No Autopsies on COVID-19 Deaths: A Missed Opportunity and the Lockdown of Science
Abstract
1. Background
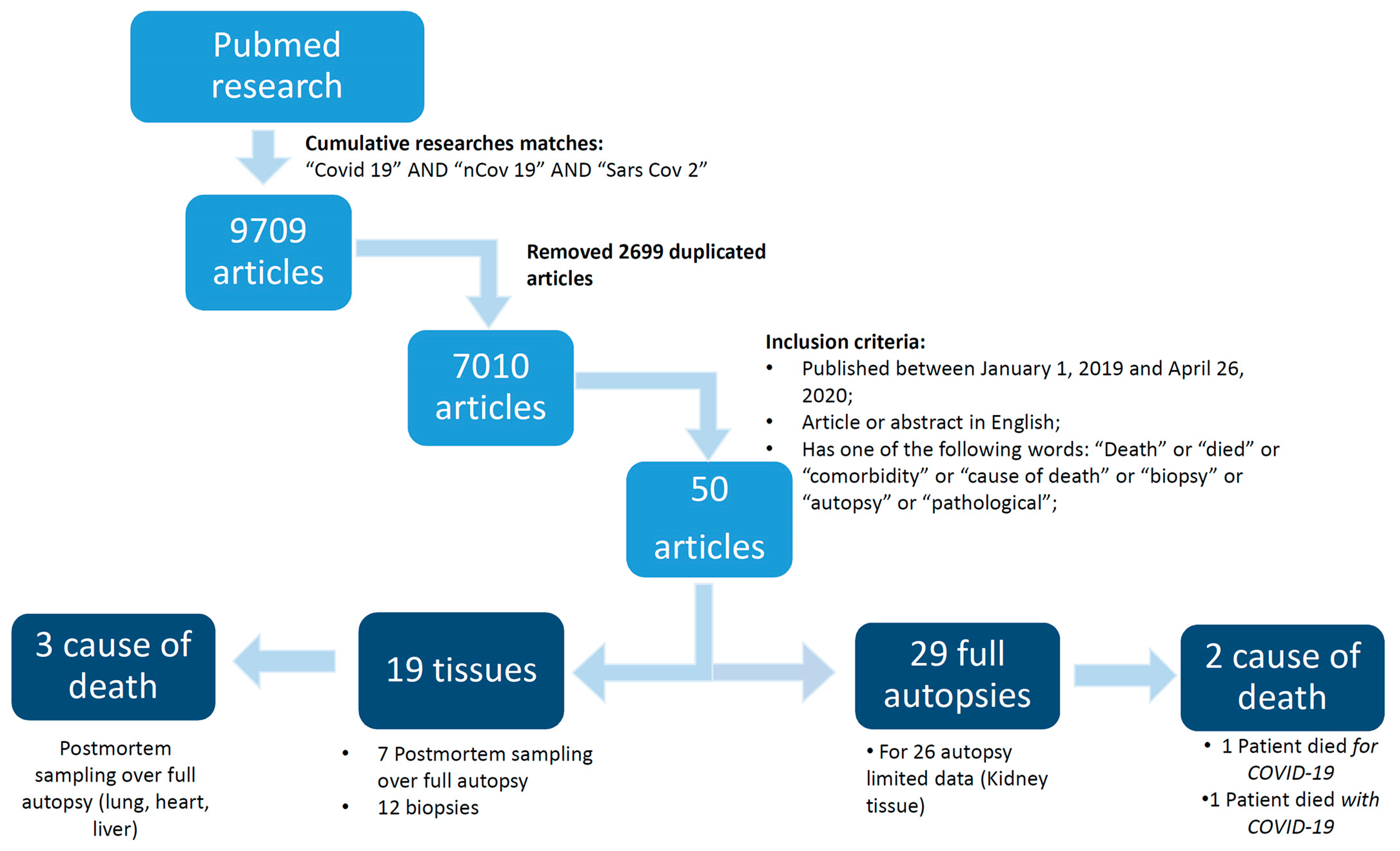
2. Methods
2.1. Database Search Terms and Timeline
2.2. Study Selection
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chan, J.F.W.; Yuan, S.; Kok, K.H.; To, K.K.W.; Chu, H.; Yang, J.; Xing, F.; Liu, J.; Yip, C.C.Y.; Poon, R.W.S.; et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet 2020. [Google Scholar] [CrossRef]
- Messina, G.; Polito, R.; Monda, V.; Cipolloni, L.; Di Nunno, N.; Di Mizio, G.; Murabito, P.; Carotenuto, M.; Messina, A.; Pisanelli, D.; et al. Functional Role of Dietary Intervention to Improve the Outcome of COVID-19: A Hypothesis of Work. Int. J. Mol. Sci. 2020, 21, 3104. [Google Scholar] [CrossRef] [PubMed]
- Nishiura, H.; Jung, S.M.; Linton, N.M.; Kinoshita, R.; Yang, Y.; Hayashi, K.; Kobayashi, T.; Yuan, B.; Akhmetzhanov, A.R. Akhmetzhanov The Extent of Transmission of Novel Coronavirus in Wuhan, China, 2020. J. Clin. Med. 2020, 9, 330. [Google Scholar] [CrossRef]
- Buja, L.M.; Barth, R.F.; Krueger, G.R.; Brodsky, S.V.; Hunter, R.L. The Importance of the Autopsy in Medicine: Perspectives of Pathology Colleagues. Acad. Pathol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, D.A.; Herman, C.J. The importance of the autopsy in emerging and reemerging infectious diseases. Clin. Infect. Dis. 1996. [Google Scholar] [CrossRef] [PubMed]
- Ng, D.L.; Al Hosani, F.; Keating, M.K.; Gerber, S.I.; Jones, T.L.; Metcalfe, M.G.; Tong, S.; Tao, Y.; Alami, N.N.; Haynes, L.M.; et al. Clinicopathologic, immunohistochemical, and ultrastructural findings of a fatal case of middle east respiratory syndrome coronavirus infection in the United Arab Emirates, April 2014. Am. J. Pathol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Callinan, L.S.; Holman, R.C.; Blau, D.M. Determinants for autopsy after unexplained deaths possibly resulting from infectious causes, United States. Emerg. Infect. Dis. 2012. [Google Scholar] [CrossRef]
- Peng, Y.D.; Meng, K.; Guan, H.Q.; Leng, L.; Zhu, R.R.; Wang, B.Y.; He, M.A.; Cheng, L.X.; Huang, K.Z.Q. Clinical characteristics and outcomes of 112 cardiovascular disease patients infected by 2019-nCoV. Chin. J. Cardiovasc. Dis. 2020, 48. [Google Scholar] [CrossRef]
- Tian, S.; Hu, W.; Niu, L.; Liu, H.; Xu, H.; Xiao, S.-Y. Pulmonary pathology of early phase 2019 novel coronavirus (COVID-19) pneumonia in two patients with lung cancer. J. Thorac. Oncol. 2020. [Google Scholar] [CrossRef]
- Guan, W.; Ni, Z.; Hu, Y.; Liang, W.; Ou, C.; He, J.; Liu, L.; Shan, H.; Lei, C.; Hui, D.S.; et al. Clinical characteristics of 2019 novel coronavirus infection in China. medRxiv 2020. [Google Scholar] [CrossRef]
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020. [Google Scholar] [CrossRef]
- Kui, L.; Fang, Y.-Y.; Deng, Y.; Liu, W.; Wang, M.-F.; Ma, J.-P.; Xiao, W.; Wang, Y.-N.; Zhong, M.-H.; Li, C.-H.; et al. Clinical characteristics of novel coronavirus cases in tertiary hospitals in Hubei Province. Chin. Med. J. (Engl) 2020. [Google Scholar] [CrossRef]
- Wang, W.; Tang, J.; Wei, F. Updated understanding of the outbreak of 2019 novel coronavirus (2019-nCoV) in Wuhan, China. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir. Med. 2020. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients with 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA - J. Am. Med. Assoc. 2020. [Google Scholar] [CrossRef]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 2020. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, W.; Xiao, M.; Li, Y.J.; Yang, Y.; Zhao, J.; Zhou, X.; Jiang, W.; Zhao, Y.Q.; Zhang, S.Y.; et al. Clinical and coagulation characteristics of 7 patients with critical COVID-2019 pneumonia and acro-ischemia. Zhonghua Xue Ye Xue Za Zhi 2020. [Google Scholar] [CrossRef]
- Analysis on 54 mortality cases of Coronavirus disease 2019 in the Republic of Korea from January 19 to March 10, 2020. J. Korean Med. Sci. 2020. [CrossRef]
- Yuan, M.; Yin, W.; Tao, Z.; Tan, W.; Hu, Y. Association of radiologic findings with mortality of patients infected with 2019 novel coronavirus in Wuhan, China. PLoS ONE 2020. [Google Scholar] [CrossRef]
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular Implications of Fatal Outcomes of Patients with Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.K.; Peng, S.; Li, L.Q.; Wang, Q.; Ping, W.; Zhang, N.; Fu, X.N. Clinical and Transmission Characteristics of Covid-19 — A Retrospective Study of 25 Cases from a Single Thoracic Surgery Department. Curr. Med. Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Wu, D.; Chen, H.; Yan, W.; Yang, D.; Chen, G.; Ma, K.; Xu, D.; Yu, H.; Wang, H.; et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: Retrospective study. BMJ 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020. [Google Scholar] [CrossRef]
- Deng, Y.; Liu, W.; Liu, K.; Fang, Y.Y.; Shang, J.; Zhou, L.; Wang, K.; Leng, F.; Wei, S.; Chen, L.; et al. Clinical characteristics of fatal and recovered cases of coronavirus disease 2019 (COVID-19) in Wuhan, China: a retrospective study. Chin. Med. J. (Engl) 2020. [Google Scholar] [CrossRef]
- Rodriguez-Morales, A.J.; Cardona-Ospina, J.A.; Gutiérrez-Ocampo, E.; Villamizar-Peña, R.; Holguin-Rivera, Y.; Escalera-Antezana, J.P.; Alvarado-Arnez, L.E.; Bonilla-Aldana, D.K.; Franco-Paredes, C.; Henao-Martinez, A.F.; et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Guan, W.; Liang, W.; Zhao, Y.; Liang, H.; Chen, Z.; Li, Y.; Liu, X.; Chen, R.; Tang, C.; Wang, T.; et al. Comorbidity and its impact on 1590 patients with Covid-19 in China: A Nationwide Analysis. Eur. Respir. J. 2020. [Google Scholar] [CrossRef]
- Bhatraju, P.K.; Ghassemieh, B.J.; Nichols, M.; Kim, R.; Jerome, K.R.; Nalla, A.K.; Greninger, A.L.; Pipavath, S.; Wurfel, M.M.; Evans, L.; et al. Covid-19 in Critically Ill Patients in the Seattle Region — Case Series. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Zhang, L.; Zhu, F.; Xie, L.; Wang, C.; Wang, J.; Chen, R.; Jia, P.; Guan, H.Q.; Peng, L.; Chen, Y.; et al. Clinical characteristics of COVID-19-infected cancer patients: A retrospective case study in three hospitals within Wuhan, China. Ann. Oncol. 2020. [Google Scholar] [CrossRef]
- Lescure, F.X.; Bouadma, L.; Nguyen, D.; Parisey, M.; Wicky, P.H.; Behillil, S.; Gaymard, A.; Bouscambert-Duchamp, M.; Donati, F.; Le Hingrat, Q.; et al. Clinical and virological data of the first cases of COVID-19 in Europe: a case series. Lancet. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Grasselli, G.; Zangrillo, A.; Zanella, A.; Antonelli, M.; Cabrini, L.; Castelli, A.; Cereda, D.; Coluccello, A.; Foti, G.; Fumagalli, R.; et al. Baseline Characteristics and Outcomes of 1591 Patients Infected with SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA - J. Am. Med. Assoc. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mi, B.; Chen, L.; Xiong, Y.; Xue, H.; Zhou, W.; Liu, G. Characteristics and Early Prognosis of COVID-19 Infection in Fracture Patients. J. Bone Jt. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Dai, Z.; Mo, P.; Li, X.; Ma, Z.; Song, S.; Chen, X.; Luo, M.; Liang, K.; Gao, S.; et al. Clinical characteristics and outcomes of older patients with coronavirus disease 2019 (COVID-19) in Wuhan, China (2019): a single-centered, retrospective study. Journals Gerontol. Ser. A 2020. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, L.; Yan, S.; Yang, F.; Xiang, L.; Zhu, J.; Shen, B.; Gong, Z. Clinical characteristics of 25 death cases with COVID-19: a retrospective review of medical records in a single medical center, Wuhan, China. Int. J. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Tu, W.J.; Cheng, W.; Yu, L.; Liu, Y.K.; Hu, X.; Liu, Q. Clinical Features and Short-term Outcomes of 102 Patients with Corona Virus Disease 2019 in Wuhan, China. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Wang, L.; He, W.; Yu, X.; Hu, D.; Bao, M.; Liu, H.; Zhou, J.; Jiang, H. Coronavirus disease 2019 in elderly patients: Characteristics and prognostic factors based on 4-week follow-up. J. Infect. 2020. [Google Scholar] [CrossRef]
- Barton, L.M.; Duval, E.J.; Stroberg, E.; Ghosh, S.; Mukhopadhyay, S. COVID-19 Autopsies, Oklahoma, USA. Am. J. Clin. Pathol. 2020. [Google Scholar] [CrossRef]
- Huang, J.; Lin, H.; Wu, Y.; Fang, Y.; Kumar, R.; Chen, G.; Lin, S. COVID-19 in posttransplant patients—report of 2 cases. Am. J. Transplant. 2020. [Google Scholar] [CrossRef]
- Ling, L.; So, C.; Shum, H.P.; Chan, P.K.S.; Lai, C.K.C.; Kandamby, D.H.; Ho, E.; So, D.; Yan, W.W.; Lui, G.; et al. Critically ill patients with COVID-19 in Hong Kong: A multicentre retrospective observational cohort study. Crit. Care Resusc. 2020, 32248675, Online ahead of print. [Google Scholar]
- Cheng, Y.; Luo, R.; Wang, K.; Zhang, M.; Wang, Z.; Dong, L.; Li, J.; Yao, Y.; Ge, S.; Xu, G. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020. [Google Scholar] [CrossRef] [PubMed]
- Du, R.H.; Liang, L.R.; Yang, C.Q.; Wang, W.; Cao, T.Z.; Li, M.; Guo, G.Y.; Du, J.; Zheng, C.L.; Zhu, Q.; et al. Predictors of Mortality for Patients with COVID-19 Pneumonia Caused by SARS-CoV-2: A Prospective Cohort Study. Eur. Respir. J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Barrasa, H.; Rello, J.; Tejada, S.; Martín, A.; Balziskueta, G.; Vinuesa, C.; Fernández-Miret, B.; Villagra, A.; Vallejo, A.; Sebastián, A.S.; et al. SARS-Cov-2 in Spanish Intensive Care: Early Experience with 15-day Survival In Vitoria. Anaesth. Crit. Care Pain Med. 2020. [Google Scholar] [CrossRef]
- Lovell, N.; Maddocks, M.; Etkind, S.N.; Taylor, K.; Carey, I.; Vora, V.; Marsh, L.; Higginson, I.J.; Prentice, W.; Edmonds, P.; et al. Characteristics, symptom management and outcomes of 101 patients with COVID-19 referred for hospital palliative care. J. Pain Symptom Manage. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, W.; Zhao, J.; Lu, Y.; Wang, X.; Yu, C.; Hu, S.; Shen, N.; Liu, W.; Sun, Z.; et al. Clinical characteristics of 80 hospitalized frontline medical workers infected with COVID-19 in Wuhan, China. J. Hosp. Infect. 2020. [Google Scholar] [CrossRef]
- Zhang, G.; Hu, C.; Luo, L.; Fang, F.; Chen, Y.; Li, J.; Peng, Z.; Pan, H. Clinical features and short-term outcomes of 221 patients with COVID-19 in Wuhan, China. J. Clin. Virol. 2020. [Google Scholar] [CrossRef]
- Magro, C.; Mulvey, J.J.; Berlin, D.; Nuovo, G.; Salvatore, S.; Harp, J.; Baxter-Stoltzfus, A.; Laurence, J. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: A report of five cases. Transl. Res. 2020. [Google Scholar] [CrossRef]
- Pereira, M.R.; Mohan, S.; Cohen, D.J.; Husain, S.A.; Dube, G.K.; Ratner, L.E.; Arcasoy, S.; Aversa, M.M.; Benvenuto, L.J.; Dadhani, D.; et al. COVID-19 in Solid Organ Transplant Recipients: Initial Report from the US Epicenter. Am. J. Transplant. 2020. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Chen, J.; Zuo, X.; Zhang, H.; Deng, A. COVID-19 infection may cause ketosis and ketoacidosis. Diabetes Obes. Metab. 2020. [Google Scholar] [CrossRef]
- Yang, F.; Shi, S.; Zhu, J.; Shi, J.; Dai, K.; Chen, X. Analysis of 92 deceased patients with COVID-19. J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Cai, Y.; Hao, Z.; Gao, Y.; Ping, W.; Wang, Q.; Peng, S.; Zhao, B.; Sun, W.; Zhu, M.; Li, K.; et al. Coronavirus Disease 2019 in the Perioperative Period of Lung Resection: A Brief Report From a Single Thoracic Surgery Department in Wuhan, People’s Republic of China. J. Thorac. Oncol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Karami, P.; Naghavi, M.; Feyzi, A.; Aghamohammadi, M.; Novin, M.S.; Mobaien, A.; Qorbanisani, M.; Karami, A.; Norooznezhad, A.H. Mortality of a pregnant patient diagnosed with COVID-19: A case report with clinical, radiological, and histopathological findings. Travel Med. Infect. Dis. 2020, 101665. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Xiong, Y.; Liu, H.; Niu, L.; Guo, J.; Liao, M.; Xiao, S.-Y. Pathological study of the 2019 novel coronavirus disease (COVID-19) through postmortem core biopsies. Mod. Pathol. 2020. [Google Scholar] [CrossRef]
- Fabre, O.; Rebet, O.; Carjaliu, I.; Radutoiu, M.; Gautier, L.; Hysi, I. Severe Acute Proximal Pulmonary Embolism and COVID-19: A Word of Caution. Ann. Thorac. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Yang, M.; Wan, C.; Yi, L.X.; Tang, F.; Zhu, H.Y.; Yi, F.; Yang, H.C.; Fogo, A.B.; Nie, X.; et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020. [Google Scholar] [CrossRef] [PubMed]
- Edrada, E.M.; Lopez, E.B.; Villarama, J.B.; Salva Villarama, E.P.; Dagoc, B.F.; Smith, C.; Sayo, A.R.; Verona, J.A.; Trifalgar-Arches, J.; Lazaro, J.; et al. First COVID-19 infections in the Philippines: A case report. Trop. Med. Health 2020. [Google Scholar] [CrossRef] [PubMed]
- Hang, L.; Yan, X.; Fan, Q.; Liu, H.; Liu, X.; Liu, Z.; Zhang, Z. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J. Thromb. Haemost. 2020. [Google Scholar] [CrossRef]
- Zheng, Y.-Y.; Ma, Y.-T.; Zhang, J.-Y.; Xie, X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020. [Google Scholar] [CrossRef]
- Rotzinger, D.C.; Beigelman-Aubry, C.; von Garnier, C.; Qanadli, S.D. Pulmonary embolism in patients with COVID-19: Time to change the paradigm of computed tomography. Thromb. Res. 2020, 190, 58–59. [Google Scholar] [CrossRef]
- Klok, F.A.; Kruip, M.J.H.A.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.A.M.P.J.; Kant, K.M.; Kaptein, F.H.J.; van Paassen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020. [Google Scholar] [CrossRef]
- World Health Organization. Infection Prevention and Control for the safe management of a dead body in the context of COVID-19. Interim guidance: 24 March 2020. Available online: https://apps.who.int/iris/bitstream/handle/10665/331538/WHO-COVID-19-lPC_DBMgmt-2020.1-eng.pdf (accessed on 27 March 2020).
- Italian Ministry of Health. Emergency Indications Related to the COVID-19 Epidemic Concerning the Funeral Sector, Cemetery, and Cremation. Circular of General Direction of Health Prevention. 2020. Available online: http://www.trovanorme.salute.gov.it/norme/renderNormsanPdf?anno=2020&codLeg=73965&parte=1%20&serie=null (accessed on 3 May 2020).
- Hanley, B.; Lucas, S.B.; Youd, E.; Swift, B.; Osborn, M. Autopsy in suspected COVID-19 cases. J. Clin. Pathol. 2020. [Google Scholar] [CrossRef]
- Pomara, C.; Volti, G.L.; Cappello, F. COVID-19 Deaths: Are We Sure It Is Pneumonia? Please, Autopsy, Autopsy, Autopsy! J. Clin. Med. 2020, 9, 1259. [Google Scholar] [CrossRef] [PubMed]
- De Cock, K.M.; Zielinski-Gutiérrez, E.; Lucas, S.B. Learning from the Dead. N. Engl. J. Med. 2019. [Google Scholar] [CrossRef]
- Vetter, P.; Fischer, W.A.; Schibler, M.; Jacobs, M.; Bausch, D.G.; Kaiser, L. Ebola Virus Shedding and Transmission: Review of Current Evidence. J. Infect. Dis. 2016. [Google Scholar] [CrossRef] [PubMed]
- Castillo, P.; Martínez, M.J.; Ussene, E.; Jordao, D.; Lovane, L.; Ismail, M.R.; Carrilho, C.; Lorenzoni, C.; Fernandes, F.; Bene, R.; et al. Validity of a Minimally Invasive Autopsy for Cause of Death Determination in Adults in Mozambique: An Observational Study. PLoS Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Kreuels, B.; Wichmann, D.; Emmerich, P.; Schmidt-Chanasit, J.; De Heer, G.; Kluge, S.; Sow, A.; Renné, T.; Günther, S.; Lohse, A.W.; et al. A case of severe Ebola virus infection complicated by gram-negative septicemia. N. Engl. J. Med. 2014. [Google Scholar] [CrossRef] [PubMed]
- Kreuels, B.; Addo, M.M.; Schmiedel, S. Severe Ebola virus infection complicated by gram-negative septicemia. N. Engl. J. Med. 2015. [Google Scholar] [CrossRef]
- Petrosillo, N.; Nicastri, E.; Lanini, S.; Capobianchi, M.R.; Di Caro, A.; Antonini, M.; Puro, V.; Lauria, F.N.; Shindo, N.; Magrini, N.; et al. Ebola virus disease complicated with viral interstitial pneumonia: A case report. BMC Infect. Dis. 2015. [Google Scholar] [CrossRef]
- Varkey, J.B.; Shantha, J.G.; Crozier, I.; Kraft, C.S.; Lyon, G.M.; Mehta, A.K.; Kumar, G.; Smith, J.R.; Kainulainen, M.H.; Whitmer, S.; et al. Persistence of ebola virus in ocular fluid during convalescence. N. Engl. J. Med. 2015. [Google Scholar] [CrossRef]
- Brief Report: Persistence of Ebola Virus in Ocular Fluid during Convalescence. N. Engl. J. Med. 2015. [CrossRef]
- Francis, T.I.; Moore, D.L.; Edington, G.M.; Smith, J.A. A clinicopathological study of human yellow fever. Bull. World Health Organ. 1972, 46, 659. [Google Scholar] [PubMed]
- Duarte-Neto, A.N.; Monteiro, R.A.; Johnsson, J.; Cunha, M.; Pour, S.Z.; Saraiva, A.C.; Ho, Y.L.; da Silva, L.F.F.; Mauad, T.; Zanotto, P.M.; et al. Ultrasound-guided minimally invasive autopsy as a tool for rapid post-mortem diagnosis in the 2018 Sao Paulo yellow fever epidemic: Correlation with conventional autopsy. PLoS Negl. Trop. Dis. 2019. [Google Scholar] [CrossRef]
- Burton, J.L.; Underwood, J. Clinical, educational, and epidemiological value of autopsy. Lancet 2007. [Google Scholar] [CrossRef]
- Franks, T.J.; Chong, P.Y.; Chui, P.; Galvin, J.R.; Lourens, R.M.; Reid, A.H.; Selbs, E.; Mcevoy, C.P.L.; Hayden, C.D.L.; Fukuoka, J.; et al. Lung pathology of severe acute respiratory syndrome (SARS): A study of 8 autopsy cases from Singapore. Hum. Pathol. 2003. [Google Scholar] [CrossRef]
- Roosen, J.; Frans, E.; Wilmer, A.; Knockaert, D.C.; Bobbaers, H. Comparison of premortem clinical diagnoses in critically ill patients and subsequent autopsy findings. Mayo Clin. Proc. 2000. [Google Scholar] [CrossRef]
- Kotovicz, F.; Mauad, T.; Saldiva, P.H.N. Clinico-pathological discrepancies in a general university hospital in São Paulo, Brazil. Clinics 2008. [Google Scholar] [CrossRef]
- Combes, A.; Mokhtari, M.; Couvelard, A.; Trouillet, J.L.; Baudot, J.; Hénin, D.; Gibert, C.; Chastre, J. Clinical and Autopsy Diagnoses in the Intensive Care Unit: A Prospective Study. Arch. Intern. Med. 2004. [Google Scholar] [CrossRef]
- Edwards, J.E. The Autopsy: Do We Still Need It? Chest 1970, 57, 113–114. [Google Scholar] [CrossRef]

| Author | Number of Patients | Patients Who Died | Average Age (Years) | Comorbidities | Severe Complications | Tissues (Biopsy or Autopsy Samples) | Autopsy |
|---|---|---|---|---|---|---|---|
| Peng Y. D. et al. [8] | 112 | 17 | Data not available | Coronary heart disease (CHD) (100%), BMI >25 (88.24%) | Data not available | 0 | 0 |
| Tian S. et al. [9] | 2 | 2 | 78.5 | lung cancer (100%), hypertension (100%), diabetes (50%) | Respiratory failure, coma, heart failure | 2 (biopsy) | 0 |
| Guan W. J. et al. [10] | 1099 | 15 | 47 | Hypertension (15%), diabetes (7.4%), CHD (2.5%), HCV (2.1%), chronic obstructive pulmonary disease (COPD) (1.1%), cancer (0.9%) | Septic shock, acute respiratory distress syndrome (ARDS), kidney failure | 0 | 0 |
| Xu Z. et al. [11] | 1 | 1 | 50 | Data not available | Respiratory failure, ARDS | 1 (autoptic sample) | 0 |
| Kui K. et al. [12] | 137 | 16 | 57 | Hypertension (9.5%), diabetes (10.2%), CHD (7.3%), COPD (1.5%), cancer (1.5%) | Data not available | 0 | 0 |
| Wang W. et al. [13] | 571 | 17 | 73 | Hypertension (41.2%), diabetes (23.5%), CHD (17.6%), stroke (17.6%), COPD (11.7%), kidney failure (11.7%), Parkinson (11.7%), cancer (5.9%), cirrhosis (5.9%) | Data not available | 0 | 0 |
| Yang X. et al. [14] | 201 | 32 | 59.7 | CHD (9%), COPD (6%), diabetes (22%), cancer (3%), stroke (22%) | ARDS, kidney failure, heart failure, liver failure | 0 | 0 |
| Huang C. et al. [15] | 41 | 6 | 49 | Diabetes (20%), hypertension (15%), cardiovascular disease (CVD) (15%) | Respiratory distress syndrome (29%), RNAaemia (15%), acute cardiac injury (12%), secondary infection (10%) | 0 | 0 |
| Wang D. et al. [16] | 138 | 6 | 56 | Hypertension (31.2%), diabetes (10.1%), CVD (14.5%), Cancer (7.2%) | ARDS, arrhythmia, shock. | 0 | 0 |
| Chen N. et al. [17] | 99 | 11 | 55.5 | CVD and cerebrovascular diseases (40%), digestive system disease (11%), endocrine system disease (13%), cancer (1%), nervous system disease (1%), respiratory system disease (1%) | ARDS (17%), kidney failure (3%), respiratory failure (8%), Septic shock (4%) | 0 | 0 |
| Zhang et al. [18] | 7 | 5 | 59 | Not indicated | Acro-ischemia presentations including finger/toe cyanosis, skin bulla and dry gangrene (100%), definite disseminated intravascular coagulation (DIC) (4 (57%)) | 0 | 0 |
| Korean Society of Infectious Diseases et al. [19] | 54 | 54 | 75.5 | CVD (59.3%); diabetes mellitus (DM) (29.6%); neurological disease (18.5%); lung disease (13.0%); malignancy (13.0%); psychologic disease (13.0%); renal disease (9.3%); hepatic disease (3.7%); kidney transplant recipient (1.9%) | Data not available | 0 | 0 |
| Yuan et al. [20] | 27 | 10 | 60 | Hypertension (19%); diabetes (22%); CVD (11%); tumor (4%); cerebral infarction (4%); chronic gastritis (4%) | ARDS (41%) | 0 | 0 |
| Guo et al. [21] | 187 | 43 | 58.5 | Hypertension (32.6%); CHD (11.2%); cardiomyopathy (8 (4.3%)); diabetes (15.0%); COPD (2.1); malignant neoplasm (7.0%); chronic kidney disease (CKD) (3.2%) | ARDS (24.6%), malignant arrhythmias (5.9%) including ventricular tachycardia/ventricular fibrillation, acute coagulopathy (34.1%), acute liver injury (15.4%) and acute kidney injury (14.6%) | 0 | 0 |
| Yang et al. [22] | 25 | 5 | 60.2 | Hypertension (15.4%); diabetes (7.7%); COPD (38.5%); CHD (30.8%) | Data not available | 0 | 0 |
| Chen et al. [23] | 274 | 113 | 62 | Hypertension (34%); diabetes (17%); CVD (8%); CHD (<1%); COPD (7%); malignancy (3%); hepatitis B (4%); cerebrovascular disease (1%); CKD (1%); gastrointestinal diseases (1%); metabolic arthritis (1%); autoimmune disease (1%) | ARDS (72%), type I respiratory failure (27%), acute cardiac injury (44%), heart failure (24%), hypoxic encephalopathy (9%), sepsis (65%), acidosis (12%), alkalosis (28%), acute kidney injury (11%), disseminated intravascular coagulation (8%), hyperkalemia (23%), shock (17%), acute liver injury (5%), gastrointestinal bleeding (<1%). | 0 | 0 |
| Zhuo et al. [24] | 191 | 54 | Data not available | Data not available | Sepsis (59%), respiratory failure (54%), ARDS (31%), heart failure (23%), septic shock (20%), coagulopathy (19%), acute cardiac injury (17%), acute kidney injury (15%), secondary infection (15%), hypoproteinemia (12%), acidosis (9%) | 0 | 0 |
| Deng et al. [25] | 225 | 109 | 69 | Hypertension (36.7%); lung disease (20.2%); diabetes (15.6%); heart disease (11.9%); malignancy (5.5%); others (28.4%) | ARDS (7.6%), acute cardiac injury (0.8%), acute kidney injury (<1%), shock (<1%), and disseminated intravascular coagulation (DIC) (<1%) | 0 | 0 |
| Rodriguez-Morales et al. [26] | 2874 | 632 | 51.97 | Hypertension (18.6%); CVD (14.4%); diabetes (11.9%); COPD (1.8%); malignancies (2.5%); chronic liver disease (CLD) (3.0%) | 20.3% who required ICU: ARDS (32.8%), cardiac injury (13.0%), acute kidney injury (7.9%), shock (6.2%), Secondary infections (5.6%). | 0 | 0 |
| Guan et al. [27] | 1590 | 50 | 48.9 | Hypertension (16.7%); CVD (53.7%), cerebrovascular disease (1.9%), diabetes (8.2%), hepatitis B (1.8%), COPD (1.5%), CKD (1.3%), malignncy (1.1%) | Data not available | 0 | 0 |
| Bhatraju et al. [28] | 24 | 12 | 64 | Asthma (14%), CKD (21%), COPD (4%), tobacco smoker (22%), diabetes (58%), | Data not available | 0 | 0 |
| Zhang et al. [29] | 28 | 8 | 65 | Cancer (100%), diabetes 4(14.3%), COPD 1 (3.6%) | ARDS 8 (28.6%), septic shock 1 (3.6%), suspected pulmonary embolism 2 (7.1%), AMI 1 (3.6%) | 0 | 0 |
| Lescure et al. [30] | 5 | 1 | 47 | Hypertension 1 (20%), cancer 1 (20%), gout 1 (20%) | Data not available | 0 | 0 |
| Wu et al. [31] | 201 | 44 | 51 | Hypertension (19.4%) diabetes (10.9%) CVD (4.0%) liver disease (3.5%) nervous system disease (3.5%) chronic lung disease (2.5%) CKD (1.0%) endocrine system disease (1.0%) tumor (0.5%) | Data not available | 0 | 0 |
| Grasselli et al. [32] | 1591 | 405 | 63 | Hypertension (49%); CVD (21%); hypercholesterolemia (18%); DM (17); malignancy (8%); COPD (4%); CKD (3%); CLD (3%); other (20%) | Data not available | 0 | 0 |
| Bobin et al. [33] | 10 | 4 | 68.4 | Fracture (100%); hypertension (40%); diabetes (30%); COPD (10%); osteoporosis (30%); CHD (10%); cirrhosis (10%), alzheimer disease (10%); brain injury (10%) | Data not available | 0 | 0 |
| Chen et al. [34] | 203 | 26 | 54 | Hypertension (21.2%); diabetes (7.9%); CVD (7.9%); cerebrovascular disease (4.4%); malignancy (3,4%); CLD (3.9%); CKD (8 (3.9%)); COPD (8 (3.9%)); Tuberculosis (4 (2.0%)); HIV (2 (0.1%)) | Cause of Death: ARDS (14%); ARDS with MOD (22%); sepsis/Shock (4%); heart failure (2%); myocardial infarction (6%); tumor (4%); intestinal bleeding (2%); | 0 | 0 |
| Li et al. [35] | 25 | 25 | 73 | Hypertension (64%); diabetes (40%); heart diseases (32%); kidney diseases (20%); cerebral infarction (16%); COPD (8%); malignant tumors (8%); acute pancreatitis (4%) | Respiratory failure (100%) | 0 | 0 |
| Cao et al. [36] | 102 | 17 | 54 | Hypertension (27.5%); diabetes (10.8%); cerebrovascular disease (5.9%); CVD (4.9%); respiratory diseases (9.8%); malignancy (3.9%); CKD (3.9%); CLD (2.0%); | Shock (9.8%); ARDS (19.6%); acute infection (16.7%); acute cardiac injury (14.7%); arrhythmia (17.6%); acute kidney injury (19.6%); acute liver injury (33.3%); lymphopenia (76.5%); Cause of Death: multiple organ dysfunction syndrome (MODS) (58.8%); ARDS (5.9%); cardiac arrest (23.5%); respiratory failure (11.8%) | 0 | 0 |
| Wang et al. [37] | 339 | 65 | 71 | Hypertension (40.8%), diabetes (16.0%), CVD (15.7%) | Lymphocytopenia (63.2%), bacterial infection (42.8%), liver enzyme abnormalities (28.7%), acute respiratory distress syndrome (21.0%) | 0 | 0 |
| Barton et al. [38] | 2 | 2 | 77 | Hypertension, splenectomy, cholelithiasis, osteoarthritis | Cause of Death: COVID-19 (ARDS—diffuse alveolar damage (DAD)) | 0 | 2 |
| 42 | Myotonic, muscular dystrophy | Complications of hepatic cirrhosis (aspiration acute bacterial bronchopneumonia) | |||||
| Huang et al. [39] | 2 | 2 | 54.5 | Transplantation (100%) | Nosocomial bacterial infection (100%); respiratory organ failure (100%); kidney organ failure (100%); heart organ failure (50%) | 0 | 0 |
| Ling et al. [40] | 8 | 1 | 64.5 | Data not available | Respiratory failure (75%); kidney failure (25%) | 0 | 0 |
| Cheng et al. [41] | 701 | 113 | 63 | Any comorbidity (42.6%); CKD (2.0%); COPD (1.9%); hypertension (33.4%); diabetes (14.3%); tumor (4.6%) | Acute kidney injury (5.1%) | 0 | 0 |
| Du et al. [42] | 179 | 21 | 57.6 | Hypertension (32.4%); CVD or cerebrovascular diseases (16.2%); diabetes (18.4%); chronic digestive disorders (11.7%); tuberculosis (4.5%); chronic hepatic or renal insufficiency (2.2%); Peripheral vascular disease (2.2%); malignancy (2.2%) | Data not available | 0 | 0 |
| Barrasa et al. [43] | 48 | 14 | 63 | Obesity (48%); arterial hypertension (44%); COPD (37%); | Hypoxemic respiratory failure (100%) | 0 | 0 |
| Lovell et al. [44] | 101 | 75 | 82 | Hypertension (54%); diabetes (36%); dementia (31%); cancer (25%); COPD (22%); renal failure (21%); congestive heart failure (18%); stroke / neurological disorder (12%); peripheral vascular disorder (4%); liver disease (2%); | Data not available | 0 | 0 |
| Wang et al. [45] | 80 | 1 | 39 | Hypertension (12.5%); diabetes (1.25%); CVD (2.5%); cerebrovascular disease (1.25%); COPD 1 (1.25%); renal disease (3.75%); liver disease (2.5%) | Data not available | 0 | 0 |
| Zhang et al. [46] | 221 | 12 | 55 | Hypertension (24.4%), diabetes (10.0%), CVD (10.0%), cerebrovascular disease (6.8%), COPD (2.7%), CKD (2.7%), CLD (3.2%), malignancy (4.1%), immunosuppression treatment (1.4%) | ARDS (21.7); arrhythmia (10.9); acute cardiac injury (7.7); shock (6.8); AKI (4.5) | 0 | 0 |
| Magro et al. [47] | 5 | 2 | 54.6 | Coronary artery disease, diabetes mellitus, heart failure, hepatitis C virus infection, end-stage renal disease, obesity (n = 2), and pre-diabetes, | Respiratory failure (n = 5); purpuric skin rash (n = 3) | 3 cases skin biopsies | 2 Cases, limited autopsy |
| Pereira et al. [48] | 90 | 16 | 57 | HTN (64%); diabetes (46%); CKD (63%); dialysis (6%); chronic lung disease (19%); HIV (1%); active cancer (3%); BMI >40 Kg/m2 (6%) | Data not available | 0 | 0 |
| Li et al. [49] | 658 | 64 | 47 | Cerebrovascular disease (8%); coronary heart disease (8.9%); heart failure (1.21%); diabetes (19.6%); hypertension (33.4%); digestive disorder (13.22%); COPD (2.88%); cancer (2.58%); CKD (2.73%); hepatitis (1.06%) | Ketosis (6.38%); acute liver injury (5.92%); septic shock (5.31%); ARDS (14.43%); diabetic ketoacidosis (DKA) (0.4%); acidosis (4.55%) | 0 | 0 |
| Yang et al. [50] | 92 | 91 | 69.8 | Hypertension (55.43%); heart disease (17.39%); cerebrovascular (10.8%); malignancy (4.34%); CLD (3.26%); CKD (2.17%); COPD (1%) | Cause of death: ARDS (79.34%), septic shock (7.6%), myocardial infarction (6.52%), heart failure (2.17%), MODS (2.17%) | 0 | 0 |
| Cai et al. [51] | 7 | 3 | 61 | COPD (28.5%); CVD (42.8%); interstitial lung disease (14.28%); hyperlipidemia (14.28%); Malignancy (100%) | Cause of death: respiratory failure 3 | 7 Biopsies | 0 |
| Karami et al. [52] | 1 | 1 | 27 | No underlying disease | Data not available | 0 | 1 |
| Tian et al. [53] | 4 | 4 | 73 | Chronic lymphocytic leukemia (CLL), cirrhosis, hypertension, diabetes, and renal transplantation | Data not available | 4 (Autopsy samples) | 0 |
| Fabre et al. [54] | 1 | 1 | 45 | Obesity (BMI 40.4), hypertension | Pulmonary Embolism | 0 | 0 |
| Su et al. [55] | 26 | 26 | 69 | History of hypertension or diabetes or both (42.3%) | Data not available | 26 (Kidney autopsy samples) | 0 |
| Edrada et al. [56] | 2 | 2 | 42 | Data not available | Data not available | 0 | 0 |
| Zhang et al. [57] | 343 | 13 | 62 | Diabetes (13.7%), hypertension (22.15%), CHD (5.5%), COPD (2.33%), cancer (2.62%), stroke history (2.33%), CLD (1.74%) | Data not available | 0 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salerno, M.; Sessa, F.; Piscopo, A.; Montana, A.; Torrisi, M.; Patanè, F.; Murabito, P.; Li Volti, G.; Pomara, C. No Autopsies on COVID-19 Deaths: A Missed Opportunity and the Lockdown of Science. J. Clin. Med. 2020, 9, 1472. https://doi.org/10.3390/jcm9051472
Salerno M, Sessa F, Piscopo A, Montana A, Torrisi M, Patanè F, Murabito P, Li Volti G, Pomara C. No Autopsies on COVID-19 Deaths: A Missed Opportunity and the Lockdown of Science. Journal of Clinical Medicine. 2020; 9(5):1472. https://doi.org/10.3390/jcm9051472
Chicago/Turabian StyleSalerno, Monica, Francesco Sessa, Amalia Piscopo, Angelo Montana, Marco Torrisi, Federico Patanè, Paolo Murabito, Giovanni Li Volti, and Cristoforo Pomara. 2020. "No Autopsies on COVID-19 Deaths: A Missed Opportunity and the Lockdown of Science" Journal of Clinical Medicine 9, no. 5: 1472. https://doi.org/10.3390/jcm9051472
APA StyleSalerno, M., Sessa, F., Piscopo, A., Montana, A., Torrisi, M., Patanè, F., Murabito, P., Li Volti, G., & Pomara, C. (2020). No Autopsies on COVID-19 Deaths: A Missed Opportunity and the Lockdown of Science. Journal of Clinical Medicine, 9(5), 1472. https://doi.org/10.3390/jcm9051472










