Sex-Differences in the Pattern of Comorbidities, Functional Independence, and Mortality in Elderly Inpatients: Evidence from the RePoSI Register
Abstract
1. Introduction
2. Material and Methods
Statistical Analysis
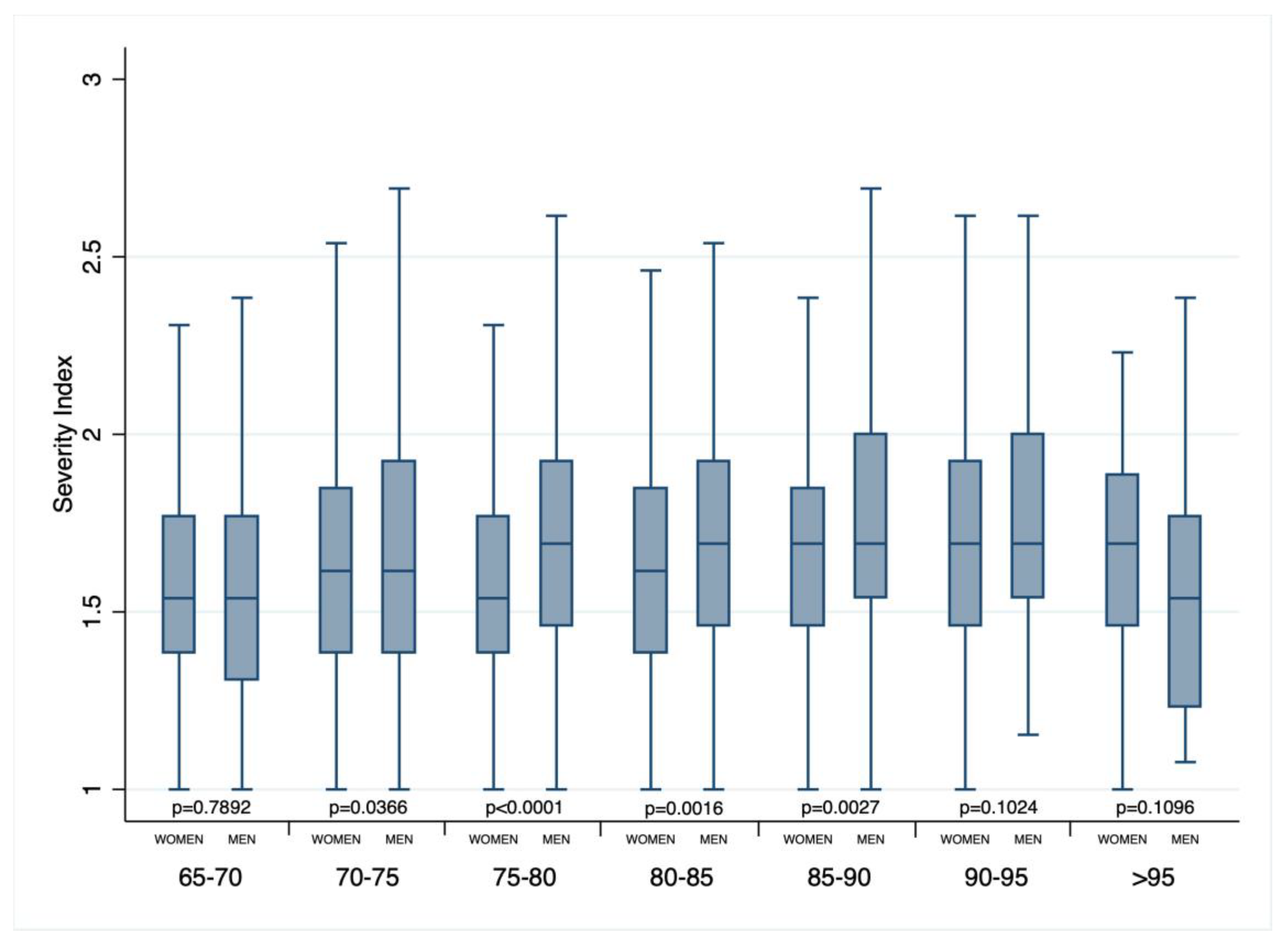
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Supplementary File 1Author Contributions
Funding
Conflicts of Interest
References
- The 2015 Aging Report European Commission. Available online: http://ec.europa.eu/economy_finance/publications/european_economy/2015/ee3_en.htm (accessed on 13 September 2018).
- Maas, A.H.; van der Schouw, Y.T.; Regitz-Zagrosek, V.; Swahn, E.; Appelman, Y.E.; Pasterkamp, G.; Ten Cate, H.; Nilsson, P.M.; Huisman, M.V.; Stam, H.C.; et al. Red alert for women’s heart: More research and knowledge on cardiovascular disease in women. Eur. Heart J. 2011, 32, 1362–1368. [Google Scholar] [CrossRef]
- Santalucia, P.; Franchi, C.; Djade, C.D.; Tettamanti, M.; Pasina, L.; Corrao, S.; Salerno, F.; Marengoni, A.; Marcucci, M.; Nobili, A.; et al. REPOSI Investigators. Gender difference in drug use in hospitalized elderly patients. Eur. J. Intern. Med. 2015, 26, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Corrao, S.; Santalucia, P.; Argano, C.; Djade, C.D.; Barone, E.; Tettamanti, M.; Pasina, L.; Franchi, C.; Kamal Eldin, T.; Marengoni, A.; et al. REPOSI Investigators. Gender-differences in disease distribution and outcome in hospitalized elderly: Data from the REPOSI study. Eur. J. Intern. Med. 2014, 25, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Nobili, A.; Licata, G.; Salerno, F.; Pasina, L.; Tettamanti, M.; Franchi, C.; De Vittorio, L.; Marengoni, A.; Corrao, S.; Iorio, A.; et al. SIMI Investigators. Polypharmacy, length of hospital stay, and in-hospital mortality among elderly patients in internal medicine wards. The REPOSI study. Eur. J. Clin. Pharmacol. 2011, 67, 507–519. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gotzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. BMJ 2007, 335, 806–808. [Google Scholar] [CrossRef] [PubMed]
- Katzman, R.; Brown, T.; Fuld, P.; Peck, A.; Schechter, R.; Schimmel, H. Validation of a short Orientation-Memory-Concentration Test of cognitive impairment. Am. J. Psychiatry 1983, 140, 734–739. [Google Scholar] [PubMed]
- Hickie, C.; Snowdon, J. Depression scales for the elderly: GDS, Gilleard, Zung. Clin. Gerontol. J. Aging Ment. Health 1987, 6, 51–53. [Google Scholar]
- Mahoney, F.I.; Barthel, D.W. Functional evaluation: The Barthel Index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar]
- Linn, B.S.; Linn, M.W.; Gurel, L. Cumulative illness rating scale. J. Am. Geriatr. Soc. 1968, 16, 622–626. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Hosmer, D.W.; Lemeshow, S. Applied Logistic Regression; J Wiley: New York, NY, USA, 2002. [Google Scholar]
- Global Population Ageing: Peril or Promise? World Economic Forum, 2012. Available online: http://demographic-challenge.com/files/downloads/6c59e8722eec82f7ffa0f1158d0f4e59/ageingbook_010612.pdf (accessed on 3 October 2018).
- Pan, A.; Okereke, O.I.; Sun, Q.; Logroscino, G.; Manson, J.E.; Willett, W.C.; Ascherio, A.; Hu, F.B.; Rexrode, K.M. Depression and incident stroke in women. Stroke 2011, 42, 2770–2775. [Google Scholar] [CrossRef] [PubMed]
- Whang, W.; Kubzansky, L.D.; Kawachi, I.; Rexrode, K.M.; Kroenke, C.H.; Glynn, R.J.; Garan, H.; Albert, C.M. Depression and risk of sudden cardiac death and coronary heart disease in women: Results from the Nurses’ Health Study. J. Am. Coll. Cardiol. 2009, 53, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Seshadri, S.; Wolf, P.A.; Beiser, A.; Au, R.; McNulty, K.; White, R.; D’Agostino, R.B. Lifetime risk of dementia and Alzheimer’s disease. Lifetime risk of dementia and Alzheimer’s disease. The impact of mortality on risk estimates in the Framingham Study. Neurology 1997, 49, 1498–1504. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.A.; Choudhury, K.R.; Rathakrishnan, B.G.; Marks, D.M.; Petrella, J.R.; Doraiswamy, P.M. Alzheimer’s Disease Neuroimaging Initiative. Marked gender differences in progression of mild cognitive impairment over 8 years. Alzheimers Dement. (N. Y.) 2015, 1, 103–110. [Google Scholar]
- Meyer, M.R.; Tschanz, J.T.; Norton, M.C.; Welsh-Bohmer, K.A.; Steffens, D.C.; Wyse, B.W.; Breitner, J.C. APOE genotype predicts when—Not whether—One is predisposed to develop Alzheimer disease. Nat. Genet. 1998, 19, 321–322. [Google Scholar] [CrossRef] [PubMed]
- Payami, H.; Zareparsi, S.; Montee, K.R.; Sexton, G.J.; Kaye, J.A.; Bird, T.D.; Yu, C.E.; Wijsman, E.M.; Heston, L.L.; Litt, M.; et al. Gender difference in apolipoprotein E-associated risk for familial Alzheimer disease: A possible clue to the higher incidence of Alzheimer disease in women. Am. J. Hum. Genet. 1996, 58, 803–811. [Google Scholar] [PubMed]
- Bretsky, P.M.; Buckwalter, J.G.; Seeman, T.E.; Miller, C.A.; Poirier, J.; Schellenberg, G.D.; Finch, C.E.; Henderson, V.W. Evidence for an interaction between apolipoprotein E genotype, gender, and Alzheimer disease. Alzheimer Dis. Assoc. Disord. 1999, 13, 216–221. [Google Scholar] [CrossRef]
- Fukumoto, N.; Fujii, T.; Combarros, O.; Kamboh, M.I.; Tsai, S.J.; Matsushita, S.; Nacmias, B.; Comings, D.E.; Arboleda, H.; Ingelsson, M.; et al. Sexually dimorphic effect of the Val66Met polymorphism of BDNF on susceptibility to Alzheimer’s disease: New data and meta-analysis. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2010, 153B, 235–242. [Google Scholar]
- Sohrabji, F.; Miranda, R.C.; Toran-Allerand, C.D. Identification of a putative estrogen response element in the gene encoding brain-derived neurotrophic factor. Proc. Natl. Acad. Sci. USA 1995, 92, 11110–11114. [Google Scholar] [CrossRef]
- Fried, L.P.; Bandeen-Roche, K.; Kasper, J.D. Guralnik JM for the Women’s Health and Aging Study collaborative research group. J. Clin. Epidemiol. 1999, 52, 27–37. [Google Scholar] [CrossRef]
- Camp, P.G.; Goring, S.M. Gender and the diagnosis, management, and surveillance of chronic obstructive pulmonary disease. Proc. Am. Thracic Soc. 2007, 4, 686–691. [Google Scholar] [CrossRef]
- Dransfield, M.T.; Washko, G.R.; Foreman, M.G.; Estepar, R.S.; Reilly, J. Gender differences in the severity of CT emphysema in COPD. Chest 2007, 132, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Molarius, A.; Janson, S. Self-rated health, chronic diseases, and symptoms among middle-aged and elderly men and women. J. Clin. Epidemiol. 2002, 55, 364–370. [Google Scholar] [CrossRef]
- Orshal, J.M.; Khalil, R.A. Gender, sex hormones, and vascular tone. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 286, R233–R249. [Google Scholar] [CrossRef]
- Oliver, J.E.; Silman, A.J. Why are women predisposed to autoimmune rheumatic diseases? Arthritis Res. Ther. 2009, 11, 252–260. [Google Scholar] [CrossRef]
- Cutolo, M.; Cappellino, S.; Sulli, A.; Serioli, B.; Secchi, M.E.; Villaggio, B.; Straub, R.H. Estrogens and autoimmune diseases. Ann. N. Y. Acad. Sci. 2006, 1089, 538–547. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Shin, H.J.; Ding, E.L.; van Dam, R.M. Adiponectin levels and risk of type 2 diabetes: A systematic review and meta-analysis. JAMA 2009, 302, 179–188. [Google Scholar] [CrossRef]
- Regitz-Zagrosek, V.; Karigas, G. Mechanistic pathways of sex differences in cardiovascular disease. Physiol. Rev. 2017, 97, 1–37. [Google Scholar] [CrossRef]
- Bello, N.; Mosca, L. Epidemiology of coronary heart disease in women. Prog. Cardiovasc. Dis. 2004, 46, 287–295. [Google Scholar] [CrossRef]
- Corrao, S.; Marengoni, A.; Natoli, G.; Nobili, A.; Mannucci, P.; Perticone, F. Clinical Severity, Age, and Sex Overcome Cardiometabolic Morbidities but Not Stroke as Predictors of Mortality in Elderly Inpatients: Data From the REgistro POliterapie Società Italiana di Medicina Interna Registry. J. Am. Geriatr. Soc. 2016, 64, 1737–1739. [Google Scholar] [CrossRef]
- Corrao, S.; Argano, C.; Natoli, G.; Nobili, A.; Corazza, G.R.; Mannucci, P.M.; Perticone, F. REPOSI Investigators. Disability, and not diabetes, is a strong predictor of mortality in oldest old patients hospitalized with pneumonia. Eur. J. Intern. Med. 2018, 54, 53–59. [Google Scholar] [CrossRef]
- Satish, S.; Freeman, D.H., Jr.; Ray, L.; Goodwin, J.S. The relationship between blood pressure and mortality in the oldest old. J. Am. Geriatr. Soc. 2001, 49, 367–374. [Google Scholar] [CrossRef]
- Odden, M.C.; Peralta, C.A.; Haan, M.N.; Covinsky, K. Rethinking the association of high blood pressure with mortality in elderly adults: The impact of frailty. Arch. Intern. Med. 2012, 172, 1162–1168. [Google Scholar] [CrossRef]





| Variables | Women | Men | p |
|---|---|---|---|
| N of subjects | 2401 | 2313 | / |
| Age (years) a | 80.3 (80–80.7) | 78.4 (78.1–78.7) | <0.0001 |
| Systolic blood pressure (mm Hg) a | 132.1 (131.2–132.9) | 131.7 (130.8–132.5) | 0.5840 |
| Diastolic blood pressure (mm Hg) a | 73.5 (73.0–73.9) | 73.5 (73.1–74.0) | 0.7881 |
| eGFR (mL/min) a | 58.1 (57.1–59.0) | 60.5 (59.5–61.6) | 0.0003 |
| Hemoglobin (g/L) a | 11.6 (11.5–11.7) | 12.1 (12.0–12.2) | <0.0001 |
| Body mass index (kg/m2) a | 26.1 (25.8–26.3) | 25.8 (25.6–25.9) | 0.7880 |
| Barthel index (disability) a | 74.5 (73.3–75.8) | 80.6 (79.5–81.8) | <0.0001 |
| Barthel index ≤ 40 (disability) (%) | 17.1 | 11.8 | <0.0001 |
| Geriatric Depression Scale (GDS) a | 1.51 (1.45–1.56) | 1.27 (1.22–1.33) | <0.0001 |
| Comorbidity index (CIRS) a | 2.92 (2.84–2.99) | 3.18 (3.10–3.26) | <0.0001 |
| Severity index (CIRS) a | 1.64 (1.63–1.65) | 1.69 (1.68–1.71) | <0.0001 |
| Length of hospital stay (days) a | 11.8 (11.3–12.3) | 11.9 (11.3–12.6) | 0.2413 |
| In-hospital mortality (%) | 4.8 | 6.1 | 0.0545 |
| 3-month mortality (%) | 13.9 | 19.7 | <0.0001 |
| 1-year mortality (%) | 46.8 | 58.2 | 0.0001 |
| Fasting glucose (mmol/L) a | 125.6 (123.2–128.0) | 129.4 (126.6–132.2) | 0.2013 |
| Creatinine (μmol/L) a | 1.12 (1.09–1.16) | 1.41 (1.37–1.45) | <0.0001 |
| Cholesterol (mmol/L) a | 168.3 (166.1–170.4) | 150.6 (148.5–152.7) | <0.0001 |
| Cognitive impairment (SBT score) a | 9.74 (9.40–10.09) | 8.50 (8.17–8.84) | <0.0001 |
| Cognitive impairment (SBT score ≥ 10) (%) | 39.0 | 32.7 | <0.0001 |
| Number of drugs at hospital admission a | 5.6 (5.5–5.7) | 5.9 (5.8–6.0) | 0.0022 |
| Number of drugs at hospital discharge a | 7.6 (7.5–7.8) | 7.7 (7.5–7.9) | 0.2524 |
| Number of drugs at follow-up 3-month a | 6.5 (6.3–6.6) | 6.7 (6.5–6.9) | 0.0983 |
| Number of drugs at follow-up 1-year a | 6.4 (6.0–6.8) | 6.5 (6.1–7.0) | 0.5517 |
| Variables | In-Hospital Mortality | Mortality at 3-Months | Mortality at 1-Year | |||
|---|---|---|---|---|---|---|
| Odds Ratio (95% C.I.) | p = | Odds Ratio (95% C.I.) | p = | Odds Ratio (95% C.I.) | p = | |
| Age | 1.05 (1.03–1.07) | <0.0001 | 1.05 (1.04–1.07) | <0.0001 | 1.05 (1.03–1.07) | <0.0001 |
| Male sex | 1.42 (1.06–1.92) | 0.02 | 1.86 (1.49–2.32) | <0.0001 | 1.78 (1.35–2.34) | <0.0001 |
| Barthel Index ≤ 40 | 3.08 (2.24–4.24) | <0.0001 | 4.19 (3.28–5.37) | <0.0001 | 3.10 (2.17–4.42) | <0.0001 |
| Severity index (CIRS) | 2.01 (1.34–3.03) | 0.001 | - | - | 1.70 (1.12–2.59) | 0.013 |
| Fasting blood glucose | 1.16 (1.04–1.28) | 0.007 | - | - | 1.10 (0.98–1.22) | 0.106 |
| Systolic blood pressure | 0.98 (0.98–0.99) | <0.0001 | 0.99 (0.98–0.99) | <0.0001 | 0.99 (0.98–0.99) | <0.0001 |
| Chronic kidney disease | - | - | 1.15 (0.90–1.47) | 0.257 | 1.05 (0.74–1.49) | 0.767 |
| Malignancy | 1.35 (0.95–1.90) | 0.09 | 2.48 (1.96–3.14) | <0.0001 | 4.39 (3.06–6.30) | <0.0001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corrao, S.; Argano, C.; Natoli, G.; Nobili, A.; Corazza, G.R.; Mannucci, P.M.; Perticone, F.; on behalf of REPOSI Investigators. Sex-Differences in the Pattern of Comorbidities, Functional Independence, and Mortality in Elderly Inpatients: Evidence from the RePoSI Register. J. Clin. Med. 2019, 8, 81. https://doi.org/10.3390/jcm8010081
Corrao S, Argano C, Natoli G, Nobili A, Corazza GR, Mannucci PM, Perticone F, on behalf of REPOSI Investigators. Sex-Differences in the Pattern of Comorbidities, Functional Independence, and Mortality in Elderly Inpatients: Evidence from the RePoSI Register. Journal of Clinical Medicine. 2019; 8(1):81. https://doi.org/10.3390/jcm8010081
Chicago/Turabian StyleCorrao, Salvatore, Christiano Argano, Giuseppe Natoli, Alessandro Nobili, Gino Roberto Corazza, Pier Mannuccio Mannucci, Francesco Perticone, and on behalf of REPOSI Investigators. 2019. "Sex-Differences in the Pattern of Comorbidities, Functional Independence, and Mortality in Elderly Inpatients: Evidence from the RePoSI Register" Journal of Clinical Medicine 8, no. 1: 81. https://doi.org/10.3390/jcm8010081
APA StyleCorrao, S., Argano, C., Natoli, G., Nobili, A., Corazza, G. R., Mannucci, P. M., Perticone, F., & on behalf of REPOSI Investigators. (2019). Sex-Differences in the Pattern of Comorbidities, Functional Independence, and Mortality in Elderly Inpatients: Evidence from the RePoSI Register. Journal of Clinical Medicine, 8(1), 81. https://doi.org/10.3390/jcm8010081








