Superiority of a Treat-to-Target Strategy over Conventional Treatment with Fixed csDMARD and Corticosteroids: A Multi-Center Randomized Controlled Trial in RA Patients with an Inadequate Response to Conventional Synthetic DMARDs, and New Therapy with Certolizumab Pegol
Abstract
:1. Introduction
2. Methods
2.1. Patients
2.2. Procedures
2.3. Therapeutic Strategies
2.3.1. T2T csDMARD Strategy
2.3.2. T2T GC Strategy
- Small joints: metacarpophalangeal (MCP), proximal interphalangeal (PIP), metatarsophalangeal joints (MTP): distal interphalangeal joints (DIP), sternoclavicular joint, acromioclavicular joint, tarsus, and distal interphalangeal joints of the feet (IP): 10 mg triamcinolone;
- Intermediate joints: carpus, elbow, ankle: 20 mg triamcinolone with 0.5 mL 1% lidocaine; big joints: knee, shoulder, hip: 40 mg triamcinolone with 4 mL 1% lidocaine.
2.3.3. Fixed Regimen Strategy
2.4. Primary Objective
2.5. Anti-Drug Antibodies
2.6. Data and Statistical Methods
2.6.1. Power Calculation
2.6.2. Statistical Analyses
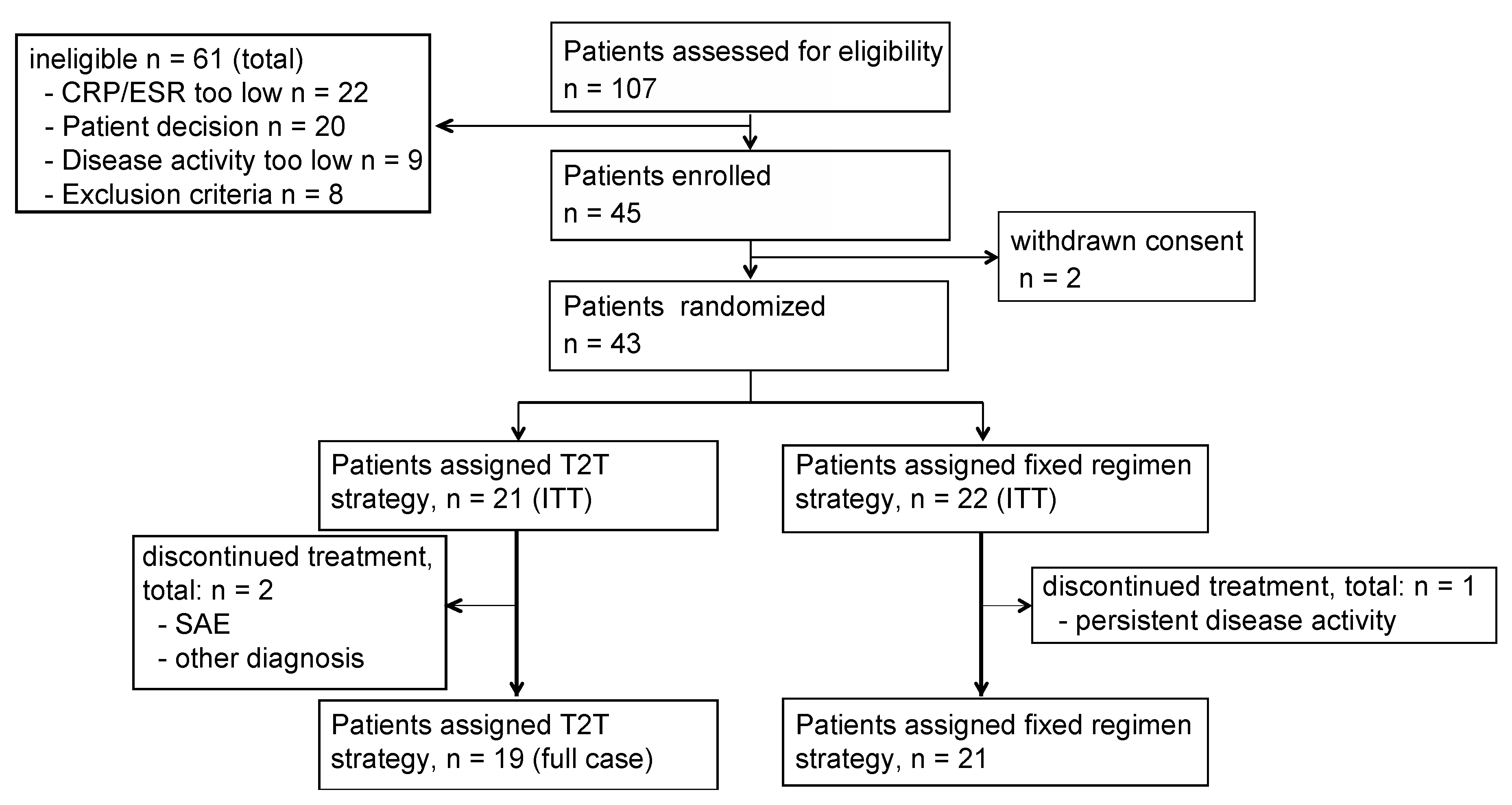
3. Results
3.1. Patient Demographics/Characteristics
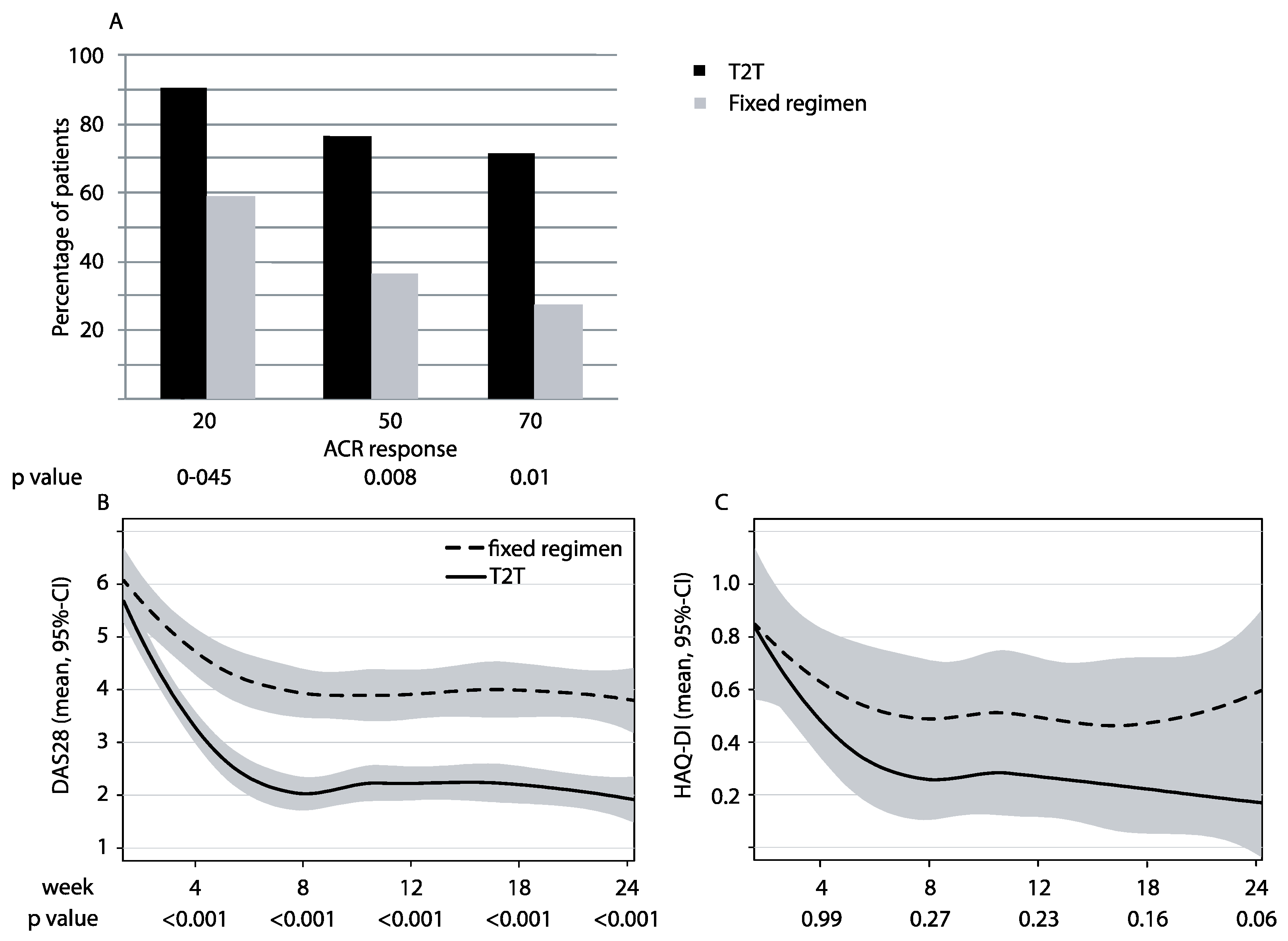
3.2. Clinical Responses
3.3. Patient-Related Outcomes
3.4. Adverse Events
3.5. Cumulative Steroid Dose
3.6. Therapeutic Changes of the csDMARD Protocol
3.7. ADA (Anti Drug Antibodies) Antibodies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Strand, V.; Cohen, S.; Schiff, M.; Weaver, A.; Fleischmann, R.; Cannon, G.; Fox, R.; Moreland, L.; Olsen, N.; Furst, D.; et al. Treatment of active rheumatoid arthritis with leflunomide compared with placebo and methotrexate. Leflunomide Rheumatoid Arthritis Investigators Group. Arch. Intern. Med. 1999, 159, 2542–2550. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, P.E.; van der Heijde, D.M.; St Clair, E.W.; Furst, D.E.; Breedveld, F.C.; Kalden, J.R.; Smolen, J.S.; Weisman, M.; Emery, P.; Feldmann, M.; et al. Infliximab and methotrexate in the treatment of rheumatoid arthritis. Anti-tumor necrosis factor trial in rheumatoid arthritis with concomitant therapy study group. N. Engl. J. Med. 2000, 343, 1594–1602. [Google Scholar] [CrossRef] [PubMed]
- Kremer, J.M.; Genant, H.K.; Moreland, L.W.; Russell, A.S.; Emery, P.; Abud-Mendoza, C.; Szechinski, J.; Li, T.; Ge, Z.; Becker, J.C.; et al. Effects of abatacept in patients with methotrexate-resistant active rheumatoid arthritis: A randomized trial. Ann. Intern. Med. 2006, 144, 865–876. [Google Scholar] [CrossRef] [PubMed]
- Keystone, E.; Emery, P.; Peterfy, C.G.; Tak, P.P.; Cohen, S.; Genoveses, M.C.; Dougados, M.; Burmester, G.R.; Greenwald, M.; Kvien, T.K.; et al. Rituximab inhibits structural joint damage in patients with rheumatoid arthritis with an inadequate response to tumour necrosis factor inhibitor therapies. Ann. Rheum. Dis. 2009, 68, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Nishimoto, N.; Hashimoto, J.; Miyasaka, N.; Yamamoto, K.; Kawai, S.; Takeuchi, T.; Murata, N.; van der Heijde, D.; Kishimoto, T. Study of active controlled monotherapy used for rheumatoid arthritis, an IL-6 inhibitor (SAMURAI): Evidence of clinical and radiographic benefit from an x ray reader-blinded randomised controlled trial of tocilizumab. Ann. Rheum. Dis. 2007, 66, 1162–1167. [Google Scholar] [CrossRef] [PubMed]
- Van der Heijde, D.; Tanaka, Y.; Fleischmann, R.; Keystone, E.; Kremer, J.; Zerbini, C.; Cardiel, M.H.; Cohen, S.; Nash, P.; Song, Y.W.; et al. Tofacitinib (CP-690,550) in patients with rheumatoid arthritis receiving methotrexate: Twelve-month data from a twenty-four-month phase III randomized radiographic study. Arthritis Rheum. 2013, 65, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.C.; Keystone, E.C.; van der Heijde, D.; Weinblatt, M.E.; Del Carmen Morales, L.; Reyes Gonzaga, J.; Yakushin, S.; Ishii, T.; Emoto, K.; Beattie, S.; et al. Baricitinib versus placebo or adalimumab in rheumatoid arthritis. N. Engl. J. Med. 2017, 376, 652–662. [Google Scholar] [CrossRef] [PubMed]
- Smolen, J.S.; Landewé, R.; Bijlsma, J.; Burmester, G.; Chatzidionysiou, K.; Dougados, M.; Nam, J.; Ramiro, S.; Voshaar, M.; van Vollenhoven, R.; et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann. Rheum. Dis. 2017, 76, 960–977. [Google Scholar] [CrossRef] [PubMed]
- Goekoop-Ruiterman, Y.P.; de Vries-Bouwstra, J.K.; Allaart, C.F.; van Zeben, D.; Kerstens, P.J.; Hazes, J.M.; Zwinderman, A.H.; Ronday, H.K.; Han, K.H.; Westedt, M.L.; et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): A randomized, controlled trial. Arthritis Rheum. 2005, 52, 3381–3390. [Google Scholar] [CrossRef] [PubMed]
- Verstappen, S.M.; Jacobs, J.W.; van der Veen, M.J.; Heurkens, A.H.; Schenk, Y.; ter Borg, E.J.; Blaauw, A.A.; Bijlsma, J.W. Intensive treatment with methotrexate in early rheumatoid arthritis: Aiming for remission. Computer assisted management in early rheumatoid arthritis (CAMERA, an open-label strategy trial). Ann. Rheum. Dis. 2007, 66, 1443–1449. [Google Scholar] [CrossRef] [PubMed]
- Dale, J.; Stirling, A.; Zhang, R.; Purves, D.; Foley, J.; Sambrook, M.; Conaghan, P.G.; van der Heijde, D.; McConnachie, A.; McInnes, I.B.; et al. Targeting ultrasound remission in early rheumatoid arthritis: The results of the TaSER study, a randomised clinical trial. Ann. Rheum. Dis. 2016, 75, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Haavardsholm, E.A.; Aga, A.B.; Olsen, I.C.; Lillegraven, S.; Hammer, H.B.; Uhliq, T.; Fremstad, H.; Madland, T.M.; Lexberg, Å.; Haukeland, H.; et al. Ultrasound in management of rheumatoid arthritis: ARCTIC randomised controlled strategy trial. BMJ 2016, 354, i4205. [Google Scholar] [CrossRef] [PubMed]
- Grigor, C.; Capell, H.; Stirling, A.; McMahon, A.D.; Lock, P.; Vallance, R.; Kincaid, W.; Porter, D. Effect of a treatment strategy of tight control for rheumatoid arthritis (the TICORA study): A single-blind randomised controlled trial. Lancet 2004, 364, 263–269. [Google Scholar] [CrossRef]
- Soubrier, M.; Puéchal, X.; Sibilia, J.; Mariette, X.; Meyer, O.; Combe, B.; Flipo, R.M.; Mulleman, D.; Berenbaum, F.; Zarnitsky, C.; et al. Evaluation of two strategies (initial methotrexate monotherapy vs. its combination with adalimumab) in management of early active rheumatoid arthritis: Data from the GUEPARD trial. Rheumatology (Oxford) 2009, 48, 1429–1434. [Google Scholar] [CrossRef] [PubMed]
- Visser, K.; van der Heijde, D. Optimal dosage and route of administration of methotrexate in rheumatoid arthritis: A systematic review of the literature. Ann. Rheum. Dis. 2009, 68, 1094–1099. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, R.; Cutolo, M.; Genovese, M.C.; Lee, E.B.; Kanik, K.S.; Sadis, S.; Connell, C.A.; Gruben, D.; Krishnaswami, S.; Wallenstein, G.; et al. Phase IIb dose-ranging study of the oral JAK inhibitor tofacitinib (CP-690,550) or adalimumab monotherapy versus placebo in patients with active rheumatoid arthritis with an inadequate response to disease-modifying antirheumatic drugs. Arthritis Rheum. 2012, 64, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Weinblatt, M.E.; Keystone, E.C.; Furst, D.E.; Moreland, L.W.; Weisman, M.H.; Birbara, C.A.; Teoh, L.A.; Fischkoff, S.A.; Chartash, E.K. Adalimumab, a fully human anti-tumor necrosis factor alpha monoclonal antibody, for the treatment of rheumatoid arthritis in patients taking concomitant methotrexate: The ARMADA trial. Arthritis Rheum. 2003, 48, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Strand, V.; Smolen, J.S.; van Vollenhoven, R.F.; Mease, P.; Burmester, G.R.; Hiepe, F.; Khanna, D.; Nikaï, E.; Coteur, G.; Schiff, M. Certolizumab pegol plus methotrexate provides broad relief from the burden of rheumatoid arthritis: Analysis of patient-reported outcomes from the RAPID 2 trial. Ann. Rheum. Dis. 2011, 70, 996–1002. [Google Scholar] [CrossRef] [PubMed]
- Weinblatt, M.E.; Fleischmann, R.; Huizinga, T.W.; Emery, P.; Pope, J.; Massarotti, E.M.; van Vollenhoven, R.F.; Wollenhaupt, J.; Bingham, C.O., 3rd; Duncan, B.; et al. Efficacy and safety of certolizumab pegol in a broad population of patients with active rheumatoid arthritis: Results from the REALISTIC phase IIIb study. Rheumatology (Oxford) 2012, 51, 2204–2214. [Google Scholar] [CrossRef] [PubMed]
- Keystone, E.; van der Heijde, D.; Mason, D., Jr.; Landewé, R.; Vollenhoven, R.V.; Combe, B.; Emery, P.; Strand, V.; Mease, P.; Desai, C.; et al. Certolizumab pegol plus methotrexate is significantly more effective than placebo plus methotrexate in active rheumatoid arthritis: Findings of a fifty-two-week, phase III, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum. 2008, 58, 3319–3329. [Google Scholar] [CrossRef] [PubMed]
- Smolen, J.S.; Emery, P.; Ferraccioli, G.F.; Samborski, W.; Berenbaum, F.; Davies, O.R.; Koetse, W.; Purcaru, O.; Bennett, B.; Burkhardt, H. Certolizumab pegol in rheumatoid arthritis patients with low to moderate activity: The CERTAIN double-blind, randomised, placebo-controlled trial. Ann. Rheum. Dis. 2015, 74, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., 3rd; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann. Rheum. Dis. 2010, 69, 1580–1588. [Google Scholar] [CrossRef] [PubMed]
- Van Schie, K.A.; Hart, M.H.; de Groot, E.R.; Kruithof, S.; Aarden, L.A.; Wolbink, G.J.; Rispens, T. Response to: ‘The antibody response against human and chimeric anti-TNF therapeutic antibodies primarily targets the TNF binding region’ by Rinaudo-Gaujous et al. Ann. Rheum. Dis. 2015, 74, e41. [Google Scholar] [CrossRef] [PubMed]
- Meikle, A.W.; Tyler, F.H. Potency and duration of action of glucocorticoids. Effects of hydrocortisone, prednisone and dexamethasone on human pituitary-adrenal function. Am. J. Med. 1977, 63, 200–207. [Google Scholar] [CrossRef]
- Maini, R.N.; Taylor, P.C.; Szechinski, J.; Pavelka, K.; Bröll, J.; Balint, G.; Emery, P.; Raemen, F.; Petersen, J.; Smolen, J.; et al. Double-blind randomized controlled clinical trial of the interleukin-6 receptor antagonist, tocilizumab, in European patients with rheumatoid arthritis who had an incomplete response to methotrexate. Arthritis Rheum. 2006, 54, 2817–2829. [Google Scholar] [CrossRef] [PubMed]
- Kremer, J.M.; Westhovens, R.; Leon, M.; Di Giorgio, E.; Alten, R.; Steinfeld, S.; Russell, A.; Dougados, M.; Emery, P.; Nuamah, I.F.; et al. Treatment of rheumatoid arthritis by selective inhibition of T-cell activation with fusion protein CTLA4Ig. N. Engl. J. Med. 2003, 349, 1907–1915. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.C.; Szczepanski, L.; Szechinski, J.; Filipowicz-Sosnowska, A.; Emery, P.; Close, D.R.; Stevens, R.M.; Shaw, T. Efficacy of B-cell-targeted therapy with rituximab in patients with rheumatoid arthritis. N. Engl. J. Med. 2004, 350, 2572–2581. [Google Scholar] [CrossRef] [PubMed]
- De Andrade, N.P.B.; da Silva Chakr, R.M.; Xavier, R.M.; Vieccili, D.; Correa, R.H.B.; de Oliveira Filho, C.M.; Brenol, C.V. Long-term outcomes of treat-to-target strategy in established rheumatoid arthritis: A daily practice prospective cohort study. Rheumatol. Int. 2017, 37, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Dickens, C.; Jackson, J.; Tomenson, B.; Hay, E.; Creed, F. Association of depression and rheumatoid arthritis. Psychosomatics 2003, 44, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Murphy, S.; Creed, F.; Jayson, M.I. Psychiatric disorder and illness behaviour in rheumatoid arthritis. Br. J. Rheumatol. 1988, 27, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Strand, V.; Mease, P.; Burmester, G.R.; Nikaï, E.; Coteur, G.; van Vollenhoven, R.; Combe, B.; Keystone, E.C.; Kavanaugh, A. Rapid and sustained improvements in health-related quality of life, fatigue, and other patient-reported outcomes in rheumatoid arthritis patients treated with certolizumab pegol plus methotrexate over 1 year: Results from the RAPID 1 randomized controlled trial. Arthritis Res. Ther. 2009, 11, R170. [Google Scholar] [PubMed]
- Bland, J.M.; Altman, D.G. Regression towards the mean. BMJ 1994, 308, 1499. [Google Scholar] [CrossRef] [PubMed]

| T2T (n = 21) | Fixed Regimen (n = 22) | |
|---|---|---|
| Age (a, mean) | 56.3 ± 15.4 | 56.8 ± 14.8 |
| Gender (% female) | 66.7% | 63.6% |
| BMI (Kg/m2, mean) | 28.6 ± 4.4 | 28.7 ± 5.6 |
| ACPA pos. (%) | 47.6% | 47.6% |
| RF pos. (%) | 66.7% | 81.0% |
| Erosive disease (%, defined by treating physician) | 26.3% | 30.0% |
| Disease duration (a, median, range) | 0.99, 3 months–10 years | 0.85, 3 months–18 years |
| Concomittant DMARD at baseline | MTX 10 mg/w, n = 2 MTX 15 mg/w, n = 9 MTX 20 mg/w, n = 4 MTX 25 mg/w, n = 3 Lef 20 mg/d, n = 3 | MTX 10 mg/w: n = 2 MTX 15 mg/w n = 6 MTX 20 mg/w n = 3 MTX 25 mg n = 5 MTX + HCQ, n = 2 15 mg/w + 200 mg/d; 20 mg/w + 400 mg/d SSZ n = 2 Lef n = 2 |
| Concomitant GC (mean dose *, number of patients) | 4.7 mg/d; n = 8 | 6.3 mg/d; n = 10 |
| Disease activity score (DAS 28) | 5.89 ± 0.98 | 6.16 ± 0.86 |
| Tender joint score (0–68) | 20.7 ± 10.3 | 23.2 ± 13.7 |
| Swollen joint score (0–66) | 18.9 ± 7.6 | 18.6 ± 10.9 |
| Pain score (0–100) | 65.3 ± 20.8 | 60.2 ± 21.0 |
| Patient global assessment | 70.1 ± 16.0 | 64.2 ± 16.9 |
| Physician global assessment (0–100) | 71.8 ± 8.8 | 67.0 ± 19.0 |
| C-reactive protein (mg/L) | 13.0 ± 16.2 | 17.1 ± 18.8 |
| Erythrocyte sedimentation rate (mm/h) | 28.7 ± 19.9 | 35.1 ± 25.2 |
| Health assessment questionnaire score * (0–3) | 0.84 ± 0.62 | 0.85 ± 0.64 |
| T2T | Fixed Regimen | p-Value | |
|---|---|---|---|
| Patient’s pain score | −53.34 | −50.51 | n.s. |
| Patient’s global assessment | −56.21 | −49.59 | n.s. |
| Physician’s global assessment | −64.62 | −31.74 | <0.001 |
| Tender joint count (68) | −19.67 | −9.9 | <0.001 |
| Swollen joint count (66) | −17.89 | −8.76 | <0.001 |
| ESR (mm/h) | −12.83 | −17.86 | <0.001 |
| CRP (mg/L) | −3.03 | −7.64 | <0.001 |
| HAQ-DI | −0.68 | −0.25 | n.s. |
| CDAI | −33.81 | −21.9 | <0.001 |
| Fixed Regimen | T2T | |
|---|---|---|
| Total (n) | 51 | 55 |
| Patients with AEs (n) | 16 | 18 |
| Intensity (mild/moderate/severe, n) | 49/1/1 | 50/4/1 |
| SAE (n) | 1 * | 2 ** |
| Related (Not, unlikely/possibly/ probably/definitely, n) | 22/3/21/4/1 | 33/3/19/0/1 |
| Cardiovascular (n) | 2 | 7 |
| Dermatological (n) | 6 | 4 |
| Gastrointestinal (n) | 3 | 4 |
| General (n) | 1 | 4 |
| Hepatology (n) | 2 | 1 |
| Infection | 10 | 13 |
| Injection site reaction to CZP (n) | 4 | 3 |
| Injury (n) | 5 | 2 |
| Joint injection reaction (n) | - | 3 |
| Musculoskeletal (n) | 4 | 3 |
| Neurological (n) | 11 | 10 |
| Ophthalmological (n) | 1 | 1 |
| Psychology (n) | 2 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mueller, R.B.; Spaeth, M.; von Restorff, C.; Ackermann, C.; Schulze-Koops, H.; von Kempis, J. Superiority of a Treat-to-Target Strategy over Conventional Treatment with Fixed csDMARD and Corticosteroids: A Multi-Center Randomized Controlled Trial in RA Patients with an Inadequate Response to Conventional Synthetic DMARDs, and New Therapy with Certolizumab Pegol. J. Clin. Med. 2019, 8, 302. https://doi.org/10.3390/jcm8030302
Mueller RB, Spaeth M, von Restorff C, Ackermann C, Schulze-Koops H, von Kempis J. Superiority of a Treat-to-Target Strategy over Conventional Treatment with Fixed csDMARD and Corticosteroids: A Multi-Center Randomized Controlled Trial in RA Patients with an Inadequate Response to Conventional Synthetic DMARDs, and New Therapy with Certolizumab Pegol. Journal of Clinical Medicine. 2019; 8(3):302. https://doi.org/10.3390/jcm8030302
Chicago/Turabian StyleMueller, Ruediger B., Michael Spaeth, Cord von Restorff, Christoph Ackermann, Hendrik Schulze-Koops, and Johannes von Kempis. 2019. "Superiority of a Treat-to-Target Strategy over Conventional Treatment with Fixed csDMARD and Corticosteroids: A Multi-Center Randomized Controlled Trial in RA Patients with an Inadequate Response to Conventional Synthetic DMARDs, and New Therapy with Certolizumab Pegol" Journal of Clinical Medicine 8, no. 3: 302. https://doi.org/10.3390/jcm8030302




