Radiomics Based on Thyroid Ultrasound Can Predict Distant Metastasis of Follicular Thyroid Carcinoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Selection and Data Collection
2.2. US Examinations and Image Evaluation
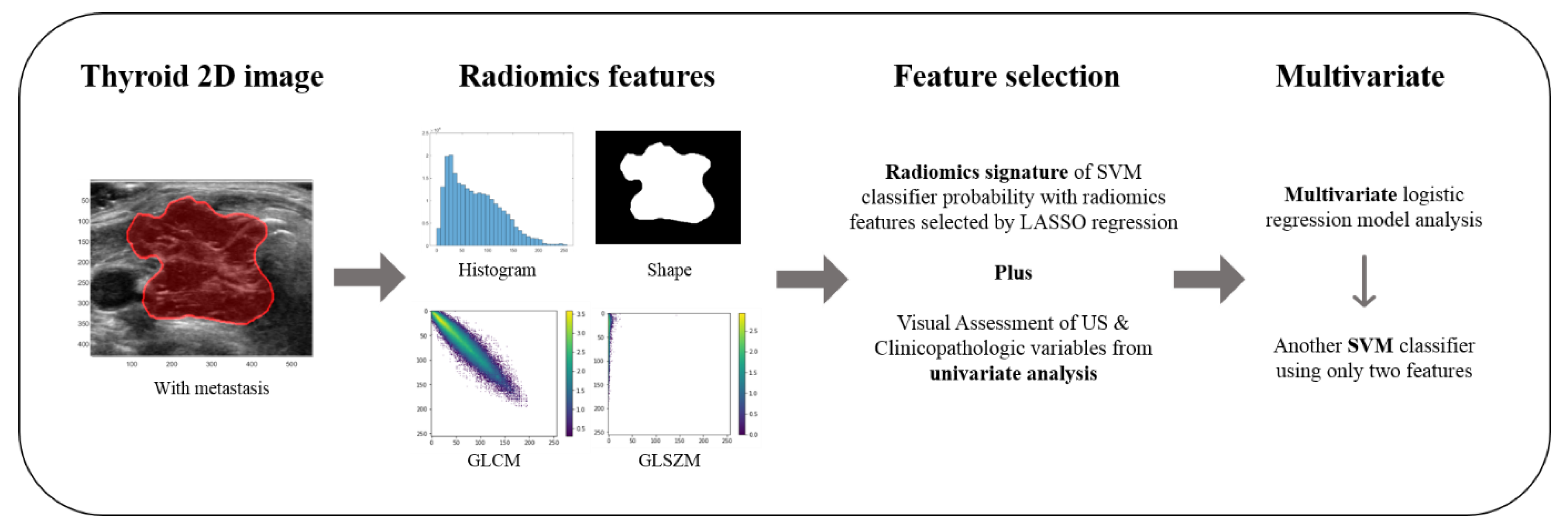
2.3. Radiomics Feature Analysis
2.4. Statistical Analysis and Combined Model
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- DeLellis, R.A.; Lloyd, R.V.; Heitz, P.U.; Eng, C. Pathology and Genetics of Tumours of Endocrine Organs, 3rd ed.; IARC Publications: Lyon, France, 2004. [Google Scholar]
- Lo, C.-Y.; Chan, W.-F.; Lam, A.K.; Wan, K.-Y. Follicular Thyroid Carcinoma. Ann. Surg. 2005, 242, 708–715. [Google Scholar] [CrossRef]
- Ito, Y.; Hirokawa, M.; Higashiyama, T.; Takamura, Y.; Miya, A.; Kobayashi, K.; Matsuzuka, F.; Kuma, K.; Miyauchi, A. Prognosis and Prognostic Factors of Follicular Carcinoma in Japan: Importance of Postoperative Pathological Examination. World J. Surg. 2007, 31, 1417–1424. [Google Scholar] [CrossRef]
- Sugino, K.; Ito, K.; Nagahama, M.; Kitagawa, W.; Shibuya, H.; Ohkuwa, K.; Yano, Y.; Uruno, T.; Akaishi, J.; Kameyama, K.; et al. Prognosis and Prognostic Factors for Distant Metastases and Tumor Mortality in Follicular Thyroid Carcinoma. Thyroid 2011, 21, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Hirokawa, M.; Masuoka, H.; Yabuta, T.; Fukushima, M.; Kihara, M.; Higashiyama, T.; Takamura, Y.; Kobayashi, K.; Miya, A.; et al. Distant metastasis at diagnosis and large tumor size are significant prognostic factors of widely invasive follicular thyroid carcinoma. Endocr. J. 2013, 60, 829–833. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.-M.; Song, D.E.; Kim, T.Y.; Sung, T.-Y.; Yoon, J.H.; Chung, K.-W.; Hong, S.J. Risk Factors for Distant Metastasis in Patients with Minimally Invasive Follicular Thyroid Carcinoma. PLoS ONE 2016, 11, e0155489. [Google Scholar] [CrossRef]
- Podda, M.; Saba, A.; Porru, F.; Reccia, I.; Pisanu, A. Follicular thyroid carcinoma: Differences in clinical relevance between minimally invasive and widely invasive tumors. World J. Surg. Oncol. 2015, 13, 193. [Google Scholar] [CrossRef] [Green Version]
- Ju, Y.W.; Kim, S.-j.; Chai, Y.J.; Yi, J.W.; Seong, C.-Y.; Kim, J.-K.; Yu, H.W.; Choi, J.Y.; Lee, K.E. Clinicopathologic Features in Minimally Invasive Follicular Thyroid Cancer Patients with Distant Metastasis. Korean J. Endocr. Surg. 2016, 16, 100–106. [Google Scholar] [CrossRef]
- Kim, H.; Shin, J.H.; Hahn, S.Y.; Oh, Y.L.; Kim, S.W.; Park, K.W.; Lim, Y. Prediction of follicular thyroid carcinoma associated with distant metastasis in the preoperative and postoperative model. Head Neck 2019, 41, 2507–2513. [Google Scholar] [CrossRef]
- Gillies, R.J.; Kinahan, P.E.; Hricak, H. Radiomics: Images Are More than Pictures, They Are Data. Radiology 2016, 278, 563–577. [Google Scholar] [CrossRef] [Green Version]
- Lambin, P.; Rios-Velazquez, E.; Leijenaar, R.; Carvalho, S.; Van Stiphout, R.G.; Granton, P.; Zegers, C.M.; Gillies, R.; Boellard, R.; Dekker, A.; et al. Radiomics: Extracting more information from medical images using advanced feature analysis. Eur. J. Cancer 2012, 48, 441–446. [Google Scholar] [CrossRef] [Green Version]
- Park, V.Y.; Han, K.; Lee, E.; Kim, E.-K.; Moon, H.J.; Yoon, J.H.; Kwak, J.Y. Association Between Radiomics Signature and Disease-Free Survival in Conventional Papillary Thyroid Carcinoma. Sci. Rep. 2019, 9, 4501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.; Lim, Y.; Ko, E.S.; Cho, H.-H.; Lee, J.E.; Han, B.-K.; Ko, E.Y.; Choi, J.S.; Park, K.W. Radiomics Signature on Magnetic Resonance Imaging: Association with Disease-Free Survival in Patients with Invasive Breast Cancer. Clin. Cancer Res. 2018, 24, 4705–4714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.; Liu, Z.; He, L.; Chen, X.; Pan, D.; Ma, Z.; Liang, C.; Tian, J.; Liang, C. Radiomics Signature: A Potential Biomarker for the Prediction of Disease-Free Survival in Early-Stage (I or II) Non—Small Cell Lung Cancer. Radiology 2016, 281, 947–957. [Google Scholar] [CrossRef]
- Huang, Y.-Q.; Liang, C.; He, L.; Tian, J.; Liang, C.-S.; Chen, X.; Ma, Z.-L.; Liu, Z. Development and Validation of a Radiomics Nomogram for Preoperative Prediction of Lymph Node Metastasis in Colorectal Cancer. J. Clin. Oncol. 2016, 34, 2157–2164. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Zhang, J.; Zhang, Q.; Xu, Y.; Zhou, J.; Liu, L. A radiomics nomogram for preoperative prediction of microvascular invasion risk in hepatitis B virus-related hepatocellular carcinoma. Diagn. Interv. Radiol. 2018, 24, 121–127. [Google Scholar] [CrossRef] [Green Version]
- Shin, J.H.; Baek, J.H.; Chung, J.; Ha, E.J.; Kim, J.-H.; Lee, Y.H.; Lim, H.K.; Moon, W.-J.; Na, D.G.; Park, J.S.; et al. Ultrasonography Diagnosis and Imaging-Based Management of Thyroid Nodules: Revised Korean Society of Thyroid Radiology Consensus Statement and Recommendations. Korean J. Radiol. 2016, 17, 370–395. [Google Scholar] [CrossRef] [Green Version]
- Tessler, F.N.; Middleton, W.D.; Grant, E.G.; Hoang, J.K.; Berland, L.L.; Teefey, S.A.; Cronan, J.J.; Beland, M.D.; Desser, T.S.; Frates, M.C.; et al. ACR Thyroid Imaging, Reporting and Data System (TI-RADS): White Paper of the ACR TI-RADS Committee. J. Am. Coll. Radiol. 2017, 14, 587–595. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, K.; Ota, H.; Hirokawa, M.; Yabuta, T.; Fukushima, M.; Masuoka, H.; Higashiyama, T.; Kihara, M.; Ito, Y.; Miya, A.; et al. “Nodule in Nodule” on Thyroid Ultrasonography: Possibility of Follicular Carcinoma Transformed from Benign Thyroid Tumor. Eur. Thyroid. J. 2016, 6, 101–107. [Google Scholar] [CrossRef] [Green Version]
- Song, G.; Xue, F.; Zhang, C. A Model Using Texture Features to Differentiate the Nature of Thyroid Nodules on Sonography. J. Ultrasound Med. 2015, 34, 1753–1760. [Google Scholar] [CrossRef]
- Nam, S.J.; Yoo, J.; Lee, H.S.; Kim, E.-K.; Moon, H.J.; Yoon, J.H.; Kwak, J.Y. Quantitative Evaluation for Differentiating Malignant and Benign Thyroid Nodules Using Histogram Analysis of Grayscale Sonograms. J. Ultrasound Med. 2016, 35, 775–782. [Google Scholar] [CrossRef]
- Chen, S.-J.; Chang, C.-Y.; Chang, K.-Y.; Tzeng, J.-E.; Chen, Y.-T.; Lin, C.-W.; Hsu, W.-C.; Wei, C.-K. Classification of the Thyroid Nodules Based on Characteristic Sonographic Textural Feature and Correlated Histopathology Using Hierarchical Support Vector Machines. Ultrasound Med. Boil. 2010, 36, 2018–2026. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, K.S.S.; Lam, A.C.L.; Pang, S.W.A.; Wang, D.; Ahuja, A.T. Feasibility Study of Texture Analysis Using Ultrasound Shear Wave Elastography to Predict Malignancy in Thyroid Nodules. Ultrasound Med. Boil. 2016, 42, 1671–1680. [Google Scholar] [CrossRef] [PubMed]
- Grani, G.; D’Alessandri, M.; Carbotta, G.; Nesca, A.; Del Sordo, M.; Alessandrini, S.; Coccaro, C.; Rendina, R.; Bianchini, M.; Prinzi, N.; et al. Grey-Scale Analysis Improves the Ultrasonographic Evaluation of Thyroid Nodules. Medicine 2015, 94, e1129. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.-R.; Shin, J.H.; Park, H.; Cho, H.; Hahn, S.Y.; Park, K.W. Radiomics Study of Thyroid Ultrasound for Predicting BRAF Mutation in Papillary Thyroid Carcinoma: Preliminary Results. Am. J. Neuroradiol. 2020, 41, 700–705. [Google Scholar] [CrossRef] [Green Version]
- Nixon, I.; Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; et al. Faculty Opinions recommendation of 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Fac. Opin.—Post-Publ. Peer Rev. Biomed. Lit. 2015, 26, 1–133. [Google Scholar] [CrossRef] [Green Version]
- Suster, S. Thyroid tumors with a follicular growth pattern: Problems in differential diagnosis. Arch. Pathol. Lab. Med. 2006, 130, 984–988. [Google Scholar]
- Sala, E.; Mema, E.; Himoto, Y.; Veeraraghavan, H.; Brenton, J.D.; Snyder, A.; Weigelt, B.; Vargas, H.A. Unravelling tumour heterogeneity using next-generation imaging: Radiomics, radiogenomics, and habitat imaging. Clin. Radiol. 2016, 72, 3–10. [Google Scholar] [CrossRef] [Green Version]
- Bedard, P.L.; Hansen, A.R.; Ratain, M.J.; Siu, L.L. Tumour heterogeneity in the clinic. Nature 2013, 501, 355–364. [Google Scholar] [CrossRef] [Green Version]
- Foley, D. Considerations of sample and feature size. IEEE Trans. Inf. Theory 1972, 18, 618–626. [Google Scholar] [CrossRef]
- Lee, S.E.; Han, K.; Kwak, J.Y.; Lee, E.; Kim, E.-K. Radiomics of US texture features in differential diagnosis between triple-negative breast cancer and fibroadenoma. Sci. Rep. 2018, 8, 13546. [Google Scholar] [CrossRef]
- Parameswaran, R.; Hu, J.S.; En, N.M.; Tan, W.; Yuan, N. Patterns of metastasis in follicular thyroid carcinoma and the difference between early and delayed presentation. Ann. R. Coll. Surg. Engl. 2017, 99, 151–154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Characteristics | Without Metastasis (n = 134) | With Metastasis (n = 35) | p-Value |
|---|---|---|---|
| Age (years) | 46.51 ± 14.09 | 59.35 ± 12.0 | <0.0001 |
| Sex | 0.214 | ||
| Female | 99 (73.9) | 30 (85.7) | |
| Male | 35 (26.1) | 5 (14.3) | |
| Size | 3.25 ± 1.52 | 4.03 ± 2.86 | 0.033 |
| Histology | <0.0001 | ||
| Minimally invasive | 129 (96.3) | 15 (42.9) | |
| Widely invasive | 5 (3.7) | 20 (57.1) | |
| Extrathyroidal extension | <0.0001 | ||
| None | 128 (95.52) | 23 (65.71) | |
| Minimal | 6 (4.48) | 8 (22.86) | |
| Gross | 0 (0) | 4 (11.43) | |
| Lymph node metastasis | 0.006 | ||
| Absence | 133 (99.25) | 31 (88.57) | |
| Presence | 1 (0.75) | 4 (11.43) |
| US Features | Without Metastasis (n = 134) | With Metastasis (n = 35) | p-Value |
|---|---|---|---|
| Internal content | 0.221 | ||
| Solid | 89 (66.42) | 28 (80.0) | |
| Predominantly solid | 44 (32.84) | 6 (17.14) | |
| Predominantly cystic | 1 (0.75) | 1 (2.86) | |
| Echogenicity | <0.0001 | ||
| Marked hypoechogenicity | 18 (13.43) | 16 (45.71) | |
| Hypoechogenicity | 49 (36.57) | 12 (34.29) | |
| Isoechogenicity | 66 (49.25) | 7 (20.0) | |
| Hyperechogenicity | 1 (0.75) | 0 (0) | |
| Shape | 0.710 | ||
| Irregular | 16 (11.94) | 5 (14.29) | |
| Oval to round | 118 (88.06) | 30 (85.71) | |
| Orientation | 0.931 | ||
| Nonparallel | 8 (5.97) | 6 (17.14) | |
| Parallel | 126 (94.03) | 29 (82.86) | |
| Margin | 0.183 | ||
| Spiculated/microlobulated | 10 (7.46) | 6 (17.14) | |
| Ill-defined | 8 (5.97) | 1 (2.86) | |
| Smooth | 116 (86.57) | 28 (80.8) | |
| Calcification | <0.0001 | ||
| No | 96 (71.64) | 6 (17.14) | |
| Microcalcification | 4 (2.99) | 2 (5.71) | |
| Macrocalcification | 12 (8.96) | 3 (8.57) | |
| Rim calcification | 22 (16.42) | 24 (68.57) | |
| Halo | 0.308 | ||
| No | 22 (16.42) | 9 (25.71) | |
| Yes | 112 (83.58) | 26 (74.29) | |
| Vascularity | 0.206 | ||
| No | 4 (2.99) | 3 (8.57) | |
| Perinodular | 18 (13.43) | 2 (5.71) | |
| Mild | 36 (36.87) | 6 (17.14) | |
| Marked | 29 (21.64) | 7 (20.0) | |
| Not done | 47 (35.07) | 17 (48.57) | |
| Nodule-in-nodule appearance | <0.0001 | ||
| No | 110 (82.09) | 13 (37.14) | |
| Yes | 24 (17.91) | 22 (62.86) | |
| K-TIRADS category | <0.0001 | ||
| 3 (low suspicion) | 76 (56.72) | 7 (20.00) | |
| 4 (intermediate suspicion) | 48 (35.82) | 18 (51.43) | |
| 5 (high suspicion) | 10 (7.46) | 10 (28.57) | |
| Suspicious lymph node | 0.037 | ||
| Presence | 1 (0.75) | 3 (8.57) | |
| Absence | 133 (99.25) | 32 (91.43) |
| Predictors | Radiomics Features | Radiomics Signature and Widely Invasive Histology | |
|---|---|---|---|
| Training performance | AUC | 0.93 | 0.94 |
| Accuracy | 0.88 | 0.88 | |
| Sensitivity | 0.92 | 0.94 | |
| Specificity | 0.87 | 0.86 | |
| Test performance | AUC | 0.90 | 0.93 |
| Accuracy | 0.85 | 0.87 | |
| Sensitivity | 0.80 | 0.91 | |
| Specificity | 0.87 | 0.86 | |
| Variables | Univariate | Multivariate (5-Fold Averaged) | ||
|---|---|---|---|---|
| Correlation Coefficient | p-Value | Beta Coefficient | p-Value | |
| Age | 0.356 | <0.0001 | 0.022 | 0.502 |
| Tumor size | 0.165 | 0.033 | −0.024 | 0.431 |
| Widely invasive histology | 0.610 | <0.0001 | 0.335 | 0.003 |
| Extrathyroidal extension | 0.417 | <0.0001 | 0.039 | 0.349 |
| Lymph node metastasis | 0.256 | 0.001 | −0.033 | 0.684 |
| Nodule-in-nodule appearance | 0.409 | <0.0001 | 0.069 | 0.361 |
| Echogenicity | −0.330 | <0.0001 | −0.003 | 0.649 |
| Orientation | −0.164 | 0.033 | 0.088 | 0.446 |
| No calcification | −0.452 | <0.0001 | −0.109 | 0.221 |
| Rim calcification | 0.475 | <0.0001 | 0.039 | 0.478 |
| K-TIRADS category | 0.342 | <0.0001 | −0.029 | 0.541 |
| Suspicious lymph node on US | 0.209 | 0.007 | 0.037 | 0.880 |
| Radiomics signature * | 0.649 | <0.0001 | 0.079 | <0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwon, M.-r.; Shin, J.H.; Park, H.; Cho, H.; Kim, E.; Hahn, S.Y. Radiomics Based on Thyroid Ultrasound Can Predict Distant Metastasis of Follicular Thyroid Carcinoma. J. Clin. Med. 2020, 9, 2156. https://doi.org/10.3390/jcm9072156
Kwon M-r, Shin JH, Park H, Cho H, Kim E, Hahn SY. Radiomics Based on Thyroid Ultrasound Can Predict Distant Metastasis of Follicular Thyroid Carcinoma. Journal of Clinical Medicine. 2020; 9(7):2156. https://doi.org/10.3390/jcm9072156
Chicago/Turabian StyleKwon, Mi-ri, Jung Hee Shin, Hyunjin Park, Hwanho Cho, Eunjin Kim, and Soo Yeon Hahn. 2020. "Radiomics Based on Thyroid Ultrasound Can Predict Distant Metastasis of Follicular Thyroid Carcinoma" Journal of Clinical Medicine 9, no. 7: 2156. https://doi.org/10.3390/jcm9072156






