Validity of Bioimpedance Equations to Evaluate Fat-Free Mass and Muscle Mass in Severely Malnourished Anorectic Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Dual-Energy X-ray Absorptiometry (DXA)
2.3. Bioelectrical Impedance Analysis (BIA)
2.4. Statistical Analysis
3. Results
3.1. Comparison of DXA and Derived BIA Values for Fat Mass and Fat-Free Mass
3.2. Comparison of DXA and Derived BIA Values for Appendicular Lean Mass
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Battle, D.E. Diagnostic and Statistical Manual of Mental Disorders (DSM). Codas 2013, 25, 191–192. [Google Scholar] [CrossRef] [PubMed]
- Tannir, H.; Itani, L.; Kreidieh, D.; El Masri, D.; Traboulsi, S.; El Ghoch, M. Body composition in adolescents and young adults with anorexia nervosa: A clinical review. Curr. Rheumatol. Rev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Lemos, T.; Gallagher, D. Current body composition measurement techniques. Curr. Opin. Endocrinol. Diabetes Obes. 2017, 24, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Andreoli, A.; Scalzo, G.; Masala, S.; Tarantino, U.; Guglielmi, G. Body composition assessment by dual-energy X-ray absorptiometry (DXA). Radiol. Med. 2009, 114, 286–300. [Google Scholar] [CrossRef] [PubMed]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Manuel Gomez, J.; Lilienthal Heitmann, B.; Kent-Smith, L.; Melchior, J.C.; Pirlich, M.; et al. Bioelectrical impedance analysis-part II: Utilization in clinical practice. Clin. Nutr. 2004, 23, 1430–1453. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorsi, G.; Bassetti, A.; Chiari, S.; Dirindelli, P.; Lorini, C.; Menicalli, C.; Santomauro, F.; Martinetti, M.G. Body composition in subjects with anorexia nervosa: Bioelectrical impedance analysis and dual-energy X-ray absorptiometry. Eat. Weight Disord. 2012, 17, e298–e303. [Google Scholar] [CrossRef]
- Mattar, L.; Godart, N.; Melchior, J.C.; Falissard, B.; Kolta, S.; Ringuenet, D.; Vindreau, C.; Nordon, C.; Blanchet, C.; Pichard, C. Underweight patients with anorexia nervosa: Comparison of bioelectrical impedance analysis using five equations to dual X-ray absorptiometry. Clin. Nutr. 2011, 30, 746–752. [Google Scholar] [CrossRef]
- Haas, V.; Stark, D.; Kohn, M.; Muller, M.J.; Clarke, S.; Blumenthal, C.; Briody, J.; Madden, S.; Gaskin, K.J. Evaluation of methods to assess reduced body fat in patients with anorexia nervosa. J. Am. Coll. Nutr. 2013, 32, 2–10. [Google Scholar] [CrossRef]
- Birmingham, C.L.; Jones, P.J.; Orphanidou, C.; Bakan, R.; Cleator, I.G.; Goldner, E.M.; Phang, P.T. The reliability of bioelectrical impedance analysis for measuring changes in the body composition of patients with anorexia nervosa. Int. J. Eat. Disord. 1996, 19, 311–315. [Google Scholar] [CrossRef]
- Achamrah, N.; Colange, G.; Delay, J.; Rimbert, A.; Folope, V.; Petit, A.; Grigioni, S.; Dechelotte, P.; Coeffier, M. Comparison of body composition assessment by DXA and BIA according to the body mass index: A retrospective study on 3655 measures. PLoS ONE 2018, 13, e0200465. [Google Scholar] [CrossRef]
- Mareschal, J.; Achamrah, N.; Norman, K.; Genton, L. Clinical Value of Muscle Mass Assessment in Clinical Conditions Associated with Malnutrition. J. Clin. Med. 2019, 8, 1040. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Jensen, G.L.; Correia, M.; Gonzalez, M.C.; Fukushima, R.; Higashiguchi, T.; Baptista, G.; Barazzoni, R.; Blaauw, R.; Coats, A.; et al. GLIM criteria for the diagnosis of malnutrition—A consensus report from the global clinical nutrition community. Clin. Nutr. 2019, 38, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Verdich, C.; Barbe, P.; Petersen, M.; Grau, K.; Ward, L.; Macdonald, I.; Sorensen, T.I.; Oppert, J.M. Changes in body composition during weight loss in obese subjects in the NUGENOB study: Comparison of bioelectrical impedance vs. dual-energy X-ray absorptiometry. Diabetes Metab. 2011, 37, 222–229. [Google Scholar] [CrossRef]
- Bedogni, G.; Marra, M.; Bianchi, L.; Malavolti, M.; Nicolai, E.; De Filippo, E.; Scalfi, L. Comparison of bioelectrical impedance analysis and dual-energy X-ray absorptiometry for the assessment of appendicular body composition in anorexic women. Eur. J. Clin. Nutr. 2003, 57, 1068–1072. [Google Scholar] [CrossRef]
- Deurenberg, P.; van der Kooy, K.; Leenen, R.; Weststrate, J.A.; Seidell, J.C. Sex and age specific prediction formulas for estimating body composition from bioelectrical impedance: A cross-validation study. Int. J. Obes. 1991, 15, 17–25. [Google Scholar]
- Kushner, R.F.; Schoeller, D.A. Estimation of total body water by bioelectrical impedance analysis. Am. J. Clin. Nutr. 1986, 44, 417–424. [Google Scholar] [CrossRef]
- Kyle, U.G.; Genton, L.; Karsegard, L.; Slosman, D.O.; Pichard, C. Single prediction equation for bioelectrical impedance analysis in adults aged 20–94 years. Nutrition 2001, 17, 248–253. [Google Scholar] [CrossRef]
- Roubenoff, R.; Baumgartner, R.N.; Harris, T.B.; Dallal, G.E.; Hannan, M.T.; Economos, C.D.; Stauber, P.M.; Wilson, P.W.; Kiel, D.P. Application of bioelectrical impedance analysis to elderly populations. J. Gerontol. Ser. A Biol. Sci. Med Sci. 1997, 52, M129–M136. [Google Scholar] [CrossRef]
- Scalfi, L.; Bedogni, G.; Marra, M.; Di Biase, G.; Caldara, A.; Severi, S.; Contaldo, F.; Battistini, N. The prediction of total body water from bioelectrical impedance in patients with anorexia nervosa. Br. J. Nutr. 1997, 78, 357–365. [Google Scholar] [CrossRef]
- Scalfi, L.; Marra, M.; Caldara, A.; Silvestri, E.; Contaldo, F. Changes in bioimpedance analysis after stable refeeding of undernourished anorexic patients. Int. J. Obes. Relat. Metab. Disord. 1999, 23, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.S.; Chumlea, W.C.; Heymsfield, S.B.; Lukaski, H.C.; Schoeller, D.; Friedl, K.; Kuczmarski, R.J.; Flegal, K.M.; Johnson, C.L.; Hubbard, V.S. Development of bioelectrical impedance analysis prediction equations for body composition with the use of a multicomponent model for use in epidemiologic surveys. Am. J. Clin. Nutr. 2003, 77, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Kyle, U.G.; Genton, L.; Hans, D.; Pichard, C. Validation of a bioelectrical impedance analysis equation to predict appendicular skeletal muscle mass (ASMM). Clin. Nutr. 2003, 22, 537–543. [Google Scholar] [CrossRef]
- Yoshida, D.; Shimada, H.; Park, H.; Anan, Y.; Ito, T.; Harada, A.; Suzuki, T. Development of an equation for estimating appendicular skeletal muscle mass in Japanese older adults using bioelectrical impedance analysis. Geriatr. Gerontol. Int. 2014, 14, 851–857. [Google Scholar] [CrossRef]
- Scafoglieri, A.; Clarys, J.P.; Bauer, J.M.; Verlaan, S.; Van Malderen, L.; Vantieghem, S.; Cederholm, T.; Sieber, C.C.; Mets, T.; Bautmans, I.; et al. Predicting appendicular lean and fat mass with bioelectrical impedance analysis in older adults with physical function decline—The PROVIDE study. Clin. Nutr. 2017, 36, 869–875. [Google Scholar] [CrossRef]
- Tengvall, M.; Ellegard, L.; Malmros, V.; Bosaeus, N.; Lissner, L.; Bosaeus, I. Body composition in the elderly: Reference values and bioelectrical impedance spectroscopy to predict total body skeletal muscle mass. Clin. Nutr. 2009, 28, 52–58. [Google Scholar] [CrossRef]
- Yu, S.C.; Powell, A.; Khow, K.S.; Visvanathan, R. The Performance of Five Bioelectrical Impedance Analysis Prediction Equations against Dual X-ray Absorptiometry in Estimating Appendicular Skeletal Muscle Mass in an Adult Australian Population. Nutrients 2016, 8, 189. [Google Scholar] [CrossRef]
- Sergi, G.; De Rui, M.; Veronese, N.; Bolzetta, F.; Berton, L.; Carraro, S.; Bano, G.; Coin, A.; Manzato, E.; Perissinotto, E. Assessing appendicular skeletal muscle mass with bioelectrical impedance analysis in free-living Caucasian older adults. Clin. Nutr. 2015, 34, 667–673. [Google Scholar] [CrossRef]
- Rangel Peniche, D.B.; Raya Giorguli, G.; Aleman-Mateo, H. Accuracy of a predictive bioelectrical impedance analysis equation for estimating appendicular skeletal muscle mass in a non-Caucasian sample of older people. Arch. Gerontol. Geriatr. 2015, 61, 39–43. [Google Scholar] [CrossRef]
- Kim, J.H.; Choi, S.H.; Lim, S.; Kim, K.W.; Lim, J.Y.; Cho, N.H.; Park, K.S.; Jang, H.C. Assessment of appendicular skeletal muscle mass by bioimpedance in older community-dwelling Korean adults. Arch. Gerontol. Geriatr. 2014, 58, 303–307. [Google Scholar] [CrossRef]
- Janssen, I.; Heymsfield, S.B.; Baumgartner, R.N.; Ross, R. Estimation of skeletal muscle mass by bioelectrical impedance analysis. J. Appl. Physiol. 2000, 89, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.I. A concordance correlation coefficient to evaluate reproducibility. Biometrics 1989, 45, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.Y.; Peng, C.H.; Hung, S.C.; Tarng, D.C. Body composition is associated with clinical outcomes in patients with non-dialysis-dependent chronic kidney disease. Kidney Int. 2018, 93, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Marra, M.; Sammarco, R.; De Filippo, E.; Caldara, A.; Speranza, E.; Scalfi, L.; Contaldo, F.; Pasanisi, F. Prediction of body composition in anorexia nervosa: Results from a retrospective study. Clin. Nutr. 2018, 37, 1670–1674. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.L.; Benavides, M.L.; Dellinger, J.R.; Adamson, B.T.; Tinsley, G.M. Segmental body composition evaluation by bioelectrical impedance analysis and dual-energy X-ray absorptiometry: Quantifying agreement between methods. Clin. Nutr. 2020, 39, 2802–2810. [Google Scholar] [CrossRef] [PubMed]
- Hubel, C.; Yilmaz, Z.; Schaumberg, K.E.; Breithaupt, L.; Hunjan, A.; Horne, E.; Garcia-Gonzalez, J.; O’Reilly, P.F.; Bulik, C.M.; Breen, G. Body composition in anorexia nervosa: Meta-analysis and meta-regression of cross-sectional and longitudinal studies. Int. J. Eat. Disord. 2019, 52, 1205–1223. [Google Scholar] [CrossRef]
- El Ghoch, M.; Calugi, S.; Lamburghini, S.; Dalle Grave, R. Anorexia nervosa and body fat distribution: A systematic review. Nutrients 2014, 6, 3895–3912. [Google Scholar] [CrossRef]
- El Ghoch, M.; Pourhassan, M.; Milanese, C.; Muller, M.J.; Calugi, S.; Bazzani, P.V.; Dalle Grave, R. Changes in lean and skeletal muscle body mass in adult females with anorexia nervosa before and after weight restoration. Clin. Nutr. 2017, 36, 170–178. [Google Scholar] [CrossRef]
- Achamrah, N.; Coeffier, M.; Dechelotte, P. Physical activity in patients with anorexia nervosa. Nutr. Rev. 2016, 74, 301–311. [Google Scholar] [CrossRef]

| Equation | |
|---|---|
| Deurenberg et al. [16] | FFM = −12.44 + (0.34 × height2/resistance) + (0.1534 × height) + (0.273 × weight) − (0.127 × age) |
| Kyle et al. [18] | FFM = −4.104 + (0.518 × height2/resistance) + (0.231 × weight) + (0.130 × reactance) |
| Sun et al. [22] | FFM = −9.529 + 0.696 × (height2/resistance) + (0.168 × weight) + (0.016 × resistance) |
| Bedogni et al. [15] | FFM = 0.6 × (height2/impedance 50kHz) + (0.2 × weight) + 3.3 |
| Roubenoff et al. [19] | FFM = 7.7435 + (0.4542 × height2/resistance) + (0.1190 × weight) + 0.0455 × reactance |
| Scalfi-1 et al. [20] | BWa = (0.434 × weight) + 6.326 |
| Scalfi-2 et al. [20] | BWa = (0.563 × height2/impedance 100kHz) + 2.695 |
| Kushner et al. [17] | BWa = (8.315 + 0.382 × height2/resistance) + 0.105 × weight |
| Equation | |
|---|---|
| Janssen et al. [31] | SMM = (height2/resistance × 0.401) + (gender × 3.825) + (age × −0.071) + 5.102 |
| Tengvall et al. [26] | SMM = −24.021 + (height × 0.33) + (resistance × −0.031) + (reactance × 0.083) + (gender × −1.58) + (weight × 0.046) |
| Scafoglieri et al. [25] | ALM = 1.821 + 0,168 × (height2/resistance) + (0,132 × weight) + (0,017 × reactance) − (gender × 1.931) |
| Kyle et al. [23] | ALM = −4.211 × (height2/resistance × 0.267) + (weight × 0.095) + (gender × 1.909) +(age × −0.012) + (reactance × 0.058) |
| Sergi et al. [28] | ALM = −3.964 + 0.227 × (resistance/height2) + (0,095 × weight) + (gender × 1.384) + (reactance × 0.064) |
| Rangel Peniche et al. [29] | ALM = −0.05376 + 0.2394 × (height2/resistance) + (2.708 × gender) + (weight × 0.065) |
| Kim et al. [30] | ALM = ((height2/resistance × 0.104) + (−0.050 × age) + (gender × 2.954) + (weight × 0.055)) + 5.663 |
| Yoshida et al. [24] | ALM = 0.221 × (height2/resistance) + (0.117 × weight) + 0.881 |
| n = 115 | Mean ± Standard Deviation | Range |
|---|---|---|
| Age (y) | 32.3 ± 14.5 | 18–67 |
| Weight (kg) | 39.9 ± 4.6 | 26.6–50.0 |
| BMI (kg/m2) | 14.6 ± 1.2 | 10.9–15.9 |
| ALM (kg) | 14.88 ± 2.04 | 9.35–20.94 |
| FM (kg) | 4.2 ± 2.4 | 1.1–12.5 |
| FM (%) | 11.0 ± 5.6 | 4.0–26.4 |
| FFM (kg) | 35.5 ± 3.8 | 24.2–43.9 |
| FFM (%) | 88.2 ± 7.2 | 73.6–96.0 |
| n = 115, Ω | Mean ± Standard Deviation | Range |
|---|---|---|
| Reactance | 61.31 ± 13.15 | 29.3–100.4 |
| Resistance | 686.2 ± 79.58 | 517.0–887.0 |
| Impedance 5 kHz | 652.6 ± 231.9 | 30–941 |
| Impedance 50 kHz | 689.3 ± 79.82 | 518–888 |
| Impedance 100 kHz | 660.0 ± 77.73 | 498–856 |
| Impedance 200 kHz | 631.0 ± 75.92 | 473–843 |
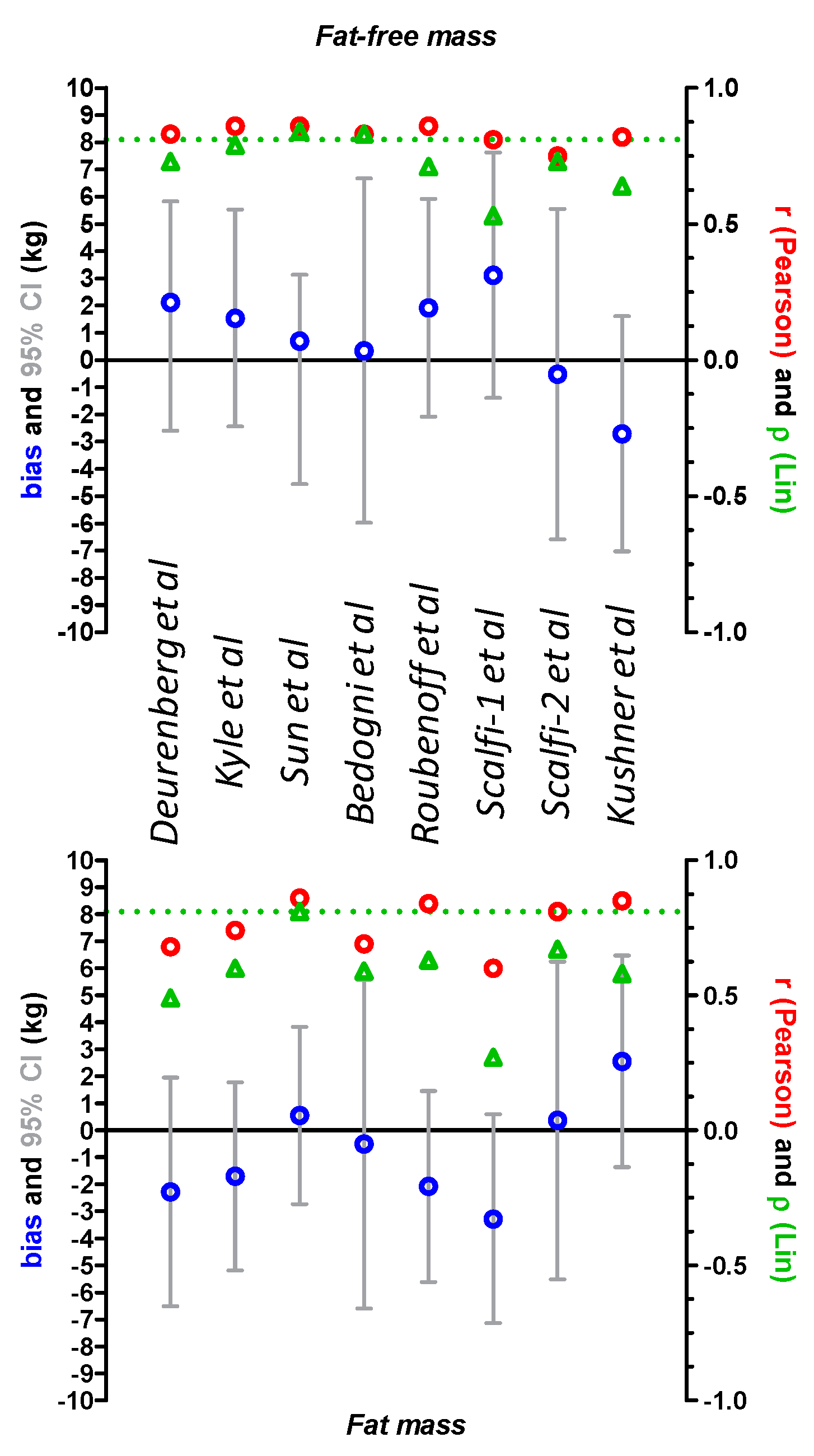
| Bias (kg) | 95% CI (kg) | Bias (%) | Accuracy (%) | Pearson r | Lin ρ | |
|---|---|---|---|---|---|---|
| Fat-free mass | ||||||
| Deurenberg et al. | 2.12 | (−2.59; 5.83) | 5.97 | 47.8 | 0.83 | 0.73 |
| Kyle et al. | 1.54 | (−2.44; 5.53) | 4.33 | 47.8 | 0.86 | 0.79 |
| Sun et al. | 0.70 | (−4.55; 3.13) | 1.97 | 66.9 | 0.86 | 0.84 |
| Bedogni et al. | 0.34 | (−5.97; 6.67) | 0.96 | 66.1 | 0.83 | 0.83 |
| Roubenoff et al. | 1.92 | (−2.07; 5.92) | 5.40 | 40.0 | 0.86 | 0.71 |
| Scalfi-1 et al. | 3.11 | (−1.39; 7.63) | 8.75 | 22.6 | 0.81 | 0.53 |
| Scalfi-2 et al. | −0.52 | (−6.59; 5.55) | −1.46 | 50.4 | 0.75 | 0.73 |
| Kushner et al. | −2.71 | (−7.03; 1.61) | −7.63 | 33.9 | 0.82 | 0.64 |
| Fat mass | ||||||
| Deurenberg et al. | −2.28 | (−6.51; 1.95) | −54.3 | 2.6 | 0.68 | 0.49 |
| Kyle et al. | −1.70 | (−5.19; 1.78) | −40.5 | 3.5 | 0.74 | 0.60 |
| Sun et al. | 0.55 | (−2.73; 3.83) | 13.1 | 13.9 | 0.86 | 0.81 |
| Bedogni et al. | −0.51 | (−6.60; 5.58) | −12.1 | 5.2 | 0.69 | 0.59 |
| Roubenoff et al. | −2.08 | (−5.61; 1.45) | −49.5 | 2.6 | 0.84 | 0.63 |
| Scalfi-1 et al. | −3.28 | (−7.14; 0.59) | −78.1 | 0.9 | 0.60 | 0.27 |
| Scalfi-2 et al. | 0.37 | (−5.52; 6.25) | 8.81 | 2.6 | 0.81 | 0.67 |
| Kushner et al. | 2.55 | (−1.37; 6.47) | 60.7 | 2.6 | 0.85 | 0.58 |
| Bias (kg) | 95% CI (kg) | Bias (%) | Accuracy (%) | Pearson r | Lin ρ | |
|---|---|---|---|---|---|---|
| Janssen et al. | −4.10 | (−7.09; −1.12) | −27.5 | 1.7 | 0.81 | 0.31 |
| Tengvall et al. | −1.24 | (−5.10; 2.62) | −8.4 | 45.2 | 0.86 | 0.69 |
| Scafoglieri et al. | −0.03 | (−2.06; 1.99) | −0.2 | 83.5 | 0.89 | 0.82 |
| Kyle et al. | 1.35 | (−0.65; 3.36) | 9.0 | 53.0 | 0.87 | 0.69 |
| Sergi et al. | 1.97 | (−0.17; 4.11) | 13.2 | 30.4 | 0.84 | 0.53 |
| Rangel Peniche et al. | 2.68 | (0.45; 4.90) | 18.0 | 10.4 | 0.83 | 0.38 |
| Kim et al. | 4.43 | (1.79; 7.09) | 29.8 | 0.0 | 0.78 | 0.14 |
| Yoshida et al. | 0.41 | (−1.62; 2.44) | 2.8 | 82.6 | 0.86 | 0.82 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coëffier, M.; Gâté, M.; Rimbert, A.; Petit, A.; Folope, V.; Grigioni, S.; Déchelotte, P.; Achamrah, N. Validity of Bioimpedance Equations to Evaluate Fat-Free Mass and Muscle Mass in Severely Malnourished Anorectic Patients. J. Clin. Med. 2020, 9, 3664. https://doi.org/10.3390/jcm9113664
Coëffier M, Gâté M, Rimbert A, Petit A, Folope V, Grigioni S, Déchelotte P, Achamrah N. Validity of Bioimpedance Equations to Evaluate Fat-Free Mass and Muscle Mass in Severely Malnourished Anorectic Patients. Journal of Clinical Medicine. 2020; 9(11):3664. https://doi.org/10.3390/jcm9113664
Chicago/Turabian StyleCoëffier, Moise, Mathilde Gâté, Agnès Rimbert, André Petit, Vanessa Folope, Sébastien Grigioni, Pierre Déchelotte, and Najate Achamrah. 2020. "Validity of Bioimpedance Equations to Evaluate Fat-Free Mass and Muscle Mass in Severely Malnourished Anorectic Patients" Journal of Clinical Medicine 9, no. 11: 3664. https://doi.org/10.3390/jcm9113664
APA StyleCoëffier, M., Gâté, M., Rimbert, A., Petit, A., Folope, V., Grigioni, S., Déchelotte, P., & Achamrah, N. (2020). Validity of Bioimpedance Equations to Evaluate Fat-Free Mass and Muscle Mass in Severely Malnourished Anorectic Patients. Journal of Clinical Medicine, 9(11), 3664. https://doi.org/10.3390/jcm9113664





