1. Introduction
Abdominal-based flaps, particularly the deep inferior epigastric perforator (DIEP) flap, have become the standard approach for autologous breast reconstruction following nipple-sparing mastectomy (NSME) or skin-sparing mastectomy (SSME) procedures [
1,
2,
3]. This technique offers several notable advantages, including an enhanced quality of life and greater patient satisfaction when compared to implant-based approaches [
4,
5,
6]. However, the trade-offs are longer surgical duration, extended hospital stay and the necessity to sacrifice an additional donor site. Considering this additional effort, our objective should be to minimize the risks associated with the additional surgery for the patients and continuously strive to enhance the surgical outcome.
Abdominal flap harvesting is performed with electrosurgical devices, using high-frequency electrical current for tissue dissection and simultaneous hemostasis [
7]. Different modalities are available, the conventional monopolar electrocautery (MPE) uses a continuous waveform of radiofrequency energy via an uninsulated metal electrode for tissue cutting through thermal ablation, operating at temperatures between 180 and 240 °C [
8]. In contrast, the PEAK PlasmaBlade (PPB) employs short (40 µs) high-frequency pulses of radiofrequency energy to generate electrical plasma along an insulated electrode’s edge and it maintains a lower operating temperature around 45 °C [
8,
9]. Previous investigations suggest that the PlasmaBlade may offer advantages over electrocautery, demonstrating reduced thermal injury depth and inflammatory responses [
8,
10,
11]. Inflammatory processes and trauma to the lymphatic network during surgical dissection are known factors contributing to postoperative seroma formation, a common complication after DIEP flap harvesting, with reported incidences ranging from 1.4% to 16.2% [
12,
13,
14,
15,
16]. Prolonged drainage duration to prevent seroma formation poses disadvantages such as extended hospitalization and the risk of ascending infections through the drain tube. Previous research has generated conflicting and inconclusive findings regarding whether the choice of the surgical dissection device significantly impacts clinical outcomes. While some studies suggest benefits associated with using the PPB, such as reduced seroma rates and shorter drain dwelling times, these studies have limitations, notably small sample sizes and none have evaluated patient-specific risk factors in this context [
11,
17,
18,
19,
20,
21,
22].
Our study seeks to evaluate the influence of the dissection device utilized during abdominal flap harvesting on clinical outcomes, while considering risk factors, such as body mass index (BMI), smoking status and previous chemotherapy.
4. Discussion
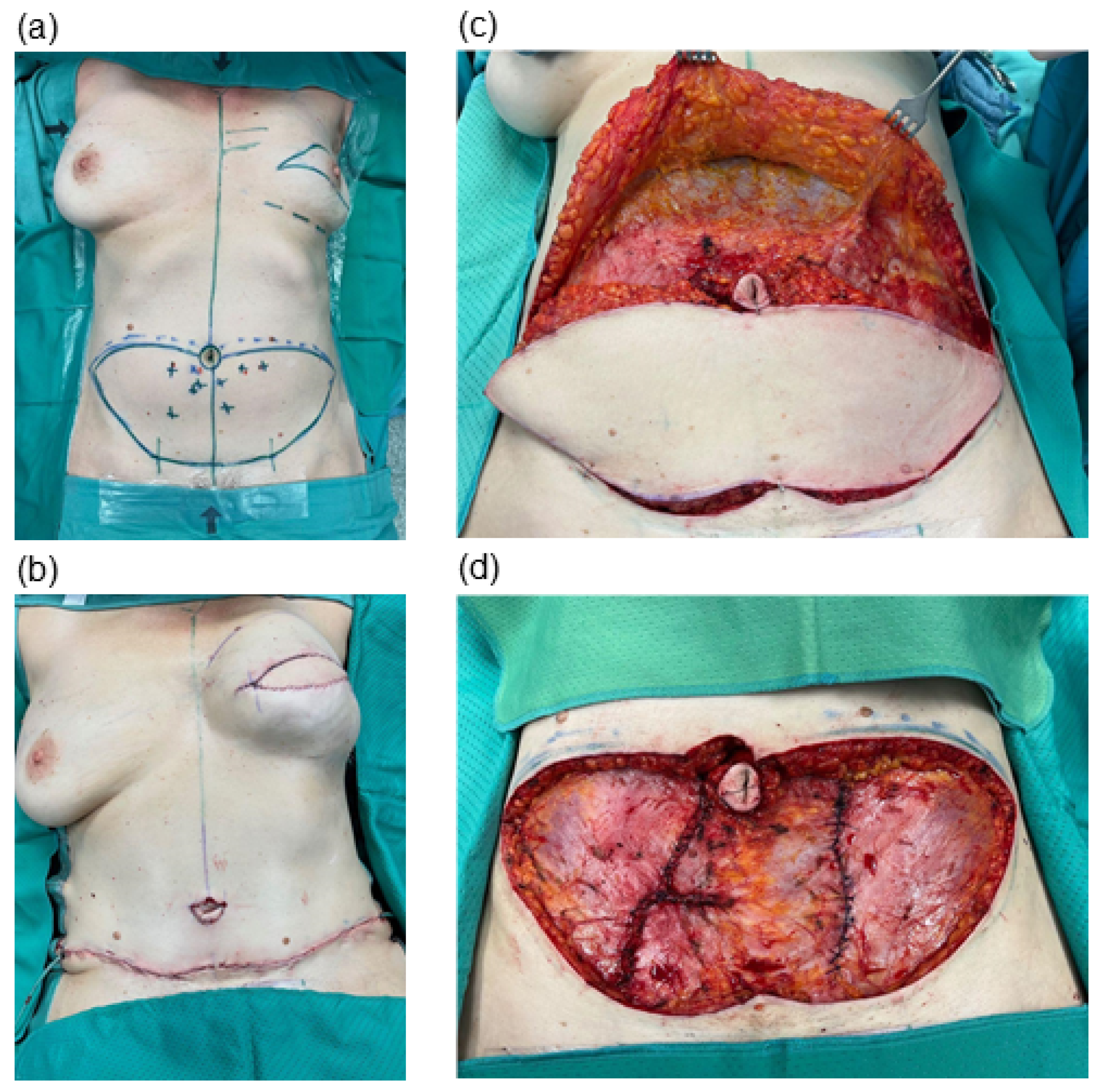
Autologous DIEP-based breast reconstruction involves extensive undermining at the abdominal donor site, resulting in sizable wound areas. Typically, flap dissection is undertaken with electrosurgical devices to allow simultaneous hemostasis and efficient operating times [
24]. We evaluated two specific dissection devices—the standard monopolar electrocautery and the newer PEAK PlasmaBlade—in the context of abdominal flap harvesting, aiming to discern their impacts on clinical outcomes.
Previous research has yielded conflicting and inconclusive results regarding whether the selection of the dissection device can genuinely lead to better clinical outcomes.
Studies assessing outcomes in extensive wound areas, like those involved in autologous breast reconstruction using the abdominal donor site, have often been limited by small sample sizes [
11,
17,
18,
19,
20,
21,
22,
25,
26]. While more extensive investigations have been undertaken in distinct surgical contexts, such as tonsillectomy [
27,
28,
29] and surgical implant replacement [
30], these findings may not be directly applicable to the specific circumstances of autologous breast reconstruction.
Our retrospective analysis sought to broaden the scope of this research by including a total of 128 patients in our study. Of these patients, 72 underwent abdominal flap preparation using conventional electrocautery, while 56 patients underwent the procedure with the PEAK PlasmaBlade. A comparison of these two patient cohorts revealed no statistically significant differences in age, body mass index, smoking status, history of neoadjuvant oncologic therapy, diabetes status and the primary indication for mastectomy. This comparability ensures the reliability and robustness of our observed outcomes.
Our analysis yielded the significant (
p* = 0.0324) finding of a higher cumulative wound fluid quantity in the monopolar electrocautery group (351.11 ± 185.96 mL) compared to the PEAK PlasmaBlade group (279.38 ± 183.38 mL). While previous research on this topic has provided inconclusive data, some studies support our finding by reporting lower total drain output following the use of the PEAK PlasmaBlade [
11,
17,
18,
19,
20,
22], whereas other authors found no difference between the two dissection devices [
21,
25,
26]. It is worth noting that, to the best of our knowledge, no prior study has reported an increase in seroma rates after utilizing the PEAK PlasmaBlade. It has previously been suggested that the reduced working temperature of the PEAK PlasmaBlade leads to lower tissue damage [
8,
10], which contributes to reduced seroma formation. One study in gender-affirming mastectomy patients has investigated histologic samples, showing a 22% reduction in thermal injury depth with the PlasmaBlade compared to conventional monopolar cautery [
18].
Similarly, the existing literature on the evaluation of hospitalization and drain dwell time yields inconsistent results. While Schlosshauer, Dogan and Sowa reported positive outcomes in the PlasmaBlade cohorts [
11,
17,
19,
20], other studies did not identify variations in terms of hospitalization and drain dwell time [
21,
25,
26]. Our data did not reveal a difference between both devices for these aspects. There is no previous work indicating inferiority of the PlasmaBlade in this regard.
We intentionally selected abdominal-based autologous breast reconstruction as our research focus. This patient group not only represents a highly standardized approach to large-scale wound preparation but also encompasses a spectrum of patient-specific risk factors, including those related to oncologic treatments, which are pertinent to our analysis. Extensive cohort studies conducted so far have failed to establish a definitive link between neoadjuvant chemotherapy and the occurrence of surgical complications during breast reconstruction [
31,
32,
33,
34,
35]. Nevertheless, we were determined to assess the role of dissection instruments in relation to neoadjuvant chemotherapy and other risk factors and its impact on surgical outcomes. We identified chemotherapy, smoking status and BMI as the primary risk factors of interest in our patient cohorts, with prevalences high enough to enable statistical analysis.
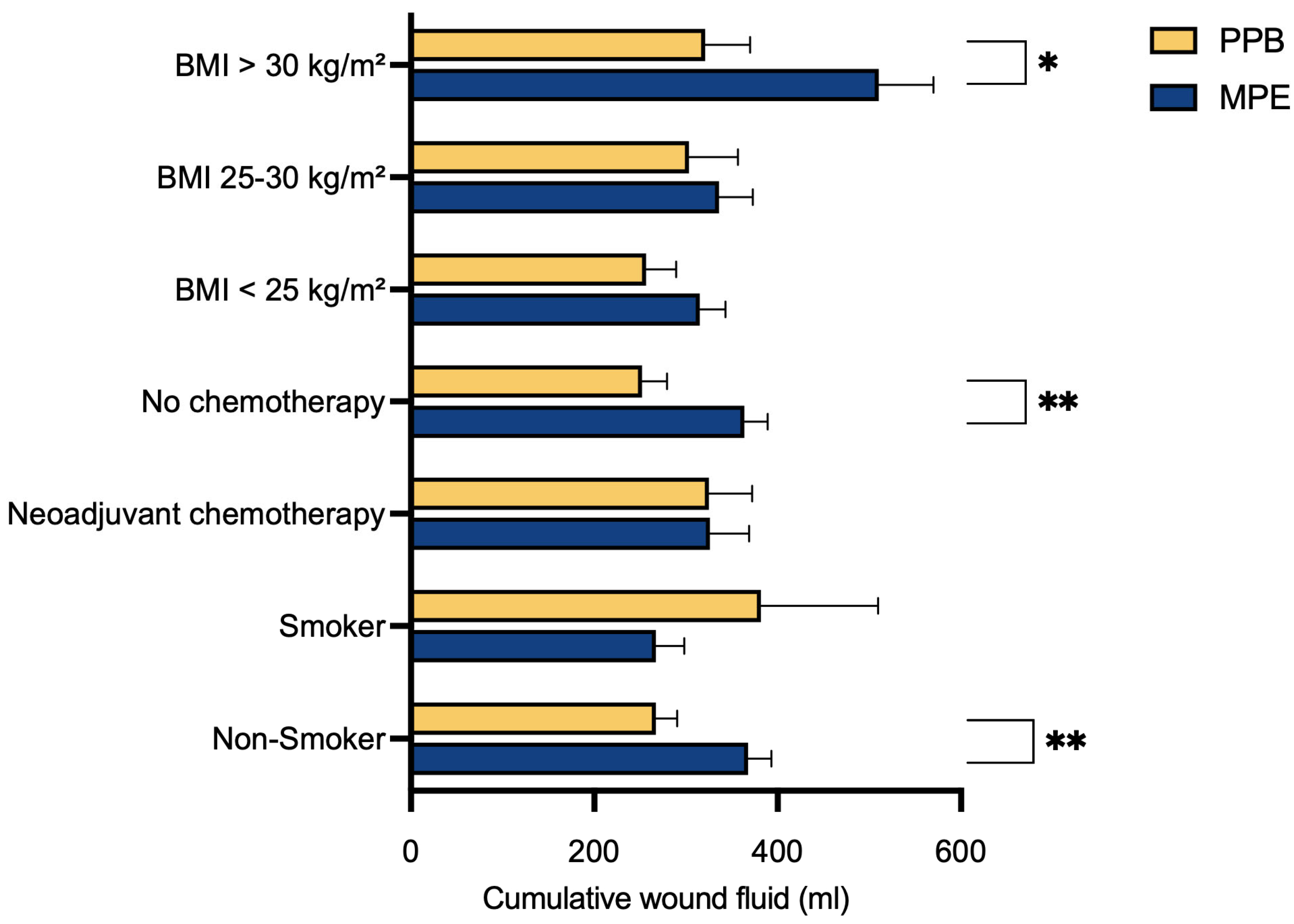
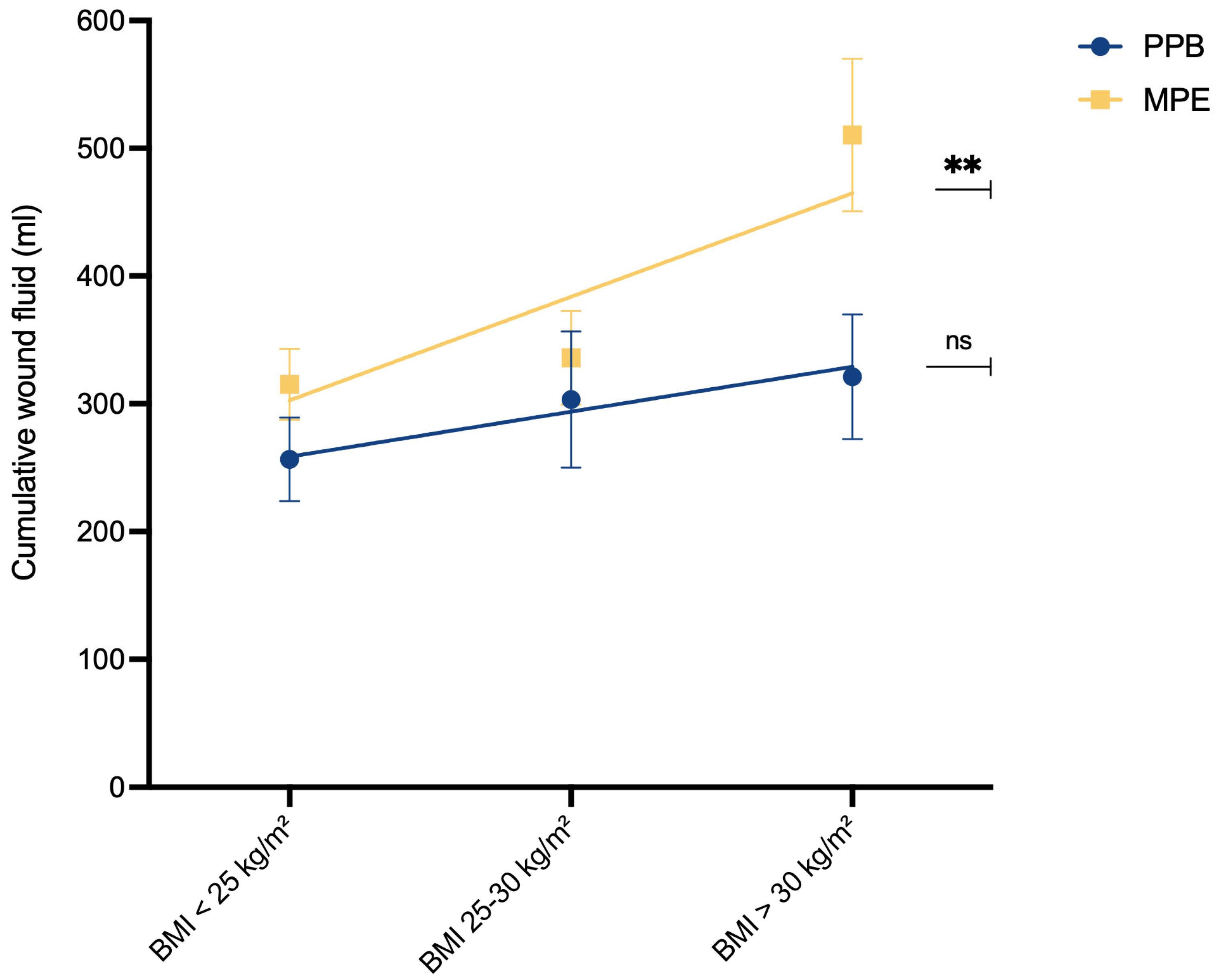
In our multivariate data analysis, we identified a substantial reduction in postoperative abdominal wound fluid production in the PEAK PlasmaBlade cohort compared to the electrocautery cohort across three specific subgroups. These groups comprised individuals without prior neoadjuvant chemotherapy (p** = 0.0041), non-smokers (p** = 0.0046) and those with a BMI exceeding 30 kg/m2 (p* = 0.0284).
It is noteworthy that these groups typically do not share the same risk profile. In standard practice, non-smokers and patients without previous neoadjuvant chemotherapy are typically regarded as low-risk individuals for surgical complications. Conversely, patients with a BMI exceeding 30 kg/m
2 are commonly perceived as a high-risk population for adverse events in the surgical context [
36]. Mani et al. previously investigated the association between BMI and the occurrence of donor-site seroma following the harvesting of DIEP flaps. Their findings revealed that obese patients (BMI > 30 kg/m
2) had the highest incidence of postoperative seroma formation, reaching a rate of 16% [
16]. Our results revealed a significant association between higher BMI and increased postoperative wound fluid production (
p** = 0.0058) within the MPE cohort (
Figure 4). In contrast, no such correlation was evident within the PPB cohort (p
ns = 0.2895), suggesting that the utilization of PPB might mitigate the risks associated with higher BMI.
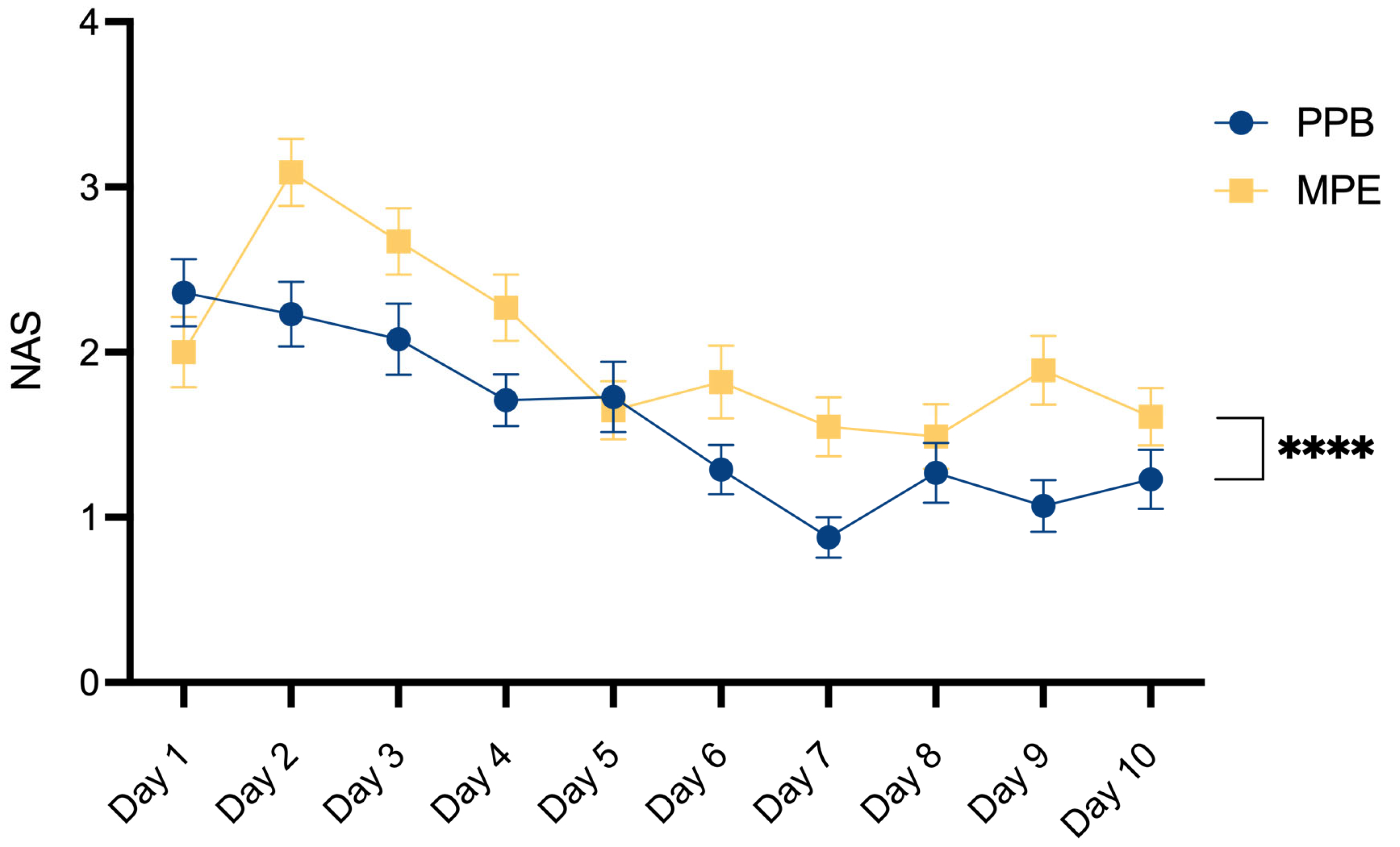
Although not statistically significant, all subgroups that displayed positive outcomes with the use of the PEAK PlasmaBlade regarding postoperative drainage volume also demonstrated a trend toward reduced drain dwell time and shorter hospital stays.
The assessment of hospitalization duration may be subject to some inaccuracies, as our department typically retains patients in the hospital until histopathological results become accessible, and the removal of the monitoring skin island is performed. Nevertheless, it is noteworthy that a tendency towards reduced drainage catheter dwell time in the PPB group may potentially be linked to our observed outcomes of a significant decrease in postoperative pain levels around day 7 and day 9 in the PPB group, attributable to the earlier removal of drainage devices.
Our data reveal a significant decrease in postoperative pain within the PEAK PlasmaBlade cohort. To our knowledge, only Friebel et al. have previously compared postoperative pain levels between both dissection devices, reporting an increase in the PPB cohort [
21]. This was attributed to the potential impact of tighter abdominal closure due to lower flap weights in this group. In our patient cohorts, both flap volume (
p = 0.8572) and the distribution of unilateral versus bilateral reconstruction (
p = 0.0560) within the two patient groups were comparable, as an analysis of our patient characteristics (
Table 1) showed. We consider this as a strength of our study since it allows the assumption that postoperative pain levels are not biased by these characteristics. There have been studies examining postoperative pain following surgical skin incisions made with electrocautery versus steel scalpel, which reported reduced pain levels and a decreased use of analgesics in the electrocautery group [
37,
38]. Chrysos explains this by highlighting that the vaporization of cells resulting from the application of pure sinusoidal current leads to immediate tissue and nerve necrosis without significantly affecting nearby structures [
38]. The varying degrees of nerve damage between electrocautery and the PEAK PlasmaBlade may also contribute to our significant finding of reduced postoperative pain in the PPB group, but such evaluations should be a focus of future studies.
Our study demonstrates also several limitations. Due to retrospective data analysis, we cannot offer detailed information concerning the adjustments of the used monopolar electrocautery. But we tried to overcome this lack of information by only including patients that were operated under the lead of one single surgeon, so it may be assumed that the same preferred settings were used. Furthermore, this study benefits from the extensive experience of this single surgeon, contributing to the reliability of the findings and minimizing the risk of performance bias. Moreover, the similarity in patient demographic characteristics across both cohorts indicates a low risk of selection bias, despite the study’s retrospective nature.