1. Introduction
Orbital lymphoproliferative tumors are composed of a heterogeneous group that includes malignant lymphomas, such as mucosa-associated lymphoid tissue (MALT) lymphoma, follicular lymphoma, and diffuse large B-cell lymphoma, as well as benign lymphoproliferative tumors, such as immunoglobulin G4-related ophthalmic disease (IgG4-ROD) and reactive lymphoid hyperplasia. It is difficult to differentiate malignant lymphoma from benign lymphoproliferative disorders, as these lesions share common clinical symptoms, imaging diagnosis, and histologic features, as well as molecular markers [
1,
2,
3,
4,
5,
6]. Clinical symptoms, imaging diagnosis, histology, molecular analysis, and flow cytometry using biopsy specimens are the most commonly used approaches for the diagnosis of orbital lymphoproliferative disorders. In particular, the kappa/lambda ratio of a biopsy specimen is useful for verifying the monoclonality of malignant B cells. However, the wide range of kappa/lambda or lambda/kappa ratios (1.4–6.0) limits its specificity, and that test is not useful in identifying and distinguishing certain cases of orbital MALT lymphoma [
7,
8,
9]. Additionally, Sato et al. [
10] detected IgG heavy-chain gene rearrangements in two of 17 cases of IgG4-ROD, further increasing the challenge in differentiating IgG4-ROD from orbital MALT lymphoma without specific analysis. Furthermore, several reports have indicated that orbital MALT lymphoma [
10,
11] and diffuse large B-cell lymphoma [
12] develop based on IgG4-ROD, suggesting that these disorders are biologically related.
Therefore, correct diagnosis of IgG4-ROD and orbital MALT lymphoma (where both are of B-cell lineage) in routine clinical practice remains complicated and challenging due to the lack of specific diagnostic biomarkers. Elevated serum IgG4 is not sufficiently sensitive or specific for this purpose [
13,
14]. Therefore, approximately one-third of patients with IgG4-ROD do not meet the clinical criteria, and are diagnosed as “possible” or “probable” IgG4-ROD, which contributes to clinical confusion and delayed diagnosis [
15,
16].
MicroRNAs (miRNAs) are small non-coding RNA molecules (approximately 22 nucleotides in length) involved in the regulation of gene expression by partial base-pairing of target messenger RNAs (mRNA) to complementary sequences, which leads to cleavage and eventual degradation of the target mRNA or translational repression [
17,
18]. Recently, the role of non-coding RNAs, of which miRNAs are the most studied, has acquired remarkable importance in the pathogenesis of many types of disease, including cancer and inflammatory diseases. miRNAs have shown promise in recent years as biomarkers for the diagnosis and prognosis of cancer because of their diverse but tissue- and cell-specific biological and pathological functions [
19]. Currently, more than 2500 miRNAs have been identified in human genomes. However, there is limited knowledge regarding the expression of miRNA. Moreover, no high-throughput miRNA expression studies have been conducted to identify miRNAs specifically associated with diseases, and no biopsy tissue and serum miRNA profiling of orbital lymphoproliferative disorders has been performed to date.
In this study, we performed a comprehensive miRNA microarray analysis using the 3D-Gene Human miRNA Oligo Chip, aiming to identify differentially expressed miRNAs and pathways using biopsy and serum specimens from patients with orbital MALT lymphoma and IgG4-ROD using machine-learning methods. Through the analysis of the targeted miRNA data, we investigated their potential implication in disease pathogenesis and their usefulness as biomarkers.
4. Discussion
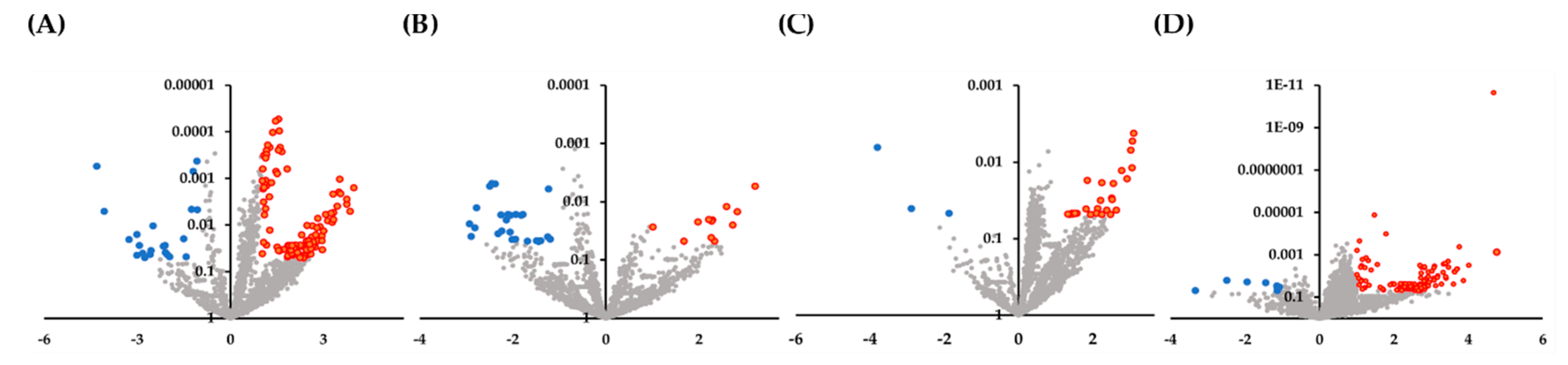
In the present study, we investigated the expression of 2565 miRNAs in both biopsy and serum specimens, and identified miRNAs as biomarkers of IgG4-ROD and orbital MALT lymphoma based on the criteria of |log FC| > 1 and p < 0.05. In ROC analysis, miRNA panels selected by RandomForest yielded AUC of 0.92 for IgG4-ROD and 0.78 for orbital MALT lymphoma, and these values are considered to be moderately accurate for the prediction of these two diseases. This study demonstrates miRNA profiles that distinguish IgG4-ROD cases from orbital MALT lymphoma and healthy volunteers, and provides a large number of miRNAs to promote comprehensive analysis of disease status. Detection of alterations in miRNAs in biopsy and serum specimens could improve current diagnostic methods for IgG4-ROD, as these data may substantiate the evidence of disease. Statistical analysis showed that orbital MALT lymphoma had significantly decreased miR-7112-3p in the biopsy and serum specimens. These results indicate that miR-7112-3p, which is important for differentiating between IgG4-ROD and orbital MALT lymphoma.
Regulatory T (Treg) cells producing IL-10 and TGFβ contribute to enhancement of IgG4 class switch recombination and the pathogenesis of IgG4-related disease (IgG4-RD) via B cells, as well as induction of fibrosis [
25]. Previous studies have confirmed the recruitment of FOXP3-positive Treg cells into the affected organs in IgG4-RD [
26]. In this analysis, miR-920 targeting FOXP3 was downregulated in the serum of IgG4-ROD patients compared with healthy individuals, indicating that miR-920 may regulate the role of Treg cells in the pathogenesis of IgG4-ROD, their function and accumulation in the orbit, as well as the degree of FOXP3 expression at the site. Moreover, pathologically, both IgG4-RD and IgG4-ROD are characterized by fibrosis [
27]. TGFβ produced by Treg cells contributes to fibrosis in IgG4-RD [
28,
29]. TGFβ regulates the expression of several miRNAs during renal fibrosis, such as miR-21, miR-29, miR-192, miR-200, and miR-433. MiR-21, miR-192, and miR-433 are positively induced by TGFβ signaling and play a pathological role in kidney diseases [
30]. In contrast, both miR-29 and miR-200 families, which are inhibited by TGFβ signaling, protect the kidney from renal fibrosis [
30]. In this study, we found that the top 10 upregulated miRNAs (including miR-3663-3p, miR-4673, and miR-4745-5p) and downregulated miRNAs (including miR-20b-5p, miR-6501-3p, miR-302c-5p, miR-758-5p, miR-193a-5p, miR-202-3p, let-7a-5p, and miR-379-5p) targeted 37 genes involved in the TGFβ-signaling pathway related to the pathogenesis of IgG4-ROD.
T follicular helper (Tfh) cells have also been considered important in the pathological processes of diseases, including IgG4-ROD and IgG4-RD. These cells are increased in active IgG4-RD, and the number of Tfh cells correlates with serum IgG4 and IL-4 levels [
31]. B-cell lymphoma 6 (BCL6) is considered to be the master transcription factor in the functioning of Tfh cells and differentiation of naïve helper T cells to Tfh cells [
32]. Strong BCL6 expression is detected in ectopic germinal centers in IgG4-RD patients [
33]. In our study, we found that the miRNA targeting BCL6 was miR-9-5p, which was downregulated in biopsy specimens of IgG4-ROD patients compared with orbital MALT lymphoma. These results provide preliminary evidence that BCL6 is targeted by miR-9-5p and is implicated in IgG4-ROD. Moreover, the decrease of miR-9-5p expression in IgG4-ROD may be a potential biomarker, leading to increased expression of BCL6.
Importantly, among all the differentially expressed serum miRNAs observed in this study, the most prominent putative biomarkers of IgG4-ROD and orbital MALT lymphoma determined through machine-learning methods (randomForest) were miR-202-3p and miR-7112-3p, respectively. The miR-202-3p level is known to correlate negatively with tumor size, transcriptionally target Gli1 and inactivate the Shh signal, and has been linked to the tumorigenesis and progression of various types of human malignancies [
34,
35,
36]. In addition, miR-202-3p inhibits gastric cancer proliferation through inducing cell apoptosis by direct interaction with Gli1 [
34]. It is difficult to compare the present results with those of previous reports considering that earlier reports did not evaluate the miRNA profiles of IgG4-RD including IgG4-ROD. Hence, additional studies are warranted to establish a relationship between miR-202-3p and the cause of IgG4-ROD. Based on the evidence provided by one published report, miR-7112-3p targets the RNA-dependent protein kinase R-like endoplasmic reticulum kinase (PERK), and inhibits the function of PERK signaling, resulting in reduced apoptosis [
37]. A significantly positive correlation between apoptotic and proliferative indices has been observed in gastrointestinal lymphomas of MALT [
38]. In this study, the expression level of miR-7112-3p was significantly downregulated in orbital MALT lymphoma patients. Further investigations are needed to confirm the relationship between miR-7112-3p and orbital MALT lymphoma.
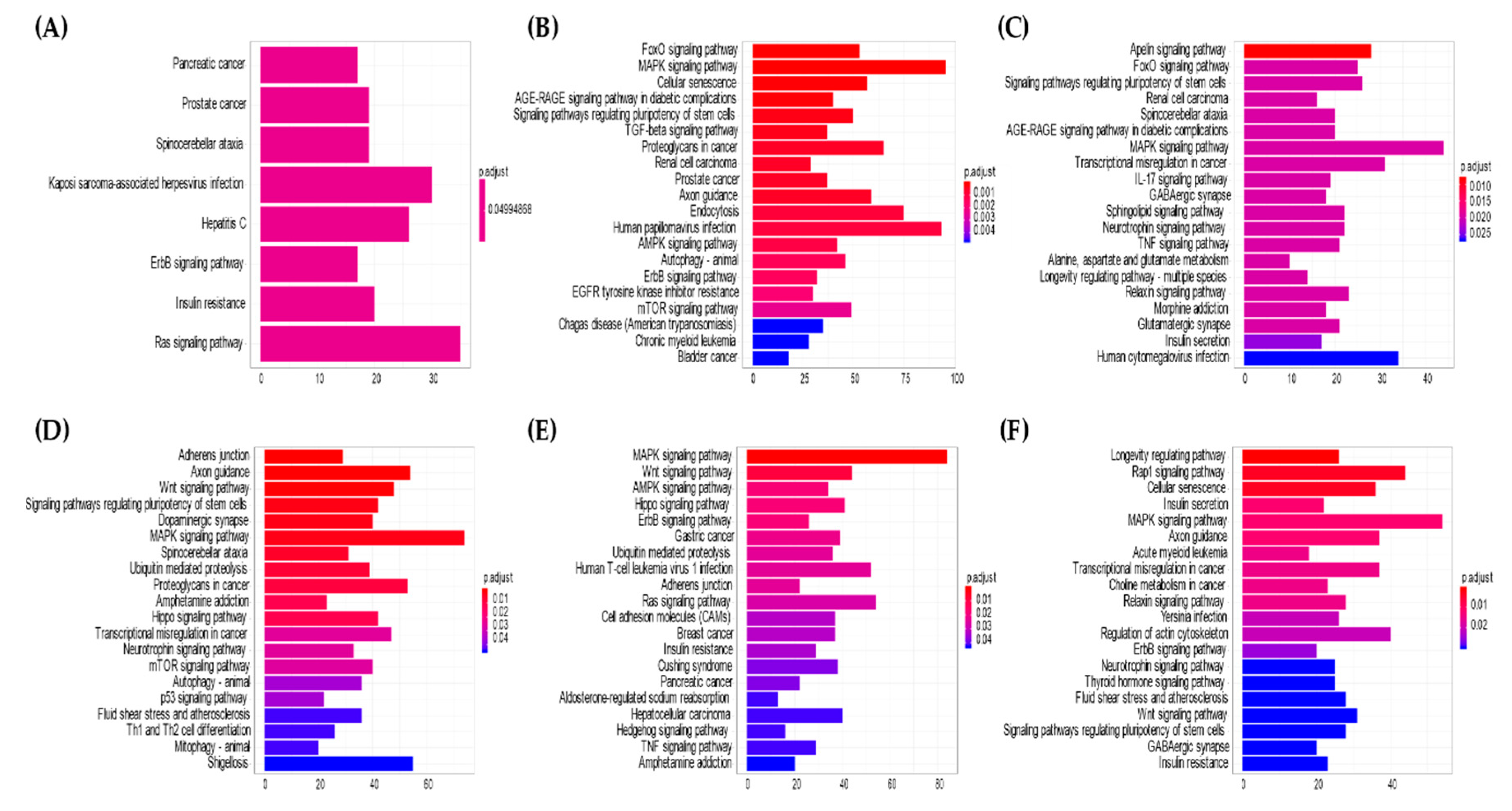
Pathways related to the top 10 downregulated miRNAs in the serum of IgG4-ROD patients compared with healthy individuals were mostly related to the FOXO signaling pathway, possibly due to its post-transcriptional regulation by miRNAs. Moreover, we observed downregulation of serum miRNAs that control the PI3K-AKT signaling pathway in IgG4-ROD. These signaling pathways are well-known, where the PI3K/AKT/FOXO pathway is involved in B-cell tumors, light chain recombination, receptor editing, and B-cell selection due to pathway constitutive activation [
39,
40]. Pathways related to the downregulated miRNAs in the serum of orbital MALT lymphoma patients compared with healthy individuals were mostly related to the adherens junction. This is a well-studied signaling pathway involved in cell adhesion of various tumors including lymphoma [
41,
42]. Furthermore, pathways related to miRNAs in biopsy specimens of IgG4-ROD patients compared with orbital MALT lymphoma were mostly related to the longevity-regulating pathway. To the best of our knowledge, this observation has not been reported previously.
Pathways related to the downregulated miRNAs in biopsy specimens of orbital MALT lymphoma patients compared with IgG4-ROD were mostly related to the MAPK signaling pathway. This signaling is a well-investigated pathway involved in inflammation due to regulation by the translocation gene of MALT lymphoma [
43]. The analysis of pathways enriched by the target genes of significantly expressed miRNAs confirms the essential role of these transcripts and their relative targeting miRNAs in the pathogenesis of IgG4-ROD and orbital MALT lymphoma.
No unique cytological or laboratory features in peripheral blood have been identified to assist the diagnosis of orbital lymphoproliferative disorders. Therefore, diagnostic biopsy is used to define and diagnose these disorders. Recently, miRNAs have been reported to be present in plasma at detectable levels, and plasma miRNAs are more stable than mRNAs in body fluids, resistant to degradation, and easily and rapidly measurable due to their small size and stem-loop structure [
44,
45,
46,
47,
48,
49]. Compared with invasive biopsy, the peripheral blood-based biomarker assay is a relatively economical and noninvasive method for the detection of IgG4-ROD and orbital MALT lymphoma, owing to its easy accessibility and the low risk associated with sample collection. However, circulating miRNAs may be affected by systemic functions, such as disease complications, physiological variation, concurrent drugs and food intake. The ROC curve analysis displayed that the levels of miR-4673 and miR-92a-2-5p in serum could discriminate with greater accuracy IgG4-ROD patients from orbital MALT lymphoma patients and healthy individuals, and that miR-7112-3p could discriminate orbital MALT lymphoma patients from IgG4-ROD patients and healthy individuals. Furthermore, analysis of multiple miRNAs selected by the randomForest method showed that five miRNAs (miR-1912, miR-202-3p, miR-320c, miR-361-5p, and miR-4755-3p) in serum could more accurately discriminate IgG4-ROD patients from orbital MALT lymphoma patients and healthy individuals. Three miRNAs (miR-4285, miR-5480-3p, and miR-7112-3p) could more accurately discriminate orbital MALT lymphoma patients from IgG4-ROD patients and healthy individuals. Moreover, these miRNA panels may exhibit higher performance than single miRNAs. To confirm these preliminary results, further evaluation is warranted. Clinically, discrimination between IgG4-ROD and orbital MALT lymphoma using serum miRNAs may support clinical decision-making and allow for the timely initiation of treatment, thereby avoiding the need for biopsy.
Recently, Blosse et al. showed that four novel miRNAs (miR-150, miR-155, miR-196a, and miR-138) played important regulatory roles in the pathogenesis of gastric MALT lymphoma caused by Helicobacter pylori infection [
50]. Consistent with the results reported for gastric MALT lymphoma, we found that miR-138 was expressed at higher levels in biopsy tissues of orbital MALT lymphoma patients compared with IgG4-ROD. However, similar results were not obtained for the other miRNAs. Orbital MALT lymphoma is rarely caused by Helicobacter pylori [
51,
52]. Therefore, the difference in miRNA profile between orbital MALT lymphoma and gastric MALT lymphoma may be attributed to the different characteristics of these two distinct types of MALT lymphoma and/or usage of different assays. Interestingly, the pathogenesis of gastric lymphoma may be associated with other risk factors, such as the hepatitis B virus, human immunodeficiency virus, Epstein–Barr virus, and human T-cell lymphotropic virus type 1 [
53]. In this study, nine (miR-3192-5p, miR-3190-3p, miR-5087, miR-601, miR-650, miR-3691-5p, miR-1255b-5p, miR-205-5p, and miR-6514-5p) of the top 10 miRNAs downregulated in biopsy specimens of orbital MALT lymphoma patients were compared with IgG4-ROD targeted genes related to the KEGG pathway of human T-cell leukemia virus 1 infection. The association of human T-cell leukemia virus 1 infection with orbital MALT lymphoma has not been reported. Further investigations are required to examine such a relationship.
To date, only two studies have reported miRNA profiles of orbital malignant lymphoma, including diffuse large B-cell lymphoma [
54,
55]. Recently, Laban et al. showed that seven (miR-140-5p, miR-148a-3p, miR193a-5p, miR-223-3p, miR-29a-3p, miR-365a-3p, and U6 snRNA) of 399 miRNAs analyzed in a panel were upregulated in the serum of patients with non-Hodgkin lymphomas, including diffuse large B-cell lymphoma. Due to differences in the study design, the disease and control characteristics, and the assay used, their data cannot be compared with our results.
The study is limited by the retrospective design and small number of cases collected from a single institution, which may cause selection bias and confounding bias. Furthermore, IgG4 levels are variable among IgG4-ROD patients. Owing to the lack of comparable studies of IgG4-ROD including IgG4-RD, it is difficult to make a direct comparison with previous studies. A prospective study to validate the present findings and examine the clinical significance of miRNAs in different manifestations of IgG4-ROD and orbital MALT lymphoma should be performed in the future using a large clinical samples from multiple centers, with the goals of diagnosing the clinical subtypes and elucidating the pathogenesis of these diseases.