Abstract
Objective: The purpose of this systematic review is to identify and appraise the evidence on the effectiveness of using wearable devices to promote physical activity and reduce pain in people with chronic musculoskeletal pain. Methods: Systematic searches of electronic databases PubMed, CINAHL, and Medline (Ovid) were undertaken for randomised control trials and observational studies of wearable-based interventions in patients with chronic musculoskeletal conditions. Result: Thirteen studies were included in this review. The methodological quality of the included articles was found to vary between moderate and high quality. Studies included patients with osteoarthritis hip/knee (number; n = 5), low back pain (n = 3), rheumatoid arthritis (n = 1), juvenile idiopathic arthritis (n = 1), inflammatory arthritis (n = 1), spondylarthritis (n = 1), and ankylosing spondylitis (n = 1). The intervention group of some of the studies included additional components associated with the use of wearable devices such as step or diet diary, motivational interviewing or counselling, goal setting, and multidimensional and tailored exercise programme interventions delivered in person, remotely, or in a hybrid format. Intervention duration ranged from 1 week to 28 weeks. There were no serious adverse events related to the use of wearables. Overall, evidence from this systematic review shows that wearable technology intervention was effective in increasing physical activity significantly, especially where extra components (counselling, coaching, prescribed physical activity, goal setting, physiotherapist) were used among clinical and non-clinical populations. However, no significant effect was found in pain reduction with the use of wearable devices. Conclusions: It is concluded that the use of wearable technology should be encouraged in patients with chronic musculoskeletal conditions. Additional research is needed, such as increasing the duration of the intervention, which may have an impact on pain.
1. Introduction
Musculoskeletal (MSK) conditions are one of the great contributors to pain and disability globally and have substantial individual, societal, and economic implications [1,2]. The broad term used to explain numerous conditions that are associated with bones, joints, ligaments, and soft tissues is MSK chronic pain conditions [3,4]. Pain is the primary sign of chronic musculoskeletal conditions. The International Association for the Study of Pain (IASP) defines pain as “an unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage” [5]. Also, pain was described by IASP as an individual experience affected by biological, psychological, and social factors [6]. However, a more relevant definition that describes the operational mechanism and clear benefit is chronic pain. Chronic pain is described as pain that has lasted three or more months or as pain persisting beyond the time of expected healing or treatment [7,8].
MSK conditions can be further explained as inflammatory and degenerative conditions that affect muscles, tendons, ligaments, joints, peripheral nerves, and supportive structures like intervertebral discs [9,10]. There are more than 100 different MSK chronic pain conditions, diseases, and syndromes that affect individuals’ ability to function and quality of life. The most common of these conditions are low back pain (LBP), osteoarthritis (OA), fibromyalgia (FM), and rheumatoid arthritis (RA) [11].
LBP is a significant public health problem, is the main cause of work absenteeism, and reduces the quality of life of patients [12]. LBP is a prevalent musculoskeletal disorder that affects up to 80% of individuals at some point in their lives. It is attributed to various etiologies, including degenerative changes, mechanical stress, and central pain sensitisation. Chronic LBP is associated with substantial disability and necessitates multimodal treatment strategies, such as exercise and patient education [13].
OA is the most common form of arthritis and chronic joint disease. OA is a degenerative joint disease characterised by cartilage degradation, joint pain, and stiffness, commonly affecting the knees and hips. It is assumed that nearly 8.75 million people in the U.K. had sought treatment in the community and hospitals for OA, and by 2035, 8.3 million people in the U.K. aged 45 years and over could have symptomatic knee OA. Management involves physical activity, weight management, and pharmacological interventions to reduce symptoms and improve joint [14].
Fibromyalgia (FM) is described or known as fibromyalgia syndrome (FMS) or fibrositis, the second most common form of non-articular rheumatism that is related to chronic musculoskeletal pain [15]. FM is a non-inflammatory syndrome characterised by widespread musculoskeletal pain. Its symptoms encompass severe fatigue, sleep disturbances, mood disruptions, and a myriad of other complaints. Effective management involves a combination of exercise, psychological therapies, and lifestyle modifications [13].
RA is an autoimmune disease characterised by systemic inflammation, predominantly affecting the joints. It results in pain, swelling, and, if left untreated, potential joint deformities. Contemporary treatment modalities include disease-modifying antirheumatic drugs (DMARDs), biological agents, and physical therapy [16].
About 2 billion people worldwide are affected by MSK conditions [1]. In the U.K., MSK conditions are treated in primary care, with referral to specialist clinics and secondary care for more complex management or specialist treatment and surgery such as rheumatology or joint replacement. Pain and disability from MSK conditions can also limit participation in physical activity (PA) [17]. In the U.K., the National Institute for Health and Care Excellence [18] guidelines recommend group- or individual-based PA for musculoskeletal patients with chronic pain.
Evidence has shown that physical activity is a core supporting factor in managing chronic conditions such as chronic musculoskeletal conditions [19,20]. The use of PA and exercise as an intervention has shown limited adverse events and is beneficial to most MSK conditions. Exercise and PA are effective in lowering LBP and enhancing physical function, which subsequently improves quality of life [21]. Participating in PA has numerous benefits, but ageing is associated with inactivity. Globally, 31% of adults are inactive as they do not meet the minimum recommended guideline of at least 30 min of moderate-intensity physical activity (MPA) on at least 5 days weekly, 20 min of vigorous-intensity physical activity (VPA) on at least 3 days weekly, or an equivalent combination achieving 600 metabolic equivalent minutes weekly [22].
Wearable technology is one of the commonly used methods to improve PA among people. The use of wearable activity tracker has increased tremendously among adults with various health problems, and this has led to increased growth of the global wearable activity tracker market over the past decade. The number of wearable activity trackers shipped globally between 2014 and 2020 has increased by approximately 144% [23]. These devices can be worn on the body or attached to the skin of a person to monitor that person’s actions continually and closely without impeding or restricting their movements [24]. The combination of wristwatch and fitness monitoring technology appeals to consumers seeking a multipurpose device that can satisfy a range of demands. Blood pressure, glucose, oxygen saturation, and sleep quality can all be measured by some of these wearable devices [25].
The growing potential of wearable technologies such as accelerometers, pedometers, fitness trackers, smartwatches, and smartphones offer new prospects to complement clinicians or health professionals in evaluating health status with objective assessments of physical activity, pain, and other health behaviours that are collected using a non-invasive method [26,27,28,29,30]. Using wearable devices promotes behavioural change techniques by encouraging self-monitoring, setting goals, and improving physical activity when used accurately [23]. The use of wearable devices has reduced the obstacles related to planned and structured forms of physical activity because of these lifestyle modifications [23]. Wearable devices give the user the ability to track their daily activities, which is extremely helpful in ensuring that they get sufficient exercise daily to maintain a healthy lifestyle [31]. The result is that it promotes self-esteem and confidence, social support, and self-efficacy [32].
Wearable technology can help people with chronic conditions such as fibromyalgia or arthritis identify triggers by tracking symptom flare-ups [33]. This enables improved decision-making and pain-relieving lifestyle changes. In supporting people with chronic pain conditions to consistently manage their pain, certain wearables come with medication reminders that make sure they take their prescribed medications on time [33].
Some wearables monitor physiological indicators (such as skin temperature, heart rate, or muscular tension) and offer biofeedback to assist users in identifying stress or discomfort before it becomes painful. Over time, reducing chronic pain may be possible through learning to control these signals [34].
Also, wearable devices have been shown to be linked with improved physiological outcomes, such as reduced BMI, reduced blood pressure, and other non-communicable diseases [22]. In a systematic review authored by [23], physical activity has been demonstrated to have antidepressant and anxiolytic effects. Wearable technology has the potential to improve psychosocial outcomes like anxiety and depression by increasing physical activity.
Wearable devices have the potential to improve the quality of treatment by allowing patients to live more independently, saving clinicians’ time, and monitoring patients remotely [35]. Additionally, the dosage of medications required for pain management can be reduced by using wearable devices [36].
The aim of our systematic review was to synthesise, identify, and appraise the evidence on the effectiveness of using wearable devices to promote physical activity and reduce pain in people with chronic musculoskeletal pain.
2. Methodology
A systematic review was conducted to identify and appraise published research on wearable devices for patients with chronic MSK conditions. The study more specifically addressed the main research questions: “Can using wearable device promote physical activity effectively in people with musculoskeletal chronic pain, and do using wearable devices reduce pain in patients with MSK chronic conditions”? This study included four interconnected processes, which consist of identification, screening, eligibility, and inclusion, to perform a comprehensive practical approach to the systematic review [37].
The literature review was conducted systematically in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) guidelines [38,39]. The related study protocol was registered ClinicalTrials.gov Protocol ID: 314666.
2.1. Search Strategy
A search strategy was identified as a predefined plan for searching for relevant evidence for the topic [40]. The literature search strategy gives the researcher a comprehensive understanding of the problem being examined from various sources that support the formulation of the research question and plan the project [41,42]. Three international databases, namely, Cumulative Index to Nursing and Allied Health Literature (CINAHL), MEDLINE (Ovid), and PubMed, were searched electronically from each database’s inception to December 2023.
Only articles published in English were included in the search, and no limit was placed on publication date. Search terms included a combination of keywords for musculoskeletal conditions, musculoskeletal diseases/disorder OR “Fibromyalgia”, OR “Low back pain” OR “Osteoarthritis” OR “Rheumatoid arthritis” OR “Arthritis”, AND “wearable technology”, OR “wearable device”, OR “wearable electronic device”. Truncation and Boolean operators were applied in the keywords that were used in the search, and the terms used are (“musculoskeletal conditions” OR “wearable technology” OR “physical activity”) AND “Exercise” AND “Chronic pain” AND (intervention OR randomised control trial OR randomised control trial OR RCT OR Observational studies), AND (MSK OR RA OR FMS OR FM).
The researcher examined the titles and abstracts of all the papers identified by the search strategy. The full article was retrieved for all papers that appeared as though they may meet the inclusion criteria. The reference list of identified papers was searched for further relevant publications. All retrieved articles were re-examined to ensure they met the inclusion criteria and to assess their methodological quality. Systematic reviews obtained from the search were also screened for potentially eligible studies.
2.2. Inclusion Criteria, Participants, and Type of Intervention
The inclusion criteria were employed using the PICOS (population, intervention, comparison, outcome, and study type) framework [43]. There were no age restrictions on study participants. Studies involving people who have had musculoskeletal chronic pain conditions/diseases for at least 3 months were included in the systematic review. The MSK chronic conditions include fibromyalgia, osteoarthritis, rheumatoid arthritis, inflammatory arthritis, low back pain, juvenile idiopathic arthritis, spondylarthritis, and ankylosing spondylitis.
Wearable technology to promote PA was the primary intervention considered. Interventions that used wearable devices such as Fitbits or commercial fitness trackers as core elements of the intervention and either as a stand-alone or in combination with education, self-management, or pharmacological treatment or rehabilitation (physiotherapist, or occupational therapist, or nurses, or cognitive and behavioural) programmes were included. These devices are easily worn and removed and do not require expert apparatus such as a harness or adhesive dressings. No restriction was placed on the healthcare professional delivering the intervention.
Randomised control trials (RCTs) and observational studies were included. Studies that assessed physical activity and pain as the outcome measures were included.
2.3. Exclusion Criteria
- Articles/reviews/studies not published in English languages;
- Studies of musculoskeletal pain in people with suspected cancer, pregnancy-related pain problems, palliative patients, and vulnerable patients (e.g., experienced trauma, cognitive impairment, dementia, terminal illness);
- Systematic review studies with meta-synthesis or protocols were excluded;
- Conference abstracts, scoping reviews, literature reviews, research letters or commentarial notes, or any other type of publication not being a report of a clinical study.
2.4. Data Extraction
The main researcher (KOE) undertook a systematic search, and then the results from the initial search were reviewed independently by the research team. The article titles and abstracts were examined by the main researcher to determine appropriate articles and information recorded. The reference lists of identified articles were checked for further relevant articles. All appropriate articles that met the inclusion criteria were retrieved, re-examined, and verified by the research team (JH and JL) to ensure they met inclusion criteria and to evaluate their methodological quality. Discrepancies were resolved by discussion between the three reviewers, and if agreement was not reached, a fourth reviewer (RL) was consulted. Reviewers ensured that guidelines on the data extraction and article types were included in the study. The following details were recorded for each included article:
- The country of origin;
- The participant study size and duration;
- Study design;
- Study objective;
- Inclusion and exclusion criteria;
- Interventions;
- Adverse events;
- Findings.
2.5. Assessment of Study Quality
The Mixed-Method Appraisal Tool (MMAT) version 2018 was used to assess studies across seven criteria [44,45]. The MMAT provides requirements for qualitative, quantitative, and mixed-methods investigations. The authors of MMAT (version 2018) advised against using an overall numerical score to represent the quality of the studies and instead provided a detailed presentation of the ratings of the criteria to represent the quality of the studies that were included. Each of the criteria was assessed on its presence (“Yes”) or absence (“No”) or unclear (“Can’t tell”). According to reports, the MMAT has moderate-to-high interrater reliability and content validity [45]. Three authors (K.E., J.H., and J.L.) critically appraised each study for quality and potential bias. Discrepancies were resolved by discussion until a consensus was reached. Regular review meetings were organised to make sure that protocol guidelines for the types of articles to be included in the study and data extraction were followed to produce a high-quality review.
3. Results
The search resulted in a total of 1407 articles, including systematic review and meta-analysis, with an additional four studies identified through reference checks. A total of 131 duplicated articles were removed, leaving 1276 articles. A total of 1219 articles were eliminated after screening of titles and abstracts. The remaining 57 articles were assessed using full text, after which 44 were excluded, with 9 articles being protocol, 10 articles using inappropriate outcome measures (e.g., postural control ability, transcutaneous electrical nerve stimulation (TENS), wearable devices mounted on a robot), and 25 articles not meeting the inclusion criteria. A total of 13 articles fulfilled the inclusion criteria and were included in this review (Figure 1: Flow diagram of selected studies).
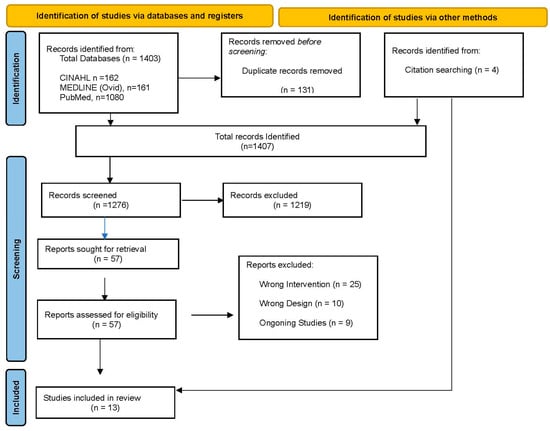
Figure 1.
PRISMA 2000 flow chart for study selection.
3.1. General Characteristics
The general characteristics of the included studies are shown in Table 1. The 13 study articles included 869 participants with a mean age ranging from 15.1 to 78.6 years across articles. All the studies were randomised control trials (RCTs) (n = 12, 92.31%), except one observational study (n = 1, 7.69%). Seven RCTs had a parallel control group [33,46,47,48,49,50,51], while four had a controlled delay group [52,53,54,55], and one had two intervention groups and a control group [56]. The geographical distribution of the studies was Canada (n = 5, 38.5%), Australia (n = 2, 15.4%), USA (n = 2, 15.4%), Sweden (n = 1, 7.7%), U.K. (n = 1, 7.7%), China (n = 1, 7.7%), and France (n = 1, 7.7%). The range of musculoskeletal chronic pain conditions included osteoarthritis with knee/hip (n = 6, 46.2%) [49,50,52,53,54,55], low back pain (n = 3, 23.1%) [46,47,48], rheumatoid arthritis (n = 1, 7.7%) [56], juvenile idiopathic arthritis (n = 1, 7.7%) [57], spondylarthritis (n = 1, 7.7%) [33], and ankylosing spondylitis (n = 1, 7.7%) [51]. The intervention period ranged from 1–12 weeks to 24–52 weeks. The follow-up duration varies from 1 to 36 weeks after intervention. The intervention format ranges from single to multidimensional, with support from health professionals.

Table 1.
Summary of each study included in the systematic review.
3.2. Quality of Studies
All studies were evaluated and allocated to an MMAT category. As shown in Table 2, the MMAT evaluation showed that each study had a precise research question and collected relevant data [33,46,47,48,49,50,51,52,53,54,55,56,57]. Randomisation processes were adequately performed in all the studies except for one study where the randomisation process was not clearly described [56]. Three studies did not include information details about participants’ adherence to wearing the assigned wearable devices during intervention [33,46,55]. The quality of the observational study was moderate, with two out of five criteria being satisfied (Table 3).

Table 2.
Mixed-Method Appraisal Tool (MMAT) for randomised control trial.

Table 3.
Mixed-Method Appraisal Tool (MMAT) for non-randomised control trial.
3.3. Wearable Technology Characteristics
Detailed description of the type and brand of wearable devices used in the studies was reported in all 13 studies (100%). Nine studies used the Fitbit®, with some variations regarding the type, with the Fitbit Flex 2® used in three studies [49,53,55], the Fitbit Flex ® in two studies [46,54], the Fitbit@ in two studies [47,52], the Fitbit zip in one study [56], and the Fitbit change HR in one study [48], respectively. Two studies used a Garmin Vivoft 4.0® [33,50]. One study used the Misfit Flash™ [57], and one study used the Medisana GmbH (Neuss, Germany) [51], respectively. Two studies also included a pedometer in the study, one for intervention [56] and one for control [48]. The devices were worn on the wrist in most of the studies (84.62% n = 11) [33,46,47,48,49,50,51,52,53,54,55]. In one study, the device was worn at multiple sites, including wrist, torso, and feet (7.7% n = 1) [57]. One study failed to mention how the wearable device was attached to the body [56]. All the wearable devices were used as a monitoring system for physical activity (n = 13 100%) [33,46,47,48,49,50,51,52,53,54,55,56,57]. Often, there was a requirement to link devices to either a smartphone or tablet with an app to display PA intensity, timing, and step count [33,46,47,48,49,50,51,52,53,54,55,56,57].
3.4. Intervention
The 13 studies used wearable technology in their intervention. Seven RCTs had a parallel control group [33,46,47,48,49,50,51], while four had a delayed control group [52,53,54,55], and one had two intervention groups and a control group [56]. Finally, an observational study only had one intervention group [57].
It should be noted that in the studies with the delayed control group design, the control group received the same multiple intervention components as the intervention group but at a delayed time [52,53,54]. For these studies, we only looked at the time point when the intervention group had already received the multiple intervention components, but the control group had not (Table 1).
The difference between intervention and control can be categorised into four situations: (1) multiple intervention components including wearable technology (WAT) [46,47,49,51,53,54,55,56]; (2) single intervention component of WAT only [33,52]; (3) one WAT vs. with another WAT [48]; and (4) one WAT plus extra components vs. the same WAT [50].
In the first situation, eight studies with multiple components, including WAT, were added to the intervention. These components included motivational interviewing/counselling [51,52,53,55], goal setting [47,49,57], tailored exercise programme [49,51], education/information booklet on PA [51,52,53,56], support from physiotherapist/occupational therapist [46,49,56], and telephone calls [47,52]. In the second situation, the only difference between the intervention group and the control group was wearable technology [33,52]. Again, it should be noted that in one study [52], the control group received the same WAT as the intervention group but at a delayed time. For this study, we only looked at the time point when the intervention group had already received it, but the control group had not (Table 1). The third situation includes one study in which the intervention group received the Fitbit while the control group received the pedometer [48]. The final situation includes one study in which both groups had the same wearable device, but the intervention group had extra components [50].
3.5. Outcome Measures (Physical Activity)
The main PA outcome measures considered are steps per day, activity counts per day, minutes per day spent performing light physical activity (LPA), moderate or vigorous physical activity (MVPA), and metabolic equivalent of task (MET). Eight of the studies [46,47,48,50,53,55,56] included data on step counts of physical activity, which was expressed as steps per day (Table 4). Six out of the eight studies showed that participants in the intervention group had a higher increase in their step count when compared with the control group measured at the end of intervention: 773 vs. 214 steps/day after 8 weeks intervention [52], 839 vs. 797 steps/day after 12 weeks intervention [53], 1148 vs. (−843) steps/day after 8 weeks intervention [55], 2649 vs. (−1585) step/day after 9 weeks intervention [46], 6 vs. (−220) steps/day after 24 weeks intervention [47], and 1432 vs. (−963) steps/day after 21 weeks intervention [56]. Among the six studies, the increase in PA was significant in the three studies with situation 1 intervention [46,55,56] but was not significant in the two studies with situation 2 intervention [33,52].

Table 4.
Physical activity outcomes.
However, two studies showed that participants in the control group had a higher step count when compared with the intervention group measured at the end of the intervention period: 2556 vs. 8241 steps/day after 12 weeks intervention [50] and 1724 vs. 1966 steps/day after 6 weeks intervention [48]. The main reasons for the different results in these two studies could be that both studies used wearable devices in both the intervention and the control groups, which means that the differences in PA might not be induced by WAT.
The metabolic equivalent of task (MET) was used by two studies to assess the amount and intensity of physical activity [49,57]. MET values are determined by dividing the work metabolic rate by the standard resting metabolic rate (RMR), which is set at 1.0 kcal·kg−1·h−1. An individual’s RMR at rest is measured as one MET [58]. There was a larger increase in MET in the intervention group (situation 1 intervention) as compared with the control group MET minutes/weekly at 12 weeks of intervention, but it was not statistically significant [49]. In the observational study, there was an MET minutes/daily increase in MET at 5 weeks [57].
Also, time spent on light physical activity (LPA) and moderate and vigorous physical activity (MVPA) was recorded in some studies. Two studies, both with situation 1 intervention, recorded time spent on LPA [46,47]. There was a significant improvement in time spent in LPA in the intervention group in two of the studies: 11.5 vs. 0.6 min [47] and 46 vs. (−58) minutes [46]. Also, six studies reported participants’ intensity and duration of MVPA. In four of the studies, the intervention group’s time spent on MVPA improved significantly when compared to the control group [46,52,54,55]. Three of the four studies employed intervention with situation 1, while one study employed intervention with situation 2, which suggests that both multiple intervention components and a single intervention component with WAT could improve MVPA. In two studies with situation 1 intervention, time spent on MVPA did not have any significant change in the intervention group, (−2.1) vs. (−4.4) [47] and 6.9 vs. 0 [53], in comparison with the control group. Also, the observational study improved post-intervention [57]. These results show that PA increased significantly when WAT was used with multiple components, but the increase was not significant in general when WAT was used as a single component.
3.6. Pain
Pain intensity was assessed by Knee Injury and OA Outcome Score (KOOS) in three studies [53,54,55], Patient Reported Outcomes Measurement Information System (PROMIS) was used in two studies [56,57], and Visual Analogue Scale (VAS) was used in two studies [46,57]. The others assessed pain intensity using the Hip injury and Osteoarthritis Outcome Score (HOOS) [49], Numerical Pain Scale (NRS) [47], McGill Pain Questionnaire [52], Arthritis Impact Measurement Scale (AIMS) [50], Physician global assessment (PhGA) [51], Pain Catastrophizing Scale (PCS) [46], Oswestry Disability Questionnaire [48], and Roland–Morris Disability Questionnaire (RMDQ) [47]. Average scores were not ascertained because of the different pain measures being used by the different studies reviewed. So, percentage change was used to compare across different studies.
In 11 of the 13 studies, pain intensity was assessed at baseline and post-intervention, while in 1 study, only baseline figures of pain were given [49] (Table 5). There was a significant improvement in pain reduction post-intervention in all studies using different pain measuring scales [46,47,48,50,51,52,53,54,55,56,57].

Table 5.
Pain outcomes.
Although the intervention group had pain reduction post-intervention, the percentage reduction was not significantly more when compared with control in 7 of the 10 randomised controlled studies: 28% vs. 22% at 25 weeks [47], 4.1% vs. 3.7% (intervention 1 vs. control), and 9% vs. 3.7% (intervention 2 vs. control) at 21 weeks [56]; 29% vs. 50% at 12 weeks [33], 18% vs. (−5%) at 9 weeks [52], 0.69% vs. 1.23% at 12 weeks [53], 7% vs. (−0.5%) at 2 months [55], and 19% vs. 13% at 6 weeks [48].
Two studies, both with situation 1 intervention, showed significantly more pain reduction in the intervention group when compared with the control: there was 75% pain reduction in the intervention group at 26 weeks compared to 40% in the control group [46], and 14% vs. 2% at 12 weeks [50], while another study found that pain reduction was significantly less in the intervention group: 37% vs. 56% at 16 weeks [51].
In summary, despite the percentage disparities, ten studies found improvement in pain reduction in both the intervention and the control groups, but the between-group differences were mostly not significant [33,46,47,48,50,51,53,54,55,56].
3.7. Adherence and Adverse Events
Overall, findings showed that participants were satisfied with the intervention, and wearable devices are a feasible intervention for patients with different musculoskeletal conditions [46,47,51,52,57].
Ten studies clearly described the procedure to monitor adherence to the intervention, which was measured consistently based on acceptance/satisfaction [52,53], wearable device usage [33,46,47,48,49,50,51,52,53,54,55,56,57] and completion of the exercise programme [46,52,53]. Their results showed that participants adhered to intervention procedures [46,47,49,51,52,53,54,55,56,57]. In four studies, there was some mentioning of monitoring participant adherence, but it was not clear how adherence was measured [33,48,50,55].
Although adherence was not clear in four of the studies, it is essential to establish that these studies showed consistency in methods used to monitor compliance with study goals. Participants demonstrated high levels of feasibility and acceptability [50]. They largely enjoyed having personalised daily steps feedback [50]. The website has high levels of usability on device usage and self-report [33,48], app analytic and regular measures of data collection [55], and engagement of participants during intervention [46,48,50,55].
Adverse events, consisting of pain/injury/illness/falls, were reported in 7 of 13 studies but were not found to be due to the use of wearable devices [51,52,53,54,55,56,57].
4. Discussion
Chronic musculoskeletal pain conditions in people are degenerative and are characterised by significant functional disability and emotional distress, which result in significantly reduced quality of life and long-term health conditions that affect most people globally [8]. There is a wide range of interventions for people with such conditions. PA remains an efficient intervention option in improving people’s quality of life as well as reducing pain [59]. This systematic review has identified and analysed evidence on the effects of interventions that used wearable technology as the main component to increase physical activity either as stand-alone or in combination with multifaceted wearable technology-related components such as goal setting and education in people with chronic musculoskeletal pain conditions. We found that wearable technology is an effective intervention to increase physical activity, especially when it is combined with such components. We also found that wearable technology intervention could reduce pain, but the amount of pain reduction was mostly not significantly different from that of the control group without wearable technology. Physical activity does not always have a positive impact on pain management. The type of physical activity, its duration, and chronic pain condition may all affect how effective physical activity interventions are for individuals with persistent musculoskeletal pain [60].
Wearable devices can offer valuable feedback, maintaining motivation and adherence to prescribed activity levels. However, people may not consistently participate in the recommended amounts of physical activity. Chronic pain is often multifaceted, involving a combination of biological, psychological, and social factors. In certain instances, merely increasing physical activity may not sufficiently address the underlying causes of pain [61].
The introduction of an education component with WAT improved PA among participants in the studies, and it was evident that education helped participants gain comprehensive knowledge of wearable device functions and usage and understand the benefits of PA [51,53,55]. It helped participants maintain exercise safety, build confidence, and gain satisfaction in their physical activities [49,50]. The education component enabled participants to successfully adopt skills to maintain behaviour change and to manage relapses, especially when the education component was delivered by health counsellors and coaches who helped in fostering social networks and providing professional feedback [50,53,55].
The inclusion of goal setting alongside WAT benefited participants by ensuring that they could receive continuous monitoring and feedback on their progress towards the PA target [56,57]. This was achieved by the researcher and participants working together to prescribe appropriate physical exercises [51], to set up strategic plans [50], to develop networking [47], and to provide professional feedback on their progress [46]. The WAT provides objective and timely analysed data on the number and duration of weekly PA session intensities of PA, based on the types of prescribed PA. It also improves engagement and empowerment of participants by providing easy access to personalised PA data [46,47]. Finally, the use of social networking, such as face-to-face or telephone counselling and professional supervision, provides support that enables participants to maintain their PA goals.
Our study found that pain levels were not significantly reduced by WAT intervention, even if WAT could improve PA significantly.
This is different from previous research suggesting that using wearable devices to promote frequent and regular physical activity can reduce chronic pain [62]. However, a previous systematic review conducted by [19] found that an increase in PA did not consistently bring about a change in self-reported pain scores. The reasons for this inconsistency may have arisen from the quality of the research, e.g., the various forms and intensity of physical activities examined in the studies and the duration of intervention. Moreover, most of the participants experienced mild–moderate pain rather than moderate–severe pain in the studies in this review.
The primary objectives of the reviewed studies are focused on increasing PA levels and intensity. The duration of the PA assessment, which ranged from 4 to 26 weeks, might not have been long enough for each participant’s pain to reduce, as the association between PA and pain is not linear [46,63]. An increase in PA might lead to an initial increase in pain in patients before it can cause pain reduction [64].
The pains experienced by patients were measured using subjective measures such as the VAS, the Numeric Pain Rating Scale, and summaries of self-administered questionnaires, which are gathered during meetings with participants and researchers. It is important to acknowledge that these assessments are susceptible to measurement error, social desirability bias, significant recall issues, and cognitive biases [62,63].
Also, participants presented different thresholds of pain associated with MSK conditions, such as juvenile idiopathic arthritis, rheumatoid arthritis, back pain, spondylarthritis, osteoarthritis, and ankylosing spondylitis. In the exploratory analyses, ref. [52] highlighted that those individuals in the PA intervention programme experience pain reduction among RA participants but not among systemic lupus erythematosus (SLE) participants. More research is required to evaluate how effectively various intervention guidelines work with those who have chronic musculoskeletal pain, even if wearables have the potential to encourage physical activity.
National Institute for Health and Care Excellence (NICE) guidelines recommend group- or individual-based physical activity for patients with musculoskeletal chronic pain. Currently, adults are expected to be involved in physical activity for at least 150 min of moderate-intensity activity, 75 min of vigorous activity, or a mixture of both [18,65,66]. Future research should try to closely follow the recommended guidelines of NICE (2021) and the Centres for Disease Control and Prevention (CDC) (2018).
4.1. Strength of the Review
The thirteen studies included in the review were published in the last six years, reflecting how these technologies are becoming important in health. Twelve of the thirteen studies included in this review were RCTs, which provided high-level evidence with low risk of biases. This systematic review included different population groups that range from adolescents to older adults. A total of six musculoskeletal conditions were covered by the 13 studies and in seven countries. This increased the generalisability of the results of the review. However, it is worth highlighting that most of the included studies only evaluated the effect of wearing devices over a short time scale, typically 3 months or less. It is therefore possible that the use of wearable devices over a longer period may lead to even greater effect sizes in terms of impact on pain or physical activity [23].
4.2. Limitations and Future Research
This systematic review has some limitations. The review is limited to quantitative methodologies, specifically controlled trials and observational studies. Although these methodologies are excellent at estimating the effectiveness of an intervention, they cannot explore patients’ experiences of using the devices. For this, qualitative research would need to be included, which was beyond the scope of this systematic review.
Studies included in this review had heterogeneous study designs, which consisted of various intervention components together with wearable devices such as telephone calls [47,52], use of a diary [56], and individual or group counselling [46,49,56]. Due to the use of these associated components in some studies being reviewed, the independent impact of wearable technology has been found difficult to establish, as these components alone could have modified participants’ physical activity lifestyle, leading to improved health outcomes.
This diversity in intervention design presents a challenge in isolating the specific impact of wearable devices on physical activity and pain outcomes. For example, while wearable devices may offer real-time tracking and feedback on activity levels, the effects of complementary elements like personalised counselling or exercise regimens could significantly influence the overall outcomes. Furthermore, educational booklets or other informational resources might help reinforce behavioural changes, making it difficult to determine whether improvements in physical activity and pain management are due primarily to the wearable device itself or to the combined effect of these multiple interventions.
Given this complexity, future research should aim to standardise intervention protocols to isolate the specific effects of wearable devices on these outcomes. Randomised controlled trials (RCTs) with a clear focus on wearable devices, either used alone or with minimal supplemental elements, would help in clarifying their true impact.
Most interventions spanned from 1 to 28 weeks, which may be inadequate to fully assess the long-term effects of wearable devices on chronic conditions. Extending the duration of these interventions is crucial to examine long-term adherence, sustained physical activity, and potential delayed effects on pain reduction.
The reviewed studies utilised a range of tools to measure physical activity (e.g., steps, METs, or MVPA minutes) and pain (e.g., VAS, KOOS, or PROMIS), hindering cross-study comparisons. Standardising outcome measures, particularly for physical activity and pain, is essential to facilitate meta-analyses and enhance the reliability of aggregated results.
The identified studies under review used a range of different wearable devices, and some of the wearables’ validity and reliability were not described in detail. The 13 studies included in this study used different types of wearable devices, which showed a lack of standardisation. Also, these different types of wearable devices have different positioning and algorithms used to verify the analysis of data. The studies did not fully describe the algorithms that were used. The algorithms are fundamental to finding a consistent procedure for the objective measurement of PA.
The 13 studies being reviewed covered a wide range of musculoskeletal conditions. There is a gap in research regarding fibromyalgia syndrome that needs to be studied because of the prevalence of fibromyalgia conditions among people with musculoskeletal chronic pain. Though the wearable device was found to have a favourable effect on individual physical activity in this systematic review, it is impossible to completely rule out the influence of publication bias because other wearable device studies may have had negative results that were not published.
5. Conclusions
In conclusion, we found that wearable technology is effective in increasing physical activity but does not significantly reduce musculoskeletal pain. This review highlighted evidence suggesting that wearable devices are acceptable to patients with MSK. The incidence of adverse events is minimal, and when they occur, they are minor. However, more research is needed to investigate the long-term effects of wearable technology and its use in other musculoskeletal conditions such as fibromyalgia syndrome. Also, how to effectively improve the effectiveness of wearable technology to reduce pain among MKS people is an important direction for future research.
Author Contributions
Conceptualisation, K.O.E., R.L., J.G.H. and J.L.; methodology, K.O.E., J.G.H. and J.L.; validation, R.L., J.G.H. and J.L.; formal analysis, K.O.E.; investigation, K.O.E.; resources, K.O.E.; data curation, K.O.E.; writing—original draft preparation, K.O.E.; writing—review and editing, K.O.E., J.G.H. and J.L.; supervision, R.L., J.G.H. and J.L; project administration, J.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the University of West London Ethics Committee, March 2022, and NHS Health Research Authority protocol code 314666, date of approval 30 January 2023.
Data Availability Statement
All data relevant to the study are included in the article.
Conflicts of Interest
There are no conflicts of interest with respect to the research, authorship, and publication of the article.
Abbreviations
| AMS | The Arthritis Impact Measurement Scale |
| AS | Ankylosing Spondylitis |
| CDC | Centers for Disease Control and Prevention |
| CINAHL | Cumulative Index to Nursing and Allied Health Literature |
| FM | Fibromyalgia |
| GBD | Global Burden of Diseases |
| HOOS | Hip injury and Osteoarthritis Outcome Score |
| JIA | Juvenile Idiopathic Arthritis |
| KOOS | Knee Injury and OA Outcome Score |
| LBP | Low back pain |
| LPA | Light physical activity |
| MET | Metabolic equivalent of task |
| MMAT | Mixed-Method Appraisal Tool |
| MSK | Musculoskeletal |
| MVPA | Moderate/vigorous physical activity |
| NICE | National Institute for Health and Care Excellence |
| NRA | Numerical Pain Scale |
| OA | Osteoarthritis |
| PA | Physical Activity |
| PCS | Pain Catastrophizing Scale |
| PhGA | Physician global assessment |
| PICOS | Population, intervention, comparison, outcome, and study |
| PRISMA | Preferred Reporting Items for Systematic Review and Meta-Analyses |
| PROMS | Patient Reported Outcomes Measurement Information System |
| RA | Rheumatoid arthritis, |
| RCT | Randomised controlled trial |
| RMDQ | Roland–Morris Disability Questionnaire |
| SLE | Systemic lupus erythematosus |
| SpA | Spondylarthritis |
| TENS | Transcutaneous electrical nerve stimulation |
| VAS | Visual Analogue Scale |
| VPA | Vigorous-intensity physical activity |
| WAT | Wearable Technology |
| WHO | World Health Organization |
References
- World Health Organization. Musculoskeletal Health. 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/musculoskeletal-conditions (accessed on 3 June 2024).
- GBD 2021 Other Musculoskeletal Disorders Collaborators. Global, regional, and national burden of other musculoskeletal disorders, 1990–2020, and projections to 2050: A systematic analysis of the Global Burden of Disease Study 2021. Lancet Rheumatol. 2023, 5, e670–e682. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.C.; Kang, S.; Benedetto, E.; Myers, H.; Blackburn, S.; Smith, S.; Dunn, K.M.; Hay, E.; Rees, J.; Beard, D.; et al. Development and initial cohort validation of the Arthritis Research UK Musculoskeletal Health Questionnaire (MSK-HQ) for use across musculoskeletal care pathways. BMJ Open 2016, 6, e012331. [Google Scholar] [CrossRef]
- Hewitt, S.; Sephton, R.; Yeowell, G. The Effectiveness of Digital Health Interventions in the Management of Musculoskeletal Conditions: Systematic Literature Review. J. Med. Internet Res. 2020, 22, e15617. [Google Scholar] [CrossRef]
- Raja, S.N.; Carr, D.B.; Cohen, M.; Finnerup, N.B.; Flor, H.; Gibson, S.; Keefe, F.J.; Mogil, J.S.; Ringkamp, M.; Sluka, K.A.; et al. The revised International Association for the Study of Pain definition of pain: Concepts, challenges, and compromises. Pain 2020, 161, 1976–1982. [Google Scholar] [CrossRef] [PubMed]
- Mallick-Searle, T.; Sharma, K.; Toal, P.; Gutman, A. Pain and Function in Chronic Musculoskeletal Pain-Treating the Whole Person. J. Multidiscip. Healthc. 2021, 14, 335–347. [Google Scholar] [CrossRef]
- Oliveira, J.S.; Pinheiro, M.B.; Fairhall, N.; Walsh, S.; Franks, T.C.; Kwok, W.; Bauman, A.; Sherrington, C. Evidence on Physical Activity and the Prevention of Frailty and Sarcopenia Among Older People: A Systematic Review to Inform the World Health Organization Physical Activity Guidelines. J. Phys. Act. Health 2020, 17, 1247–1258. [Google Scholar] [CrossRef]
- El-Tallawy, S.N.; Nalamasu, R.; Salem, G.I.; LeQuang, J.A.K.; Pergolizzi, J.V.; Christo, P.J. Management of Musculoskeletal Pain: An Update with Emphasis on Chronic Musculoskeletal Pain. Pain Ther. 2021, 10, 181–209. [Google Scholar] [CrossRef]
- Briggs, A.M.; Cross, M.J.; Hoy, D.G.; Sànchez-Riera, L.; Blyth, F.M.; Woolf, A.D.; March, L. Musculoskeletal Health Conditions Represent a Global Threat to Healthy Aging: A Report for the 2015 World Health Organization World Report on Ageing and Health. Gerontologist 2016, 56 (Suppl. 2), 243. [Google Scholar] [CrossRef]
- Tavakkol, R.; Kavi, E.; Hassanipour, S.; Rabiei, H.; Malakoutikhah, M. The global prevalence of musculoskeletal disorders among operating room personnel: A systematic review and meta-analysis. Clin. Epidemiol. Glob. Health 2020, 8, 1053–1061. [Google Scholar] [CrossRef]
- Williams, A.; Kamper, S.J.; Wiggers, J.H.; O’Brien, K.M.; Lee, H.; Wolfenden, L.; Yoong, S.L.; Robson, E.; McAuley, J.H.; Hartvigsen, J.; et al. Musculoskeletal conditions may increase the risk of chronic disease: A systematic review and meta-analysis of cohort studies. BMC Med. 2018, 16, 167. [Google Scholar] [CrossRef]
- Petit, A.; Begue, C.; Richard, I.; Roquelaure, Y. Factors influencing physiotherapists’ attitudes and beliefs toward chronic low back pain: Impact of a care network belonging. Physiother. Theory Pract. 2019, 35, 437–443. [Google Scholar] [CrossRef]
- De la Corte-Rodriguez, H.; Roman-Belmonte, J.M.; Resino-Luis, C.; Madrid-Gonzalez, J.; Rodriguez-Merchan, E.C. The Role of Physical Exercise in Chronic Musculoskeletal Pain: Best Medicine-A Narrative Review. Healthcare 2024, 12, 242. [Google Scholar] [CrossRef]
- Krakowski, P.; Rejniak, A.; Sobczyk, J.; Karpinski, R. Cartilage Integrity: A Review of Mechanical and Frictional Properties and Repair Approaches in Osteoarthritis. Healthcare 2024, 12, 1648. [Google Scholar] [CrossRef] [PubMed]
- Denche-Zamorano, A.; Pereira-Payo, D.; Collado-Mateo, D.; Adsuar-Sala, J.C.; Tomas-Carus, P.; Parraca, J.A. Physical Function, Self-Perceived Physical Fitness, Falls, Quality of Life and Degree of Disability According to Fear and Risk of Falling in Women with Fibromyalgia. J. Funct. Morphol. Kinesiol. 2024, 9, 174. [Google Scholar] [CrossRef] [PubMed]
- Jahid, M.; Khan, K.U.; Rehan-Ul-Haq Ahmed, R.S. Overview of Rheumatoid Arthritis and Scientific Understanding of the Disease. Mediterr. J. Rheumatol. 2023, 34, 284–291. [Google Scholar] [CrossRef]
- Nuesch, E.; Dieppe, P.; Reichenbach, S.; Williams, S.; Iff, S.; Juni, P. All cause and disease specific mortality in patients with knee or hip osteoarthritis: Population based cohort study. BMJ 2011, 342, d1165. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence, (NICE). Chronic Pain (Primary and Secondary) in Over 16s: Assessment of All Chronic Pain and Management of Chronic Primary Pain; NICE: London, UK, 2021; pp. 1–40. [Google Scholar]
- Geneen, L.J.; Moore, R.A.; Clarke, C.; Martin, D.; Colvin, L.A.; Smith, B.H. Physical activity and exercise for chronic pain in adults: An overview of Cochrane Reviews. Cochrane Database Syst. Rev. 2017, 4, CD011279. [Google Scholar] [PubMed]
- Jordan, K.P.; Kadam, U.T.; Hayward, R.; Porcheret, M.; Young, C.; Croft, P. Annual consultation prevalence of regional musculoskeletal problems in primary care: An observational study. BMC Musculoskelet. Disord. 2010, 11, 144. [Google Scholar] [CrossRef]
- Kirsch Micheletti, J.; Blafoss, R.; Sundstrup, E.; Bay, H.; Pastre, C.M.; Andersen, L.L. Association between lifestyle and musculoskeletal pain: Cross-sectional study among 10,000 adults from the general working population. BMC Musculoskelet. Disord. 2019, 20, 609. [Google Scholar] [CrossRef]
- Brickwood, K.; Watson, G.; O’Brien, J.; Williams, A.D. Consumer-Based Wearable Activity Trackers Increase Physical Activity Participation: Systematic Review and Meta-Analysis. JMIR Mhealth Uhealth 2019, 7, e11819. [Google Scholar] [CrossRef]
- Ferguson, T.; Olds, T.; Curtis, R.; Blake, H.; Crozier, A.J.; Dankiw, K.; Dumuid, D.; Kasai, D.; O’Connor, E.; Virgara, R.; et al. Effectiveness of wearable activity trackers to increase physical activity and improve health: A systematic review of systematic reviews and meta-analyses. Lancet Digit. Health 2022, 4, e615–e626. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, M.M.; Alon, G.; Pai, V.M.; Conroy, R.S. Wearable technologies for active living and rehabilitation: Current research challenges and future opportunities. J. Rehabil. Assist. Technol. Eng. 2019, 6, 2055668319839607. [Google Scholar] [CrossRef]
- Mahmood, A.; Kim, H.; Kedia, S.; Dillon, P. Wearable Activity Tracker Use and Physical Activity Among Informal Caregivers in the United States: Quantitative Study. JMIR Mhealth Uhealth 2022, 10, e40391. [Google Scholar] [CrossRef]
- Kos, M.; Pijnappel, E.N.; Buffart, L.M.; Balvers, B.R.; Kampshoff, C.S.; Wilmink, J.W.; van Laarhoven, H.W.M.; van Oijen, M.G.H. The association between wearable activity monitor metrics and performance status in oncology: A systematic review. Support Care Cancer 2021, 29, 7085–7099. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, M.; Donno, L.; Cimolin, V.; Galli, M. Cervical Range of Motion Assessment through Inertial Technology: A Validity and Reliability Study. Sensors 2023, 23, 6013. [Google Scholar] [CrossRef] [PubMed]
- Porciuncula, F.; Roto, A.V.; Kumar, D.; Davis, I.; Roy, S.; Walsh, C.J.; Awad, L.N. Wearable Movement Sensors for Rehabilitation: A Focused Review of Technological and Clinical Advances. PM&R 2018, 10, S220–S232. [Google Scholar]
- Saygin, D.; Rockette-Wagner, B.; Oddis, C.; Neiman, N.; Koontz, D.; Moghadam-Kia, S.; Aggarwal, R. Consumer-based activity trackers in evaluation of physical activity in myositis patients. Rheumatology 2022, 61, 2951–2958. [Google Scholar] [CrossRef] [PubMed]
- Degroote, L.; Hamerlinck, G.; Poels, K.; Maher, C.; Crombez, G.; De Bourdeaudhuij, I.; Vandendriessche, A.; Curtis, R.G.; DeSmet, A. Low-Cost Consumer-Based Trackers to Measure Physical Activity and Sleep Duration Among Adults in Free-Living Conditions: Validation Study. JMIR Mhealth Uhealth 2020, 8, e16674. [Google Scholar] [CrossRef] [PubMed]
- Haghi, M.; Thurow, K.; Stoll, R. Wearable Devices in Medical Internet of Things: Scientific Research and Commercially Available Devices. Healthc. Inform. Res. 2017, 23, 4–15. [Google Scholar] [CrossRef]
- Kandola, A.; Ashdown-Franks, G.; Hendrikse, J.; Sabiston, C.M.; Stubbs, B. Physical activity and depression: Towards understanding the antidepressant mechanisms of physical activity. Neurosci. Biobehav. Rev. 2019, 107, 525–539. [Google Scholar] [CrossRef]
- Labat, G.; Hayotte, M.; Bailly, L.; Fabre, R.; Brocq, O.; Gerus, P.; Breuil, V.; Fournier-Mehouas, M.; Zory, R.; D’Arripe-Longueville, F.; et al. Impact of a Wearable Activity Tracker on Disease Flares in Spondyloarthritis: A Randomized Controlled Trial. J. Rheumatol. 2022, 49, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Wang, Y.; Xu, D.; Nossent, J.; Pavlos, N.J.; Xu, J. Rheumatoid arthritis: Pathological mechanisms and modern pharmacologic therapies. Bone Res. 2018, 6, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Piwek, L.; Ellis, D.A.; Andrews, S.; Joinson, A. The Rise of Consumer Health Wearables: Promises and Barriers. PLoS Med. 2016, 13, e1001953. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.C. No exchange, same pain, no gain: Risk-reward of wearable healthcare disclosure of health personally identifiable information for enhanced pain treatment. Health Inform. J. 2019, 25, 1675–1691. [Google Scholar] [CrossRef] [PubMed]
- Petticrew, M.; Roberts, H. Systematic reviews—Do they ’work’ in informing decision-making around health inequalities? Health Econ. Policy Law 2008, 3, 197–211. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.D.; Quatman, C.E.; Manring, M.M.; Siston, R.A.; Flanigan, D.C. How to write a systematic review. Am. J. Sports Med. 2014, 42, 2761–2768. [Google Scholar] [CrossRef]
- Taylor, J.; Jesson, J.K.; Matheson, L.; Lacey, F.M. Doing Your Literature Review—Traditional and Systematic Techniques; SAGE Publications Ltd.: London, UK, 2011; p. 192. [Google Scholar]
- Bramer, W.M.; de Jonge, G.B.; Rethlefsen, M.L.; Mast, F.; Kleijnen, J. A systematic approach to searching: An efficient and complete method to develop literature searches. J. Med. Libr. Assoc. 2018, 106, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Whittaker, A. Succeeding in Literature Reviews and Research Project Plans for Nursing Students; Learning Matters: Exeter, UK, 2017. [Google Scholar]
- Methley, A.M.; Campbell, S.; Chew-Graham, C.; McNally, R.; Cheraghi-Sohi, S. PICO, PICOS and SPIDER: A comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv. Res. 2014, 14, 579. [Google Scholar] [CrossRef] [PubMed]
- Hong, Q.N.; Gonzalez-Reyes, A.; Pluye, P. Improving the usefulness of a tool for appraising the quality of qualitative, quantitative and mixed methods studies, the Mixed Methods Appraisal Tool (MMAT). J. Eval. Clin. Pract. 2018, 24, 459–467. [Google Scholar] [CrossRef]
- Pace, R.; Pluye, P.; Bartlett, G.; Macaulay, A.C.; Salsberg, J.; Jagosh, J.; Seller, R. Testing the reliability and efficiency of the pilot Mixed Methods Appraisal Tool (MMAT) for systematic mixed studies review. Int. J. Nurs. Stud. 2012, 49, 47–53. [Google Scholar] [CrossRef]
- Alzahrani, H.; Mackey, M.; Stamatakis, E.; Shirley, D. Wearables-based walking program in addition to usual physiotherapy care for the management of patients with low back pain at medium or high risk of chronicity: A pilot randomized controlled trial. PLoS ONE 2021, 16, e0256459. [Google Scholar] [CrossRef] [PubMed]
- Amorim, A.B.; Pappas, E.; Simic, M.; Ferreira, M.L.; Jennings, M.; Tiedemann, A.; Carvalho-e-Silva, A.P.; Caputo, E.; Kongsted, A.; Ferreirra, P.H. Integrating Mobile-health, health coaching, and physical activity to reduce the burden of chronic low back pain trial (IMPACT): A pilot randomised controlled trial. BMC Musculoskelet. Disord. 2019, 20, 71. [Google Scholar] [CrossRef]
- Gordon, R.; Bloxham, S. Influence of the Fitbit Charge HR on physical activity, aerobic fitness and disability in non-specific back pain participants. J. Sports Med. Phys. Fit. 2017, 57, 1669–1675. [Google Scholar] [CrossRef]
- Ostlind, E.; Eek, F.; Stigmar, K.; Sant’Anna, A.; Hansson, E.E. Promoting work ability with a wearable activity tracker in working age individuals with hip and/or knee osteoarthritis: A randomized controlled trial. BMC Musculoskelet. Disord. 2022, 23, 112. [Google Scholar] [CrossRef] [PubMed]
- Plumb Vilardaga, J.C.; Kelleher, S.A.; Diachina, A.; Riley, J.; Somers, T.J. Linking physical activity to personal values: Feasibility and acceptability randomized pilot of a behavioral intervention for older adults with osteoarthritis pain. Pilot Feasibility Stud. 2022, 8, 164–170. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, X.; Wang, W.; Shi, Y.; Ji, X.; Hu, L.; Wang, L.; Yin, Y.; Xie, S.; Zhu, J.; et al. Adherence, Efficacy, and Safety of Wearable Technology-Assisted Combined Home-Based Exercise in Chinese Patients with Ankylosing Spondylitis: Randomized Pilot Controlled Clinical Trial. J. Med. Internet Res. 2022, 24, e29703. [Google Scholar] [CrossRef]
- Li, L.C.; Feehan, L.M.; Xie, H.; Lu, N.; Shaw, C.; Gromala, D.; Aviña-Zubieta, J.A.; Koehn, C.; Hoens, A.M.; English, K.; et al. Efficacy of a Physical Activity Counseling Program with Use of a Wearable Tracker in People with Inflammatory Arthritis: A Randomized Controlled Trial. Arthritis Care Res. 2020, 72, 1755–1765. [Google Scholar] [CrossRef]
- Li, L.C.; Feehan, L.M.; Xie, H.; Lu, N.; Shaw, C.D.; Gromala, D.; Zhu, S.; Aviña-Zubieta, J.A.; Hoens, A.M.; Koehn, C.; et al. Effects of a 12-Week Multifaceted Wearable-Based Program for People with Knee Osteoarthritis: Randomized Controlled Trial. JMIR Mhealth Uhealth 2020, 8, e19116. [Google Scholar] [CrossRef]
- Li, L.C.; Sayre, E.C.; Xie, H.; Clayton, C.; Feehan, L.M. A Community-Based Physical Activity Counselling Program for People with Knee Osteoarthritis: Feasibility and Preliminary Efficacy of the Track-OA Study. JMIR Mhealth Uhealth 2017, 5, e86. [Google Scholar] [CrossRef]
- Li, L.C.; Sayre, E.C.; Xie, H.; Falck, R.S.; Best, J.R.; Liu-Ambrose, T.; Grewal, N.; Hoens, A.M.; Noonan, G.; Feehan, L.M. Efficacy of a Community-Based Technology-Enabled Physical Activity Counseling Program for People with Knee Osteoarthritis: Proof-of-Concept Study. J. Med. Internet Res. 2018, 20, e159. [Google Scholar] [CrossRef]
- Katz, P.; Margaretten, M.; Gregorich, S.; Trupin, L. Physical Activity to Reduce Fatigue in Rheumatoid Arthritis: A Randomized Controlled Trial. Arthritis Care Res. 2018, 70, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Heale, L.D.; Dover, S.; Goh, Y.I.; Maksymiuk, V.A.; Wells, G.D.; Feldman, B.M. A wearable activity tracker intervention for promoting physical activity in adolescents with juvenile idiopathic arthritis: A pilot study. Pediatr. Rheumatol. Online J. 2018, 16, 66–75. [Google Scholar] [CrossRef]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R., Jr.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. 2011 Compendium of Physical Activities: A second update of codes and MET values. Med. Sci. Sports Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef] [PubMed]
- Posadzki, P.; Pieper, D.; Bajpai, R.; Makaruk, H.; Könsgen, N.; Neuhaus, A.L.; Semwal, M. Exercise/physical activity and health outcomes: An overview of Cochrane systematic reviews. BMC Public Health 2020, 20, 1724. [Google Scholar] [CrossRef] [PubMed]
- Naugle, K.M.; Fillingim, R.B.; Riley, J.L., III. A meta-analytic review of the hypoalgesic effects of exercise. J. Pain 2012, 13, 1139–1150. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, I.; Pavic, J.; Filipovic, B.; Ozimec Vulinec, S.; Ilic, B.; Petek, D. Integrated Approach to Chronic Pain-The Role of Psychosocial Factors and Multidisciplinary Treatment: A Narrative Review. Int. J. Environ. Res. Public Health 2024, 21, 1135. [Google Scholar] [CrossRef]
- Leroux, A.; Rzasa-Lynn, R.; Crainiceanu, C.; Sharma, T. Wearable Devices: Current Status and Opportunities in Pain Assessment and Management. Digit. Biomark. 2021, 5, 89–102. [Google Scholar] [CrossRef]
- Naugle, K.M.; Riley, J.L., III. Self-reported physical activity predicts pain inhibitory and facilitatory function. Med. Sci. Sports Exerc. 2014, 46, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Lima, L.V.; Abner, T.S.S.; Sluka, K.A. Does exercise increase or decrease pain? Central mechanisms underlying these two phenomena. J. Physiol. 2017, 595, 4141–4150. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Action Plan on Physical Activity 2018–2030: More Active People for a Healthier World; WHO: Geneva, Switzerland, 2018; pp. 3–43. [Google Scholar]
- Centers for Disease Control and Prevention, (CDC). Physical Activity Guidelines for Americans; CDC: Atlanta, GA, USA, 2018.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).

