, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Ly, C.L.; Kataru, R.P.; Mehrara, B.J. Inflammatory Manifestations of Lymphedema. Int. J. Mol. Sci. 2017, 18, 171. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Grada, A.A.; Phillips, T.J. Lymphedema: Pathophysiology and clinical manifestations. J. Am. Acad. Dermatol. 2017, 77, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.H.; Loh, C.Y.Y.; Lin, C.Y. Outcomes of Vascularized Lymph Node Transfer and Lymphovenous Anastomosis for Treatment of Primary Lymphedema. Plast. Reconstr. Surg. Glob. Open 2018, 6, e2056. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Losco, L.; Bolletta, A.; de Sire, A.; Chen, S.H.; Sert, G.; Aksoyler, D.; Velazquez-Mujica, J.; Invernizzi, M.; Cigna, E.; Chen, H.C. The Combination of Lymph Node Transfer and Excisional Procedures in Bilateral Lower Extremity Lymphedema: Clinical Outcomes and Quality of Life Assessment with Long-Term Follow-Up. J. Clin. Med. 2022, 11, 570. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gabriele, G.; Nigri, A.; Chisci, G.; Massarelli, O.; Cascino, F.; Komorowska-Timek, E.; Kazuki, K.; Hara, H.; Mihara, M.; Gennaro, P. Combination of Supramicrosurgical Lymphatico-Venular Anastomosis (sLVA) and Lymph-Sparing Liposuction in Treating Cancer-Related Lymphedema: Rationale for a Regional One-Stage Approach. J. Clin. Med. 2024, 13, 2872. https://doi.org/10.3390/jcm13102872
Gabriele G, Nigri A, Chisci G, Massarelli O, Cascino F, Komorowska-Timek E, Kazuki K, Hara H, Mihara M, Gennaro P. Combination of Supramicrosurgical Lymphatico-Venular Anastomosis (sLVA) and Lymph-Sparing Liposuction in Treating Cancer-Related Lymphedema: Rationale for a Regional One-Stage Approach. Journal of Clinical Medicine. 2024; 13(10):2872. https://doi.org/10.3390/jcm13102872
Chicago/Turabian StyleGabriele, Guido, Andrea Nigri, Glauco Chisci, Olindo Massarelli, Flavia Cascino, Ewa Komorowska-Timek, Kikuchi Kazuki, Hisako Hara, Makoto Mihara, and Paolo Gennaro. 2024. "Combination of Supramicrosurgical Lymphatico-Venular Anastomosis (sLVA) and Lymph-Sparing Liposuction in Treating Cancer-Related Lymphedema: Rationale for a Regional One-Stage Approach" Journal of Clinical Medicine 13, no. 10: 2872. https://doi.org/10.3390/jcm13102872
APA StyleGabriele, G., Nigri, A., Chisci, G., Massarelli, O., Cascino, F., Komorowska-Timek, E., Kazuki, K., Hara, H., Mihara, M., & Gennaro, P. (2024). Combination of Supramicrosurgical Lymphatico-Venular Anastomosis (sLVA) and Lymph-Sparing Liposuction in Treating Cancer-Related Lymphedema: Rationale for a Regional One-Stage Approach. Journal of Clinical Medicine, 13(10), 2872. https://doi.org/10.3390/jcm13102872