The Prognostic Value of High Platelet Reactivity in Ischemic Stroke Depends on the Etiology: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Clinical Outcome
2.3. Ethics Statement
2.4. Platelet Function Testing
2.5. Statistical Analysis
3. Results
3.1. All Subjects
3.2. Two Etiological Subgroups
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Naghavi, M.; Wang, H.; Lozano, R.; Davis, A.; Liang, X.; Zhou, M.; Vollset, S.E.; Ozgoren, A.A.; Abdalla, S.; Abd-Allah, F.; et al. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 385, 117–171. [Google Scholar]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeove, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brwon, M.; Demaerschalk, B.M.; Hoh, B.; et al. 2018 Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association. Stroke 2018, 49, e46–e110. [Google Scholar] [CrossRef] [PubMed]
- Rondina, M.T.; Weyrich, A.S.; Zimmerman, G.A. Platelets as cellular effectors of inflammation in vascular diseases. Circ. Res. 2013, 112, 1506–1519. [Google Scholar] [CrossRef] [PubMed]
- Linden, M.D.; Jackson, D.E. Platelets: Pleiotropic roles in atherogenesis and atherothrombosis. Int. J. Biochem. Cell Biol. 2010, 42, 1762–1766. [Google Scholar] [CrossRef] [PubMed]
- Paniccia, R.; Priora, R.; Liotta, A.A.; Agatina, A. Platelet function tests: A comparative review. Vasc. Health Risk Manag. 2015, 11, 133–148. [Google Scholar] [CrossRef]
- Marulanda-Londono, E.; Chaturvedi, S. Stroke due to large vessel atherosclerosis. Neurol. Clin. Pract. 2016, 6, 252–258. [Google Scholar] [CrossRef]
- Wiśniewski, A.; Sikora, J.; Filipska, K.; Kozera, G. Assessment of the relationship between platelet reactivity, vascular risk factors and gender in cerebral ischaemia patients. Neurol. Neurochir. Pol. 2019, 53, 258–264. [Google Scholar] [CrossRef]
- Wiśniewski, A.; Sikora, J.; Sławińska, A.; Filipska, K.; Karczmarska-Wódzka, A.; Serafin, Z.; Kozera, G. High On-Treatment Platelet Reactivity Affects the Extent of Ischemic Lesions in Stroke Patients Due to Large-Vessel Disease. J. Clin. Med. 2020, 9, 251. [Google Scholar] [CrossRef]
- Oh, M.S.; Yu, K.H.; Lee, J.H.; Jung, S.; Kim, C.; Jang, M.U.; Lee, J.; Lee, B.C. Aspirin resistance is associated with increased stroke severity and infarct volume. Neurology 2016, 86, 1808–1817. [Google Scholar] [CrossRef]
- Agayeva, N.; Topcuoglu, M.A.; Arsava, E.M. The Interplay between Stroke Severity, Antiplatelet Use, and Aspirin Resistance in Ischemic Stroke. J. Stroke Cerebrovasc. Dis. 2016, 25, 397–403. [Google Scholar] [CrossRef]
- Zheng, A.S.; Churilov, L.; Colley, R.E.; Goh, C.; Davis, S.M.; Yan, B. Association of aspirin resistance with increased stroke severity and infarct size. JAMA Neurol. 2013, 70, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Xie, N.C.; Hu, H.L.; Chen, C.; Lian, Y.J. Biochemical aspirin resistance is associated with increased stroke severity and infarct volumes in ischemic stroke patients. Oncotarget 2017, 8, 77086–77095. [Google Scholar] [CrossRef] [PubMed]
- Sacco, R.L.; Kasner, S.E.; Broderick, J.P.; Caplan, L.R.; Connors, J.J.; Culebras, A.; Elkind, M.S.; George, M.G.; Hamdan, A.D.; Higashida, R.T.; et al. An updated definition of stroke for the 21st century: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013, 44, 2064–2089. [Google Scholar] [CrossRef] [PubMed]
- Wojczal, J.; Tomczyk, T.; Luchowski, P. Standards in neurosonology. J. Ultrason. 2016, 16, 44–45. [Google Scholar] [CrossRef] [PubMed]
- Wardlaw, J.M.; Smith, E.E.; Biessels, G.J.; Cordonnier, C.; Fazekas, F.; Frayne, R.; Lindley, R.I.; O’Brien, J.T.; Barkhof, F.; Benavente, O.R.; et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 2013, 12, 822–838. [Google Scholar] [CrossRef]
- Sibbing, D.; Braun, S.; Jawansky, S.; Vogt, W.; Mehilli, J.; Schömig, A.; Kastrati, A.; von Beckerath, N. Assessment of ADP-induced platelet aggregation with light transmission aggregometry and multiplate electrode platelet aggregometry before and after clopidogrel treatment. Thromb. Haemost. 2008, 99, 121–126. [Google Scholar]
- Tóth, O.; Calatzis, A.; Penz, S.; Losonczy, H.; Siess, W. Multiple electrode aggregometry: A new device to measure platelet aggregation in whole blood. Thromb. Haemost. 2006, 96, 781–788. [Google Scholar]
- Kim, J.T.; Heo, S.H.; Lee, J.S.; Choi, M.J.; Choi, K.H.; Nam, T.S.; Lee, S.H.; Park, M.S.; Kim, B.C.; Kim, M.K.; et al. Aspirin resistance in the acute stages of acute ischemic stroke is associated with the development of new ischemic lesions. PLoS ONE 2015, 10, e0120743. [Google Scholar] [CrossRef]
- Lai, P.T.; Chen, S.Y.; Lee, Y.S.; Ho, Y.P.; Chiang, Y.Y.; Hsu, H.Y. Relationship between acute stroke outcome, aspirin resistance, and humoral factors. J. Chin. Med. Assoc. 2012, 75, 513–518. [Google Scholar] [CrossRef]
- Englyst, N.A.; Horsfield, G.; Kwan, J.; Byrne, C.D. Aspirin resistance is more common in lacunar strokes than embolic strokes and is related to stroke severity. J. Cereb. Blood Flow Metab. 2008, 28, 1196–1203. [Google Scholar] [CrossRef]
- Yip, H.K.; Liou, C.W.; Chang, H.W.; Lan, M.Y.; Liu, J.S.; Chen, M.C. Link between platelet activity and outcomes after an ischemic stroke. Cerebrovasc. Dis. 2005, 20, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Sobol, A.B.; Mochecka, A.; Selmaj, K.; Loba, J. Is there a relationship between aspirin responsiveness and clinical aspects of ischemic stroke? Adv. Clin. Exp. Med. 2009, 18, 473–479. [Google Scholar]
- Amy, Y.X.; Keezer, M.R.; Zhu, B.; Wolfson, C.; Côté, R. Pre-stroke use of antihypertensives, antiplatelets, or statins and early ischemic stroke outcomes. Cerebrovasc. Dis. 2009, 27, 398–402. [Google Scholar]
- Wilterdink, J.L.; Bendixen, B.; Adams, H.P., Jr.; Woolson, R.F.; Clarke, W.R.; Hansen, M.D. Effect of prior aspirin use on stroke severity in the trial of Org 10172 in acute stroke treatment (TOAST). Stroke 2001, 32, 2836–2840. [Google Scholar] [CrossRef] [PubMed]
- Tsai, N.W.; Chang, W.N.; Shaw, C.F.; Jan, C.R.; Chang, H.W.; Huang, C.R.; Chen, S.D.; Chuang, Y.C.; Lee, L.H.; Wang, H.C.; et al. Levels and value of platelet activation markers in different subtypes of acute non-cardio-embolic ischemic stroke. Thromb. Res. 2009, 124, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Kinsella, J.A.; Tobin, W.O.; Hamilton, G.; McCabe, D.J. Platelet activation, function, and reactivity in atherosclerotic carotid artery stenosis: A systematic review of the literature. Int. J. Stroke 2013, 8, 451–464. [Google Scholar] [CrossRef]
- Dawson, J.; Quinn, T.; Lees, K.R.; Walters, M.R. Microembolic signals and aspirin resistance in patients with carotid stenosis. Cardiovasc. Ther. 2012, 30, 234–239. [Google Scholar] [CrossRef]
- Kinsella, J.A.; Tobin, W.A.; Tierney, S. Assessment of ‘on-treatment platelet reactivity’ and relationship with cerebral micro-embolic signals in asymptomatic and symptomatic carotid stenosis. J. Neurol. Sci. 2017, 376, 133–139. [Google Scholar] [CrossRef]
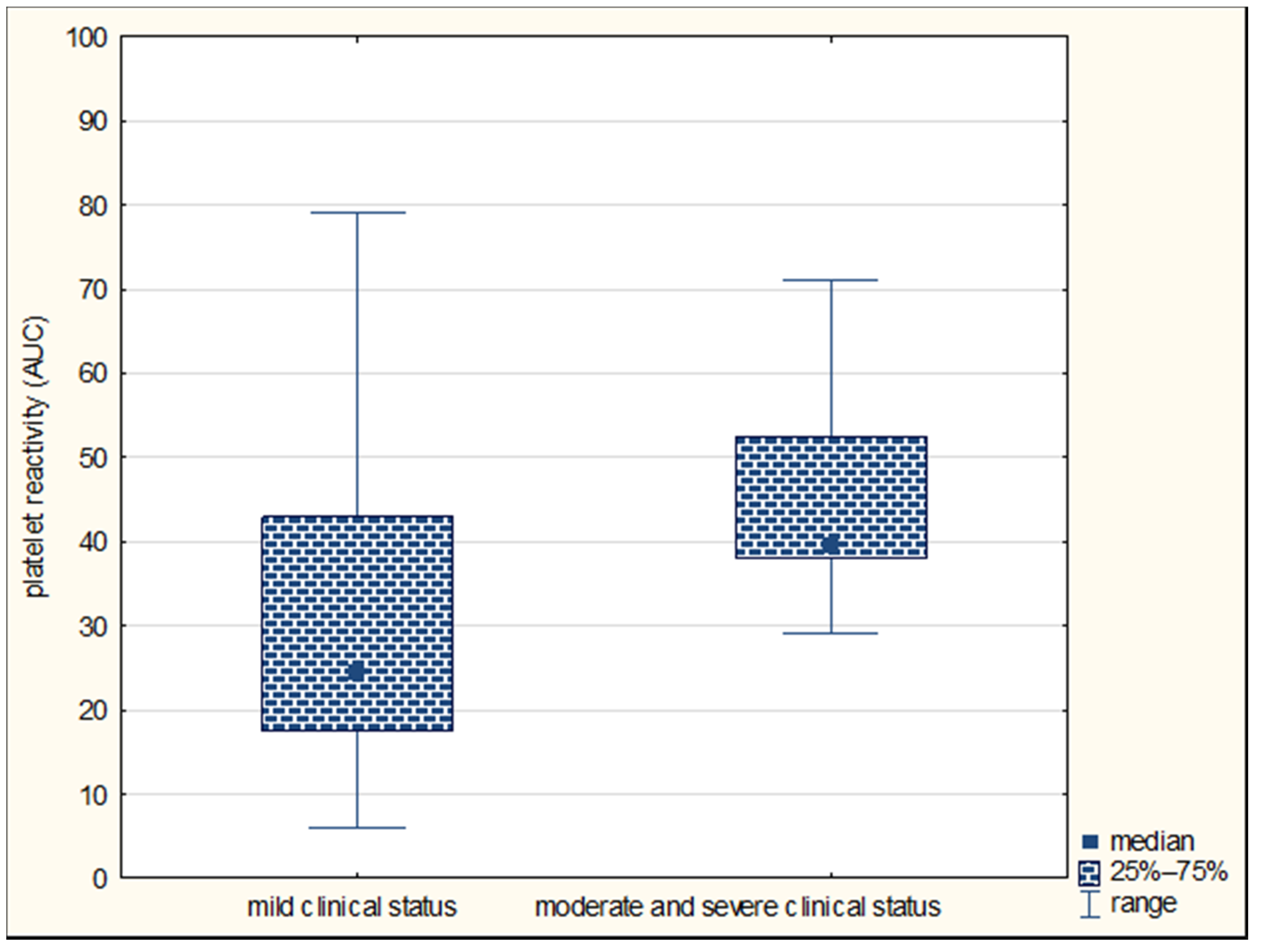
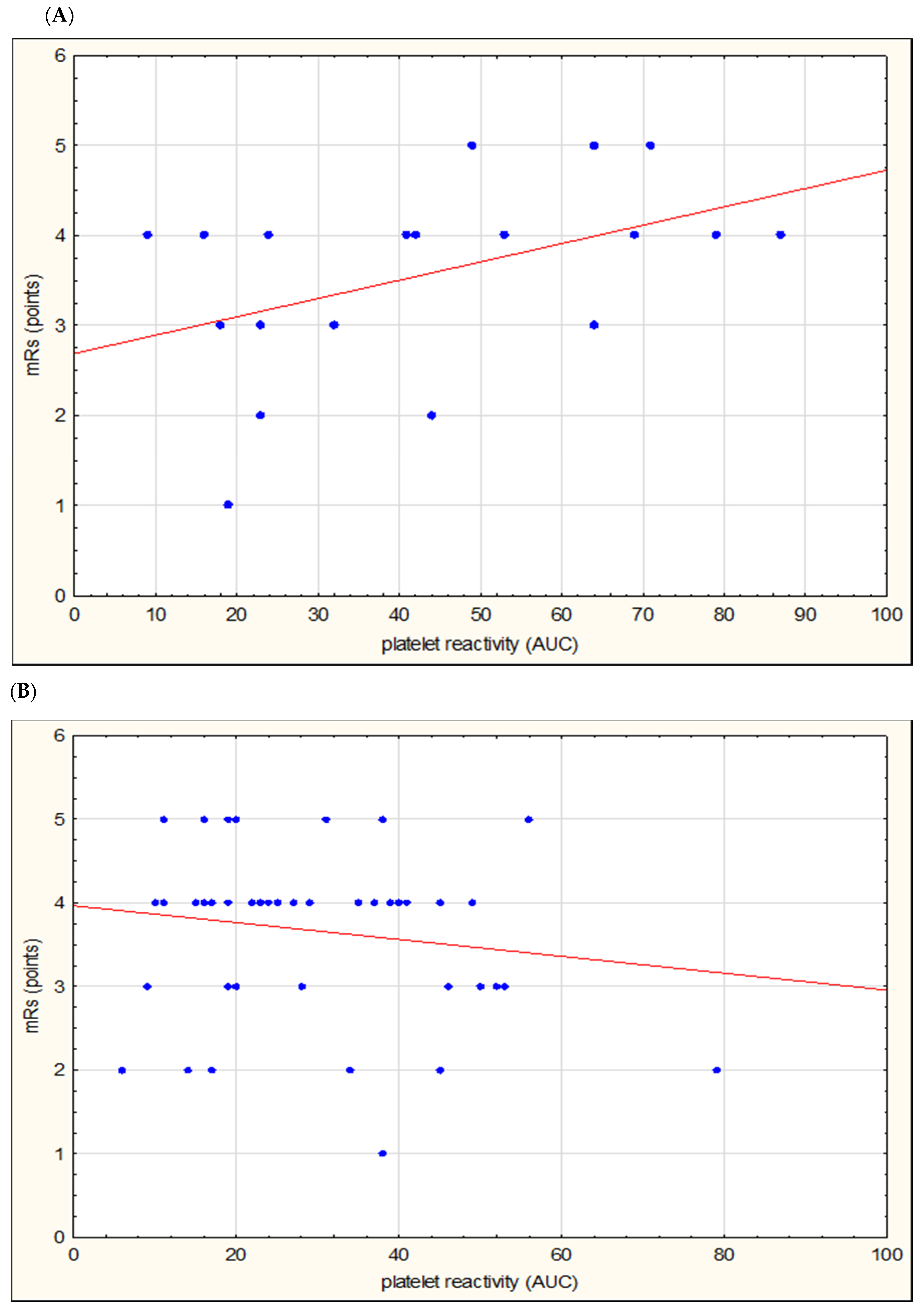
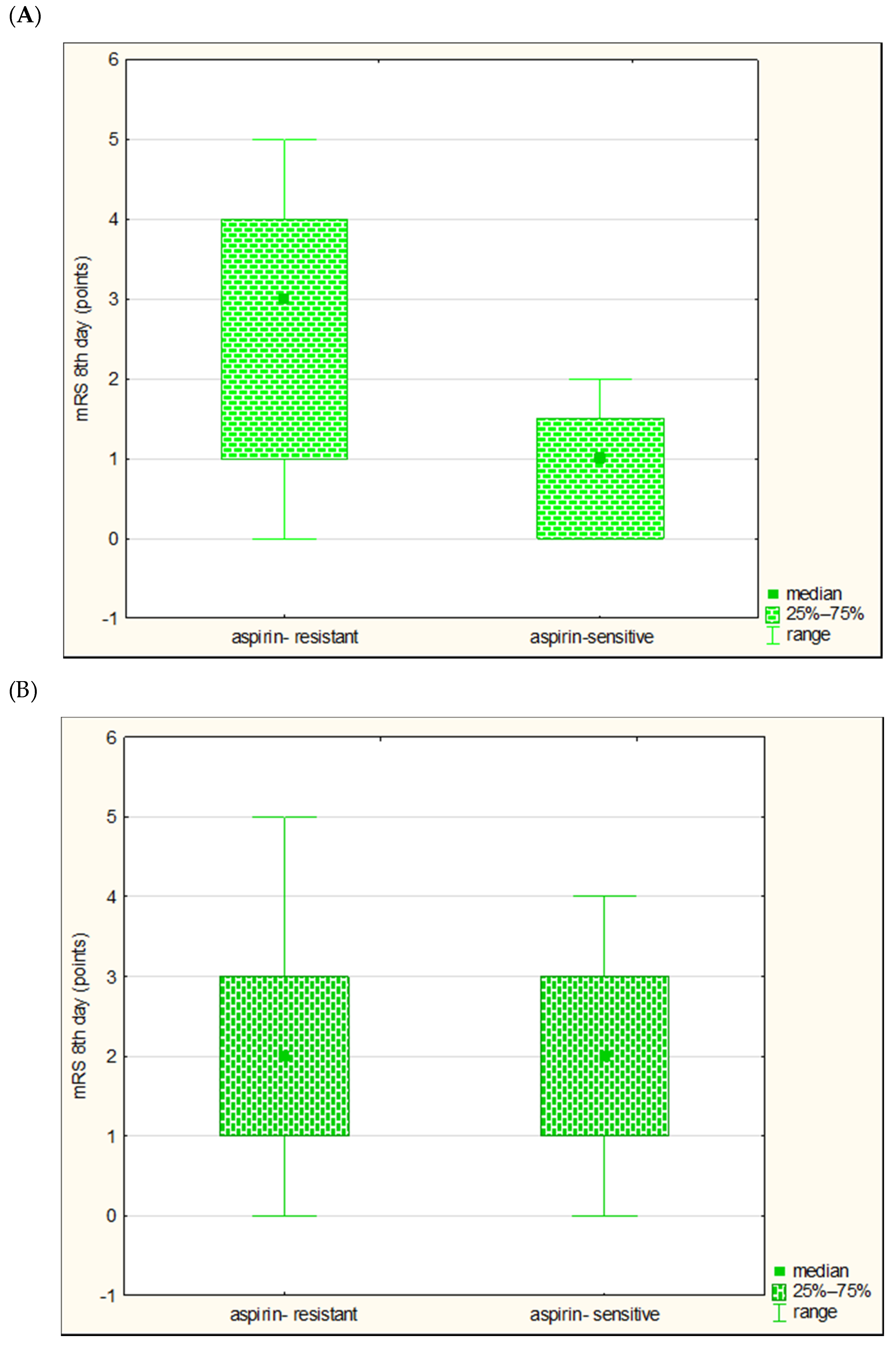

| Parameter | Large Vessel Disease n = 20 | Small Vessel Disease n = 49 | p-Value |
|---|---|---|---|
| Age median (range) * | 67 (45–85) | 68 (40–89) | 0.7761 |
| Male N, (%) ** | 14 (70%) | 21 (42.9%) | 0.0408 |
| Platelet reactivity: optical aggregometry (AUC) median (range) * | 17.1 (0–208.6) | 20.4 (0–154.2) | 0.7147 |
| Platelet reactivity: impedance aggregometry (AUC) median (range) * | 42 (9–101) | 27.5 (6–108) | 0.0622 |
| NIHSS 1 day median (range) * | 5 (2–17) | 5 (1–17) | 0.6770 |
| NIHSS 8 day median (range) * | 2 (0–10) | 2 (0–10) | 0.8324 |
| NIHSS 90 day median (range) * | 1 (0–8) | 2 (0–10) | 0.6625 |
| mRS 1 day median (range) * | 4 (1–5) | 4 (1–5) | 0.7304 |
| mRS 8 day median (range) * | 1 (0–5) | 2 (0–4) | 0.4999 |
| mRS 90 day median (range) * | 1 (0–4) | 2 (0–4) | 0.5740 |
| General Population | Large Vessel Disease | |||||||
|---|---|---|---|---|---|---|---|---|
| Multiplate® | LTA | Multiplate® | LTA | |||||
| R | p | R | p | R | p | R | p | |
| NIHSS 1 day | 0.0713 | 0.5603 | 0.0010 | 0.9948 | 0.4908 | 0.0728 | 0.0010 | 0.9947 |
| NIHSS 8 days | 0.0473 | 0.6996 | 0.1472 | 0.3462 | 0.2636 | 0.2614 | 0.1472 | 0.3462 |
| NIHSS 90 days | 0.0781 | 0.5233 | 0.0859 | 0.5838 | 0.2801 | 0.2017 | 0.0859 | 0.5837 |
| mRS 1 day | 0.0273 | 0.8240 | 0.0170 | 0.9139 | 0.4526 | 0.0451 | 0.01698 | 0.9139 |
| mRS 8 days | 0.1233 | 0.3128 | 0.0781 | 0.6186 | 0.4068 | 0.0750 | 0.0781 | 0.6186 |
| mRS 90 days | 0.0968 | 0.4288 | 0.1099 | 0.4829 | 0.3676 | 0.1108 | 0.1099 | 0.4826 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiśniewski, A.; Filipska, K.; Sikora, J.; Ślusarz, R.; Kozera, G. The Prognostic Value of High Platelet Reactivity in Ischemic Stroke Depends on the Etiology: A Pilot Study. J. Clin. Med. 2020, 9, 859. https://doi.org/10.3390/jcm9030859
Wiśniewski A, Filipska K, Sikora J, Ślusarz R, Kozera G. The Prognostic Value of High Platelet Reactivity in Ischemic Stroke Depends on the Etiology: A Pilot Study. Journal of Clinical Medicine. 2020; 9(3):859. https://doi.org/10.3390/jcm9030859
Chicago/Turabian StyleWiśniewski, Adam, Karolina Filipska, Joanna Sikora, Robert Ślusarz, and Grzegorz Kozera. 2020. "The Prognostic Value of High Platelet Reactivity in Ischemic Stroke Depends on the Etiology: A Pilot Study" Journal of Clinical Medicine 9, no. 3: 859. https://doi.org/10.3390/jcm9030859
APA StyleWiśniewski, A., Filipska, K., Sikora, J., Ślusarz, R., & Kozera, G. (2020). The Prognostic Value of High Platelet Reactivity in Ischemic Stroke Depends on the Etiology: A Pilot Study. Journal of Clinical Medicine, 9(3), 859. https://doi.org/10.3390/jcm9030859






