The Possible Effect of β-Blocker Use on the Circulating MMP-2/TIMP-2 System in Patients with Chronic Kidney Disease on Conservative Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Laboratory Methods
2.3. Statistical Analysis
3. Results
3.1. The Characteristics of Patients with CKD Treated and Not Treated with β-Blockers
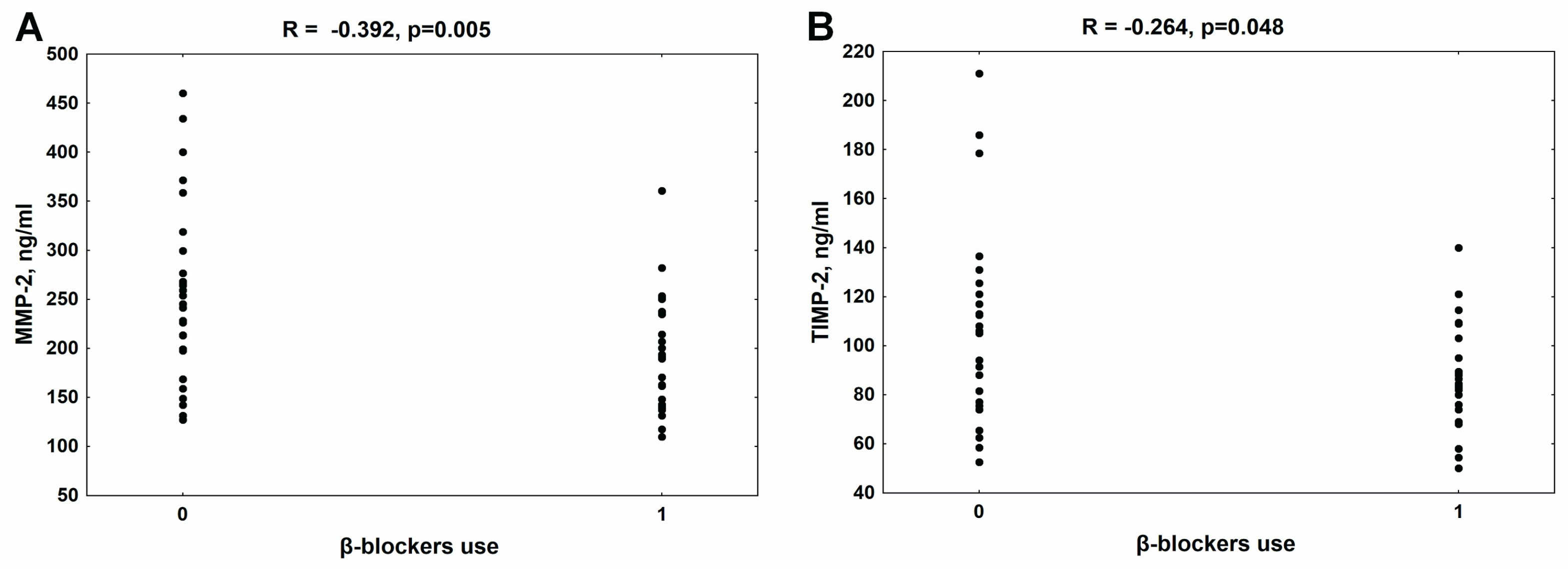
3.2. The Relationship between the Use of β-Blockers and MMP-2 and TIMP-2 Concentrations in the Whole Group of Patients with CKD
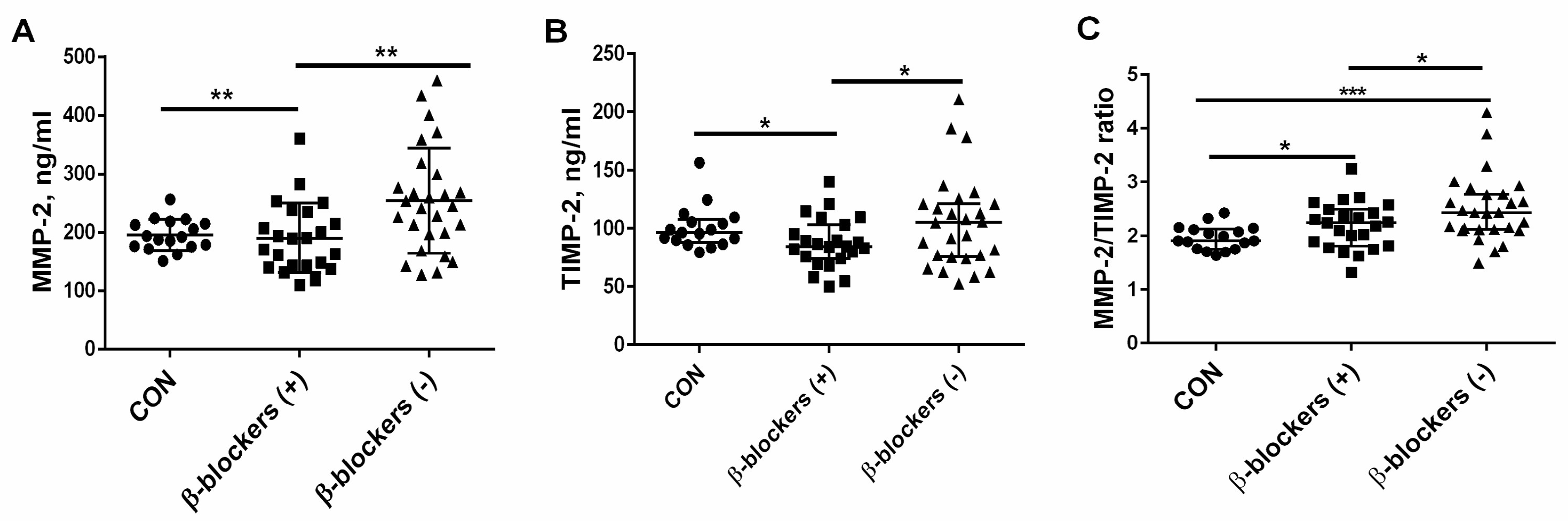
3.3. The Effect of β-Blocker Treatment on MMP-2 and TIMP-2 Levels and the MMP-2/TIMP-2 Ratio in Patients with CKD
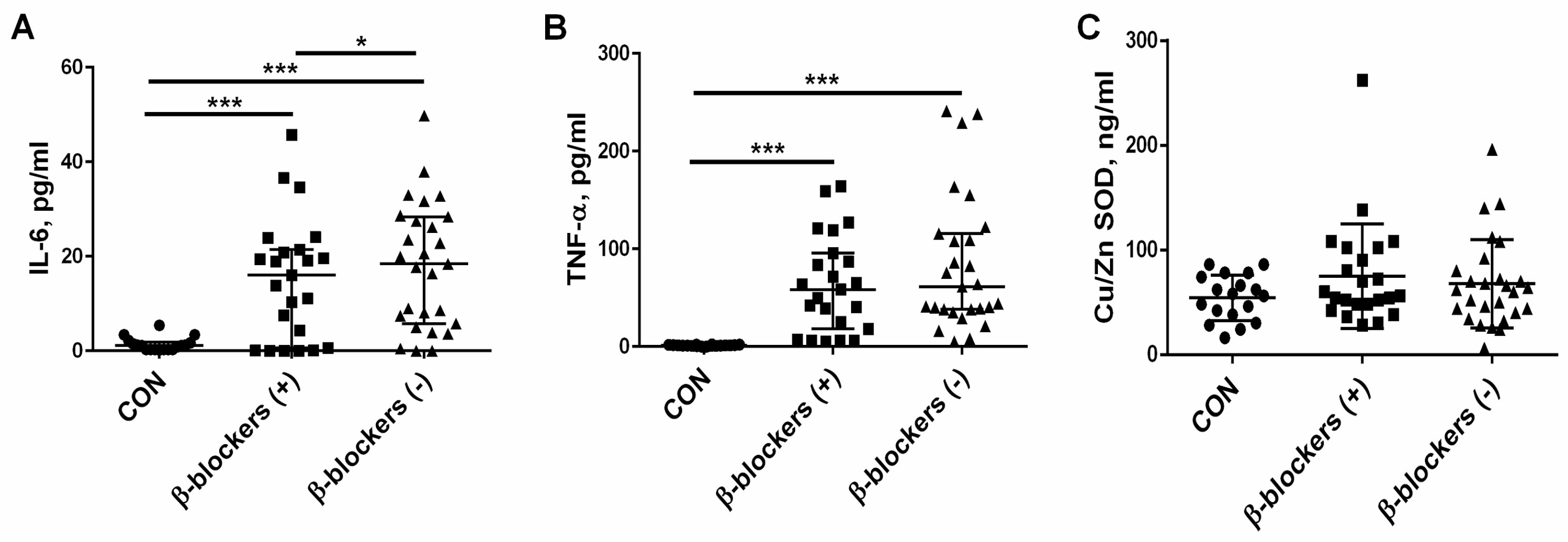
3.4. The Effect of β-Blocker Treatment on Proinflammatory Cytokines and the Marker of Oxidative Stress—Cu/Zn SOD— Levels
3.5. The Factors Affecting the MMP-2/TIMP-2 System in the β-Blockers (+) Group and β-Blockers (−) Group
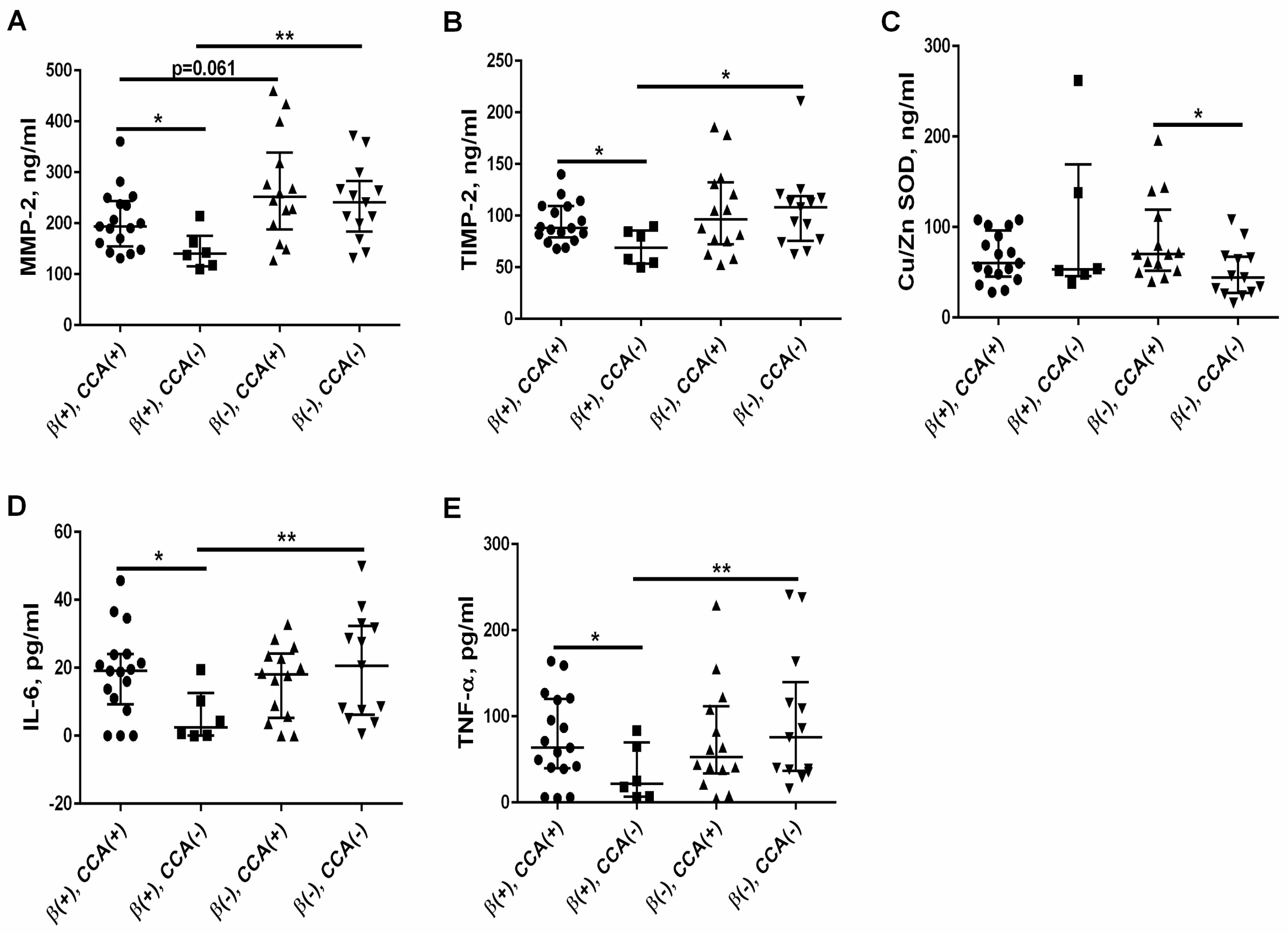
3.6. The Effect of Calcium Channel Antagonists (CCA) on the MMP-2/TIMP-2 System, Oxidative Status, and Proinflammatory Cytokines in the β-Blockers (+) Group and β-Blockers (−) Group
3.7. Variables Independently Predicting the MMP-2/TIMP-2 System in the β-Blockers Group (+) and β-Blockers (−) Group
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gupta, A.; Nagaraju, S.P.; Bhojaraja, M.V.; Swaminathan, S.M.; Mohan, P.B. Hypertension in Chronic Kidney Disease: An Update on Diagnosis and Management. South. Med. J. 2023, 116, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Pugh, D.; Gallacher, P.J.; Dhaun, N. Management on Hypertension in Chronic Kidney Disease. Drugs 2019, 79, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Burnier, M.; Damianaki, A. Hypertension as Cardiovascular Risk Factor in Chronic Kidney Disease. Circ. Res. 2023, 132, 1050–1063. [Google Scholar] [CrossRef] [PubMed]
- Georgianos, P.I.; Agarwal, R. Hypertension in chronic kidney disease-treatment standard 2023. Nephrol. Dial. Transplant. 2023, 38, 2694–2703. [Google Scholar] [CrossRef] [PubMed]
- Converse, R.L., Jr.; Jacobsen, T.N.; Toto, R.D.; Jost, C.M.; Cosentino, F.; Fouad-Tarazi, F.; Victor, R.G. Sympathetic overactivity in patients with chronic renal failure. N. Engl. J. Med. 1992, 327, 1912–1918. [Google Scholar] [CrossRef] [PubMed]
- Shina, A.D.; Agarwal, R. Clinical Pharmacology of Antihypertensive Therapy for the Treatment of Hypertension in CKD. Clin. J. Am. Soc. Nephrol. 2019, 14, 757–764. [Google Scholar]
- AlHabeeb, W.; Mrabeti, S.; Abdelsalam, A.A.I. Therapeutic Properties of Highly Selective β-blockers With or Without Additional Vasodilator Properties: Focus on Bisoprolol and Nebivolol in Patients With Cardiovascular Disease. Cardiovasc. Drugs Ther. 2022, 36, 959–971. [Google Scholar] [CrossRef]
- Mookerjee, R.P.; Pavesi, M.; Thomsen, K.L.; Mehta, G.; Macnaughtan, J.; Bendtsen, F.; Coenraad, M.; Sperl, J.; Gines, P.; Moreau, R.; et al. Treatment with non-selective beta blockers is associated with reduced severity of systemic inflammation and improved survival of patients with acute-on chronic liver failure. J. Hepatol. 2016, 64, 574–582. [Google Scholar] [CrossRef]
- Le, D.E.; Pascotto, M.; Leong-Poi, H.; Sari, I.; Micari, A.; Kaul, S. Antiinflammatory and pro-angiogenic effects of beta blockers in a canine model of chronic ischemic cardiomyopathy: Comparison between carvedilol and metoprolol. Basic. Res. Cardiol. 2013, 108, 384. [Google Scholar] [CrossRef]
- Lin, T.T.; Sung, Y.L.; Syu, J.Y.; Lin, K.Y.; Hsu, H.J.; Liao, M.T.; Liu, Y.B.; Lin, S.F. Anti-Inflammatory and Antiarrhythmic Effects of Beta Blocker in a Rat Model of Rheumatoid Arthritis. J. Am. Heart Assoc. 2020, 9, e016084. [Google Scholar] [CrossRef]
- Serg, M.; Kampus, P.; Kals, J.; Zagura, M.; Zilmer, M.; Zilmer, K.; Kullisaar, T.; Eha, J. Nebivolol and metoprolol: Long-term effects on inflammation and oxidative stress in essential hypertension. Scand. J. Clin. Lab. Investig. 2012, 72, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Kukin, M.L.; Kalman, J.; Charney, R.H.; Levy, D.K.; Buchholz-Varley, C.; Ocampo, O.N.; Eng, C. Prospective, randomized comparison of effect of long-term treatment with metoprolol or carvedilol on symptoms, exercise, ejection fraction, and oxidative stress in heart failure. Circulation 1999, 99, 2645–2651. [Google Scholar] [CrossRef] [PubMed]
- Rizzi, E.; Guimaraes, D.A.; Ceron, C.S.; Prado, C.M.; Pinheiro, L.C.; Martins-Oliveira, A.; Gerlach, R.F.; Tanus-Santos, J.E. β1-Adrenergic blockers exert antioxidant effects, reduce matrix metalloproteinase activity, and improve renovascular hypertension-induced cardiac hypertrophy. Free Radic. Biol. Med. 2014, 73, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.C.; Chen, Y.H.; Leu, H.B.; Chen, Y.L.; Lin, F.Y.; Lin, S.J.; Chen, J.W. Carvedilol, a pharmacological antioxidant, inhibits neointimal matrix metalloproteinase-2 and -9 in experimental atherosclerosis. Free Radic. Biol. Med. 2007, 43, 1508–1522. [Google Scholar] [CrossRef] [PubMed]
- Skrzypiec-Spring, M.; Haczkiewicz, K.; Sapa, A.; Piasecki, T.; Kwiatkowska, J.; Ceremuga, I.; Wozniak, M.; Biczysko, W.; Kobierzycki, C.; Dziegiel, P.; et al. Carvedilol Inhibits Matrix Metalloproteinase-2 Activation in Experimental Autoimmune Myocarditis: Possibilities of Cardioprotective Application. J. Cardiovasc. Pharmacol. Ther. 2018, 23, 89–97. [Google Scholar] [CrossRef]
- Ersan, S.; Tanrısev, M.; Cavdar, Z.; Celık, A.; Unlu, M.; Kocak, A.; Kose, T. Pretreatment with nebivolol attenuates level and expression of matrix metalloproteinases in a rat model of renal ischaemia-reperfusion injury. Nephrology 2017, 22, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Hardy, E.; Hardy-Sosa, A.; Fernandez-Patron, C. MMP-2: Is too low as bad as too high in the cardiovascular system? Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H1332–H1340. [Google Scholar] [CrossRef]
- Strongin, A.Y.; Marmer, B.L.; Grant, G.A.; Goldberg, G.I. Plasma membrane-dependent activation of the 72-kDa type IV collagenase is prevented by complex formation with TIMP-2. J. Biol. Chem. 1993, 268, 14033–14039. [Google Scholar] [CrossRef]
- Kandalam, V.; Basu, R.; Moore, L.; Fan, D.; Wang, X.; Jaworski, D.M.; Oudit, G.Y.; Kassiri, Z. Lack of tissue inhibitor of metalloproteinases 2 leads to exacerbated left ventricular dysfunction and adverse extracellular matrix remodeling in response to biomechanical stress. Circulation 2011, 124, 2094–2105. [Google Scholar] [CrossRef]
- Pawlak, K.; Mysliwiec, M.; Pawlak, D. Peripheral blood level alterations of MMP-2 and MMP-9 in patients with chronic kidney disease on conservative treatment and on hemodialysis. Clin. Biochem. 2011, 44, 838–843. [Google Scholar] [CrossRef]
- Pawlak, K.; Pawlak, D.; Mysliwiec, M. Extrinsic coagulation pathway activation and metalloproteinase-2/TIMPs system are related to oxidative stress and atherosclerosis in hemodialysis patients. Thromb. Haemost. 2004, 92, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, K.; Pawlak, D.; Mysliwiec, M. Urokinase-type plasminogen activator and metalloproteinase-2 are independently related to the carotid atherosclerosis in haemodialysis patients. Thromb. Res. 2008, 121, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, K.; Tankiewicz, J.; Mysliwiec, M.; Pawlak, D. Systemic levels of MMP2/TIMP2 and cardiovascular risk in CAPD patients. Nephron. Clin. Pract. 2010, 115, c251–c258. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Sinha, A.D.; Pappas, M.K.; Abraham, T.N.; Tegegne, G.G. Hypertension in hemodialysis patients treated with atenolol or lisinopril: A randomized controlled trial. Nephrol. Dial. Transplant. 2014, 29, 672–681. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M.A.; Pilmore, H.L.; Ierino, F.L.; Badve, S.V.; Cass, A.; Garg, A.X.; Isbel, N.M.; Krum, H.; Pascoe, E.M.; Perkovic, V.; et al. The beta-blocker to lower cardiovascular dialysis events (BLOCADE) Feasibility study: A randomized controlled trial. Am. J. Kidney Dis. 2016, 67, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Guo, X.; Yu, Q. Efects of beta-blockers on cardiovascular events and mortality in dialysis patients: A systematic review and meta-analysis. Blood Purif. 2019, 48, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Sim, J.J.; Shi, J.; Shaw, S.F.; Lee, M.-S.; Neyer, J.R.; Kovesdy, C.P.; Kalantar-Zadeh, K.; Jacobsen, S.J. β-Blocker use and risk of mortality in heart failure patients initiating maintenance dialysis. Am. J. Kidney Dis. 2021, 77, 704–712. [Google Scholar] [CrossRef]
- Dong, H.; Zhou, L.; Yang, L.; Lu, H.; Cao, S.; Song, H.; Fu, S. β-Blockers could improve the 28-day and 3-year survival of patients with end-stage renal disease: A retrospective cohort study. Int. Urol. Nephrol. 2023, 55, 1597–1607. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Pawlak, K.; Pawlak, D.; Mysliwiec, M. Cu/Zn superoxide dismutase plasma levels as a new useful clinical biomarker of oxidative stress in patients with end-stage renal disease. Clin. Biochem. 2005, 38, 700–705. [Google Scholar] [CrossRef]
- Washio, K.; Inagaki, M.; Tsuji, M.; Morio, Y.; Akiyama, S.; Gotoh, H.; Gotoh, T.; Gotoh, Y.; Oguchi, K. Oral vitamin C supplementation in hemodialysis patients and its effect on the plasma level of oxidized ascorbic acid and Cu/Zn superoxide dismutase, an oxidative stress marker. Nephron Clin. Pract. 2008, 109, c49–c54. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, L.M.; Pansani, T.N.; Hebling, J.; de Souza Costa, C.A.; Basso, F.G. Chemotherapy drugs and inflammatory cytokines enhance matrix metalloproteinases expression by oral mucosa cells. Arch. Oral Biol. 2021, 127, 105159. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, M.; Ishida, Y.; Kimura, A.; Kuninaka, Y.; Taruya, A.; Ozaki, M.; Tanaka, A.; Mukaida, N.; Kondo, T. Crucial Involvement of IL-6 in Thrombus Resolution in Mice via Macrophage Recruitment and the Induction of Proteolytic Enzymes. Front. Immunol. 2020, 10, 3150. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, J.J. Collagenases and tissue inhibitors of metalloproteinases: A functional balance in tissue degradation. Oral Dis. 1996, 2, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Kobusiak-Prokopowicz, M.; Krzysztofik, J.; Kaaz, K.; Jolda-Mydlowska, B.; Mysiak, A. MMP-2 and TIMP-2 in patients with heart failure and chronic kidney disease. Open Med. 2018, 13, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Squire, I.B.; Evans, J.; Ng, L.L.; Loftus, I.M.; Thompson, M.M. Plasma MMP-9 and MMP-2 following acute myocardial infarction in man: Correlation with echocardiographic and neurohumoral parameters of left ventricular dysfunction. J. Card. Fail. 2004, 10, 228–233. [Google Scholar] [CrossRef]
- Hsu, T.W.; Kuo, K.L.; Hung, S.C.; Huang, P.H.; Chen, J.W.; Tarng, D.C. Progression of kidney disease in non-diabetic patients with coronary artery disease: Predictive role of circulating matrix metalloproteinase-2,-3, and -9. PLoS ONE 2013, 8, e70132. [Google Scholar] [CrossRef] [PubMed]
- Nagano, M.; Fukami, K.; Yamagishi, S.; Ueda, S.; Kaida, Y.; Matsumoto, T.; Yoshimura, J.; Hazama, T.; Takamiya, Y.; Kusumoto, T.; et al. Circulating matrix metalloproteinase-2 is an independent correlate of proteinuria in patients with chronic kidney disease. Am. J. Nephrol. 2009, 29, 109–115. [Google Scholar] [CrossRef]
- Borden, P.; Heller, R.A. Transcriptional control of matrix metalloproteinases and the tissue inhibitors of matrix metalloproteinases. Crit. Rev. Eukaryot. Gene Expr. 1997, 7, 159–178. [Google Scholar] [CrossRef]
- Valentin, F.; Bueb, J.L.; Kieffer, A.P.; Tschirhart, B.E.; Atkinson, A.J. Oxidative stress activates MMP-2 in cultured human coronary smooth muscle cells. Fundam. Clin. Pharmacol. 2005, 19, 661–667. [Google Scholar] [CrossRef]
- Tan, S.; Zhou, F.; Zhang, Z.; Wang, J.; Xu, J.; Zhuang, Q.; Meng, Q.; Xi, Q.; Jiang, Y.; Wu, G. Beta-1 blocker reduces inflammation and preserves intestinal barrier function after open abdominal surgery. Surgery 2021, 169, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Ackland, G.L.; Yao, S.T.; Rudiger, A.; Dyson, A.; Stidwill, R.; Poputnikov, D.; Singer, M.; Gourine, A.V. Cardioprotection, attenuated systemic inflammation, and survival benefit of beta1-adrenoceptor blockade in severe sepsis in rats. Crit. Care Med. 2010, 38, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Kamimura, D.; Hirano, T. Pleiotropy and specificity: Insights from the interleukin 6 family of cytokines. Immunity 2019, 50, 812–831. [Google Scholar] [CrossRef] [PubMed]
- Jagadeesh, G.S.; Nagoor Meeran, M.F.; Selvaraj, P. Activation of β1-adrenoceptor triggers oxidative stress mediated myocardial membrane destabilization in isoproterenol induced myocardial infarcted rats: 7-hydroxycoumarin and its counter action. Eur. J. Pharmacol. 2016, 777, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Seeland, U.; Selejan, S.; Engelhardt, S.; Muller, P.; Lohse, M.J.; Bohm, M. Interstitial remodelingin β1-adrenergic receptor transgenic mice. Basic Res. Cardiol. 2007, 102, 183–193. [Google Scholar] [CrossRef]
- Sorrentino, S.A.; Doerries, C.; Manes, C.; Speer, T.; Dessy, C.; Lobysheva, I.; Mohmand, W.; Akbar, R.; Bahlmann, F.; Besler, C.; et al. Nebivolol exerts beneficial effects on endothelial function, early endothelial progenitor cells, myocardial neovascularization, and left ventricular dysfunction early after myocardial infarction beyond conventional β1-blockade. J. Am. Coll. Cardiol. 2011, 57, 601–611. [Google Scholar] [CrossRef]
- Fang, Y.; Nicol, L.; Harouki, N.; Monteil, C.; Wecker, D.; Debunne, M.; Bauer, F.; Lallemand, F.; Richard, V.; Thuillez, C.; et al. Improvement of left ventricular diastolic function induced by beta-blockade: A comparison between nebivolol and metoprolol. J. Mol. Cell. Cardiol. 2011, 51, 168–176. [Google Scholar] [CrossRef]
- Stenvinkel, P.; Barany, P. Anaemia, rHuEPO resistance, and cardiovascular disease in end-stage renal failure: Links to inflammation and oxidative stress. Nephrol. Dial. Transplant. 2002, 17, 32–37. [Google Scholar] [CrossRef]
- Kaysen, G.A. The microinflammatory state in uremia: Causes and potential consequences. J. Am. Soc. Nephrol. 2001, 12, 1549–1557. [Google Scholar] [CrossRef]
- Rysz, J.; Banach, M.; Stolarek, R.A.; Pasnik, J.; Cialkowska-Rysz, A.; Koktysz, R.; Piechota, M.; Baj, Z. Serum matrix metalloproteinases MMP-2 and MMP-9 and metalloproteinase tissue inhibitors TIMP-1 and TIMP-2 in diabetic nephropathy. J. Nephrol. 2007, 20, 444–452. [Google Scholar]
- Kim, S.S.; Shin, N.; Bae, S.S.; Lee, M.Y.; Rhee, H.; Kim, I.Y.; Seong, E.Y.; Lee, D.W.; Lee, S.B.; Kwak, I.S.; et al. Enhanced expression of two discrete isoforms of matrix metalloproteinase-2 in experimental and human diabetic nephropathy. PLoS ONE 2017, 12, e0171625. [Google Scholar] [CrossRef] [PubMed]
- Endo, T.; Nakabayashi, K.; Sekiuchi, M.; Kuroda, T.; Soejima, A.; Yamada, A. Matrix metalloproteinase-2, matrix metalloproteinase-9, and tissue inhibitor of metalloproteinase-1 in the peripheral blood of patients with various glomerular diseases and their implication in pathogenetic lesions: Study based on an enzyme-linked assay and immunohistochemical staining. Clin. Exp. Nephrol. 2006, 10, 253–261. [Google Scholar] [PubMed]
- Marçal, D.M.; Rizzi, E.; Martins-Oliveira, A.; Ceron, C.S.; Guimaraes, D.A.; Gerlach, R.F.; Tanus-Santos, J.E. Comparative study on antioxidant effects and vascular matrix metalloproteinase-2 downregulation by dihydropyridines in renovascular hypertension. Naunyn Schmiedebergs Arch. Pharmacol. 2011, 383, 35–44. [Google Scholar] [CrossRef]
- Mendes, A.S.; Blascke de Mello, M.M.; Parente, J.M.; Omoto, A.C.M.; Neto-Neves, E.M.; Fazan, R., Jr.; Tanus-Santos, J.E.; Castro, M.M. Verapamil decreases calpain-1 and matrix metalloproteinase-2 activities and improves hypertension-induced hypertrophic cardiac remodeling in rats. Life Sci. 2020, 244, 117153. [Google Scholar] [CrossRef]
- Martinez, M.L.; Castro, M.M.; Rizzi, E.; Fernandes, K.; Demacq, C.; Bendhack, L.M.; Gerlach, R.F.; Tanus-Santos, J.E. Lercanidipine reduces matrix metalloproteinase-2 activity and reverses vascular dysfunction in renovascular hypertensive rats. Eur. J. Pharmacol. 2008, 591, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Zervoudaki, A.; Economou, E.; Pitsavos, C.; Vasiliadou, K.; Aggeli, C.; Tsioufis, K.; Toutouza, M.; Stefanadis, C.; Toutouzas, P. The effect of Ca2+ channel antagonists on plasma concentrations of matrix metalloproteinase-2 and -9 in essential hypertension. Am. J. Hypertens. 2004, 17, 273–276. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zervoudaki, A.; Economou, E.; Stefanadis, C.; Pitsavos, C.; Tsioufis, K.; Aggeli, C.; Vasiliadou, K.; Toutouza, M.; Toutouzas, P. Plasma levels of active extracellular matrix metalloproteinases 2 and 9 in patients with essential hypertension before and after antihypertensive treatment. J. Hum. Hypertens. 2003, 17, 119–124. [Google Scholar] [CrossRef]

| β-Blockers (+), n = 23 | β-Blockers (−), n = 27 | p Value | |
|---|---|---|---|
| Age, years | 53.43 ± 12.70 | 53.48 ± 17.74 | 0.897 |
| Male sex, % | 57 | 59 | 0.886 |
| eGFR, mL/min/1.73 m2 | 17.5 (9.7–42.6) | 18.7 (12.8–33.8) | 0.535 |
| CKD, stage 1, % | 0 | 4 | 0.332 |
| CKD, stage 2, % | 17 | 15 | 0.849 |
| CKD, stage 3, % | 17 | 19 | 0.856 |
| CKD, stage 4, % | 17 | 19 | 0.856 |
| CKD, stage 5, % | 48 | 42 | 0.673 |
| SBP, mm Hg | 136.90 ± 11.23 | 134.23 ± 14.26 | 0.471 |
| DBP, mm Hg | 88.57 ± 7.27 | 84.62 ± 7.20 | 0.695 |
| Heart rate, bpm | 73.3 ± 8.9 | 68.8 ± 9.5 | 0.562 |
| RBC, 1012/L | 3.63 ± 0.72 | 3.45 ± 0.57 | 0.334 |
| Hemoglobin, g/dL | 11.13 ± 2.35 | 10.70 ± 1.98 | 0.490 |
| WBC, 109/L | 6.41 ± 1.95 | 6.49 ± 2.62 | 0.560 |
| Glucose, mg/dL | 90.0 (81.0–100.0) | 93.5 (85.0–117.0) | 0.474 |
| Albumin, g/dL | 3.12 ± 0.71 | 3.07 ± 0.77 | 0.814 |
| Total protein, g/dL | 6.10 ± 1.22 | 6.10 ± 1.17 | 0.999 |
| Total cholesterol, mg/dL | 208.0 (175.0–223.0) | 190.0 (170.0–219.0) | 0.338 |
| Triglycerides, mg/dL | 170 (115–265) | 152 (80–291) | 0.448 |
| Cardiovascular disease, % | 48 | 37 | 0.432 |
| Smokers, % | 9 | 19 | 0.316 |
| Diabetic nephropathy, % | 37 | 30 | 0.652 |
| Glomerulonephritis, % | 37 | 47 | 0.475 |
| Polycystic kidney disease, % | 17 | 19 | 0.854 |
| Hypertensive nephropathy, % | 9 | 4 | 0.234 |
| Diuretics, % | 26 | 15 | 0.166 |
| Converting enzyme inhibitors/sartans % | 65 | 52 | 0.176 |
| Calcium channel antagonists, % | 74 | 52 | 0.110 |
| α-adrenoceptor antagonists, % | 13 | 4 | 0.123 |
| Statin, % | 9 | 19 | 0.316 |
| β-Blockers (+) | β-Blockers (−) | |||
|---|---|---|---|---|
| MMP-2 | TIMP-2 | MMP-2 | TIMP-2 | |
| Sex, male = 1 | R = 0.031 NS | R = 0.008 NS | χ2 = 4.518 p = 0.034 | χ2 = 2.706 NS |
| SBP | R = 0.163 NS | R = 0.463 p = 0.035 | R = 0.180 NS | R = 0.150 NS |
| RBC | R = −0.500 p = 0.015 | R = −0.509 p = 0.013 | R = −0.142 NS | R = −0.075 NS |
| Hemoglobin | R = −0.510 p = 0.013 | R = −0.556 p = 0.006 | R = −0.102 NS | R = −0.046 NS |
| Interleukin 6 | R = 0.458 p = 0.028 | R = 0.390 p = 0.066 | R = 0.101 NS | R = 0.061 NS |
| TNF-α | R = 0.419 p = 0.046 | R = 0.230 NS | R = 0.042 NS | R = 0.062 NS |
| Cu/Zn SOD | R = −0.066 NS | R = 0.077 NS | R = 0.382 p = 0.049 | R = 0.549 p = 0.003 |
| Albumin | R = −0.586 p = 0.002 | R = −0.290 NS | R = −0.471 p = 0.030 | R = −0.300 NS |
| Total protein | R = −0.544 p = 0.008 | R = −0.082 NS | R = −0.273 NS | R = −0.056 NS |
| Diabetic nephropathy | χ2 = 0.016 NS | χ2 = 0.020 NS | χ2 = 6.497 p = 0.011 | χ2 = 6.618 p = 0.010 |
| Glomerulonephritis | χ2 = 4.422 p = 0.035 | χ2 = 2.782 NS | χ2 = 6.253 p = 0.010 | χ2 = 6.367 p = 0.012 |
| Calcium channel antagonists | χ2 = 6.199 p = 0.013 | χ2 = 7.475 p = 0.006 | χ2 = 0.514 NS | χ2 = 0.019 NS |
| Independent Variable | Regression Coefficient (β) | Standard Error | p Values | |
|---|---|---|---|---|
| MMP-2 | albumin | −0.553 | 0.181 | <0.001 |
| RBC | −0.512 | 0.178 | 0.016 | |
| TIMP-2 * | MMP-2 | 0.740 | 0.132 | <0.001 |
| SBP | 0.297 | 0.132 | 0.038 |
| Independent Variable | Regression Coefficient (β) | Standard Error | p Values | |
|---|---|---|---|---|
| MMP-2 | Cu/Zn SOD | 0.734 | 0.208 | 0.005 |
| albumin | −0.682 | 0.242 | 0.018 | |
| TIMP-2 * | MMP-2 | 0.739 | 0.113 | <0.001 |
| glomerulonephritis | −0.264 | 0.112 | 0.028 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopańko, M.; Zabłudowska, M.; Pawlak, D.; Sieklucka, B.; Krupa, A.; Sokołowska, K.; Ziemińska, M.; Pawlak, K. The Possible Effect of β-Blocker Use on the Circulating MMP-2/TIMP-2 System in Patients with Chronic Kidney Disease on Conservative Treatment. J. Clin. Med. 2024, 13, 1847. https://doi.org/10.3390/jcm13071847
Kopańko M, Zabłudowska M, Pawlak D, Sieklucka B, Krupa A, Sokołowska K, Ziemińska M, Pawlak K. The Possible Effect of β-Blocker Use on the Circulating MMP-2/TIMP-2 System in Patients with Chronic Kidney Disease on Conservative Treatment. Journal of Clinical Medicine. 2024; 13(7):1847. https://doi.org/10.3390/jcm13071847
Chicago/Turabian StyleKopańko, Magdalena, Magdalena Zabłudowska, Dariusz Pawlak, Beata Sieklucka, Anna Krupa, Katarzyna Sokołowska, Marta Ziemińska, and Krystyna Pawlak. 2024. "The Possible Effect of β-Blocker Use on the Circulating MMP-2/TIMP-2 System in Patients with Chronic Kidney Disease on Conservative Treatment" Journal of Clinical Medicine 13, no. 7: 1847. https://doi.org/10.3390/jcm13071847
APA StyleKopańko, M., Zabłudowska, M., Pawlak, D., Sieklucka, B., Krupa, A., Sokołowska, K., Ziemińska, M., & Pawlak, K. (2024). The Possible Effect of β-Blocker Use on the Circulating MMP-2/TIMP-2 System in Patients with Chronic Kidney Disease on Conservative Treatment. Journal of Clinical Medicine, 13(7), 1847. https://doi.org/10.3390/jcm13071847






