Early and Mid-Term Outcomes of Isolated Type 2 Endoleak Refractory to an Embolization Procedure
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
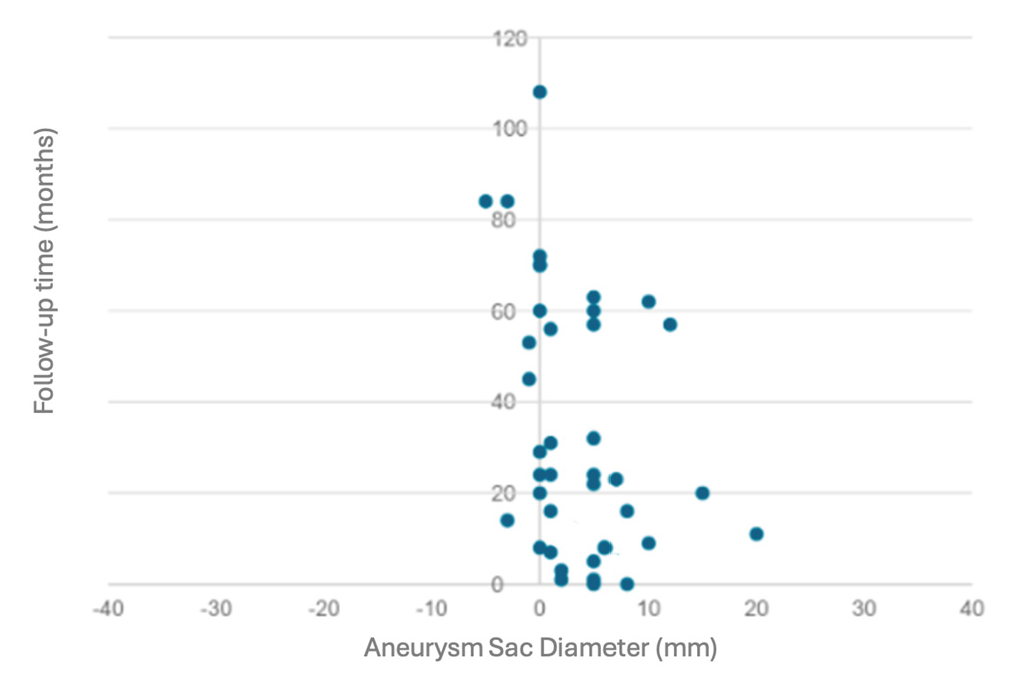
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patel, R.; Sweeting, M.J.; Powell, J.T.; Greenhalgh, R.M. Endovascular versus Open Repair of Abdominal Aortic Aneurysm in 15-Years’ Follow-up of the UK Endovascular Aneurysm Repair Trial 1 (EVAR Trial 1): A Randomised Controlled Trial. Lancet 2016, 388, 2366–2374. [Google Scholar] [CrossRef]
- De Bruin, J.L.; Baas, A.F.; Buth, J.; Prinssen, M.; Verhoeven, E.L.G.; Cuypers, P.W.M.; van Sambeek, M.R.H.M.; Balm, R.; Grobbee, D.E.; Blankensteijn, J.D. Long-Term Outcome of Open or Endovascular Repair of Abdominal Aortic Aneurysm. N. Engl. J. Med. 2010, 362, 1881–1889. [Google Scholar] [CrossRef]
- Blankensteijn, J.D.; de Jong, S.E.C.A.; Prinssen, M.; van der Ham, A.C.; Buth, J.; van Sterkenburg, S.M.M.; Verhagen, H.J.M.; Buskens, E.; Grobbee, D.E. Two-Year Outcomes after Conventional or Endovascular Repair of Abdominal Aortic Aneurysms. N. Engl. J. Med. 2005, 352, 2398–2405. [Google Scholar] [CrossRef]
- Becquemin, J.-P.; Pillet, J.-C.; Lescalie, F.; Sapoval, M.; Goueffic, Y.; Lermusiaux, P.; Steinmetz, E.; Marzelle, J. A Randomized Controlled Trial of Endovascular Aneurysm Repair versus Open Surgery for Abdominal Aortic Aneurysms in Low- to Moderate-Risk Patients. J. Vasc. Surg. 2011, 53, 1167–1173.e1. [Google Scholar] [CrossRef]
- The United Kingdom EVAR Trial Investigators. Endovascular versus Open Repair of Abdominal Aortic Aneurysm. N. Engl. J. Med. 2010, 362, 1863–1871. [Google Scholar] [CrossRef]
- Lederle, F.A.; Kyriakides, T.C.; Stroupe, K.T.; Freischlag, J.A.; Padberg, F.T.; Matsumura, J.S.; Huo, Z.; Johnson, G.R. Open versus Endovascular Repair of Abdominal Aortic Aneurysm. N. Engl. J. Med. 2019, 380, 2126–2135. [Google Scholar] [CrossRef]
- Antoniou, G.A.; Antoniou, S.A.; Torella, F. Editor’s Choice—Endovascular vs. Open Repair for Abdominal Aortic Aneurysm: Systematic Review and Meta-Analysis of Updated Peri-Operative and Long Term Data of Randomised Controlled Trials. Eur. J. Vasc. Endovasc. Surg. 2020, 59, 385–397. [Google Scholar] [CrossRef]
- Cuozzo, S.; Miceli, F.; Brizzi, V.; Marzano, A.; Gattuso, R.; Sbarigia, E. Ovation Alto Stent-Graft Implantation in Patient with TASC D Aorto-Iliac Lesion and Coexistent AAA—Case Report. Ann. Vasc. Surg. Brief Rep. Innov. 2023, 3, 100156. [Google Scholar] [CrossRef]
- Girolamo, A.D.; Ascione, M.; Miceli, F.; Mohseni, A.; Pranteda, C.; Sirignano, P.; Taurino, M.; di Marzo, L.; Mansour, W. Clinical and Radiological Outcomes of Accessory Renal Artery Exclusion during Endovascular Repair of Abdominal Aortic Aneurysms. Diagnostics 2024, 14, 864. [Google Scholar] [CrossRef]
- van Marrewijk, C.; Buth, J.; Harris, P.L.; Norgren, L.; Nevelsteen, A.; Wyatt, M.G. Significance of Endoleaks after Endovascular Repair of Abdominal Aortic Aneurysms: The EUROSTAR Experience. J. Vasc. Surg. 2002, 35, 461–473. [Google Scholar] [CrossRef]
- Ultee, K.H.J.; Büttner, S.; Huurman, R.; Bastos Gonçalves, F.; Hoeks, S.E.; Bramer, W.M.; Schermerhorn, M.L.; Verhagen, H.J.M. Editor’s Choice—Systematic Review and Meta-Analysis of the Outcome of Treatment for Type II Endoleak Following Endovascular Aneurysm Repair. Eur. J. Vasc. Endovasc. Surg. 2018, 56, 794–807. [Google Scholar] [CrossRef]
- Mozes, G.D.; Pather, K.; Oderich, G.S.; Mirza, A.; Colglazier, J.J.; Shuja, F.; Mendes, B.C.; Kalra, M.; Bjarnason, H.; Bower, T.C.; et al. Outcomes of Onyx® Embolization of Type II Endoleaks After Endovascular Repair of Abdominal Aortic Aneurysms. Ann. Vasc. Surg. 2020, 67, 223–231. [Google Scholar] [CrossRef]
- Scallan, O.; Kribs, S.; Power, A.H.; DeRose, G.; Duncan, A.; Dubois, L. Onyx versus Coil Embolization for the Treatment of Type II Endoleaks. J. Vasc. Surg. 2021, 73, 1966–1972. [Google Scholar] [CrossRef]
- Wanhainen, A.; Van Herzeele, I.; Bastos Goncalves, F.; Bellmunt Montoya, S.; Berard, X.; Boyle, J.R.; D’Oria, M.; Prendes, C.F.; Karkos, C.D.; Kazimierczak, A.; et al. Editor’s Choice—European Society for Vascular Surgery (ESVS) 2024 Clinical Practice Guidelines on the Management of Abdominal Aorto-Iliac Artery Aneurysms. Eur. J. Vasc. Endovasc. Surg. 2024, 67, 192–331. [Google Scholar] [CrossRef]
- Hobo, R.; Buth, J. Secondary Interventions Following Endovascular Abdominal Aortic Aneurysm Repair Using Current Endografts. A EUROSTAR Report. J. Vasc. Surg. 2006, 43, 896–902.e1. [Google Scholar] [CrossRef]
- Hatzl, J.; Wang, V.; Hakimi, M.; Uhl, C.; Rengier, F.; Bruckner, T.; Böckler, D. Persisting Type 2 Endoleaks Following EVAR for AAA Are Associated With AAA Expansion. J. Endovasc. Ther. 2023, 30, 372–381. [Google Scholar] [CrossRef]
- Esposito, D.; Fargion, A.T.; Dorigo, W.; Melani, A.; Capone, A.; Di Domenico, R.; Villani, F.; Speziali, S.; Pratesi, C. Stability of the Aneurysmatic Sac Post-EVAR Could No Longer Be a Reliable Criterion of Healing. J. Cardiovasc. Surg. 2022, 63, 155–159. [Google Scholar] [CrossRef]
- Mathlouthi, A.; Guajardo, I.; Al-Nouri, O.; Malas, M.; Barleben, A. Prophylactic Aneurysm Embolization during EVAR Is Safe, Improves Sac Regression and Decreases the Incidence of Type II Endoleak. Ann. Vasc. Surg. 2021, 74, 36–41. [Google Scholar] [CrossRef]
- Niklas, N.; Malec, M.; Gutowski, P.; Kazimierczak, A.; Rynio, P. Effectiveness of Inferior Mesenteric Artery Embolization on Type II Endoleak-Related Complications after Endovascular Aortic Repair (EVAR): Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 11, 5491. [Google Scholar] [CrossRef]
- Taurino, M.; Visco, V.; Raffa, S.; Ricci, B.; Ruggiero, M.; Torrisi, M.R.; Fiorani, P. Matrix Metalloproteinase 9 Activity in Patients before and after Endovascular or Surgical Repair of Abdominal Aortic Aneurysms. Vascular 2004, 12, 312–317. [Google Scholar] [CrossRef]
- Sirignano, P.; Mangialardi, N.; Nespola, M.; Aloisi, F.; Orrico, M.; Ronchey, S.; Del Porto, F.; Taurino, M. Incidence and Fate of Refractory Type II Endoleak after EVAR: A Retrospective Experience of Two High-Volume Italian Centers. J. Pers. Med. 2022, 12, 339. [Google Scholar] [CrossRef]
- Burger, P.M.; Pradhan, A.D.; Dorresteijn, J.A.N.; Koudstaal, S.; Teraa, M.; de Borst, G.J.; van der Meer, M.G.; Mosterd, A.; Ridker, P.M.; Visseren, F.L.J. C-Reactive Protein and Risk of Cardiovascular Events and Mortality in Patients with Various Cardiovascular Disease Locations. Am. J. Cardiol. 2023, 197, 13–23. [Google Scholar] [CrossRef]
- Piazza, M.; Squizzato, F.; Miccoli, T.; Lepidi, S.; Menegolo, M.; Grego, F.; Antonello, M. Definition of Type II Endoleak Risk Based on Preoperative Anatomical Characteristics. J. Endovasc. Ther. 2017, 24, 566–572. [Google Scholar] [CrossRef]
- Jones, J.E.; Atkins, M.D.; Brewster, D.C.; Chung, T.K.; Kwolek, C.J.; LaMuraglia, G.M.; Hodgman, T.M.; Cambria, R.P. Persistent Type 2 Endoleak after Endovascular Repair of Abdominal Aortic Aneurysm Is Associated with Adverse Late Outcomes. J. Vasc. Surg. 2007, 46, 1–8. [Google Scholar] [CrossRef]
- El Batti, S.; Cochennec, F.; Roudot-Thoraval, F.; Becquemin, J.-P. Type II Endoleaks after Endovascular Repair of Abdominal Aortic Aneurysm Are Not Always a Benign Condition. J. Vasc. Surg. 2013, 57, 1291–1297. [Google Scholar] [CrossRef]
- Timaran, C.H.; Ohki, T.; Rhee, S.J.; Veith, F.J.; Gargiulo, N.J.; Toriumi, H.; Malas, M.B.; Suggs, W.D.; Wain, R.A.; Lipsitz, E.C. Predicting Aneurysm Enlargement in Patients with Persistent Type II Endoleaks. J. Vasc. Surg. 2004, 39, 1157–1162. [Google Scholar] [CrossRef]
- Goudeketting, S.R.; Fung Kon Jin, P.H.P.; Ünlü, Ç.; de Vries, J.-P.P.M. Systematic Review and Meta-Analysis of Elective and Urgent Late Open Conversion after Failed Endovascular Aneurysm Repair. J. Vasc. Surg. 2019, 70, 615–628.e7. [Google Scholar] [CrossRef]
- Arya, S.; Coleman, D.M.; Knepper, J.; Henke, P.K.; Upchurch, G.R.; Rectenwald, J.E.; Criado, E.; Eliason, J.L.; Gallagher, K.A. Outcomes After Late Explantation of Aortic Endografts Depend on Indication for Explantation. Ann. Vasc. Surg. 2013, 27, 865–873. [Google Scholar] [CrossRef]
- Perini, P.; Gargiulo, M.; Silingardi, R.; Bonardelli, S.; Bellosta, R.; Bonvini, S.; Michelagnoli, S.; Tusini, N.; Capelli, P.; Freyrie, A.; et al. Twenty-Two Year Multicentre Experience of Late Open Conversions after Endovascular Abdominal Aneurysm Repair. Eur. J. Vasc. Endovasc. Surg. 2020, 59, 757–765. [Google Scholar] [CrossRef]
- Perini, P.; Gargiulo, M.; Silingardi, R.; Piccinini, E.; Capelli, P.; Fontana, A.; Migliari, M.; Masi, G.; Scabini, M.; Tusini, N.; et al. Late Open Conversions after Endovascular Abdominal Aneurysm Repair in an Urgent Setting. J. Vasc. Surg. 2019, 69, 423–431. [Google Scholar] [CrossRef]
- Faccenna, F.; Alunno, A.; Castiglione, A.; Felli, M.M.G.; Venosi, S.; Gattuso, R.; Gossetti, B. Persistent Type II Endoleak: Two Cases of Successful Sacotomy. Ann. Vasc. Surg. 2013, 27, 240.e9–240.e11. [Google Scholar] [CrossRef] [PubMed]
- Nabi, D.; Murphy, E.H.; Pak, J.; Zarins, C.K. Open Surgical Repair after Failed Endovascular Aneurysm Repair: Is Endograft Removal Necessary? J. Vasc. Surg. 2009, 50, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Mangialardi, N.; Ronchey, S.; Orrico, M.; Serrao, E.; Alberti, V.; Fazzini, S.; Pecoraro, F.; Setacci, C. Surgical Conversion with Graft Salvage as a Definitive Treatment for Persistent Type II Endoleak Causing Sac Enlargement. J. Vasc. Surg. 2015, 62, 1437–1441. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Demographics and Comorbidities | Group A (EL2 Resolution After the First Embolization) Tot. n. 19 | Group B (Refractory EL2) Tot. n. 38 | p |
|---|---|---|---|
| Mean age (years ± SD) | 78.45 ± 7.02 | 77.43 ± 8.43 | 0.6540 |
| Male Sex (%, n) | 89.4% (17) | 92.1% (35) | >0.05 |
| Smoking history | 17 | 29 | |
| Arterial Hypertension (%, n) | 73.6% (14) | 84.2% (31) | 0.49 |
| Dyslipidemia (%, n) | 78.9% (15) | 84.2% (32) | 0.62 |
| CAD (%, n) | 26.3% (5) | 57.9% (22) | 0.002 |
| COPD (%, n) | 31.5% (6) | 23.7% (9) | 0.52 |
| Diabetes Mellitus (%, n) | 21.1% (4) | 15.8% (6) | 0.62 |
| Mean time of EL2 diagnosis (days ± SD) | 38 ± 5.2 | 192 ± 22.7 | 0.0001 |
| Standard EVAR (%, n) | 21.1% (4) | 73.7% (28) | 0.0001 |
| EVAR + IMA embolization (%, n) | 42.10% (8) | 5.26% (2) | 0.0013 |
| EVAR + Sac embolization (%, n) | 36.8% (7) | 21% (8) | 0.2199 |
| Gore Excluder (%, n) | 42% (8) | 50% (19) | 0.5825 |
| Endurant Medtronic (%, n) | 31.6% (6) | 26.3% (10) | 0.7583 |
| Incraft Cordis (%, n) | 15.8% (3) | 13.1% (5) | >0.05 |
| Endologix AFX (%, n) | 5.3% (1) | 5.3% (2) | >0.05 |
| Endologix Ovation (%, n) | 5.3% (1) | 5.3% (2) | >0.05 |
| Refractory Type 2 EL | ||
|---|---|---|
| Included Variables | p | Error Relative |
| Preoperative AAA diameter > 65 mm at first EVAR procedure | <0.001 | 12.95 (34–440.28) |
| Sac embolization | <0.001 | 0.182 (6.171–34) |
| IMA embolization | <0.001 | 0.055 (1.886–34) |
| Group A (EL2 Resolution After the First Embolization) Tot. n 19 | Group B (Refractory EL2) Tot. n 38 | p | |
|---|---|---|---|
| Mean aneurysm diameter before EVAR (mm) | 54.5 ± 12.06 | 73.3 ± 10.86 | 0.0001 |
| Mean aneurysm diameter at first embolization (mm) | 63.4 ± 11.23 | 83.3 ± 12.08 | 0.0001 |
| Mean aneurysm diameter at second intervention | - | 87.2 ± 10.04 |
| Aortic Branches | Group A (EL2 Resolution After the First Embolization) Tot. n. 19 | Group B (Refractory EL2) Tot. n. 38 | p |
|---|---|---|---|
| IMA patency (n, %) | 8 (42.10%) | 28 (73.68%) | 0.01 |
| Patent lumbar arteries (n, %) | |||
| 2 (10.52%) | 2 (5.26%) | 0.46 |
| 6 (31.57%) | 8 (21.05%) | 0.38 | |
| 4 (21.05%) | 24 (63.1%) | 0.002 | |
| 1 (5.26%) | 12 (31.57%) | 0.02 |
| First Embolization Procedure | Group A (EL2 Resolution After the First Embolization) Tot. n. 19 | Group B (Refractory EL2) Tot. n. 38 | p |
|---|---|---|---|
| IMA embolization | 3 (15.7%) | 7 (18.42%) | >0.05 |
| Sac embolization | 1 (5.26%) | 25 (68.9%) | 0.000082 |
| Lumbar embolization | 8 (42.1%) | 2 (5.26%) | 0.00001 |
| Sac + IMA embolization | 2 (10.5%) | 2 (5.26%) | - |
| Sac + lumbar embolization | 5 (26.3%) | 2 (5.26%) | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miceli, F.; Dajci, A.; Di Girolamo, A.; Nardis, P.; Ascione, M.; Cangiano, R.; Gattuso, R.; Sterpetti, A.; di Marzo, L.; Mansour, W. Early and Mid-Term Outcomes of Isolated Type 2 Endoleak Refractory to an Embolization Procedure. J. Clin. Med. 2025, 14, 502. https://doi.org/10.3390/jcm14020502
Miceli F, Dajci A, Di Girolamo A, Nardis P, Ascione M, Cangiano R, Gattuso R, Sterpetti A, di Marzo L, Mansour W. Early and Mid-Term Outcomes of Isolated Type 2 Endoleak Refractory to an Embolization Procedure. Journal of Clinical Medicine. 2025; 14(2):502. https://doi.org/10.3390/jcm14020502
Chicago/Turabian StyleMiceli, Francesca, Ada Dajci, Alessia Di Girolamo, Piergiorgio Nardis, Marta Ascione, Rocco Cangiano, Roberto Gattuso, Antonio Sterpetti, Luca di Marzo, and Wassim Mansour. 2025. "Early and Mid-Term Outcomes of Isolated Type 2 Endoleak Refractory to an Embolization Procedure" Journal of Clinical Medicine 14, no. 2: 502. https://doi.org/10.3390/jcm14020502
APA StyleMiceli, F., Dajci, A., Di Girolamo, A., Nardis, P., Ascione, M., Cangiano, R., Gattuso, R., Sterpetti, A., di Marzo, L., & Mansour, W. (2025). Early and Mid-Term Outcomes of Isolated Type 2 Endoleak Refractory to an Embolization Procedure. Journal of Clinical Medicine, 14(2), 502. https://doi.org/10.3390/jcm14020502








