Mindfulness-Based Program Plus Amygdala and Insula Retraining (MAIR) for the Treatment of Women with Fibromyalgia: A Pilot Randomized Controlled Trial
Abstract
:1. Introduction
2. Experimental Section
2.1. Research Design
2.2. Study Sample
2.3. Procedure
2.4. Treatments
2.4.1. Relaxation Therapy (RT)
2.4.2. Mindfulness + Amygdala and Insula Retraining (MAIR)
2.5. Measures
2.5.1. Socio-Demographic Characteristics
2.5.2. Primary Outcome
2.5.3. Secondary Outcomes
2.5.4. Biomarkers Outcomes
2.6. Data Analyses
3. Results
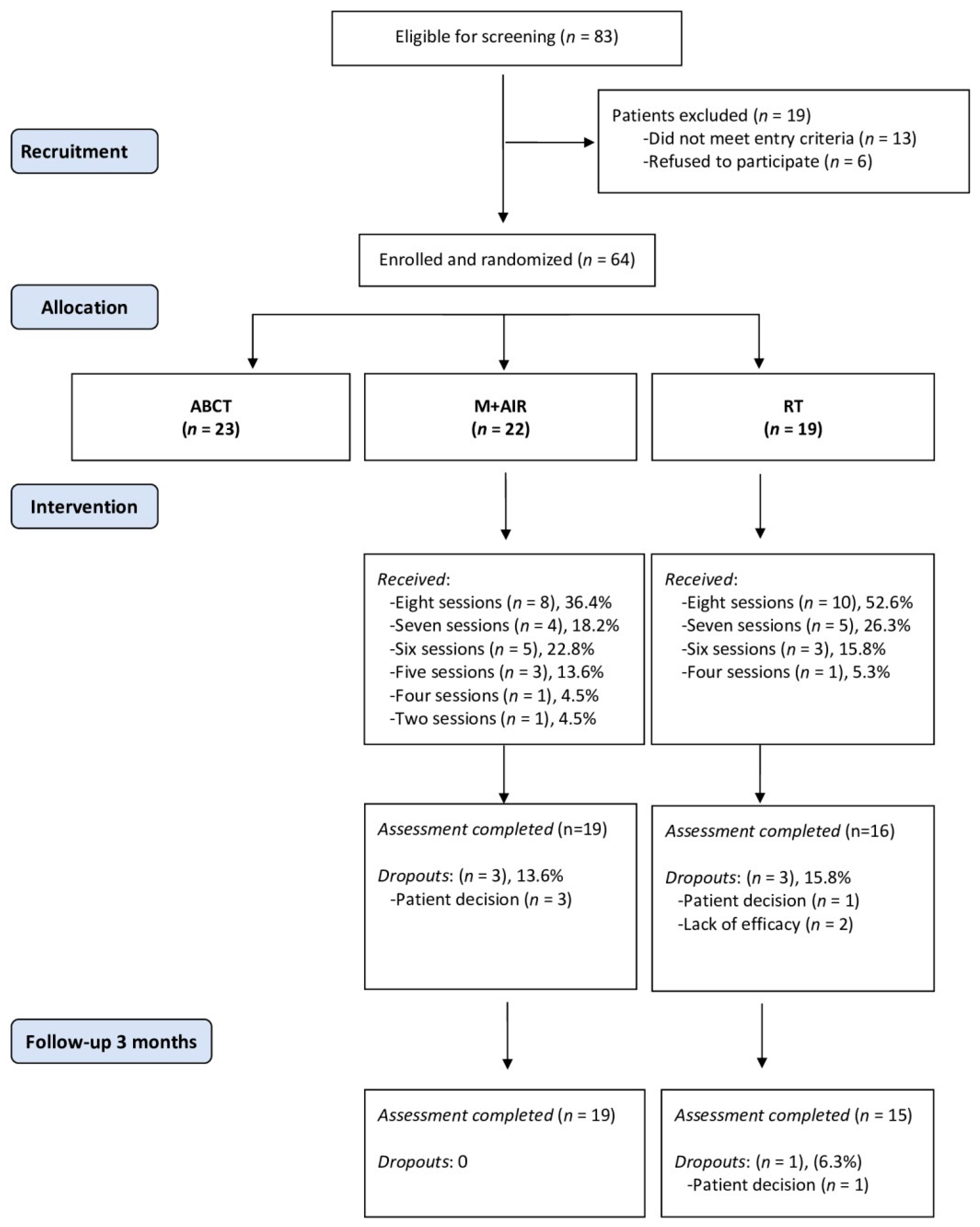
3.1. Patients Flow and Compliance
3.2. Baseline Socio-Demographic and Clinical Characteristics of Patients
3.3. Effects on Primary and Secondary Outcomes
3.4. Effects on Biomarkers
3.5. Absolute Risk Reduction and Number Needed to Treat (NNT)
3.6. Patient Preferences and Credibility of Therapies
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Wolfe, F.; Clauw, D.J.; FitzCharles, M.; Goldenberg, D.L.; Häuser, W.; Katz, R.L.; Mease, P.J.; Russell, A.S.; Russell, I.J.; Walitt, B. Revisions to the 2010/2011 Fibromyalgia diagnostic criteria. Arthritis Rheum. 2016, 46, 319–329. [Google Scholar] [CrossRef]
- Leadley, R.M.; Armstrong, N.; Lee, Y.C.; Allen, A.; Kleijnen, J. Chronic diseases in the European Union: The prevalence and health cost implications of chronic pain. J. Pain Palliat. Care Pharm. 2012, 26, 310–325. [Google Scholar] [CrossRef]
- Häuser, W.; Ablin, J.; Fitzcharles, M.A.; Littlejohn, G.; Luciano, J.V.; Usui, C.; Walitt, B. Fibromyalgia. Nat. Rev. Dis. 2015, 1, 15022. [Google Scholar] [CrossRef]
- Häuser, W.; Ablin, J.; Perrot, S.; Fitzcharles, M.A. Management of fibromyalgia: Practical guides from recent evidence-based guidelines. Pol. Arch. Med. Wewn. 2017, 127, 47–56. [Google Scholar] [CrossRef] [Green Version]
- Sharpe, L.; Jones, E.; Ashton-James, C.E.; Nicholas, M.K.; Refshauge, K. Necessary components of psychological treatment in chronic pain management programs: A Delphi study. Eur. J. Pain 2020. [Google Scholar] [CrossRef]
- Häuser, W.; Bernardy, K.; Arnold, B.; Offenbächer, M.; Schiltenwolf, M. Efficacy of multicomponent treatment in fibromyalgia syndrome: A meta-analysis of randomized controlled clinical trials. Arthritis Rheum. 2009, 61, 216–224. [Google Scholar] [CrossRef]
- Bernardy, K.; Klose, P.; Welsch, P.; Häuser, W. Efficacy, acceptability, and safety of cognitive behavioural therapies in fibromyalgia syndrome–a systematic review and meta-analysis of randomized controlled trials. Eur. J. Pain 2018, 22, 242–260. [Google Scholar] [CrossRef] [Green Version]
- Sosa, M.D.; Núñez, S.; Gallego, T.; Pecos, D.; Monserrat, J.; Álvarez, M. Effectiveness of therapeutic exercise in fibromyalgia syndrome: A systematic review and meta-analysis of randomized clinical trials. Biomed. Res. Int. 2017. [Google Scholar] [CrossRef]
- Amer-Cuenca, J.J.A.; Pecos-Martín, D.; Martínez-Merinero, P.; Lluch, E.; Nijs, J.; Meeus, M.; Peña, R.F.; Fernández-Carnero, J. How much is needed? Comparison of the effectiveness of different pain education dosages in patients with fibromyalgia. Pain Med. 2019, 21, 1–12. [Google Scholar] [CrossRef]
- Serrat, M.; Sanabria-Mazo, J.P.; García-Troiteiro, E.; Fontcuberta, A.; Mateo-Canedo, C.; Almirall, M.; Feliu-Soler, A.; Méndez-Ulrich, J.L.; Sanz, A.; Luciano, J.V. Efficacy of a multicomponent intervention for fibromyalgia based on pain neuroscience education, exercise therapy, psychological support, and nature exposure (NAT-FM): Study protocol of a randomized controlled trial. Int. J. Environ. Res. Public Health 2020, 17, 634. [Google Scholar] [CrossRef] [Green Version]
- Lauche, R.; Cramer, H.; Dobos, G.; Langhorst, J.; Schmidt, S. A systematic review and meta-analysis of mindfulness-based stress reduction for the fibromyalgia syndrome. J. Psychosom. Res. 2013, 75, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Langhorst, J.; Häuser, W.; Bernardy, K.; Lucius, H.; Settan, M.; Winkelmann, A.; Musial, F. Complementary and alternative therapies for fibromyalgia syndrome. Systematic review, meta-analysis and guideline. Schmerz 2012, 26, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Andrés-Rodriguez, L.; Borras, X.; Feliu-Soler, A.; Pérez-Aranda, A.; Rozadilla-Sacanell, A.; Montero-Marin, J.; Maes, M.; Luciano, J.V. Immune-inflammatory pathways and clinical changes in fibromyalgia patients treated with Mindfulness-Based Stress Reduction (MBSR): A randomized, controlled clinical trial. Brain Behav. Immun. 2019, 80, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Aranda, A.; Feliu-Soler, A.; Montero-Marín, J.; García-Campayo, J.; Andrés-Rodríguez, L.; Borràs, X.; Rozadilla-Sacanell, A.; Peñarrubia-Maria, M.T.; Angarita-Osorio, N.; McCracken, L.M.; et al. A randomized controlled efficacy trial of mindfulness-based stress reduction compared with an active control group and usual care for fibromyalgia: The EUDAIMON study. Pain 2019, 160, 2508–2523. [Google Scholar] [CrossRef]
- Cash, E.; Salmon, P.; Weissbecker, I.; Rebholz, W.N.; Bayley-Veloso, R.; Zimmaro, L.A.; Floyd, A.; Dedert, E.; Sephton, S.E. Mindfulness meditation alleviates fibromyalgia symptoms in women: Results of a randomized clinical trial. Ann. Behav. Med. 2015, 49, 319–330. [Google Scholar] [CrossRef] [Green Version]
- Toussaint, L.; Whipple, M.O.; Abboud, L.L.; Vincent, A.; Wahner-Roedler, D.L. A mind-body technique for symptoms related to fibromyalgia and chronic fatigue. Explore 2012, 8, 92–98. [Google Scholar] [CrossRef]
- Gupta, A. Can amygdala retraining techniques improve the wellbeing of patients with chronic fatigue syndrome: A clinical audit of subjective outcomes in a small sample. J. Holist. Health 2010, 7, 12–15. [Google Scholar]
- Gupta, A. Unconscious amygdalar fear conditioning in a subset of chronic fatigue syndrome patients. Med. Hypotheses 2002, 59, 727–735. [Google Scholar] [CrossRef]
- Quirk, G.J.; Garcia, R.; González-Lima, F. Prefrontal mechanisms in extinction of conditioned fear. Biol. Psychiatry 2006, 60, 337–343. [Google Scholar] [CrossRef]
- Gotink, R.A.; Meijboom, R.; Vernooij, M.W.; Smits, M.; Hunink, M.M. 8-week mindfulness-based stress reduction induces brain changes similar to traditional long-term meditation practice–a systematic review. Brain Cogn. 2016, 108, 32–41. [Google Scholar] [CrossRef]
- Monroe, T.B.; Gore, J.C.; Bruehl, S.P.; Benningfield, M.M.; Dietrich, M.S.; Chen, L.M.; Newhouse, P.; Fillingim, R.; Chodkowski, B.A.; Atalla, S.; et al. Sex differences in psychophysical and neurophysiological responses to pain in older adults: A cross-sectional study. Biol. Sex Differ. 2015, 6, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lutz, J.; Jäger, L.; de Quervain, D.; Krauseneck, T.; Padberg, F.; Wichnalek, M.; Beyer, A.; Stahl, R.; Zirngibl, B.; Morhard, D.; et al. White and gray matter abnormalities in the brain of patients with fibromyalgia: A diffusion-tensor and volumetric imaging study. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 2008, 58, 3960–3969. [Google Scholar] [CrossRef] [PubMed]
- Pyke, T.L.; Osmotherly, P.G.; Baines, S. Measuring glutamate levels in the brains of fibromyalgia patients and a potential role for glutamate in the pathophysiology of fibromyalgia symptoms. Clin. J. Pain 2017, 33, 944–954. [Google Scholar] [CrossRef] [PubMed]
- Sawaddiruk, P.; Paiboonworachat, S.; Chattipakorn, N.; Chattipakorn, S.C. Alterations of brain activity in fibromyalgia patients. J. Clin. Neurosci. 2017, 38, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Gracely, R.H.; Ambrose, K.R. Neuroimaging of fibromyalgia. Best Pract. Res. Clin. Rheumatol. 2011, 25, 271–284. [Google Scholar] [CrossRef]
- Laske, C.; Stransky, E.; Eschweiler, G.W.; Klein, R.; Wittorf, A.; Leyhe, T.; Richartz, E.; Köhler, N.; Bartels, M.; Buchkremer, G.; et al. Increased BDNF serum concentration in fibromyalgia with or without depression or antidepressants. J. Psychiatr. Res. 2007, 41, 600–605. [Google Scholar] [CrossRef]
- Nugraha, B.; Karst, M.; Engeli, S.; Gutenbrunner, C. Brain-derived neurotrophic factor and exercise in fibromyalgia syndrome patients: A mini review. Rheumatol. Int. 2012, 32, 2593–2599. [Google Scholar] [CrossRef]
- Morgan, N.; Irwin, M.R.; Chung, M.; Wang, C. The effects of mind-body therapies on the immune system: Meta-analysis. PLoS ONE 2014, 9, e100903. [Google Scholar] [CrossRef] [Green Version]
- Montero-Marin, J.; Andrés-Rodríguez, L.; Tops, M.; Luciano, J.V.; Navarro-Gil, M.; Feliu-Soler, A.; López-del-Hoyo, Y.; García-Campayo, J. Effects of attachment-based compassion therapy (ABCT) on brain-derived neurotrophic factor and low-grade inflammation among fibromyalgia patients: A randomized controlled trial. Sci. Rep. 2019, 9, 15639. [Google Scholar] [CrossRef] [Green Version]
- Montero-Marin, J.; Navarro-Gil, M.; Puebla-Guedea, M.; Luciano, J.V.; Van Gordon, W.; Shonin, E.; Garcia-Campayo, J. Efficacy of ‘Attachment Based Compassion Therapy’ (ABCT) in the treatment of fibromyalgia: A randomized controlled trial. Front. Psychiatry 2018, 8, 307. [Google Scholar] [CrossRef] [Green Version]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomized trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef] [PubMed]
- Dworkin, R.H.; Turk, D.C.; Wyrwich, K.W.; Beaton, D.; Cleeland, C.S.; Farrar, J.T.; Haythornthwaite, J.A.; Jensen, M.P.; Kerns, R.D.; Ader, D.N.; et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J. Pain 2008, 9, 105–121. [Google Scholar] [CrossRef] [PubMed]
- García-Campayo, J.; Rodero, B.; del Hoyo, D.L.; Luciano, J.V.; Aida, M.; Gili, M. Validation of a Spanish language version of the pain self-perception scale in patients with fibromyalgia. BMC Musculoskelet. Disord. 2010, 11, 255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rivera, J.; González, T. The fibromyalgia impact questionnaire: A validated Spanish version to assess the health status in women with fibromyalgia. Clin. Exp. Rheumatol. 2004, 22, 554–560. [Google Scholar] [PubMed]
- Díaz-Marsá, M.; Palomares, N.; Morón, M.D.; Tajima, K.; Fuentes, M.E.; López-Ibor, J.J.; Carrasco, J.L. Psychological factors affecting response to antidepressant drugs in fibromyalgia. Psychosomatics 2011, 52, 237–244. [Google Scholar] [CrossRef] [PubMed]
- García-Campayo, J.; Rodero, B.; Alda, M.; Sobradiel, N.; Montero, J.; Moreno, S. Validation of the Spanish version of the pain catastrophizing scale in fibromyalgia. Med. Clin. 2008, 131, 487–492. [Google Scholar]
- Vallejo, M.A.; Rivera, J.; Esteve-Vives, J.; Rodríguez-Muñoz, M.F. Use of the Hospital Anxiety and Depression Scale (HADS) to evaluate anxiety and depression in fibromyalgia patients. Rev. Psiquiatr. Salud Ment. 2012, 5, 107–114. [Google Scholar] [CrossRef]
- Badia, X.; Roset, M.; Montserrat, S.; Herdman, M.; Segura, A. The Spanish version of EuroQol: A description and its applications. European Quality of Life scale. Med. Clin. 1999, 112, 79–85. [Google Scholar]
- Ruiz, F.J.; Langer-Herrera, A.I.; Luciano, C.; Cangas, A.J.; Beltrán, I. Measuring experiential avoidance and psychological inflexibility: The Spanish version of the acceptance and action questionnaire—II. Psicothema 2013, 25, 123–129. [Google Scholar]
- Aguado, J.; Luciano, J.V.; Cebolla, A.; Serrano-Blanco, A.; Soler, J.; García-Campayo, J. Bifactor analysis and construct validity of the five facet mindfulness questionnaire (FFMQ) in non-clinical Spanish samples. Front. Psychol. 2015, 6, 404. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Campayo, J.; Navarro-Gil, M.; Andrés, E.; Montero-Marin, J.; López-Artal, L.; Demarzo, M.M.P. Validation of the Spanish versions of the long (26 items) and short (12 items) forms of the Self-Compassion Scale (SCS). Health Qual. Life Outcomes 2014, 12, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Egbewale, B.E.; Lewis, M.; Sim, J. Bias, precision and statistical power of analysis of covariance in the analysis of randomized trials with baseline imbalance: A simulation study. BMC Med. Res. Methodol. 2014, 14, 49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morris, S.B. Estimating effect sizes from pretest-posttest-control group designs. Organ. Res. Methods 2008, 11, 364–386. [Google Scholar] [CrossRef]
- Glickman, M.E.; Rao, S.R.; Schultz, M.R. False discovery rate control is a recommended alternative to Bonferroni-type adjustments in health studies. J. Clin. Epidemiol. 2014, 67, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Bennett, R. The Fibromyalgia Impact Questionnaire (FIQ): A review of its development, current version, operating characteristics and uses. Clin. Exp. Rheumatol. 2005, 23, S154–S162. [Google Scholar] [PubMed]
- Pérez-Aranda, A.; Andrés-Rodríguez, L.; Feliu-Soler, A.; Núñez, C.; Stephan-Otto, C.; Pastor-Mira, M.A.; López-Roig, S.; Peñacoba, C.; Calandre, E.P.; Slim, M.; et al. Clustering a large Spanish sample of patients with fibromyalgia using the Fibromyalgia Impact Questionnaire–Revised: Differences in clinical outcomes, economic costs, inflammatory markers, and gray matter volumes. Pain 2019, 160, 908–921. [Google Scholar] [CrossRef] [PubMed]
- Angarita-Osorio, N.; Pérez-Aranda, A.; Feliu-Soler, A.; Andrés-Rodríguez, L.; Borràs, X.; Suso-Ribera, C.; Slim, M.; Herrera-Mercadal, P.; Fernández-Vergel, R.; Blanco, M.; et al. Patients with fibromyalgia reporting severe pain but low impact of the syndrome: Clinical and pain-related cognitive features. Pain Pract. 2019, 20, 255–261. [Google Scholar] [CrossRef]
- Sanada, K.; Díez, M.A.; Valero, M.S.; Pérez-Yus, M.C.; Demarzo, M.M.P.; García-Toro, M.; García-Campayo, J. Effects of non-pharmacological interventions on inflammatory biomarker expression in patients with fibromyalgia: A systematic review. Arthritis Res. 2015, 17, 272. [Google Scholar] [CrossRef] [Green Version]
- Andrés-Rodríguez, L.; Borràs, X.; Feliu-Soler, A.; Pérez-Aranda, A.; Angarita-Osorio, N.; Moreno-Peral, P.; Luciano, J.V. Peripheral immune aberrations in fibromyalgia: A systematic review, meta-analysis and meta-regression. Brain Behav. Immun. 2019, 87, 881–889. [Google Scholar] [CrossRef]
- Rodriguez-Pinto, I.; Agmon-Levin, N.; Howard, A.; Shoenfeld, Y. Fibromyalgia and cytokines. Immunol. Lett. 2014, 161, 200–203. [Google Scholar] [CrossRef]
- Deitos, A.; Dussán-Sarria, J.A.; Souza, A.D.; Medeiros, L.; Tarragô-Mda, G.; Sehn, F.; Chassot, M.; Zanette, S.; Schwertner, A.; Fregni, F.; et al. Clinical Value of Serum Neuroplasticity Mediators in Identifying the Central Sensitivity Syndrome in Patients with Chronic Pain with and Without Structural Pathology. Clin. J. Pain 2015, 31, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Haas, L.; Portela, L.V.; Böhmer, A.E.; Oses, J.P.; Lara, D.R. Increased plasma levels of brain derived neurotrophic factor (BDNF) in patients with fibromyalgia. Neurochem. Res. 2010, 35, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Jha, M.K.; Trivedi, M.H. Personalized Antidepressant Selection and Pathway to Novel Treatments: Clinical Utility of Targeting Inflammation. Int. J. Mol. Sci. 2018, 19, 233. [Google Scholar] [CrossRef] [Green Version]
- Zanette, S.A.; Dussan-Sarria, J.A.; Souza, A.; Deitos, A.; Torres, I.L.; Caumo, W. Higher serum S100B and BDNF levels are correlated with a lower pressure-pain threshold in fibromyalgia. Mol. Pain 2014, 10, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bazzichi, L.; Da-Valle, Y.; Rossi, A.; Giacomelli, C.; Sernissi, F.; Giannaccini, G.; Betti, L.; Ciregia, F.; Giusti, L.; Scarpellini, P.; et al. multidisciplinary approach to study the effects of balneotherapy and mud-bath therapy treatments on fibromyalgia. Clin. Exp. Rheumatol. 2013, 31, S111–S120. [Google Scholar] [PubMed]
- Molendijk, M.L.; Bus, B.A.; Spinhoven, P.; Penninx, B.W.; Kenis, G.; Prickaerts, J.; Elzinga, B.M. Serum levels of brain-derived neurotrophic factor in major depressive disorder: State–trait issues, clinical features and pharmacological treatment. Mol. Psychiatry 2011, 16, 1088–1095. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bjelland, I.; Dahl, A.A.; Haug, T.T.; Neckelmann, D. The validity of the Hospital Anxiety and Depression Scale: An updated literature review. J. Psychosom. Res. 2002, 52, 69–77. [Google Scholar] [CrossRef]
| Session | RT | MAIR |
|---|---|---|
| 1 | Visualizations I. Presentation of the different relaxation techniques and their usefulness. | General overview. Theoretical aspects of the brain, the limbic system, fear, conditioning and reconditioning. Visualization of 100% recovery. |
| 2 | Visualizations II. Deepening in guided relaxation through imagination training. Noticing the effects of relaxation in the body and mind and learning when to use it. | Developing FM. How stress triggers the nervous system. Mindfulness and self-awareness. Anchoring presence in the body and breathing. |
| 3 | Visualizations III. Working with emotions through imagination. Emotional burdens are symbolically released, reducing emotional discomfort. | Amygdala technique. Reconditioning. Breaking negative thoughts, meanings, and emotions, and somatic answers in the body. Breathing and meditation. |
| 4 | Autogenic relaxation I. Autogenic relaxation initiation. Fostering sensations of relaxation through imagining a ball of light and heat. | Regulation of negative emotions and symptoms. The “soften and flow” self-regulation through mindfulness practice. Body scan meditation. |
| 5 | Autogenic relaxation II. Deepening in autogenic relaxation by learning when and how to use it. Working on heaviness sensations. | The accelerator of MAIR. Internal dialogue, patterns and behaviors related to illness in FM. Importance of mindfulness as a daily practice. Walking meditation. |
| 6 | Progressive relaxation. Initiation to the progressive relaxation. Tensing and relaxing the muscles to become aware of the change in body sensations. | Dealing with stress. Awareness of negative thoughts related to external stimuli. Over-reactions of the nervous system. Mindfulness in daily activities. |
| 7 | Breathing I. Learning to use breathing exercises. Knowing its benefits. Deep inspiration and exhalation. Using breathing to calm anxiety. | Awareness of limiting beliefs. How to identify and change them through reconditioning. Motivation and sense of life. Meditation on values. |
| 8 | Breathing II. Deepening in breathing exercises by learning different deep-breathing exercises. | Recovery, cycles, and stages and returning to regular life. Fear of failure in terms of recovery. Positive visualizations of the future. Review and summary of the protocol. |
| Characteristics at Baseline | RT (n = 19) | MAIR (n = 22) | (p) |
|---|---|---|---|
| Socio-demographics | |||
| Sex, female | 19 (100) | 22 (100) | − |
| Age | 52.21 (5.95) | 52.77 (13.45) | 0.86 |
| Marital status, stable relationship | 13 (68.4) | 12 (54.5) | 0.39 |
| Residence, urban | 19 (100) | 22 (100) | 0.27 |
| Dwelling, own home | 17 (89.5) | 18 (81.8) | 0.76 |
| Education | 0.66 | ||
| Primary | 4 (21.1) | 7 (31.8) | |
| Secondary | 8 (42.1) | 8 (36.4) | |
| University | 7 (36.8) | 7 (31.8) | |
| Employment | 0.43 | ||
| Employed | 5 (26.2) | 3 (13.6) | |
| Sick leave/inability | 8 (42.2) | 8 (36.4) | |
| Unemployed | 6 (31.6) | 11 (50.0) | |
| Clinical measures | |||
| Fibromyalgia impact | |||
| FIQ (0–100) | 62.83 (18.41) | 70.61 (17.21) | 0.17 |
| Clinical severity | |||
| CGI-S (1–7) | 4.32 (1.16) | 4.59 (1.14) | 0.45 |
| Pain catastrophizing | |||
| PCS (0–52) | 25.00 (10.92) | 29.50 (9.73) | 0.17 |
| Anxiety and depression | |||
| HADS-A (0–21) | 11.37 (5.40) | 12.32 (3.48) | 0.52 |
| HADS-D (0–21) | 8.05 (6.03) | 9.73 (5.19) | 0.35 |
| Perceived health | |||
| EQ-VAS (0–100) | 54.00 (20.19) | 48.18 (17.01) | 0.32 |
| Psychological inflexibility | |||
| AAQ-II (10–70) | 37.32 (13.34) | 41.00 (10.45) | 0.33 |
| Mindfulness facets | |||
| FFMQ (39–195) | 118.74 (14.96) | 116.32 (18.48) | 0.65 |
| Self-compassion | |||
| SCS (6–30) | 16.81 (4.13) | 16.63 (3.81) | 0.89 |
| Taking pharmacological treatment | |||
| Non-opioid analgesics | 2 (10.5) | 3 (13.6)) | 0.76 |
| Opioids | 2 (10.5) | 4 (18.2) | 0.49 |
| NSAIDs | 2 (10.5) | 2 (9.1) | 0.88 |
| Antidepressants | 2 (10.5) | 8 (36.4) | 0.06 |
| Anticonvulsants | 2 (10.5) | 4 (18.2) | 0.49 |
| Benzodiazepines | 6 (31.6) | 5 (22.7) | 0.52 |
| Antipsychotics | 0 (0.0) | 1 (4.5) | 0.35 |
| Total number of medications | 0.84 (0.96) | 1.23 (0.97) | 0.21 |
| RT (n = 15) M (SD) | MAIR (n = 19) M (SD) | d | B (95% CI) | z (p) | |
|---|---|---|---|---|---|
| FIQ (0–100) | |||||
| Baseline | 61.12 (20.21) | 68.03 (17.02) | |||
| Post-treatment | 61.22 (25.90) | 42.84 (20.57) | −1.34 | −26.38 (−40.87–−11.89) | −3.57 (<0.001) |
| Follow-up | 67.82 (17.77) | 51.05 (16.30) | −1.25 | −23.99 (−38.64–−9.33) | −3.21 (0.001) |
| CGI-S (1–7) | |||||
| Baseline | 4.27 (1.28) | 4.47 (1.12) | |||
| Post-treatment | 4.33 (0.82) | 3.79 (0.86) | −0.62 | −0.72 (−1.52–0.08) | −1.76 (0.078) |
| Follow-up | 4.07 (0.80) | 3.32 (1.00) | −0.79 | −0.98 (−1.79–−0.18) | −2.39 (0.017) |
| PCS (0–52) | |||||
| Baseline | 25.93 (10.14) | 30.13 (8.40) | |||
| Post-treatment | 23.47 (14.49) | 22.67 (13.14) | −0.52 | −3.48 (−10.07–3.12) | −1.03 (0.302) |
| Follow-up | 23.53 (13.58) | 16.20 (9.83) | −1.20 | −10.00 (−16.41–−3.59) | −3.06 (0.002) |
| HADS-A (0–21) | |||||
| Baseline | 11.53 (6.06) | 12.42 (3.73) | |||
| Post-treatment | 10.53 (5.24) | 8.05 (3.60) | −0.68 | −3.15 (−5.37–−0.93) | −2.78 (0.005) |
| Follow-up | 9.80 (4.84) | 6.84 (1.54) | −0.78 | −3.75 (−6.00–−1.50) | −3.27 (0.001) |
| HADS-D (0–21) | |||||
| Baseline | 8.33 (6.67) | 9.32 (5.11) | |||
| Post-treatment | 7.53 (4.81) | 5.05 (3.70) | −0.59 | −4.07 (−6.54–−1.60) | −3.23 (0.001) |
| Follow-up | 7.80 (5.99) | 5.47 (3.57) | −0.56 | −3.66 (−6.16–−1.15) | −2.86 (0.004) |
| EQ-VAS (0–100) | |||||
| Baseline | 53.07 (21.71) | 47.89 (16.10) | |||
| Post-treatment | 56.87 (18.95) | 64.74 (16.87) | 0.69 | 12.26 (−0.55–25.08) | 1.88 (0.061) |
| Follow-up | 61.67 (15.66) | 70.63 (14.29) | 0.75 | 14.08 (1.11–27.04) | 2.13 (0.033) |
| AAQ-II (10–70) | |||||
| Baseline | 38.00 (14.23) | 40.95 (11.20) | |||
| Post-treatment | 39.07 (13.90) | 34.79 (11.65) | −0.57 | −7.64 (−15.43–0.15) | −1.92 (0.055) |
| Follow-up | 37.00 (12.78) | 26.21 (4.74) | −1.08 | −14.06 (−21.95–−6.18) | −3.49 (<0.001) |
| FFMQ (39–195) | |||||
| Baseline | 120.07 (16.36) | 117.16 (18.41) | |||
| Post-treatment | 121.87 (23.71) | 131.79 (17.95) | 0.71 | 13.44 (2.87–24.01) | 2.49 (0.013) |
| Follow-up | 122.67 (19.88) | 132.32 (13.06) | 0.70 | 12.46 (1.76–23.16) | 2.28 (0.022) |
| SCS (6–30) | |||||
| Baseline | 17.23 (4.40) | 16.31 (3.64) | |||
| Post-treatment | 17.19 (4.57) | 20.31 (4.25) | 0.99 | 3.80 (1.45–6.15) | 3.17 (0.002) |
| Follow-up | 17.14 (4.53) | 23.28 (3.35) | 1.73 | 6.78 (4.40–9.16) | 5.58 (<0.001) |
| RT (n = 16) M (SD) | MAIR (n = 19) M (SD) | d | B (95% CI) | z (p) | |
|---|---|---|---|---|---|
| BDNF | |||||
| Pre- | 19.34 (6.62) | 22.72 (8.24) | |||
| Post- | 21.54 (7.08) | 20.47 (6.13) | −0.58 | −5.94 (−9.65–−2.23) | −3.13 (0.002) |
| hs-CRP | |||||
| Pre- | 3.54 (4.36) | 4.68 (6.42) | |||
| Post- | 4.00 (4.12) | 3.85 (5.50) | −0.23 | −0.54 (−1.19–0.11) | −1.64 (0.101) |
| TNF-α | |||||
| Pre- | 5.99 (2.74) | 5.92 (1.65) | |||
| Post- | 5.93 (4.13) | 5.60 (2.30) | −0.12 | −0.06 (−0.25–0.12) | −0.68 (0.499) |
| IL-6 | |||||
| Pre- | 3.04 (1.18) | 3.35 (2.63) | |||
| Post- | 3.14 (1.87) | 3.44 (1.12) | −0.01 | 0.12 (−0.24–0.49) | 0.65 (0.514) |
| IL-10 | |||||
| Pre- | 5.13 (0.43) | 5.59 (1.44) | |||
| Post- | 5.02 (1.41) | 5.31 (0.53) | −0.15 | −0.07 (−0.16–0.02) | −1.51 (0.132) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanabria-Mazo, J.P.; Montero-Marin, J.; Feliu-Soler, A.; Gasión, V.; Navarro-Gil, M.; Morillo-Sarto, H.; Colomer-Carbonell, A.; Borràs, X.; Tops, M.; Luciano, J.V.; et al. Mindfulness-Based Program Plus Amygdala and Insula Retraining (MAIR) for the Treatment of Women with Fibromyalgia: A Pilot Randomized Controlled Trial. J. Clin. Med. 2020, 9, 3246. https://doi.org/10.3390/jcm9103246
Sanabria-Mazo JP, Montero-Marin J, Feliu-Soler A, Gasión V, Navarro-Gil M, Morillo-Sarto H, Colomer-Carbonell A, Borràs X, Tops M, Luciano JV, et al. Mindfulness-Based Program Plus Amygdala and Insula Retraining (MAIR) for the Treatment of Women with Fibromyalgia: A Pilot Randomized Controlled Trial. Journal of Clinical Medicine. 2020; 9(10):3246. https://doi.org/10.3390/jcm9103246
Chicago/Turabian StyleSanabria-Mazo, Juan P., Jesus Montero-Marin, Albert Feliu-Soler, Virginia Gasión, Mayte Navarro-Gil, Héctor Morillo-Sarto, Ariadna Colomer-Carbonell, Xavier Borràs, Mattie Tops, Juan V. Luciano, and et al. 2020. "Mindfulness-Based Program Plus Amygdala and Insula Retraining (MAIR) for the Treatment of Women with Fibromyalgia: A Pilot Randomized Controlled Trial" Journal of Clinical Medicine 9, no. 10: 3246. https://doi.org/10.3390/jcm9103246
APA StyleSanabria-Mazo, J. P., Montero-Marin, J., Feliu-Soler, A., Gasión, V., Navarro-Gil, M., Morillo-Sarto, H., Colomer-Carbonell, A., Borràs, X., Tops, M., Luciano, J. V., & García-Campayo, J. (2020). Mindfulness-Based Program Plus Amygdala and Insula Retraining (MAIR) for the Treatment of Women with Fibromyalgia: A Pilot Randomized Controlled Trial. Journal of Clinical Medicine, 9(10), 3246. https://doi.org/10.3390/jcm9103246








