Impact of Galectin-3 Circulating Levels on Frailty in Elderly Patients with Systolic Heart Failure
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Frailty Assessment
2.3. Biomarker Assessments
2.4. Statistical Analysis
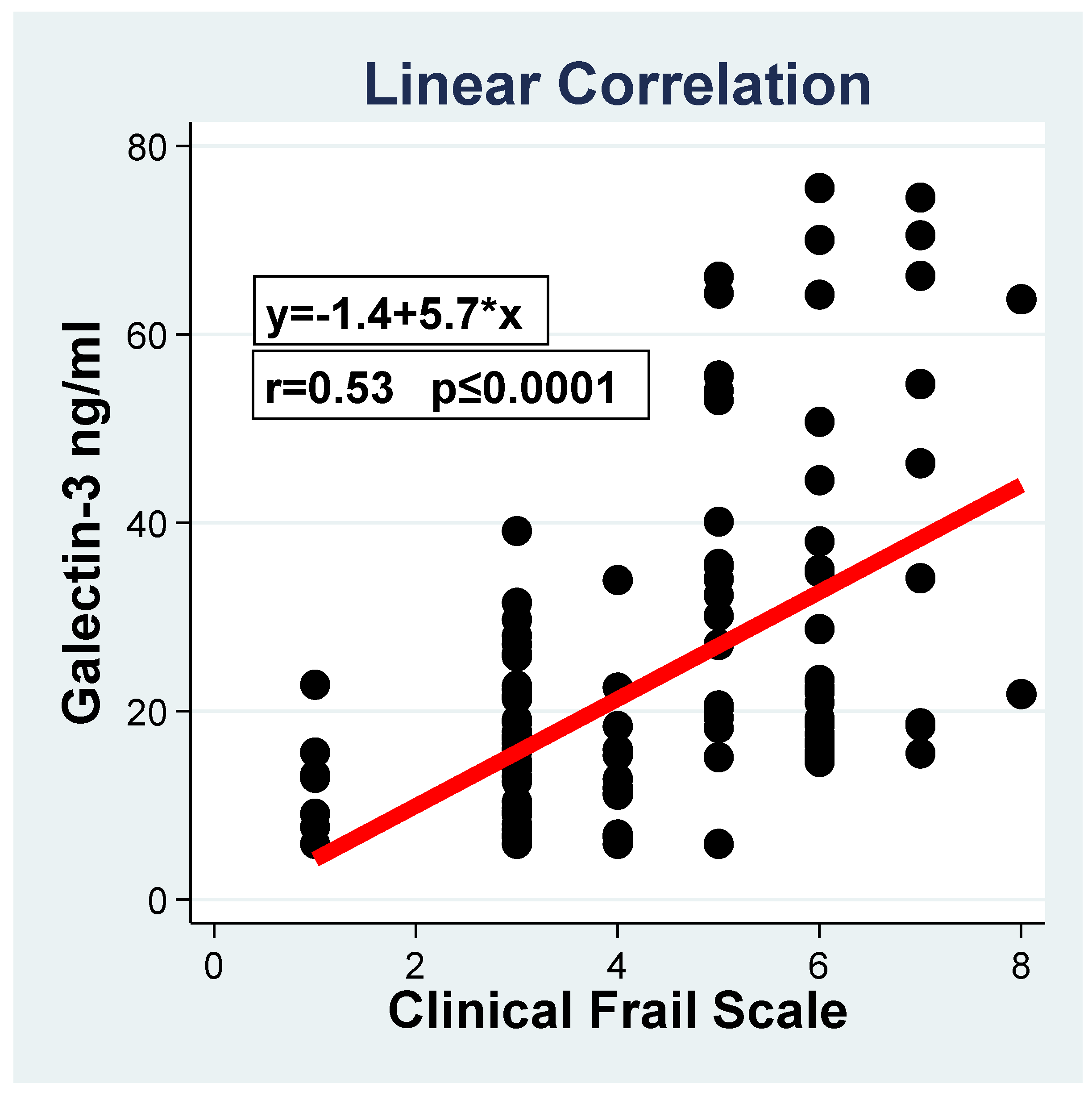
3. Results
3.1. Characteristics of the Overall Study Population and of Subgroups According to Frailty Status
3.2. Clinical and Laboratory Factors Associated with Frailty
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. Ser. A 2001, 56, M146–M157. [Google Scholar] [CrossRef] [PubMed]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K.; Iliffe, S. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef] [Green Version]
- Bandeen-Roche, K.; Seplaki, C.L.; Huang, J.; Buta, B.; Kalyani, R.R.; Varadhan, R.; Xue, Q.-L.; Walston, J.D.; Kasper, J.D. Frailty in Older Adults: A Nationally Representative Profile in the United States. J. Gerontol. Ser. A 2015, 70, 1427–1434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Caoimh, R.; Galluzzo, L.; Rodríguez-Laso, Á.; Van Der Heyden, J.; Ranhoff, A.H.; Lamprini-Koula, M.; Ciutan, M.; López-Samaniego, L.; Carcaillon-Bentata, L.; Kennelly, S.; et al. Prevalence of frailty at population level in European ADVANTAGE Joint Action Member States: A systematic review and meta-analysis. Annali dell’Istituto Superiore di Sanità 2018, 54, 226–238. [Google Scholar] [PubMed]
- Wang, J.; Maxwell, C.A.; Yu, F. Biological Processes and Biomarkers Related to Frailty in Older Adults: A State-of-the-Science Literature Review. Boil. Res. Nurs. 2018, 21, 80–106. [Google Scholar] [CrossRef]
- O’Connor, C.M.; Stough, W.G.; Gallup, D.S.; Hasselblad, V.; Gheorghiade, M. Demo-graphics, clinical characteristics, and outcomes of patients hospitalized for decom-pensated heart failure: Observations from the IMPACT-HF registry. J. Card Fail. 2005, 11, 200–205. [Google Scholar] [CrossRef]
- Sze, S.; Pellicori, P.; Zhang, J.; Weston, J.; Clark, A.L. Identification of Frailty in Chronic Heart Failure. JACC Hear. Fail. 2019, 7, 291–302. [Google Scholar] [CrossRef]
- Astiz, M.T.V.; Blaya-Novakova, V.; Sánchez, E.; Ortiz, J.; Serra-Rexach, J.A.; Bueno, H. Prevalence and prognostic impact of frailty and its components in non-dependent elderly patients with heart failure. Eur. J. Hear. Fail. 2016, 18, 869–875. [Google Scholar] [CrossRef] [Green Version]
- Sanders, N.A.; Supiano, M.A.; Lewis, E.F.; Liu, J.; Claggett, B.; Pfeffer, M.A.; Desai, A.S.; Sweitzer, N.K.; Solomon, S.D.; Fang, J.C. The frailty syndrome and outcomes in the TOPCAT trial. Eur. J. Hear. Fail. 2018, 20, 1570–1577. [Google Scholar] [CrossRef] [Green Version]
- Rockwood, K.; Song, X.; Macknight, C.; Bergman, H.; Hogan, D.B.; McDowell, I.; Mitnitski, A. A global clinical measure of fitness and frailty in elderly people. Can. Med Assoc. J. 2005, 173, 489–495. [Google Scholar] [CrossRef] [Green Version]
- Madan, S.A.; Fida, N.; Barman, P.; Sims, D.; Shin, J.; Verghese, J.; Piña, I.; Jorde, U.; Patel, S.R. Frailty Assessment in Advanced Heart Failure. J. Card. Fail. 2016, 22, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Adamo, L.; Rocha-Resende, C.; Prabhu, S.D.; Mann, D.L. Reappraising the role of inflammation in heart failure. Nat. Rev. Cardiol. 2020, 17, 269–285. [Google Scholar] [CrossRef] [PubMed]
- Leng, S.X.; Chen, X.; Mao, G. Frailty syndrome: An overview. Clin. Interv. Aging 2014, 9, 433–441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zampino, M.; Ferrucci, L.; Semba, R.D. Biomarkers in the path from cellular senescence to frailty. Exp. Gerontol. 2019, 129, 110750. [Google Scholar] [CrossRef] [PubMed]
- Soysal, P.; Stubbs, B.; Lucato, P.; Luchini, C.; Solmi, M.; Peluso, R.; Sergi, G.; Isik, A.T.; Manzato, E.; Maggi, S.; et al. Inflammation and frailty in the elderly: A systematic review and meta-analysis. Ageing Res. Rev. 2016, 31, 1–8. [Google Scholar] [CrossRef]
- Wang, Z.-D.; Yao, S.; Shi, G.-P.; Wang, Y.; Shi, J.-M.; Guo, J.-H.; Zhu, Y.-S.; Jiang, X.-Y.; Chu, X.-F.; Wang, X. Frailty index is associated with increased risk of elevated BNP in an elderly population: The Rugao Longevity and Ageing Study. Aging Clin. Exp. Res. 2019, 32, 305–311. [Google Scholar] [CrossRef]
- Lok, D.J.A.; Van Der Meer, P.; De La Porte, P.W.B.-A.; Lipsic, E.; Van Wijngaarden, J.; Hillege, H.L.; Van Veldhuisen, D.J. Prognostic value of galectin-3, a novel marker of fibrosis, in patients with chronic heart failure: Data from the DEAL-HF study. Clin. Res. Cardiol. 2010, 99, 323–328. [Google Scholar] [CrossRef] [Green Version]
- Sharma, U.C.; Pokharel, S.; van Brakel, T.J.; van Berlo, J.H.; Cleutjens, J.P.; Schroen, B.; Andre, S.; Crijns, H.J.; Gabius, H.J.; Maessen, J.; et al. Galectin-3 Marks Activated Macrophages in Failure-Prone Hypertrophied Hearts and Contributes to Cardiac Dysfunction. Circulation 2004, 110, 3121–3128. [Google Scholar] [CrossRef]
- Yu, L.; Ruifrok, W.P.; Meissner, M.; Bos, E.M.; van Goor, H.; Sanjabi, B.; van der Harst, P.; Pitt, B.; Goldstein, I.J.; Koerts, J.A.; et al. Genetic and phar- macological inhibition of galectin-3 pre- vents cardiac remodeling by interfering with myocardial fibrogenesis. Circ. Heart Fail. 2013, 6, 107–117. [Google Scholar] [CrossRef] [Green Version]
- Keng, B.M.; Gao, F.; Ewe, S.H.; Tan, R.S.; Teo, L.L.; Xie, B.Q.; Koh, W.; Koh, A.S. Galectin–3 as a candidate upstream biomarker for quantifying risks of myocardial ageing. ESC Hear. Fail. 2019, 6, 1068–1076. [Google Scholar] [CrossRef] [Green Version]
- Cardoso, A.L.; Fernandes, A.; Aguilar-Pimentel, J.A.; De Angelis, M.H.; Guedes, J.R.; Brito, M.A.; Ortolano, S.; Pani, G.; Athanasopoulou, S.; Gonos, E.S.; et al. Towards frailty biomarkers: Candidates from genes and pathways regulated in aging and age-related diseases. Ageing Res. Rev. 2018, 47, 214–277. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A New Equation to Estimate Glomerular Filtration Rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Afilalo, J. The Clinical Frailty Scale: Upgrade Your Eyeball Test. Circulation 2017, 135, 2025–2027. [Google Scholar] [CrossRef] [PubMed]
- Fortin, M.; Hudon, C.; Dubois, M.-F.; Almirall, J.; Lapointe, L.; Soubhi, H. Comparative assessment of three different indices of multimorbidity for studies on health-related quality of life. Heal. Qual. Life Outcomes 2005, 3, 74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rochon, P.A.; Katz, J.N.; Morrow, L.A.; McGlinchey-Berroth, R.; Alquist, M.M.; Sarkaretai, M.; Minaher, K.L. Co-morbid illness is associated with survival and length of hospital stay in patients with chronic disability. A prospective comparison of the comordbidity indices. Med. Care 1996, 34, 1093–1101. [Google Scholar] [CrossRef] [PubMed]
- Felker, G.M.; Fiuzat, M.; Shaw, L.K.; Clare, R.; Whellan, D.J.; Bettari, L.; Shirolkar, S.C.; Donahue, M.; Kitzman, D.W.; Zannad, F.; et al. Galectin-3 in ambulatory patients with heart failure: Results from the HF-ACTION study. Circ. Hear. Fail. 2011, 5, 72–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCullough, P.A.; Olobatoke, A.; Vanhecke, T.E. Galectin-3: A novel blood test for the evaluation and management of patients with heart failure. Rev. Cardiovasc. Med. 2011, 12, 200–210. [Google Scholar] [PubMed]
- Vittinghoff, E.; McCulloch, C.E. Relaxing the Rule of Ten Events per Variable in Logistic and Cox Regression. Am. J. Epidemiol. 2007, 165, 710–718. [Google Scholar] [CrossRef] [Green Version]
- Royston, P.; Ambler, G.; Sauerbrei, W. The use of fractional polynomials to model continuous risk variables in epidemiology. Int. J. Epidemiol. 1999, 28, 964–974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vickers, A.J.; Cronin, A.M.; Elkin, E.B.; Gönen, M. Extensions to decision curve analysis, a novel method for evaluating diagnostic tests, prediction models and molecular markers. BMC Med. Inform. Decis. Mak. 2008, 8, 53. [Google Scholar] [CrossRef] [Green Version]
- Lanzillo, B.; Piscosquito, G.; Marcuccio, L.; Lanzillo, A.; Vitale, D.F. Prognosis of severe acquired brain injury: Short and long-term outcome determinants and their potential clinical relevance after rehabilitation. A comprehensive approach to analyze cohort studies. PLoS ONE 2019, 14, e0216507. [Google Scholar] [CrossRef] [PubMed]
- Komici, K.; Vitale, D.F.; Leosco, D.; Mancini, A.; Corbi, G.; Bencivenga, L.; Mezzani, A.; Trimarco, B.; Morisco, C.; Ferrara, N.; et al. Pressure injuries in elderly with acute myocardial infarction. Clin. Interv. Aging 2017, 12, 1495–1501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Denfeld, Q.E.; Winters-Stone, K.; Mudd, J.O.; Gelow, J.M.; Kurdi, S.; Lee, C.S. The prevalence of frailty in heart failure: A systematic review and meta-analysis. Int. J. Cardiol. 2017, 236, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Jha, S.R.; Ha, H.S.K.; Hickman, L.D.; Hannu, M.; Davidson, P.M.; Macdonald, P.S.; Newton, P.J. Frailty in advanced heart failure: A systematic review. Hear. Fail. Rev. 2015, 20, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Vigorito, C.; Abreu, A.; Ambrosetti, M.; Belardinelli, R.; Corrà, U.; Cupples, M.E.; Davos, C.H.; Hoefer, S.; Iliou, M.-C.; Schmid, J.-P.; et al. Frailty and cardiac rehabilitation: A call to action from the EAPC Cardiac Rehabilitation Section. Eur. J. Prev. Cardiol. 2016, 24, 577–590. [Google Scholar] [CrossRef] [Green Version]
- Pandey, A.; Kitzman, D.; Reeves, G. Frailty Is Intertwined with Heart Failure: Mechanisms, Prevalence, Prognosis, Assessment, and Management. JACC Hear. Fail. 2019, 7, 1001–1011. [Google Scholar] [CrossRef]
- Yang, X.; Lupón, J.; Vidán, M.T.; Ferguson, C.; Gastelurrutia, P.; Newton, P.J.; Macdonald, P.S.; Bueno, H.; Bayés–Genís, A.; Woo, J.; et al. Impact of Frailty on Mortality and Hospitalization in Chronic Heart Failure: A Systematic Review and Meta–Analysis. J. Am. Hear. Assoc. 2018, 7. [Google Scholar] [CrossRef] [Green Version]
- Nagaratnam, N.; Gayagay, G. Validation of the Cumulative Illness Rating Scale (CIRS) in hospitalized nonagenarians. Arch. Gerontol. Geriatr. 2007, 44, 29–36. [Google Scholar] [CrossRef]
- Lenti, M.V.; Pasina, L.; Cococcia, S.; Cortesi, L.; Miceli, E.; Dominioni, C.C.; Pisati, M.; Mengoli, C.; Perticone, F.; Nobili, A.; et al. Mortality rate and risk factors for gastrointestinal bleeding in elderly patients. Eur. J. Intern. Med. 2019, 61, 54–61. [Google Scholar] [CrossRef]
- Milani, P.; Rajkumar, S.V.; Merlini, G.; Kumar, S.; Gertz, M.A.; Palladini, G.; Lacy, M.Q.; Buadi, F.K.; Hayman, S.R.; Leung, N.; et al. N–terminal fragment of the type–B natriuretic peptide (NT–proBNP) contributes to a simple new frailty score in patients with newly diagnosed multiple myeloma. Am. J. Hematol. 2016, 91, 1129–1134. [Google Scholar] [CrossRef] [Green Version]
- Baylis, D.; Bartlett, D.B.; Syddall, H.E.; Ntani, G.; Gale, C.R.; Cooper, C.; Lord, J.; Sayer, A.A. Immune-endocrine biomarkers as predictors of frailty and mortality: A 10-year longitudinal study in community-dwelling older people. AGE 2012, 35, 963–971. [Google Scholar] [CrossRef] [PubMed]
- Puts, M.T.E.; Visser, M.; Twisk, J.W.R.; Deeg, R.J.H.; Lips, P. Endocrine and inflammatory markers as predictors of frailty. Clin. Endocrinol. 2005, 63, 403–411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boxer, R.S.; Dauser, D.A.; Walsh, S.J.; Hager, W.D.; Kenny, A.M. The Association Between Vitamin D and Inflammation with the 6-Minute Walk and Frailty in Patients with Heart Failure. J. Am. Geriatr. Soc. 2008, 56, 454–461. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, E.; Picca, A.; Marini, F.; Biancolillo, A.; Coelho-Junior, H.J.; Gervasoni, J.; Bossola, M.; Cesari, M.; Onder, G.; Landi, F.; et al. Inflammatory signatures in older persons with physical frailty and sarcopenia: The frailty “cytokinome” at its core. Exp. Gerontol. 2019, 122, 129–138. [Google Scholar] [CrossRef]
- Mitnitski, A.; Collerton, J.; Martin-Ruiz, C.; Jagger, C.; Von Zglinicki, T.; Rockwood, K.; Kirkwood, T.B.L. Age-related frailty and its association with biological markers of ageing. BMC Med. 2015, 13, 161. [Google Scholar] [CrossRef] [Green Version]
- Vickers, A.J.; Van Calster, B.; Steyerberg, E.W. Net benefit approaches to the evaluation of prediction models, molecular markers, and diagnostic tests. BMJ 2016, 352, i6. [Google Scholar] [CrossRef] [Green Version]
- Rabinovich, G.A.; Toscano, M.A. Turning ‘sweet’ on immunity: Galectin–glycan interactions in immune tolerance and inflammation. Nat. Rev. Immunol. 2009, 9, 338–352. [Google Scholar] [CrossRef]
- Komici, K.; Rengo, G.; Leosco, D.; Ferrara, N. Cardiac fibrosis in heart failure. JGG 2017, 65, 177–183. [Google Scholar]
- Sciacchitano, S.; Lavra, L.; Morgante, A.; Ulivieri, A.; Magi, F.; De Francesco, G.P.; Bellotti, C.; Salehi, L.B.; Ricci, A. Galectin-3: One Molecule for an Alphabet of Diseases, from A to Z. Int. J. Mol. Sci. 2018, 19, 379. [Google Scholar] [CrossRef] [Green Version]
- Varricchi, G.; Bencivenga, L.; Poto, R.; Pecoraro, A.; Shamji, M.H.; Rengo, G. The emerging role of T follicular helper (TFH) cells in aging: Influence on the immune frailty. Ageing Res. Rev. 2020, 61, 101071. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-J.; Lee, H.-W.; Kang, H.G.; La, S.-H.; Choi, I.J.; Ro, J.Y.; Bresalier, R.S.; Song, J.; Chun, K.-H. Ablation of galectin-3 induces p27(KIP1)-dependent premature senescence without oncogenic stress. Cell Death Differ. 2014, 21, 1769–1779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Openo, K.P.; Kadrofske, M.M.; Patterson, R.J.; Wang, J.L. Galectin-3 Expression and Subcellular Localization in Senescent Human Fibroblasts. Exp. Cell Res. 2000, 255, 278–290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srivatsan, V.; George, M.; Shanmugam, E. Utility of galectin-3 as a prognostic biomarker in heart failure: Where do we stand? Eur. J. Prev. Cardiol. 2014, 22, 1096–1110. [Google Scholar] [CrossRef] [PubMed]
- Testa, M.; Rosso, G.L.; Ferreri, C.; Feola, M. The Predictive Value of Plasma Brain Natriuretic Peptide and Galectin-3 in Elderly Patients Admitted for Heart Failure †. Diseases 2018, 6, 88. [Google Scholar] [CrossRef] [Green Version]

| Characteristics | All Population (N = 128) | Frail (N = 54) | Non- Frail (N = 74) | p-Value |
|---|---|---|---|---|
| Age, years | 69.2 ± 4.8 | 70.5 ± 5.4 | 68.2 ± 4.2 | 0.008 |
| Gender M, N (%) | 112 (87.5) | 45/54 (83.4) | 66/73 (90.4) | 0.28 |
| BMI, kg/m2 | 25.4 ± 4.3 | 24.5 ± 4.6 | 26.0 ± 4.2 | 0.07 |
| WBC/µL, | 7833 ± 2623.5 | 8104.7 ± 4145.9 | 7430.6 ± 2099.6 | 0.78 |
| Hbg g/dL | 12.8 ± 1.5 | 12.2 ± 1.6 | 13.6 ± 1.7 | 0.001 |
| Fibrinogen | 400.2 ± 65.8 | 405.2 ± 112.4 | 394.8 ± 108.3 | 0.75 |
| Na mmol/L | 138.4 ± 3.2 | 138.3 ± 4.5 | 138.7 ± 3.8 | 0.68 |
| GFR mL/kg | 65.2 ± 18.4 | 59.1 ± 20.4 | 70.2 ± 14.8 | 0.001 |
| CRP mg/L | 7.9 ± 10.7 | 13.4 ± 13.8 | 3.7 ± 4.2 | <0.0001 |
| LVEF % | 28.7 ± 8.5 | 26.7 ± 6.1 | 30.2 ± 10.2 | 0.02 |
| NT proBNP pg/mL | 5922.4 ± 15,099.9 | 11,427.9 ± 21,803.4 | 1856.4 ± 3570.1 | 0.002 |
| Galectin-3 ng/mL | 22.8 ± 16.9 | 34.4 ± 19.3 | 14.3 ± 7.6 | <0.0001 |
| Hypertension, N (%) | 80 (62.5) | 37/54 (68.5) | 43/74 (58.1) | 0.23 |
| Dyslipidemia, N (%) | 108 (84.3) | 43/54 (79.6) | 65/74 (87.8) | 0.21 |
| NYHA class III,IV N(%) | 58 (54.7) | 35/54 (64.8) | 23/74 (31.1) | <0.0001 |
| CIRS-CI | 3.73 ± 2.2 | 4.6 ± 2.2 | 3.1 ± 1.9 | <0.0001 |
| CFS | 4.27 ±1.7 | 5.9 ± 0.8 | 3.1 ± 0.8 | <0.0001 |
| ACEInhib/ARBs | 104 (81.2) | 41/54 (75.9) | 63/74 (85.1) | 0.19 |
| Beta-blockers | 85 (66.4) | 40/54 (74.1) | 45/74 (60.8) | 0.12 |
| Diuretics * | 112 (87.5) | 48/54 (88.9) | 64/74 (86.4) | 1.0 |
| Drugs Number | 4.84 ± 1.4 | 4.9 ± 1.2 | 4.8 ± 1.4 | 0.34 |
| Global Pseudo R2 * = 0.5 | ||||
|---|---|---|---|---|
| Variables | Odd’s Ratio | 95% CI | p-Value | Partial Contribution to Global R2 |
| Age (decades) | 3.29 | 1.03–10.55 | 0.045 | 6.3% |
| Gender | 0.86 | 0.19–4.03 | 0.854 | NA |
| BMI | 0.95 | 0.83–1.10 | 0.415 | NA |
| CKD | 1.47 | 0.34–6.36 | 0.605 | NA |
| CIRS-CI (SD units) | 1.85 | 1.03–3.32 | 0.039 | 10.3% |
| NYHA Class III, IV | 1.53 | 0.46–5.13 | 0.456 | NA |
| LVEF | 0.93 | 0.84–1-03 | 0.172 | NA |
| NT-proBNP (SD units) | 2.39 | 1.22–4.73 | 0.012 | 24.5% |
| Hgb | 0.95 | 0.64–1.40 | 0.828 | NA |
| CRP (SD units) | 3.73 | 1.24–11.22 | 0.019 | 19.5% |
| Gal-3 (SD units) | 5.65 | 1.97–16.22 | 0.019 | 39.4% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Komici, K.; Gnemmi, I.; Bencivenga, L.; Vitale, D.F.; Rengo, G.; Di Stefano, A.; Eleuteri, E. Impact of Galectin-3 Circulating Levels on Frailty in Elderly Patients with Systolic Heart Failure. J. Clin. Med. 2020, 9, 2229. https://doi.org/10.3390/jcm9072229
Komici K, Gnemmi I, Bencivenga L, Vitale DF, Rengo G, Di Stefano A, Eleuteri E. Impact of Galectin-3 Circulating Levels on Frailty in Elderly Patients with Systolic Heart Failure. Journal of Clinical Medicine. 2020; 9(7):2229. https://doi.org/10.3390/jcm9072229
Chicago/Turabian StyleKomici, Klara, Isabella Gnemmi, Leonardo Bencivenga, Dino Franco Vitale, Giuseppe Rengo, Antonino Di Stefano, and Ermanno Eleuteri. 2020. "Impact of Galectin-3 Circulating Levels on Frailty in Elderly Patients with Systolic Heart Failure" Journal of Clinical Medicine 9, no. 7: 2229. https://doi.org/10.3390/jcm9072229







