Percutaneous Endoscopic Cervical Discectomy versus Anterior Cervical Discectomy and Fusion: A Comparative Cohort Study with a Five-Year Follow-Up
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. Outcome Measurements
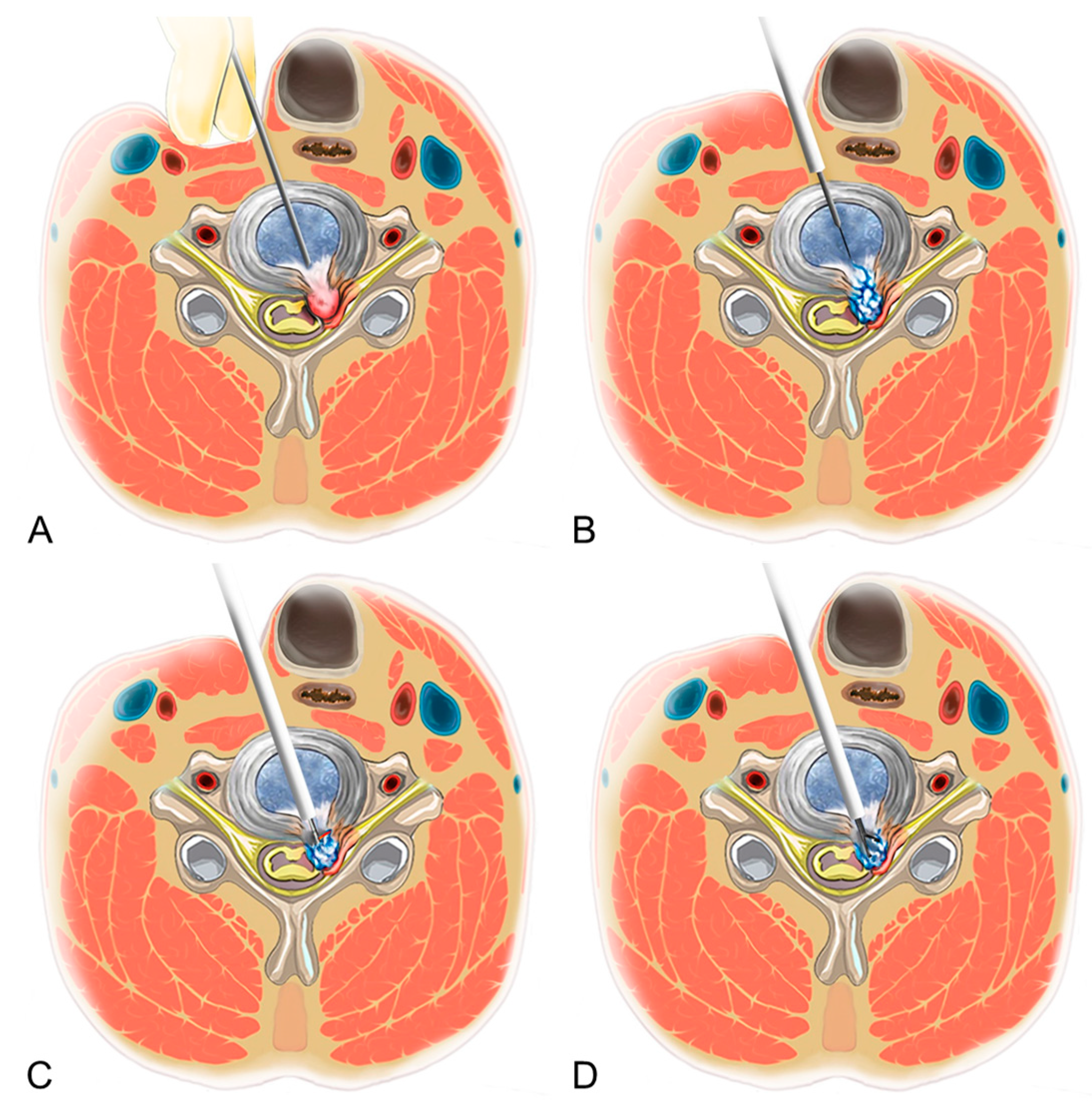
2.3. Surgical Techniques
2.4. Statistical Analysis
3. Results
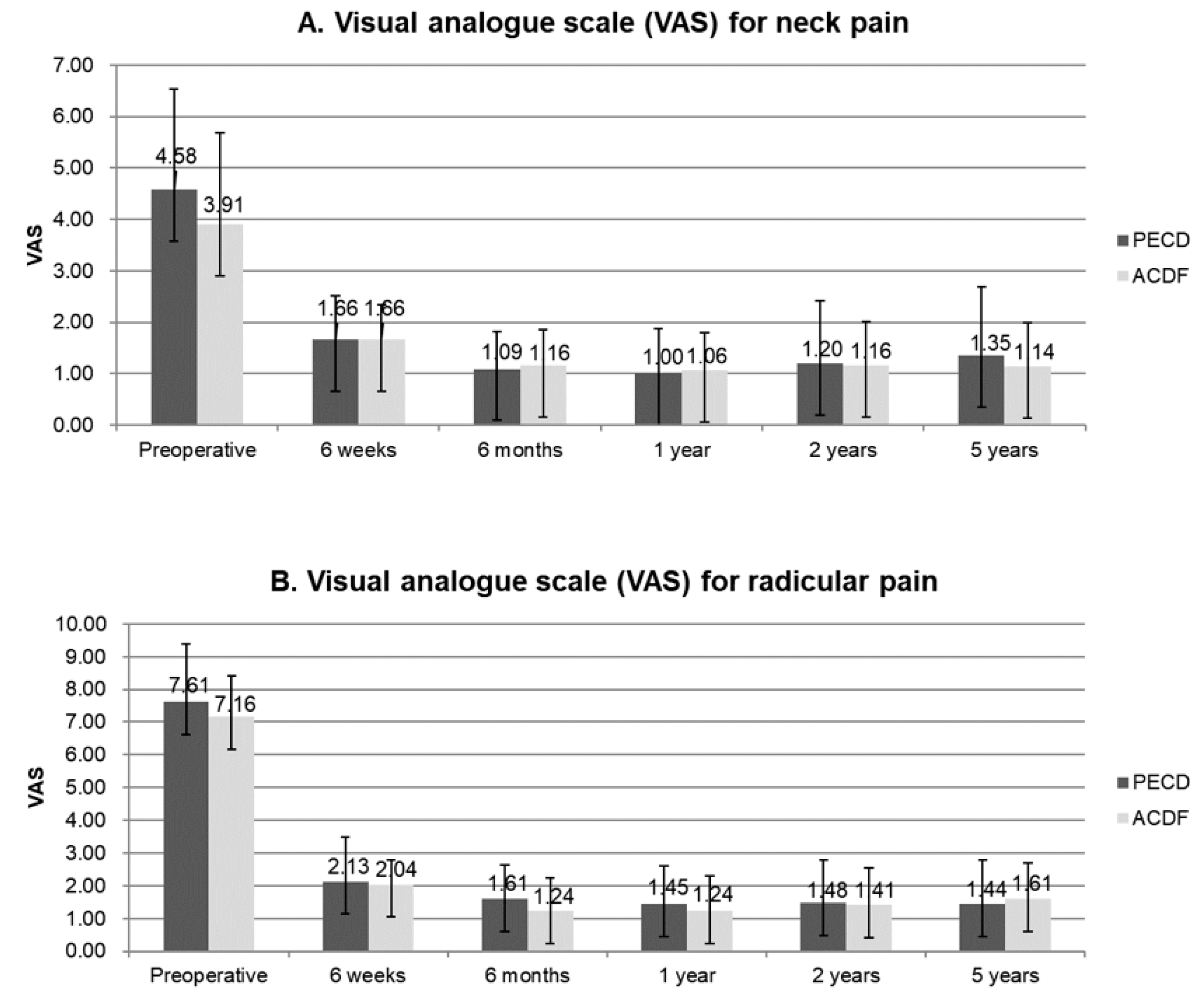
3.1. Demographics and Clinical Outcomes
3.2. Perioperative Data
3.3. Complications and Revisions
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Flynn, T.B. Neurologic complications of anterior cervical interbody fusion. Spine (Phila Pa 1976) 1982, 7, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Bulger, R.F.; Rejowski, J.E.; Beatty, R.A. Vocal cord paralysis associated with anterior cervical fusion: Considerations for prevention and treatment. J. Neurosurg. 1985, 62, 657–661. [Google Scholar] [CrossRef]
- Pedram, M.; Castagnera, L.; Carat, X.; Macouillard, G.; Vital, J.M. Pharyngolaryngeal lesions in patients undergoing cervical spine surgery through the anterior approach: Contribution of methylprednisolone. Eur. Spine J. 2003, 12, 84–90. [Google Scholar] [CrossRef]
- Wang, M.C.; Chan, L.; Maiman, D.J.; Kreuter, W.; Deyo, R.A. Complications and mortality associated with cervical spine surgery for degenerative disease in the United States. Spine (Phila Pa 1976) 2007, 32, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Maiman, D.J.; Kumaresan, S.; Yoganandan, N.; Pintar, F.A. Biomechanical effect of anterior cervical spine fusion on adjacent segments. Biomed. Mater. Eng. 1999, 9, 27–38. [Google Scholar]
- Epstein, N.E. A review of laminoforaminotomy for the management of lateral and foraminal cervical disc herniations or spurs. Surg. Neurol. 2002, 57, 226–233. [Google Scholar] [CrossRef]
- Kulkarni, V.; Rajshekhar, V.; Raghuram, L. Accelerated spondylotic changes adjacent to the fused segment following central cervical corpectomy: Magnetic resonance imaging study evidence. J. Neurosurg. Spine 2004, 100 (Suppl. 1), 2–6. [Google Scholar] [CrossRef] [Green Version]
- Hijikata, S. Percutaneous nucleotomy. A new concept technique and 12 years’ experience. Clin. Orthop. 1989, 238, 9–23. [Google Scholar] [CrossRef]
- Kambin, P.; Sampson, S. Posterolateral percutaneous suction-excision of herniated lumbar intervertebral discs. Report of interim results. Clin. Orthop. Relat. Res. 1986, 207, 37–43. [Google Scholar]
- Lee, J.H.; Lee, S.H. Clinical and radiographic changes after percutaneous endoscopic cervical discectomy: A long-term follow-up. Photomed. Laser Surg. 2014, 32, 663–668. [Google Scholar] [CrossRef]
- Ahn, Y. Percutaneous endoscopic cervical discectomy using working channel endoscopes. Expert Rev. Med. Devices 2016, 13, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Courtheoux, F.; Theron, J. Automated percutaneous nucleotomy in the treatment of cervicobrachial neuralgia due to disc herniation. J. Neuroradiol. 1992, 19, 211–216. [Google Scholar] [PubMed]
- Bonaldi, G.; Minonzio, G.; Belloni, G.; Dorizzi, A.; Fachinetti, P.; Marra, A.; Goddi, A. Percutaneous cervical diskectomy: Preliminary experience. Neuroradiology 1994, 36, 483–486. [Google Scholar] [CrossRef] [PubMed]
- Hoogland, T.; Scheckenbach, C. Low-dose chemonucleolysis combined with percutaneous nucleotomy in herniated cervical disks. J. Spinal Disord. 1995, 8, 228–232. [Google Scholar] [CrossRef]
- Siebert, W. Percutaneous laser discectomy of cervical discs: Preliminary clinical results. J. Clin. Laser Med. Surg. 1995, 13, 205–207. [Google Scholar] [CrossRef]
- Hellinger, J. Technical aspects of the percutaneous cervical and lumbar laser-disc-decompression and nucleotomy. Neurol. Res. 1999, 21, 99–102. [Google Scholar] [CrossRef]
- Knight, M.T.; Goswami, A.; Patko, J.T. Cervical percutaneous laser disc decompression: Preliminary results of an ongoing prospective outcome study. J. Clin. Laser Med. Surg. 2001, 19, 3–8. [Google Scholar] [CrossRef]
- Chiu, J.C.; Clifford, T.J.; Greenspan, M.; Richley, R.C.; Lohman, G.; Sison, R.B. Percutaneous microdecompressive endoscopic cervical discectomy with laser thermodiskoplasty. Mt. Sinai J. Med. 2000, 67, 278–282. [Google Scholar]
- Ahn, Y.; Lee, S.H.; Lee, S.C.; Shin, S.W.; Chung, S.E. Factors predicting excellent outcome of percutaneous cervical discectomy: Analysis of 111 consecutive cases. Neuroradiology 2004, 46, 378–384. [Google Scholar] [CrossRef]
- Ahn, Y.; Lee, S.H.; Shin, S.W. Percutaneous endoscopic cervical discectomy: Clinical outcome and radiographic changes. Photomed. Laser Surg. 2005, 23, 362–368. [Google Scholar] [CrossRef]
- Ahn, Y.; Lee, S.H.; Chung, S.E.; Park, H.S.; Shin, S.W. Percutaneous endoscopic cervical discectomy for discogenic cervical headache due to soft disc herniation. Neuroradiology 2005, 47, 924–930. [Google Scholar] [CrossRef]
- Ruetten, S.; Komp, M.; Merk, H.; Godolias, G. Full-endoscopic anterior decompression versus conventional anterior decompression and fusion in cervical disc herniations. Int. Orthop. 2009, 33, 1677–1682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, K.J.; Choi, B.W.; Choi, B.R.; Seo, G.B. Cross-cultural adaptation and validation of the Korean version of the neck disability index. Spine (Phila Pa 1976) 2010, 35, E1045–E1049. [Google Scholar] [CrossRef] [PubMed]
- Tsou, P.M.; Yeung, A.T. Transforaminal endoscopic decompression for radiculopathy secondary to intracanal noncontained lumbar disc herniations: Outcome and technique. Spine J. 2002, 2, 41–48. [Google Scholar] [CrossRef]
- Hees, H.L.; Nieuwenhuijsen, K.; Koeter, M.W.; Bültmann, U.; Schene, A.H. Towards a new definition of return-to-work outcomes in common mental disorders from a multi-stakeholder perspective. PLoS ONE 2012, 7, e39947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steenstra, I.A.; Lee, H.; de Vroome, E.M.; Busse, J.W.; Hogg-Johnson, S.J. Comparing current definitions of return to work: A measurement approach. J. Occup. Rehabil. 2012, 22, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Casal-Moro, R.; Castro-Menéndez, M.; Hernández-Blanco, M.; Bravo-Ricoy, J.A.; Jorge-Barreiro, F.J. Long-term outcome after microendoscopic diskectomy for lumbar disk herniation: A prospective clinical study with a 5-year follow-up. Neurosurgery 2011, 68, 1568–1575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.C.; Zhong, C.F.; Deng, G.B.; Liang, R.W.; Huang, C.M. Full-Endoscopic Procedures Versus Traditional Discectomy Surgery for Discectomy: A Systematic Review and Meta-analysis of Current Global Clinical Trials. Pain Physician 2016, 19, 103–118. [Google Scholar]
- Quillo-Olvera, J.; Lin, G.X.; Kim, J.S. Percutaneous endoscopic cervical discectomy: A technical review. Ann. Transl. Med. 2018, 6, 100. [Google Scholar] [CrossRef] [Green Version]



| PECD | ACDF | p Value | |
|---|---|---|---|
| Number of Patients | 51 | 64 | |
| Sex (M:F) | 28:23 | 36:28 | 1 |
| Mean Age (Years) | 42.2 (25–67) | 47.5 (20–75) | 0.083 |
| Mean BMI (kg/m2) | 24.06 | 23.71 | 0.535 |
| Operative Level | 0.273 | ||
| C3–4 | 3 | 5 | |
| C4–5 | 10 | 6 | |
| C5–6 | 22 | 37 | |
| C6–7 | 16 | 16 | |
| Motor Deficit | 0.682 | ||
| Normal | 30 (58.8%) | 34 (53.1%) | |
| Mild (Grade 4) | 16 (31.4%) | 25 (39.1%) | |
| Moderate (≤Grade 3) | 5 (9.8%) | 5 (7.8%) | |
| Sensory Disturbance | 0.687 | ||
| Hypesthesia | 8 (15.7%) | 12 (18.8%) | |
| Numbness | 32 (69.6%) | 39 (60.9%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahn, Y.; Keum, H.J.; Shin, S.H. Percutaneous Endoscopic Cervical Discectomy versus Anterior Cervical Discectomy and Fusion: A Comparative Cohort Study with a Five-Year Follow-Up. J. Clin. Med. 2020, 9, 371. https://doi.org/10.3390/jcm9020371
Ahn Y, Keum HJ, Shin SH. Percutaneous Endoscopic Cervical Discectomy versus Anterior Cervical Discectomy and Fusion: A Comparative Cohort Study with a Five-Year Follow-Up. Journal of Clinical Medicine. 2020; 9(2):371. https://doi.org/10.3390/jcm9020371
Chicago/Turabian StyleAhn, Yong, Han Joong Keum, and Sang Ha Shin. 2020. "Percutaneous Endoscopic Cervical Discectomy versus Anterior Cervical Discectomy and Fusion: A Comparative Cohort Study with a Five-Year Follow-Up" Journal of Clinical Medicine 9, no. 2: 371. https://doi.org/10.3390/jcm9020371
APA StyleAhn, Y., Keum, H. J., & Shin, S. H. (2020). Percutaneous Endoscopic Cervical Discectomy versus Anterior Cervical Discectomy and Fusion: A Comparative Cohort Study with a Five-Year Follow-Up. Journal of Clinical Medicine, 9(2), 371. https://doi.org/10.3390/jcm9020371





