1. Introduction
Heart failure is characterized by autonomic dysfunction [
1,
2]. Heart rate recovery (HRR) after graded exercise stress testing is one of the commonly used indexes reflecting autonomic activity [
1,
2]. HRR is defined as the fall in heart rate during the first minute after exercise and is easily calculated as the difference in heart rate between the peak of exercise and one minute after exercise cessation. HRR is a marker of vagal tone, thanks to sympathetic deactivation and parasympathetic reactivation [
1,
2]. The first minute of post-exercise recovery is predominantly secondary to parasympathetic reactivation [
1,
3,
4,
5]. However, even though vagal-related HRR is parasympathetically mediated, previous studies suggest that it might represent independent aspects, but complementary information, with regard to parasympathetic function [
6].
In patients with heart failure (HF), the total sympathetic nervous activity is augmented owing to loss of baroreflex sensitivity and to increased sympathetic afferent activity [
7,
8,
9]. The sympathetic nervous system negatively impacts the cardiovascular system in HF; down-regulation of beta1-receptors and direct toxic effects on the myocardium contribute to cardiac remodeling and life-threatening arrhythmias [
7,
8,
9].
Sacubitril/valsartan (S/V) is a treatment for HF with reduced ejection fraction (HFrEF), which impressively impact cardiovascular prognosis by reducing major adverse cardiovascular events such as cardiovascular mortality and sudden death [
10]. Moreover, S/V significantly improves cardiopulmonary functional capacity and ventilatory parameters [
11,
12,
13,
14].
The present study aims at evaluating the effects of S/V on autonomic function (as evaluated by HRR) and its correlation with cardiopulmonary indexes in HFrEF patients.
3. Results
The study population consisted of 134 HFrEF patients (87% male, mean age 57.9 ± 9.6 years). Baseline clinical and demographic characteristics of the study population and stratified by ARNI dose are given in
Table 1 and
Table 2, respectively. Overall, mean LVEF was 28.1% ± 5.9% and, at baseline, the majority of patients had NYHA class II (62%) or class III (34%), whereas no patients had NYHA class IV (
Table 1). At 12-month follow-up, a significant improvement in NYHA class was observed: 104 patients (77.2%) had NYHA class II and 15 patients (11.4%) had NYHA class III (
Table 1).
Major comorbidity was represented by hypertension (54%), dyslipidemia (59%), diabetes (34%), chronic obstructive pulmonary disease (COPD) (16%), and atrial fibrillation (16%) (
Table 1). At study entry, 6 patients (4.5%) had pacemaker (PMK), 102 patients (76%) had PMK plus implantable cardioverter-defibrillator (ICD), and 23 patients (17%) had cardiac resynchronization therapy (CRT) devices (
Table 1). At 12-month follow-up, 103 patients (77%) had PMK-ICD and a total of 25 (18%) patients had CRT devices. Forty-four patients were active smokers (
Table 1).
Clinical and demographic characteristics of the study population stratified by ARNI dose are given in
Table 3 At study entry, 90 (67.2%) HFrEF patients were taking ARNI
1 dose, 40 (31.3%) HFrEF patients were taking ARNI
2 dose, and 2 (1.5%) HFrEF patients were taking ARNI
3 dose; whereas at 12-month follow-up, 39 (29.1%) HFrEF patients were taking ARNI
1 dose, 48 (35.8%) HFrEF patients were taking ARNI
2 dose, and 47 (35.1%) HFrEF patients were taking ARNI
3 dose.
Baseline and 12-month follow-up Doppler-echocardiography parameters in overall population and stratified by ARNI dose are reported in
Table 3 and
Table 4, respectively. A significant improvement in LVEF was observed at 12-month follow-up (from 28% ± 5.8% to 31.8% ± 7.3%,
p < 0.0001) (
Table 3). LVEF improvement was greater among patients treated with higher ARNI doses (from 28.4% ± 6.2% to 29.2% ± 7.2%,
p = 0.415 in ARNI
1 group; from 27.4% ± 6.4% to 32.1% ± 6.5%,
p = 0.006 in ARNI
2 group; and from 28.2% ± 4.9% to 33.8% ± 8.0%,
p = 0.001 in ARNI
3 group).
Baseline and 12-month follow-up CPET parameters in the overall population and stratified by ARNI dose are reported in
Table 5 and
Table 6, respectively. Overall, S/V therapy exerted beneficial effects on cardiopulmonary functional capacity (
Table 5). Significant changes in VO
2peak and predicted VO
2peak (%) were observed at 12-month follow-up (from 15.3 ± 3.7 to 17.8 ± 4.2 mL/kg/min,
p < 0.0001; and from 56.4% ± 13.9% to 64.8% ± 17.8%,
p < 0.0001, respectively) (
Table 5). Mean VO
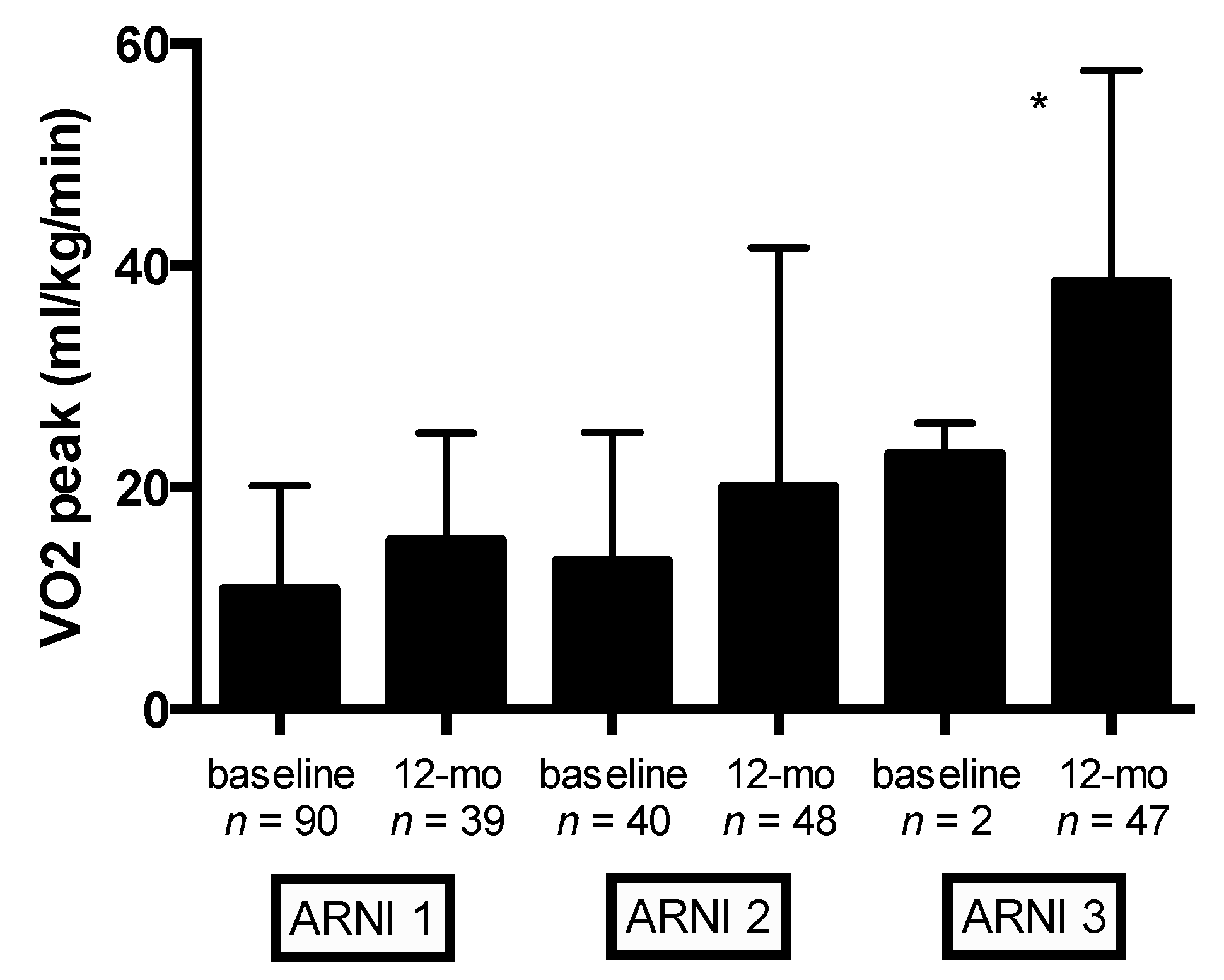
2peak (mL/kg/min) values according to ARNI dose at baseline and 12-month follow-up are shown in
Figure 1. Compared with the lowest ARNI treatment dose, HFrEF patients receiving the higher ARNI dose (97/103 mg bid) had greater VO
2peak improvement (
p < 0.0001) (
Figure 1). Compared with baseline, significant improvement in other cardiopulmonary functional capacity parameters were observed at 12-month follow-up: maximal workload and ΔVO2/Δwork significantly improved from 72.8 ± 25.1 to 90.5 ± 28.1 watts,
p < 0.0001, and from 9.2 ± 1.6 to 10.2 ± 1.5 mL/min/watt,
p < 0.0001, respectively (
Table 5).
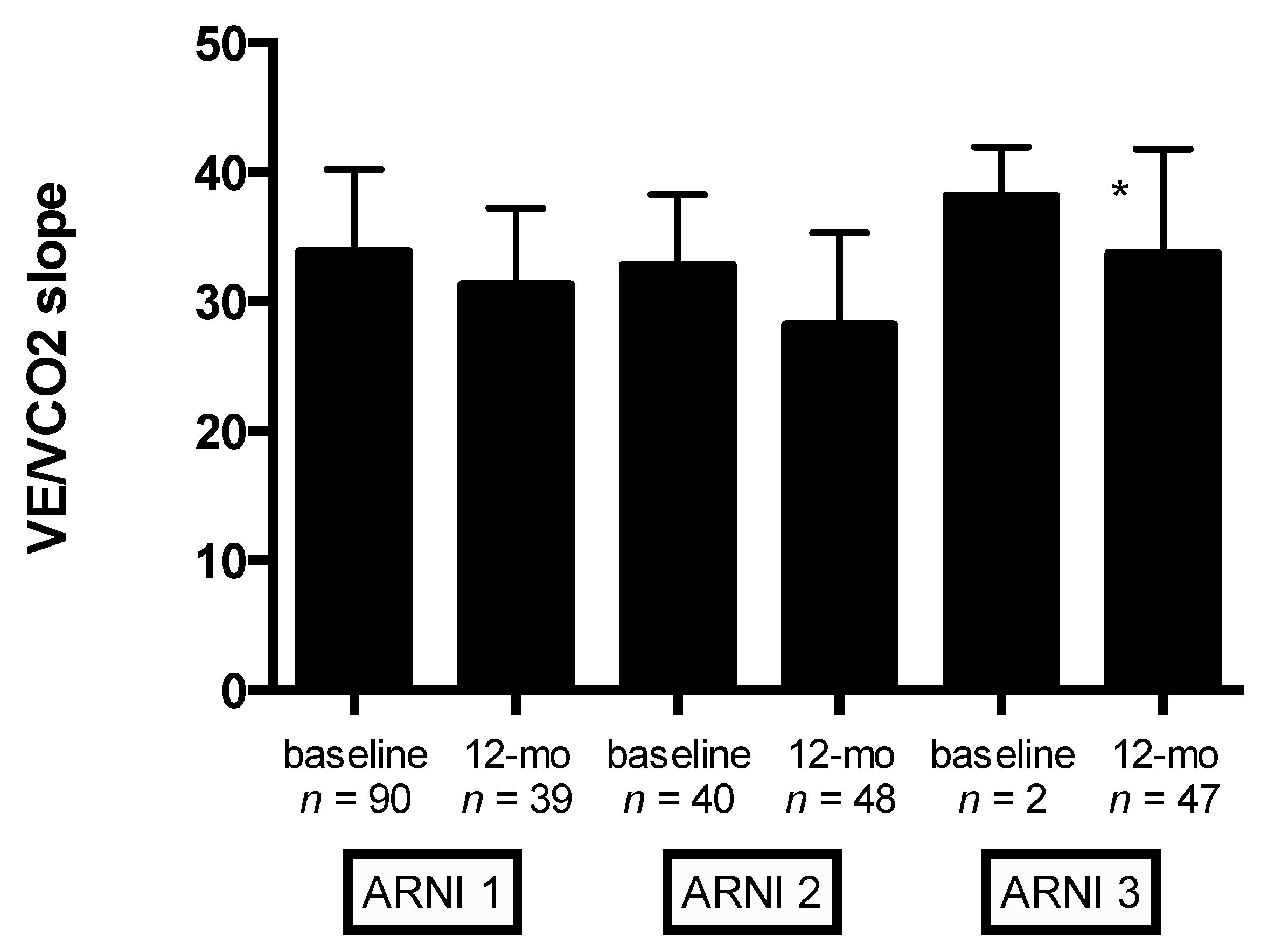
Moreover, significant improvement in VE/VCO
2slope was observed at 12-month follow-up (from 33.4 ± 6.2 to 30.3 ± 6.5,
p < 0.0001) (
Table 5). Mean changes in VE/VCO
2slope according to ARNI dose at baseline and 12-month follow-up are shown in
Figure 2. Compared with the lowest ARNI treatment dose, HFrEF patients receiving the higher ARNI dose (97/103 mg bid) had greater VE/VCO
2slope improvement (
p < 0.0001) (
Figure 2). Notably, significant improvement in other ventilatory parameters was observed at 12-month follow-up: peak ventilation and respiratory rate significantly increase from baseline (from 48.1 ± 12.3 to 61.4 ± 18.9 L/min,
p < 0.0001; and from 30.8 ± 6.3 to 33.6 ± 7.4,
p < 0.0001, respectively) (
Table 5). In addition, HFrEF patients treated with S/V showed a significant reduction in EOV detection at CPET (28 EOV detected at baseline CPET vs. 9 EOV detected at 12-month follow-up,
p < 0.001). HRR at baseline CPET was a significant predictor of EOV at 12-month follow-up (B = −2.065, SE = 0.354,
p < 0.001).
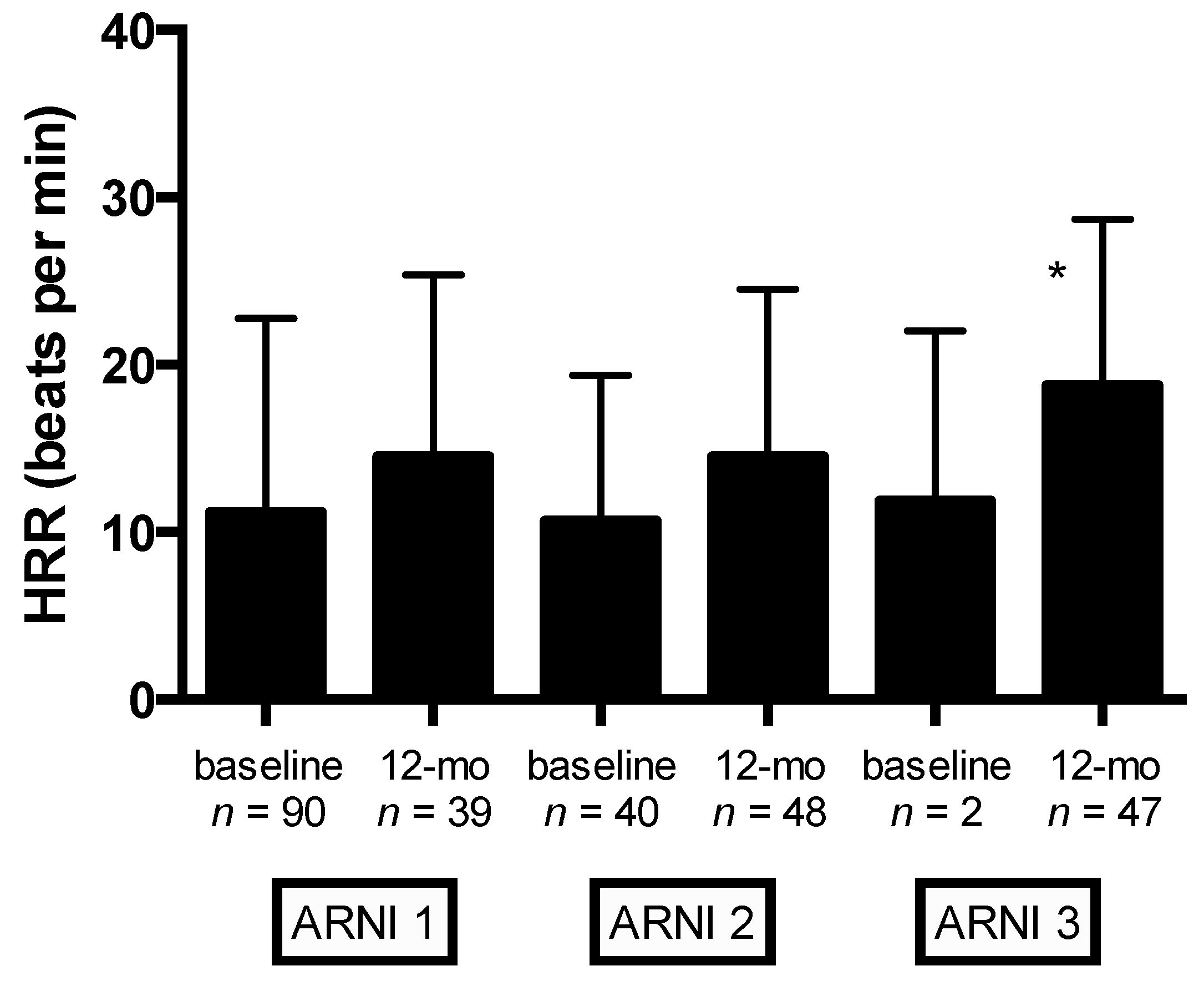
Significant changes in autonomic function (as evaluated by HRR) were observed at 12-month follow-up (from 11.4 ± 9.5 to 17.4 ± 15.1 bpm,
p = 0.004) (
Table 5). Mean changes in HRR stratified by ARNI dose at baseline and 12-month follow-up are shown in
Figure 3. Compared with the lowest ARNI treatment dose, HFrEF patients receiving the higher ARNI dose (97/103 mg bid) had greater HRR improvement (
p = 0.009) (
Figure 3).
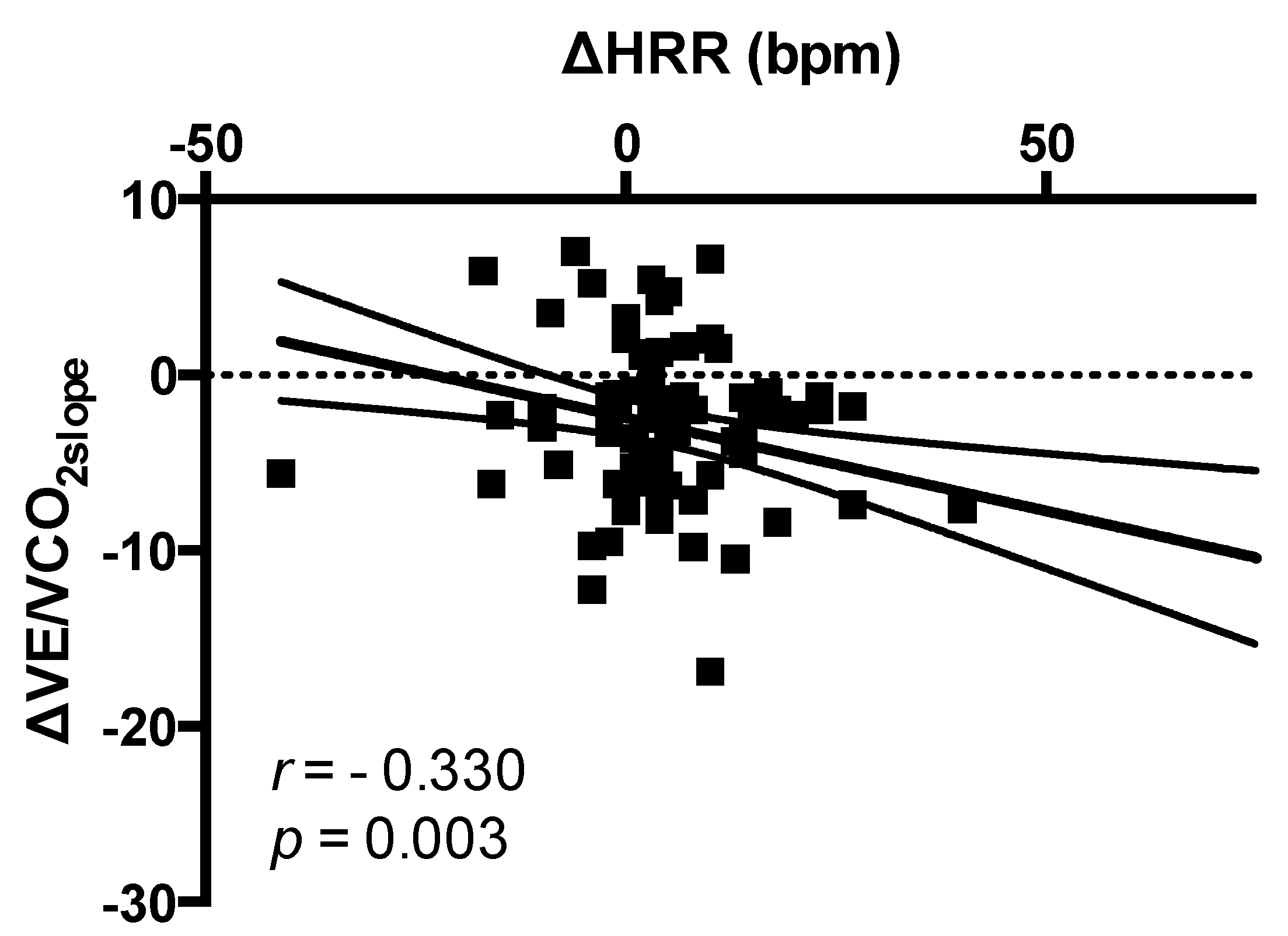
No correlation between changes in HRR (bpm) and changes in systolic blood pressure (SBP) (mmHg) was found (
r = −0.022,
p = 0.850). Changes in HRR (bpm) were significantly correlated to changes in VE/VCO
2slope (
r = −0.330;
p = 0.003) (
Figure 4). After adjusting for age, NYHA class, diabetes, atrial fibrillation, CRT/pacemaker, NT-proBNP levels, drug therapy (beta-blockers, ivabradine, and loop diuretics use), maximal ARNI dose reached, and changes in VO
2peak, multivariate analysis showed that changes in HRR were significantly associated to changes in VE/VCO
2slope (B = −0.975, SE = 0.364, Bstd = −0.304,
p = 0.009).
Finally, at a median follow-up of 26 ± 6.7 months, the mortality rate was 10% (13 cardiovascular deaths), and 42 HF hospitalizations occurred. No significant association between ARNI dose or changes in HRR and these hard endpoints was found.
4. Discussion
This study showed a significant improvement in autonomic function as measured by HRR in HFrEF patients undergoing S/V treatment for 1 year. Autonomic function ameliorating, as expressed by the increase in HRR, was associated to the improvement in cardiopulmonary function.
To the best of our knowledge, this is the first study reporting beneficial effects on autonomic function in HFrEF patients undergoing S/V therapy. Previous studies showed that autonomic function is favorably improved by structured exercise training in patients after acute myocardial infarction or chronic heart failure [
27,
28,
29,
30]. However, exercise-induced improvement in cardiopulmonary and autonomic function is lost when exercise sessions is abandoned [
28]. In a recent meta-analysis including patients with coronary artery disease, exercise-based cardiac resynchronization (CR) improved post-exercise parasympathetic function, with greater effects in younger patients with coronary artery disease and in patients who have undergone percutaneous intervention [
30]. Notably, the overall effect size showed significant differences in HRR (MD = +5.35; 95% confidence interval (CI) = 4.08–6.61 bpm) in favor of the exercise-based CR group [
30].
In this study, HFrEF patients on S/V therapy showed a significant increase in HRR (+6.0 beats/min, p = 0.004) that could likely be ascribed to the effect of S/V therapy. Future studies are encouraged in order to investigate whether combined strategies (S/V therapy plus structured exercise training) might improve outcome in HFrEF patients.
HRR is a powerful predictor of all-cause mortality in patients with chronic heart failure [
31,
32,
33,
34]. The mechanisms involved in mortality reduction are probably multifactorial, and likely include a central mechanism activated by the release of inhibitory commands from the motor cortex to the parasympathetic center [
35], or afferent stimulation from baroreflex or chemoreflex functions [
36]. Imai et al. [
37] demonstrated that a release of inhibitory central command rather than baro- or chemo-receptor stimulation may play a key role in vagal reactivation after exercise, because the heart rate response early during recovery (i.e., the first 30 s) minimally depends on exercise intensity.
Both enhanced post-infarction inflammatory response [
38,
39] and autonomic dysfunction [
40,
41,
42,
43] are associated with LV remodelling and poor clinical outcomes. Postinfarction healing is a highly regulated process. When post-infarction inflammatory response is efficient, a scar with tensile strength is properly formed, preventing the expansion of the infarction area, with consequent lower incidence of decompensating heart failure. Despite the key role of the healing process in post-infarction LV remodeling, the mechanisms underlying the initiation and regulation of these processes remain to be elucidated. Experimental models showed that S/V attenuates cardiac remodeling and dysfunction after myocardial infarction by reducing cardiac fibrosis and hypertrophy [
44,
45]. The same mechanicistic pathways might be at the basis of the favorable effect of S/V therapy on cardiac remodeling and inflammatory status in HFrEF patients and, consequently, responsible for the impressive outcome improvement [
10].
Although this cohort of HFrEF patients did not undergo structured exercise-based cardiac rehabilitation programs, they were encouraged to maintain an adequate level of physical activity according to guidelines. It is possible that S/V therapy by ameliorating exercise tolerance could favor the engagement of leisure time physical activity with a potential protective beneficial effect on arrhythmias and mortality. The PARADIGM-HF trial [
10] showed a reduced risk for sudden death (HR = 0.80, 95% CI = 0.68–0.94,
p = 0.008) in HFrEF patients treated with S/V. Because only 15% of HF patients from PARADIGM-HF trial in both arms had implantable cardioverter-defibrillator (ICD), it could be hypothesized that the improvement in sudden death could be related to the improvement in autonomic function and its related protective antiarrhytmic effect. In our cohort, 76% of HFrEF patients had ICD and no major arrhythmic episodes were detected at device check.
EOV, a cyclic fluctuation of minute ventilation and expired gas kinetics occurring during exercise, is a marker of HF severity, and predicts either mortality or morbidity [
26]. Interestingly, S/V showed a significant reduction in EOV detection at CPET (28 EOV detected at baseline CPET vs. 9 EOV detected at 12-month follow-up,
p < 0.001). It could be hypothesized that the beneficial effect on autonomic function may translate to a more efficient and coordinated ventilation pattern. Future studies are eagerly awaited in order to establish whether EOV improvement might improve prognosis in HFrEF patients treated with S/V.
The relatively small sample size of patients, predominantly adult males, might limit the conclusion of the study. In addition, HRR is only an indirect, although widely accepted, reflection of sympathovagal balance. Furthermore, this study had no statistical power to analyze the relationship between HRR and prognosis. Conversely, the present study for the first time reported beneficial effects on autonomic function (as measured by HRR) in HFrEF patients undergoing S/V therapy.