Changes in Spinal and Corticospinal Excitability in Patients with Chronic Ankle Instability: A Systematic Review with Meta-Analysis
Abstract
1. Introduction
2. Experimental Section
2.1. Eligibility Criteria
2.2. Literature Search Strategy
2.3. Study Selection and Data Extraction
2.4. Assessment of Methodological Quality
2.5. Data Analysis
3. Results
3.1. Search Findings
3.2. Methodological Quality
3.3. Study Characteristics
3.3.1. Study Design
3.3.2. Participants
3.3.3. Outcome Measures
3.4. Neural Excitability Meta-Analysis
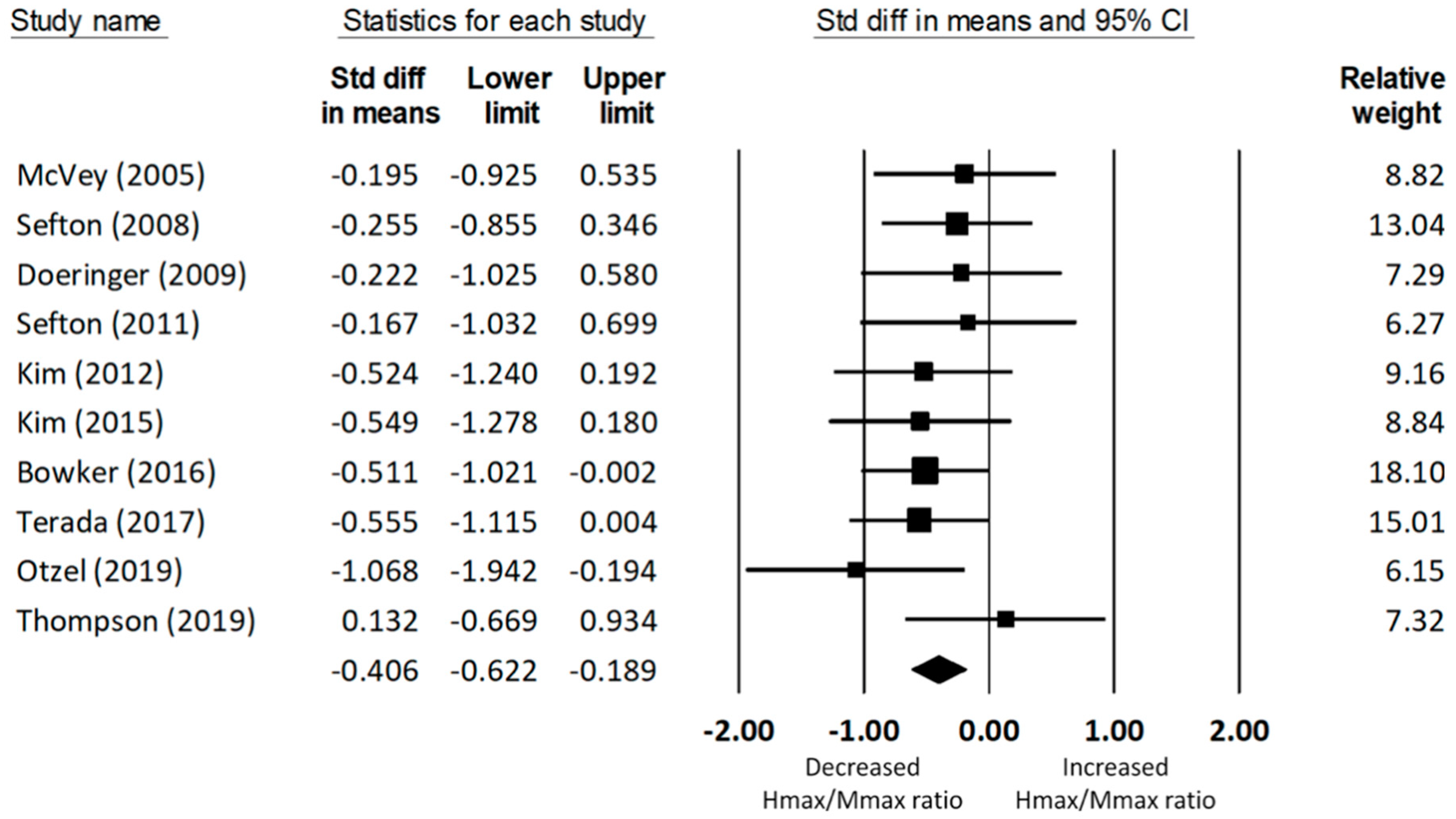
3.4.1. Soleus Hmax/Mmax Ratio
3.4.2. Fibular Longus Hmax/Mmax Ratio
3.4.3. Soleus Cortical Motor Threshold
3.4.4. Fibularis Longus Cortical Motor Threshold
3.5. Risk of Bias across Studies
4. Discussion
4.1. Spinal Reflex Excitability Associated with CAI
4.1.1. Functional Consequences of Reflex Inhibition in CAI Patients
4.1.2. Mechanisms of Reflex Inhibition in CAI Patients
4.1.3. Reduced Spinal Reflex Excitability as a Sign of “Arthrogenic Muscle Inhibition” in CAI Patients?
4.2. Corticospinal Excitability Associated with CAI
4.3. Limitations
4.4. Recommendations for Future Research
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Gribble, P.A.; Bleakley, C.M.; Caulfield, B.M.; Docherty, C.L.; Fourchet, F.; Fong, D.T.; Hertel, J.; Hiller, C.E.; Kaminski, T.W.; McKeon, P.O.; et al. Evidence review for the 2016 International Ankle Consortium consensus statement on the prevalence, impact and long-term consequences of lateral ankle sprains. Br. J. Sports Med. 2016, 50, 1496–1505. [Google Scholar] [CrossRef] [PubMed]
- Gribble, P.A.; Delahunt, E.; Bleakley, C.M.; Caulfield, B.; Docherty, C.L.; Fong, D.T.; Fourchet, F.; Hertel, J.; Hiller, C.E.; Kaminski, T.W.; et al. Selection criteria for patients with chronic ankle instability in controlled research: A position statement of the International Ankle Consortium. J. Athl. Train. 2014, 49, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Attenborough, A.S.; Sinclair, P.J.; Sharp, T.; Greene, A.; Stuelcken, M.; Smith, R.M.; Hiller, C.E. A snapshot of chronic ankle instability in a cohort of netball players. J. Sci. Med. Sport 2016, 19, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Tanen, L.; Docherty, C.L.; Van Der Pol, B.; Simon, J.; Schrader, J. Prevalence of chronic ankle instability in high school and division I athletes. Foot Ankle Spec. 2014, 7, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Attenborough, A.S.; Hiller, C.E.; Smith, R.M.; Stuelcken, M.; Greene, A.; Sinclair, P.J. Chronic ankle instability in sporting populations. Sports Med. 2014, 44, 1545–1556. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.; Hall, E.; Docherty, C. Prevalence of chronic ankle instability and associated symptoms in university dance majors: An exploratory study. J. Dance Med. Sci. 2014, 18, 178–184. [Google Scholar] [CrossRef]
- Hubbard-Turner, T.; Turner, M.J. Physical Activity Levels in College Students with Chronic Ankle Instability. J. Athl. Train. 2015, 50, 742–747. [Google Scholar] [CrossRef]
- Hubbard-Turner, T.; Wikstrom, E.A.; Guderian, S.; Turner, M.J. An Acute Lateral Ankle Sprain Significantly Decreases Physical Activity across the Lifespan. J. Sports Sci. Med. 2015, 14, 556–561. [Google Scholar] [PubMed]
- Houston, M.N.; Hoch, J.M.; Hoch, M.C. Patient-Reported Outcome Measures in Individuals with Chronic Ankle Instability: A Systematic Review. J. Athl. Train. 2015, 50, 1019–1033. [Google Scholar] [CrossRef]
- Lee, M.; Kwon, J.W.; Choi, W.J.; Lee, J.W. Comparison of Outcomes for Osteochondral Lesions of the Talus with and Without Chronic Lateral Ankle Instability. Foot Ankle Int. 2015, 36, 1050–1057. [Google Scholar] [CrossRef]
- Bischof, J.E.; Spritzer, C.E.; Caputo, A.M.; Easley, M.E.; DeOrio, J.K.; Nunley, J.A., 2nd; DeFrate, L.E. In vivo cartilage contact strains in patients with lateral ankle instability. J. Biomech. 2010, 43, 2561–2566. [Google Scholar] [CrossRef] [PubMed]
- Valderrabano, V.; Horisberger, M.; Russell, I.; Dougall, H.; Hintermann, B. Etiology of ankle osteoarthritis. Clin. Orthop. Relat. Res. 2009, 467, 1800–1806. [Google Scholar] [CrossRef] [PubMed]
- Valderrabano, V.; Hintermann, B.; Horisberger, M.; Fung, T.S. Ligamentous posttraumatic ankle osteoarthritis. Am. J. Sports Med. 2006, 34, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Hertel, J. Functional Anatomy, Pathomechanics, and Pathophysiology of Lateral Ankle Instability. J. Athl. Train. 2002, 37, 364–375. [Google Scholar] [PubMed]
- Hertel, J. Sensorimotor deficits with ankle sprains and chronic ankle instability. Clin. Sports Med. 2008, 27, 353–370. [Google Scholar] [CrossRef] [PubMed]
- Aman, J.E.; Elangovan, N.; Yeh, I.L.; Konczak, J. The effectiveness of proprioceptive training for improving motor function: A systematic review. Front. Hum. Neurosci. 2014, 8, 1075. [Google Scholar] [CrossRef] [PubMed]
- Schiftan, G.S.; Ross, L.A.; Hahne, A.J. The effectiveness of proprioceptive training in preventing ankle sprains in sporting populations: A systematic review and meta-analysis. J. Sci. Med. Sport 2015, 18, 238–244. [Google Scholar] [CrossRef]
- Rios, J.L.; Gorges, A.L.; dos Santos, M.J. Individuals with chronic ankle instability compensate for their ankle deficits using proximal musculature to maintain reduced postural sway while kicking a ball. Hum. Mov. Sci. 2015, 43, 33–44. [Google Scholar] [CrossRef]
- Terada, M.; Ball, L.M.; Pietrosimone, B.G.; Gribble, P.A. Altered visual focus on sensorimotor control in people with chronic ankle instability. J. Sports Sci. 2016, 34, 171–180. [Google Scholar] [CrossRef]
- Terada, M.; Bowker, S.; Thomas, A.C.; Pietrosimone, B.; Hiller, C.E.; Rice, M.S.; Gribble, P.A. Alterations in stride-to-stride variability during walking in individuals with chronic ankle instability. Hum. Mov. Sci. 2015, 40, 154–162. [Google Scholar] [CrossRef]
- Yen, S.C.; Corkery, M.B.; Donohoe, A.; Grogan, M.; Wu, Y.N. Feedback and Feedforward Control During Walking in Individuals with Chronic Ankle Instability. J. Orthop. Sports Phys. Ther. 2016, 46, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Hertel, J.; Olmsted-Kramer, L.C. Deficits in time-to-boundary measures of postural control with chronic ankle instability. Gait Posture 2007, 25, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Delahunt, E.; Monaghan, K.; Caulfield, B. Altered neuromuscular control and ankle joint kinematics during walking in subjects with functional instability of the ankle joint. Am. J. Sports Med. 2006, 34, 1970–1976. [Google Scholar] [CrossRef] [PubMed]
- Doherty, C.; Bleakley, C.; Hertel, J.; Caulfield, B.; Ryan, J.; Sweeney, K.; Patterson, M.R.; Delahunt, E. Lower Limb Interjoint Postural Coordination One Year after First-Time Lateral Ankle Sprain. Med. Sci. Sports Exerc. 2015, 47, 2398–2405. [Google Scholar] [CrossRef] [PubMed]
- Hass, C.J.; Bishop, M.D.; Doidge, D.; Wikstrom, E.A. Chronic ankle instability alters central organization of movement. Am. J. Sports Med. 2010, 38, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Terada, M.; Pietrosimone, B.G.; Gribble, P.A. Alterations in neuromuscular control at the knee in individuals with chronic ankle instability. J. Athl. Train. 2014, 49, 599–607. [Google Scholar] [CrossRef] [PubMed]
- McVey, E.D.; Palmieri, R.M.; Docherty, C.L.; Zinder, S.M.; Ingersoll, C.D. Arthrogenic muscle inhibition in the leg muscles of subjects exhibiting functional ankle instability. Foot Ankle Int. 2005, 26, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Palmieri-Smith, R.M.; Hopkins, J.T.; Brown, T.N. Peroneal activation deficits in persons with functional ankle instability. Am. J. Sports Med. 2009, 37, 982–988. [Google Scholar] [CrossRef] [PubMed]
- Harkey, M.; McLeod, M.M.; Terada, M.; Gribble, P.A.; Pietrosimone, B.G. Quadratic Association between Corticomotor and Spinal-Reflexive Excitability and Self-Reported Disability in Participants with Chronic Ankle Instability. J. Sport Rehabil. 2016, 25, 137–145. [Google Scholar] [CrossRef]
- Kim, K.M.; Hart, J.M.; Saliba, S.A.; Hertel, J. Relationships between self-reported ankle function and modulation of Hoffmann reflex in patients with chronic ankle instability. Phys. Ther. Sport 2016, 17, 63–68. [Google Scholar] [CrossRef]
- Kim, K.M.; Hart, J.M.; Saliba, S.A.; Hertel, J. Modulation of the Fibularis Longus Hoffmann Reflex and Postural Instability Associated with Chronic Ankle Instability. J. Athl. Train. 2016, 51, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Thompson, C.S.; Hiller, C.E.; Schabrun, S.M. Altered spinal-level sensorimotor control related to pain and perceived instability in people with chronic ankle instability. J. Sci. Med. Sport 2019, 22, 425–429. [Google Scholar] [CrossRef] [PubMed]
- Kosik, K.B.; Terada, M.; Drinkard, C.P.; McCann, R.S.; Gribble, P.A. Potential Corticomotor Plasticity in Those with and without Chronic Ankle Instability. Med. Sci. Sports Exerc. 2017, 49, 141–149. [Google Scholar] [CrossRef] [PubMed]
- McLeod, M.M.; Gribble, P.A.; Pietrosimone, B.G. Chronic Ankle Instability and Neural Excitability of the Lower Extremity. J. Athl. Train. 2015, 50, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Pietrosimone, B.G.; Gribble, P.A. Chronic ankle instability and corticomotor excitability of the fibularis longus muscle. J. Athl. Train. 2012, 47, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Terada, M.; Bowker, S.; Thomas, A.C.; Pietrosimone, B.; Hiller, C.E.; Gribble, P.A. Corticospinal Excitability and Inhibition of the Soleus in Individuals with Chronic Ankle Instability. PM R 2016, 8, 1090–1096. [Google Scholar] [CrossRef] [PubMed]
- Needle, A.R.; Lepley, A.S.; Grooms, D.R. Central Nervous System Adaptation after Ligamentous Injury: A Summary of Theories, Evidence, and Clinical Interpretation. Sports Med. 2017, 47, 1271–1288. [Google Scholar] [CrossRef]
- Doeringer, J.R.; Hoch, M.C.; Krause, B.A. The Effect of Focal Ankle Cooling on Spinal Reflex Activity in Individuals with Chronic Ankle Instability. Athl. Train. Sports Health Care 2009, 1, 59–64. [Google Scholar] [CrossRef]
- Doeringer, J.R.; Hoch, M.C.; Krause, B.A. Ice application effects on peroneus longus and tibialis anterior motoneuron excitability in subjects with functional ankle instability. Int. J. Neurosci. 2010, 120, 17–22. [Google Scholar] [CrossRef]
- Needle, A.R.; Palmer, J.A.; Kesar, T.M.; Binder-Macleod, S.A.; Swanik, C.B. Brain regulation of muscle tone in healthy and functionally unstable ankles. J. Sport Rehabil. 2013, 22, 202–211. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, R.M.; Ingersoll, C.D.; Hoffman, M.A. The hoffmann reflex: Methodologic considerations and applications for use in sports medicine and athletic training research. J. Athl. Train. 2004, 39, 268–277. [Google Scholar] [PubMed]
- Rossini, P.M.; Burke, D.; Chen, R.; Cohen, L.G.; Daskalakis, Z.; Di Iorio, R.; Di Lazzaro, V.; Ferreri, F.; Fitzgerald, P.B.; George, M.S.; et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin. Neurophysiol. 2015, 126, 1071–1107. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Ingersoll, C.D.; Hertel, J. Altered postural modulation of Hoffmann reflex in the soleus and fibularis longus associated with chronic ankle instability. J. Electromyogr. Kinesiol. 2012, 22, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef]
- Simpson, J.D.; Stewart, E.M.; Macias, D.M.; Chander, H.; Knight, A.C. Individuals with chronic ankle instability exhibit dynamic postural stability deficits and altered unilateral landing biomechanics: A systematic review. Phys. Ther. Sport 2018. [Google Scholar] [CrossRef]
- Moisan, G.; Descarreaux, M.; Cantin, V. Effects of chronic ankle instability on kinetics, kinematics and muscle activity during walking and running: A systematic review. Gait Posture 2017, 52, 381–399. [Google Scholar] [CrossRef]
- McKeon, J.M.; McKeon, P.O. Evaluation of joint position recognition measurement variables associated with chronic ankle instability: A meta-analysis. J. Athl. Train. 2012, 47, 444–456. [Google Scholar] [CrossRef]
- Munn, J.; Sullivan, S.J.; Schneiders, A.G. Evidence of sensorimotor deficits in functional ankle instability: A systematic review with meta-analysis. J. Sci. Med. Sport 2010, 13, 2–12. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: London, UK, 2013. [Google Scholar]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. Introduction to Meta-Analysis; Wiley: New York, NY, USA, 2009; p. 452. [Google Scholar]
- Kim, K.M.; Ingersoll, C.D.; Hertel, J. Facilitation of Hoffmann reflexes of ankle muscles in prone but not standing positions by focal ankle-joint cooling. J. Sport Rehabil. 2015, 24, 130–139. [Google Scholar] [CrossRef]
- Sefton, J.M.; Hicks-Little, C.A.; Hubbard, T.J.; Clemens, M.G.; Yengo, C.M.; Koceja, D.M.; Cordova, M.L. Segmental spinal reflex adaptations associated with chronic ankle instability. Arch. Phys. Med. Rehabil. 2008, 89, 1991–1995. [Google Scholar] [CrossRef] [PubMed]
- Sefton, J.M.; Yarar, C.; Hicks-Little, C.A.; Berry, J.W.; Cordova, M.L. Six weeks of balance training improves sensorimotor function in individuals with chronic ankle instability. J. Orthop. Sports Phys. Ther. 2011, 41, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Bowker, S.; Terada, M.; Thomas, A.C.; Pietrosimone, B.G.; Hiller, C.E.; Gribble, P.A. Neural Excitability and Joint Laxity in Chronic Ankle Instability, Coper, and Control Groups. J. Athl. Train. 2016, 51, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Otzel, D.M.; Hass, C.J.; Wikstrom, E.A.; Bishop, M.D.; Borsa, P.A.; Tillman, M.D. Motoneuron Function Does Not Change Following Whole-Body Vibration in Individuals with Chronic Ankle Instability. J. Sport Rehabil. 2019. [Google Scholar] [CrossRef] [PubMed]
- Terada, M.; Bowker, S.; Hiller, C.E.; Thomas, A.C.; Pietrosimone, B.; Gribble, P.A. Quantifying levels of function between different subgroups of chronic ankle instability. Scand. J. Med. Sci. Sports 2017, 27, 650–660. [Google Scholar] [CrossRef] [PubMed]
- Sefton, J.M.; Hicks-Little, C.A.; Hubbard, T.J.; Clemens, M.G.; Yengo, C.M.; Koceja, D.M.; Cordova, M.L. Sensorimotor function as a predictor of chronic ankle instability. Clin. Biomech. 2009, 24, 451–458. [Google Scholar] [CrossRef]
- Diekfuss, J.A.; Grooms, D.R.; Yuan, W.; Dudley, J.; Barber Foss, K.D.; Thomas, S.; Ellis, J.D.; Schneider, D.K.; Leach, J.; Bonnette, S.; et al. Does brain functional connectivity contribute to musculoskeletal injury? A preliminary prospective analysis of a neural biomarker of ACL injury risk. J. Sci. Med. Sport 2019, 22, 169–174. [Google Scholar] [CrossRef]
- Pelletier, R.; Bourbonnais, D.; Higgins, J. Nociception, pain, neuroplasticity and the practice of Osteopathic Manipulative Medicine. Int. J. Osteopath. Med. 2018, 27, 34–44. [Google Scholar] [CrossRef]
- Rio, E.; Kidgell, D.; Moseley, G.L.; Gaida, J.; Docking, S.; Purdam, C.; Cook, J. Tendon neuroplastic training: Changing the way we think about tendon rehabilitation: A narrative review. Br. J. Sports Med. 2016, 50, 209–215. [Google Scholar] [CrossRef]
- Grooms, D.; Appelbaum, G.; Onate, J. Neuroplasticity following anterior cruciate ligament injury: A framework for visual-motor training approaches in rehabilitation. J. Orthop. Sports Phys. Ther. 2015, 45, 381–393. [Google Scholar] [CrossRef]
- Gokeler, A.; Neuhaus, D.; Benjaminse, A.; Grooms, D.R.; Baumeister, J. Principles of Motor Learning to Support Neuroplasticity after ACL Injury: Implications for Optimizing Performance and Reducing Risk of Second ACL Injury. Sports Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Deckers, K.; De Smedt, K.; Mitchell, B.; Vivian, D.; Russo, M.; Georgius, P.; Green, M.; Vieceli, J.; Eldabe, S.; Gulve, A.; et al. New Therapy for Refractory Chronic Mechanical Low Back Pain-Restorative Neurostimulation to Activate the Lumbar Multifidus: One Year Results of a Prospective Multicenter Clinical Trial. Neuromodulation 2018, 21, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Deckers, K.; Eldabe, S.; Kiesel, K.; Gilligan, C.; Vieceli, J.; Crosby, P. Muscle Control and Non-specific Chronic Low Back Pain. Neuromodulation 2018, 21, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Freeman, S.; Mascia, A.; McGill, S. Arthrogenic neuromusculature inhibition: A foundational investigation of existence in the hip joint. Clin. Biomech. 2013, 28, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Sonnery-Cottet, B.; Saithna, A.; Quelard, B.; Daggett, M.; Borade, A.; Ouanezar, H.; Thaunat, M.; Blakeney, W.G. Arthrogenic muscle inhibition after ACL reconstruction: A scoping review of the efficacy of interventions. Br. J. Sports Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Rice, D.A.; McNair, P.J. Quadriceps arthrogenic muscle inhibition: Neural mechanisms and treatment perspectives. Semin. Arthritis Rheum. 2010, 40, 250–266. [Google Scholar] [CrossRef] [PubMed]
- Taube, W.; Gruber, M.; Gollhofer, A. Spinal and supraspinal adaptations associated with balance training and their functional relevance. Acta Physiol. 2008, 193, 101–116. [Google Scholar] [CrossRef] [PubMed]
- Rudomin, P.; Schmidt, R.F. Presynaptic inhibition in the vertebrate spinal cord revisited. Exp. Brain Res. 1999, 129, 1–37. [Google Scholar] [CrossRef]
- Hopkins, J.T.; Ingersoll, C.D. Arthrogenic muscle inhibition: A limiting factor in joint rehabilitation. J. Sport Rehabil. 2000, 9, 135–159. [Google Scholar] [CrossRef]
- Becker, R.; Berth, A.; Nehring, M.; Awiszus, F. Neuromuscular quadriceps dysfunction prior to osteoarthritis of the knee. J. Orthop. Res. 2004, 22, 768–773. [Google Scholar] [CrossRef]
- Taube, W.; Gruber, M.; Beck, S.; Faist, M.; Gollhofer, A.; Schubert, M. Cortical and spinal adaptations induced by balance training: Correlation between stance stability and corticospinal activation. Acta Physiol. 2007, 189, 347–358. [Google Scholar] [CrossRef]
- Trimble, M.H.; Koceja, D.M. Modulation of the triceps surae H-reflex with training. Int. J. Neurosci. 1994, 76, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Gruber, M.; Taube, W.; Gollhofer, A.; Beck, S.; Amtage, F.; Schubert, M. Training-specific adaptations of H- and stretch reflexes in human soleus muscle. J. Mot. Behav. 2007, 39, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Crayton, J.W.; King, S. Inter-individual variability of the H-reflex in normal subjects. Electromyogr. Clin. Neurophysiol. 1981, 21, 183–200. [Google Scholar] [PubMed]
- Kido, A.; Tanaka, N.; Stein, R.B. Spinal excitation and inhibition decrease as humans age. Can. J. Physiol. Pharmacol. 2004, 82, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Priori, A.; Polidori, L.; Rona, S.; Manfredi, M.; Berardelli, A. Spinal and cortical inhibition in Huntington’s chorea. Mov. Disord. 2000, 15, 938–946. [Google Scholar] [CrossRef]
- Toft, E.; Sinkjaer, T. H-reflex changes during contractions of the ankle extensors in spastic patients. Acta Neurol. Scand. 1993, 88, 327–333. [Google Scholar] [CrossRef]
- Hodapp, M.; Klisch, C.; Mall, V.; Vry, J.; Berger, W.; Faist, M. Modulation of soleus H-reflexes during gait in children with cerebral palsy. J. Neurophysiol. 2007, 98, 3263–3268. [Google Scholar] [CrossRef][Green Version]
- Gilbert, D.L.; Isaacs, K.M.; Augusta, M.; Macneil, L.K.; Mostofsky, S.H. Motor cortex inhibition: A marker of ADHD behavior and motor development in children. Neurology 2011, 76, 615–621. [Google Scholar] [CrossRef]
- Flamand, V.H.; Nadeau, L.; Schneider, C. Brain motor excitability and visuomotor coordination in 8-year-old children born very preterm. Clin. Neurophysiol. 2012, 123, 1191–1199. [Google Scholar] [CrossRef]
- Papegaaij, S.; Taube, W.; Hogenhout, M.; Baudry, S.; Hortobagyi, T. Age-related decrease in motor cortical inhibition during standing under different sensory conditions. Front. Aging Neurosci. 2014, 6, 126. [Google Scholar] [CrossRef] [PubMed]
- Boorman, G.; Becker, W.J.; Morrice, B.L.; Lee, R.G. Modulation of the soleus H-reflex during pedalling in normal humans and in patients with spinal spasticity. J. Neurol. Neurosurg. Psychiatry 1992, 55, 1150–1156. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, G.R.; Knutzen, K.M. Soleus H-reflex gain in healthy elderly and young adults when lying, standing, and balancing. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 57, B321–B329. [Google Scholar] [CrossRef] [PubMed]
- Butefisch, C.M.; Boroojerdi, B.; Chen, R.; Battaglia, F.; Hallett, M. Task-dependent intracortical inhibition is impaired in focal hand dystonia. Mov. Disord. 2005, 20, 545–551. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, M.N.; Orekhov, Y.; Ziemann, U. The role of GABA(B) receptors in intracortical inhibition in the human motor cortex. Exp. Brain Res. 2006, 173, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Inghilleri, M.; Berardelli, A.; Cruccu, G.; Manfredi, M. Silent period evoked by transcranial stimulation of the human cortex and cervicomedullary junction. J. Physiol. 1993, 466, 521–534. [Google Scholar] [PubMed]
- Terada, M.; Johnson, N.; Kosik, K.; Gribble, P. Quantifying Brain White Matter Microstructure of People with Lateral Ankle Sprain. Med. Sci. Sports Exerc. 2019, 51, 640–646. [Google Scholar] [CrossRef]
- Lauber, B.; Gollhofer, A.; Taube, W. Differences in motor cortical control of the soleus and tibialis anterior. J. Exp. Biol. 2018, 221. [Google Scholar] [CrossRef]
- Papegaaij, S.; Baudry, S.; Negyesi, J.; Taube, W.; Hortobagyi, T. Intracortical inhibition in the soleus muscle is reduced during the control of upright standing in both young and old adults. Eur. J. Appl. Physiol. 2016, 116, 959–967. [Google Scholar] [CrossRef]
- Remaud, A.; Bilodeau, M.; Tremblay, F. Age and muscle-dependent variations in corticospinal excitability during standing tasks. PLoS ONE 2014, 9, e110004. [Google Scholar] [CrossRef]
- Petersen, N.T.; Pyndt, H.S.; Nielsen, J.B. Investigating human motor control by transcranial magnetic stimulation. Exp. Brain Res. 2003, 152, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.E.; Docherty, C.L. Health-related quality of life is decreased in middle-aged adults with chronic ankle instability. J. Sci. Med. Sport 2018, 21, 1206–1209. [Google Scholar] [CrossRef] [PubMed]




| Author (Year) | Study Design | Inclusion Criteria | Participant Characteristics | Stimulation Settings | Outcome | ||
|---|---|---|---|---|---|---|---|
| CAI Group | Comparison Group | CAI Group | Comparison Group | ||||
| McVey (2005) | Case- control | ≥5 “yes” responses on AII | Uninjured: No history of ankle injury or significant lower extremity injury or surgery | 15 unilateral CAI patients (8 females, 26.5 ± 126.5 years, 173 ± 6.8 cm, 70 ± 7.2 kg) | 14 uninjured (13 females, 21.3 ± 2.5 years,166 ± 5.4 cm, 61 ± 6.4 kg) | Unipolar stimulating electrode to stimulate the sciatic nerve with 1 ms squared wave pulse that were 10 s apart by increasing stimulus intensity in 0.2 V until Hmax and Mmax were obtained | Hmax:Mmax ratios of soleus, fibularis longus, and tibialis anterior measured in a prone position |
| Sefton (2008) | Case- control | >1 ankle sprain in the previous year, recurring symptoms, and difficulty in >1 area in the FADI or 2 areas in the FADI-Sport | Uninjured: No history of ankle injury and no incidence of acute or chronic lower extremity injuries | 22 CAI patients (17 females, 22.3 years, 167.6 cm, 69.8 kg) Measures of standard deviation were not reported | 21 uninjured (16 females, 21.9 years, 166.0 cm, 64.1 kg) Measures of standard deviation were not reported | Unipolar stimulating electrode placed over the popliteal fossa to stimulate the posterior tibial nerve with 1 ms squared wave pulse that were 10 to 20 s apart by slowly increasing stimulus intensity | Hmax:Mmax ratio of soleus during bipedal stance and percent changes in paired reflex depression and recurrent inhibition of soleus measured both in unipedal and bipedal stances |
| Doeringer (2009) | Case-control with crossover | A history of ankle sprain, episodes of “giving way” and feelings of instability (≥3 “yes” responses on AII) | Uninjured: No history of ankle sprain or other lower extremity injuries to the tested limb | 12 CAI patients (9 females, 23 ± 1 years, 168.7 ± 9.8 cm, 73.4 ± 20.0 kg) | 12 uninjured (7 females, 23 ± 1 years, 171.7 ± 7.0 cm, 77.9 ± 14.9 kg) | Bipolar (bar) stimulating electrode placed over the popliteal fossa to stimulate the posterior tibial nerve with 1 ms squared wave pulses that were 20 s apart by increasing stimulus intensity in 0.2 to 0.5 V increments until Hmax and Mmax were obtained | Hmax:Mmax ratio of soleus measured in a reclining position with 120° of hip flexion and 60° of knee flexion, and the neutral position of the ankle |
| Palmieri-Smith (2009) | Case-control | All CAI subjects that were physically active (Tegner score of 5 or 6) met the criteria set forth by both Functional Ankle Instability Questionnaire and AII. | Uninjured: All control subjects were physically active (Tegner score of 5 or 6), but other criteria were not specified. | 21 unilateral CAI patients (18 females, 21 ± 2 years, 171 ± 7 cm, 65 ± 9 kg) | 21 uninjured (18 females, 21 ± 3 years, 169 ± 9 cm, 64 ± 10 kg) | Unipolar stimulating electrode placed over the popliteal fossa to stimulate the sciatic nerve with 1 ms squared wave pulses that were 10 s apart by increasing stimulus intensity in 0.2 V increments until Hmax and Mmax were obtained | Hmax:Mmax ratio of fibularis longus measured in prone position |
| Doeringer (2010) | Case-control with crossover | A history of ankle sprain, episodes of “giving way” and feelings of instability (≥3 “yes” responses on AII) | Uninjured: No history of ankle sprain or other lower extremity injuries to the tested limb | 12 CAI patients (9 females, 23 ± 1 years, 168.7 ± 9.8 cm, 73.4 ± 20.0 kg) | 12 uninjured (7 females, 23 ± 1 years, 171.7 ± 7.0 cm, 77.9 ± 14.9 kg) | Bipolar (bar) stimulating electrode placed over the popliteal fossa to stimulate the sciatic nerve with 1 ms squared wave pulses that were 20 s apart by increasing stimulus intensity in 0.2 to 0.5 V increments until Hmax and Mmax were obtained | Hmax:Mmax ratio of fibularis longus and tibialis anterior measured in a reclining position with 120° of hip flexion, 60° of knee flexion, and the neutral position of the ankle |
| Sefton (2011) | Case- control with repeated measures | >1 ankle sprain in the previous year, recurring symptoms, and difficulty in >1 area in the FADI or 2 areas in the FADI-Sport | Uninjured: No history of ankle injury and no incidence of acute or chronic lower extremity injuries | 12 CAI patients (8 females, 21.2 ± 2.1 years, 165.1 ± 8.9 cm, 67.2 ± 9.4 kg) | 9 uninjured (6 females, 20.8 ± 1.3 years, 167.3 ± 7.9 cm, 62.8 ± 10.3 kg) | Unipolar stimulating electrode placed over the popliteal fossa to stimulate the posterior tibial nerve with 1 ms squared wave pulse that were 10 to 20 s apart by slowly increasing stimulus intensity | Hmax:Mmax ratio of soleus during bipedal stance and percent changes in paired reflex depression and recurrent inhibition of soleus measured both in unipedal and bipedal stances |
| Kim (2012) | Case-control | A history of at least 1 lateral ankle sprain (1-yr old or greater), episodes of “giving way”, feelings of instability (≥4 “yes” responses on AII), and self-reported ankle disability (≤90% on FAAM and ≤80% on the FAAM-Sport) | Uninjured: No history of ankle injury or significant lower extremity injury or surgery, and any limitation of ankle function | 16 unilateral CAI patients (6 females, 21.0 ± 6.9 years, 173.9 ± 7.4 cm, 72.6 ± 11.9 kg) | 15 uninjured (6 females 19.9 ± 4.3 years, 175.8 ± 9.7 cm, 71.3 ± 17.8 kg) | Unipolar stimulating electrode placed over the superior popliteal fossa to stimulate the sciatic nerve with 1 ms squared wave pulses that were at least 12 s apart by increasing stimulus intensity in 0.2 V increments until Hmax was obtained, then 1.0 V increments until Mmax plateaued | Hmax:Mmax ratio of soleus and fibularis longus measured in 3 body positions: prone, bipedal, and unipedal stances |
| Pietro- simone (2012) | Case-control | A history of at least 2 unilateral ankle sprains and self-reported function (<90% on FADI, <80% on FADI-Sport) | Uninjured: No ankle instability and self-reported functions (>95% on FADI, >85% on FADI-Sport) | 10 unilateral CAI patients (6 females, 21.2 ± 1.2 years, 175.1 ± 9.7 cm, 77.1 ± 13.6 kg) | 10 uninjured (6 females 21.2 ± 2.3 years, 172.3 ± 8.9 cm, 73.4 ± 7.2 kg) | Double-cone coil placed over the contralateral vertex of the cranium relative to the involved limb to deliver a single magnetic pulse of a maximum magnetic stimulus of 1.4 Tesla that were 15 s apart between trials | Resting motor threshold expressed as a percentage of 2 Tesla of fibularis longus measured in the seated position with 85° of hip flexion, 10° of knee flexion, and 10° of ankle plantar flexion |
| Needle (2013) | Case- control | A history of at least 1 unilateral ankle sprain (≤25 on CAIT) | Uninjured: No history of ankle injury and a score of >27 on CAIT) | 12 unilateral CAI patients (6 females, 20.9 ± 4.1 years, 170.6 ± 10.1 cm, 72.5 ± 15.0 kg) | 12 uninjured (6 females 21.2 ± 2.6 years, 172.7 ± 8.5 cm, 70.0 ± 15.0 kg) | A figure-8 coil placed over the contralateral vertex of the cranium relative to the involved limb to deliver a single magnetic pulse of a maximum magnetic stimulus of 1.4 Tesla that were 5 s apart between trials | AMT of soleus, fibularis longus, and tibialis anterior measured in the seated position with pronation of ankle at 15% of maximal effort of fibularis longus activity |
| Kim (2015) | Case-control with crossover | A history of at least 1 lateral ankle sprain (1-yr old or greater), episodes of “giving way”, feelings of instability (≥4 “yes” responses on AII), and self-reported ankle disability (≤90% on FAAM and ≤80% on the FAAM-Sport) | Uninjured: No history of ankle injury or lower extremity injury or surgery and any limitation of ankle function | 15 CAI patients (6 females, 22.6 ± 5.8 years, 174.7 ± 8.1 cm, 74.9 ± 12.8 kg) | 15 uninjured (6 females, 23.8 ± 5.8 years, 171.9 ± 9.9 cm, 68.9 ± 15.5 kg) | Unipolar stimulating electrode placed over the superior popliteal fossa to stimulate the sciatic nerve with 1 ms squared wave pulses that were at least 12 s apart by increasing stimulus intensity in 0.2 V increments until Hmax was obtained, then 1.0 V increments until Mmax plateaued | Hmax:Mmax ratio of soleus and fibularis longus measured in 3 body positions: prone, bipedal and unipedal stances |
| McLeod (2015) | Case-control | A history of at least 1 acute lateral ankle sprain, resulting in swelling, pain, and/or temporary loss of function but not within the 3 months) and >2 episodes of the ankle “giving way” in the 6 months (≤80% on the FAAM-Sport) | Uninjured: No history of ankle injury and a score of 100% on the FAAM-Sport | 21 CAI patients (12 females, 20.8 ± 1.6 years, 171.6 ± 11.4 cm, 68.8 ± 11.9 kg) | 24 uninjured (17 females, 22.5 ± 2.9 years, 172.4 ± 10.9 cm, 69.2 ± 12.3 kg) | For H-reflex testing, the unipolar stimulating electrode to stimulate (1) the sciatic nerve for fibularis longus and (2) the femoral nerve for vastus medialis separately by increasing stimulus intensity in 0.2 V increments until Hmax was obtained For TMS testing, the double-cone coil placed over the contralateral vertex of the cranium relative to the involved limb to deliver a brief magnetic stimulus of a maximum magnetic stimulus of 1.4 Tesla | Hmax:Mmax ratios of both fibularis longus and vastus medialis measured in a supine position For the fibularis longus AMT and 5 MEP responses at varying levels of intensity, measured in the seated position with 85° of hip flexion, 10° of knee flexion, and 10° of ankle plantar flexion For the vastus medialis, AMT and 5 MEP responses at varying levels of intensity, measured in the seated position with 85° of hip flexion and 90° of knee flexion |
| Bowker (2016) | Case-control | A history of at least 1 ankle sprain, resulting in swelling, pain, and/or temporary loss of function), ≥2 episodes of the ankle “giving way” within the 6 months, perceived ankle instability, and dysfunction during daily living activities (≥5 “yes” responses on AII and scores of ≥11 on IdFAI) | Uninjured: No history of ankle sprain and a score of 0 on both AII and IdFAI Copers: History of ankle sprains, but no reported episode of the ankle “giving way”, perceived instability, or loss of function without modifying physical activity, <5 “yes” responses on AII, and scores of <11 on IdFAI | 37 CAI patients (19 females, 22 ± 3.5 years, 25.2 ± 3.8 body mass index) | 26 uninjured (17 females, 21.6 ± 3.2 years, 23.7 ± 2.8 body mass index 30 copers (17 females, 21.9 ± 4.3 years, 26.2 ± 6.3 body mass index) | Unipolar stimulating electrode placed over the proximal lateral popliteal fossa to stimulate the posterior tibial nerve with 1 ms squared wave pulse that were 10 s apart by increasing or decreasing the stimulus intensity in 0.2 V increments until Hmax was obtained, then 1.0 V increments until Mmax plateaued | Hmax:Mmax ratio of soleus measured in the seated position with 90° of hip flexion, 90° of knee flexion, 90° of ankle plantar flexion |
| Terada (2016) | Case-control | A history of at least 2 significant ankle sprains, resulting in swelling, pain, and/or temporary loss of function, ≥2 episodes of the ankle “giving way” within the 6 months, perceived ankle instability, and dysfunction during daily living activities (≥4 “yes” responses on AII and scores of ≥11 on IdFAI) | Uninjured: No history of ankle sprain and a score of 0 on both the AII and IdFAI | 16 CAI patients (6 females, 22.2 ± 3.6 years, 25.8 ± 2.5 body mass index) | 17 uninjured (9 females, 21.2 ± 3.0 years, 24.7 ± 3.1 body mass index) | For Mmax of the fibularis longus, the unipolar stimulating electrode placed over the proximal lateral popliteal fossa to stimulate the posterior tibial nerve with 1 ms squared wave pulse by increasing the stimulus intensity in 1.0 V increments until Mmax plateaued For TMS testing, the double-cone coil placed over the contralateral vertex of the cranium relative to the involved limb to deliver a series of magnetic stimuli of 1.0 Tesla | AMT, MEP120%:Mmax ratio, and CSP: MEP120% ratio of soleus measured in the seated position with 90° of knee flexion, 90° of ankle dorsiflexion |
| Kosik (2017) | Case-control | A history of at least 1 acute lateral ankle sprain, resulting in swelling, pain, and/or temporary loss of function and ≥2 episodes of the ankle “giving way” within the 6 months (≥5 “yes” responses on AII and scores of ≥11 on IdFAI ≤24 on CAIT) | Uninjured: No history of lower extremity injuries and a score 0 on both the AII and IdFAI, and 30 on CAIT | 18 CAI patients (14 females, 23.8 ± 3.6 years, 169.6 ± 7.5 cm, 73.1 ± 12.0 kg) | 16 uninjured (10 females, 21.1 ± 2.2 years, 168.6 ± 13.4 cm, 66.5 ± 10.2 kg) | For H-reflex testing, the unipolar stimulating electrode to stimulate the proximal common fibular nerve with 1 ms squared wave pulse by increasing or decreasing the stimulus intensity in 0.2 V increments until Hmax was obtained, then 1.0 V increments until Mmax plateaued For TMS testing, The double-cone coil placed over the contralateral vertex of the cranium relative to the involved limb to deliver a series of magnetic stimuli of 1 Tesla that were 10 s apart | Hmax:Mmax ratio of fibularis longus measured in the prone position MEP100%:Mmax ratio and corticomotor map outcomes for fibularis longus: (1) size of corticomotor map area, (2) volume of corticomotor map, (3) location of cortical representation, measured in the seated position with 75° of hip flexion, 60° of knee flexion, 80° of ankle plantar flexion |
| Terada (2017) | Case- control | CAI subgroups: (1) PI-RAS: A history of at least 2 previous ankle sprains and ≥2 episodes of the ankle “giving way” within the 6 months (≥5 “yes” responses on AII and scores of ≥11 on IdFAI) (2) PI: A history of one previous ankle sprain and ≥2 episodes of the ankle “giving way” within the 6 months (≥5 “yes” responses on AII and scores of ≥11 on IdFAI) (3) RAS: A history of at least 2 previous ankle sprains, but no episode of the ankle “giving way”, <5 “yes” responses on AII, and scores of <11 on IdFAI | Uninjured: No history of ankle sprain and a score 0 on both the AII and IdFAI Copers: A history of one previous ankle sprain, report of returning to full activity for at least 12 months following an initial ankle sprain without recurrent injury, no reported episode of the ankle “giving way”, perceived instability, or loss of function without modifying physical activity, <5 “yes” responses on AII, and scores of <11 on IdFAI | 25 PI-RAS patients (11 females, 22.5 ± 4.0 years, 171.4 ± 8.7 cm, 76.2 ± 14.8 kg, 25.8 ± 3.6 body mass index) 13 PI patients (9 females, 20.8 ± 1.6 years, 165.8 ± 6.5 cm, 65.7 ± 11.8 kg, 23.9 ± 3.8 body mass index) 12 RAS patients (6 females, 22.2 ± 4.8 years, 171.0 ± 10.3 cm, 79.1 ± 7.8 kg, 26.7 ± 7.6 body mass index) | 26 uninjured (17 females, 21.6 ± 3.2 years, 166.1 ± 8.1 cm, 66.2 ± 13.1 kg, 23.8 ± 3.0 body mass index) 18 copers (11 females, 21.6 ± 4.0 years, 169.6 ± 11.3 cm, 72.4 ± 17.3 kg, 24.9 ± 3.8 body mass index) | For H-reflex testing, the unipolar stimulating electrode to stimulate the posterior tibial nerve with 1 ms squared wave pulse that were 10 s apart by increasing the stimulus intensity in 0.2 V increments until Hmax was obtained, then 1.0 V increments until Mmax plateau | Hmax:Mmax ratio of soleus measured in the seated position with 90° of hip flexion, 90° of knee flexion, and 90° of ankle plantar flexion |
| Otzel (2019) | Case-control with crossover | A history of at least 1 moderate ankle sprain requiring immobilization, no formal rehabilitation, at least one recurrent ankle sprain 3-6 months prior to participation, perceived pain, ankle instability or weakness, and self-reported functional limitations (≤90% on FADI and ≤80% on the FADI-Sport) | Uninjured age-matched control Specific inclusion criteria not reported | 10 CAI patients (6 females, 20.7 ± 1.3 years, 169.4 ± 10.7 cm, 66.0 ± 10.1 kg) | 10 uninjured (7 females, 19.8 ± 0.7 years, 165.6 ± 9.2 cm, 59.1 ± 10.7 kg) | For H-reflex testing, the unipolar stimulating electrode to stimulate the posterior tibial nerve with 1 ms squared wave pulse that were 10 s apart by increasing the stimulus intensity in 0.2 V increments until Hmax was obtained, then continued until Mmax plateau | Hmax:Mmax ratio of soleus measured in the seated position with 30° of hip flexion, 90° of knee flexion, 90° of ankle plantar flexion |
| Thompson (2019) | Case-control | A history of at least 1 significant ankle sprain, causing inflammatory symptoms and disrupted activity), the most recent ankle sprain occurred less than 3 months prior to study participation, reports of episodes of the ankle “giving way” and/or recurrent pain and/or perceived ankle instability, and dysfunction during daily living activities (≥5 “yes” responses on AII and scores <24 on CAIT) | Uninjured: No history of an ankle sprain Copers: History of ankle sprains, but no report of recurrent injuries, episode of the ankle “giving way”, and/or perceived instability | 12 CAI patients (4 females, 25.2 ± 3.7 years, 177.7 ± 8.1 cm, 75.8 ± 14.8 kg) | 12 uninjured (4 females, 23.3 ± 4.5 years, 171.6 ± 6.2 cm, 74.3 ± 10.2 kg) 12 copers (4 females, 24.2 ± 4.7 years, 172.7 ± 8.2 cm, 71.4 ± 6.9 kg) | For H-reflex testing, the unipolar stimulating electrode to stimulate the posterior tibial nerve with 1 ms squared wave pulse that was 10–15 s apart | Soleus Hmax:Mmax ratio and slope of recruitment curve during bipedal stance Soleus H50%:Mmax ratio and percent changes in presynaptic inhibition and recurrent inhibition, measured both in unipedal and bipedal stances |
| Study | Reporting | External Validity | Internal Validity Bias | Internal Validity Confounding | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Quality Index Score (%) | 1. Hypothesis Clearly Described? | 2. Main Outcomes Clearly Described? | 3. Characteristics of the Patients included Clearly Described? | 5. Distribution of Principle Confounder of Each Group Clearly Described? | 6. Main Findings Clearly Described? | 7. Estimates of Random Variability Provided for the Main Outcomes? | 10. Actual Probability Values Reported for Main Outcomes? | 11. Were the Subjects Asked to Participate Representative of the Entire Population? | 12. Were the Subjects who Were Prepared to Participate Representative of the Entire Population? | 16. Was it Clear if the Results Were Based on “Data Dredging’? | 18. Were the Statistical Tests Appropriate? | 20. Were the Main Outcome Measures Valid and Reliable? | 21. Were all Patients and Controls Recruited from the Same Population? | 22. Were all Patients and Controls Recruited over the Same Time Period? | |
| McVey (2005) | 60.0 | + | + | - | + | + | + | + | - | - | + | + | + | - | - |
| Sefton (2008) | 66.7 | - | + | - | ++ | + | + | + | - | - | + | + | + | + | - |
| Doeringer (2009) | 66.7 | + | + | - | + | + | + | + | - | - | + | + | + | + | - |
| Palmieri-Smith (2009) | 66.7 | + | + | - | ++ | + | + | + | - | - | + | + | + | - | - |
| Doeringer (2010) | 66.7 | + | + | - | + | + | + | + | - | - | + | + | + | + | - |
| Sefton (2011) | 60.0 | + | + | - | + | + | + | + | - | - | + | + | + | - | - |
| Kim (2012) | 73.3 | + | + | + | ++ | + | + | + | - | - | + | + | + | - | - |
| Pietro- simone (2012) | 66.7 | + | + | - | ++ | + | + | + | - | - | + | + | + | - | - |
| Needle (2013) | 46.7 | - | - | - | ++ | - | + | + | - | - | + | + | - | + | - |
| Kim (2015) | 73.3 | + | + | + | ++ | + | + | + | - | - | + | + | + | - | - |
| McLeod (2015) | 60.0 | + | + | - | + | + | + | + | - | - | + | + | + | - | - |
| Bowker (2016) | 73.3 | + | + | + | + | + | + | + | - | - | + | + | + | + | - |
| Terada (2016) | 73.3 | + | + | + | + | + | + | + | - | - | + | + | + | + | - |
| Kosik (2017) | 73.3 | + | + | + | + | + | + | + | - | - | + | + | + | + | - |
| Terada (2017) | 73.3 | + | + | + | + | + | + | + | - | - | + | + | + | + | - |
| Otzel (2019) | 60.0 | + | + | + | + | + | + | + | - | - | + | + | - | - | - |
| Thompson (2019) | 60.0 | + | + | + | + | + | + | + | - | - | + | + | - | - | - |
| Average (SD) | 65.9 (7.4) | ||||||||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.-M.; Kim, J.-S.; Cruz-Díaz, D.; Ryu, S.; Kang, M.; Taube, W. Changes in Spinal and Corticospinal Excitability in Patients with Chronic Ankle Instability: A Systematic Review with Meta-Analysis. J. Clin. Med. 2019, 8, 1037. https://doi.org/10.3390/jcm8071037
Kim K-M, Kim J-S, Cruz-Díaz D, Ryu S, Kang M, Taube W. Changes in Spinal and Corticospinal Excitability in Patients with Chronic Ankle Instability: A Systematic Review with Meta-Analysis. Journal of Clinical Medicine. 2019; 8(7):1037. https://doi.org/10.3390/jcm8071037
Chicago/Turabian StyleKim, Kyung-Min, Joo-Sung Kim, David Cruz-Díaz, Seungho Ryu, Minsoo Kang, and Wolfgang Taube. 2019. "Changes in Spinal and Corticospinal Excitability in Patients with Chronic Ankle Instability: A Systematic Review with Meta-Analysis" Journal of Clinical Medicine 8, no. 7: 1037. https://doi.org/10.3390/jcm8071037
APA StyleKim, K.-M., Kim, J.-S., Cruz-Díaz, D., Ryu, S., Kang, M., & Taube, W. (2019). Changes in Spinal and Corticospinal Excitability in Patients with Chronic Ankle Instability: A Systematic Review with Meta-Analysis. Journal of Clinical Medicine, 8(7), 1037. https://doi.org/10.3390/jcm8071037






