Association between Left Atrial Deformation and Brain Involvement in Patients with Anderson-Fabry Disease at Diagnosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Echocardiography
2.3. Brain MRI Data Acquisition and Interpretation
- −
- deep white matter:
- (1)
- no WMLs;
- (2)
- punctate foci of hyperintensity;
- (3)
- beginning confluent WMLs;
- (4)
- large confluent WMLs;
- −
- periventricular white matter:
- (1)
- no WMLs;
- (2)
- periventricular caps;
- (3)
- smooth periventricular “halo”;
- (4)
- irregular periventricular WMLs extending into deep white matter.
2.4. Statistical Analysis
3. Results
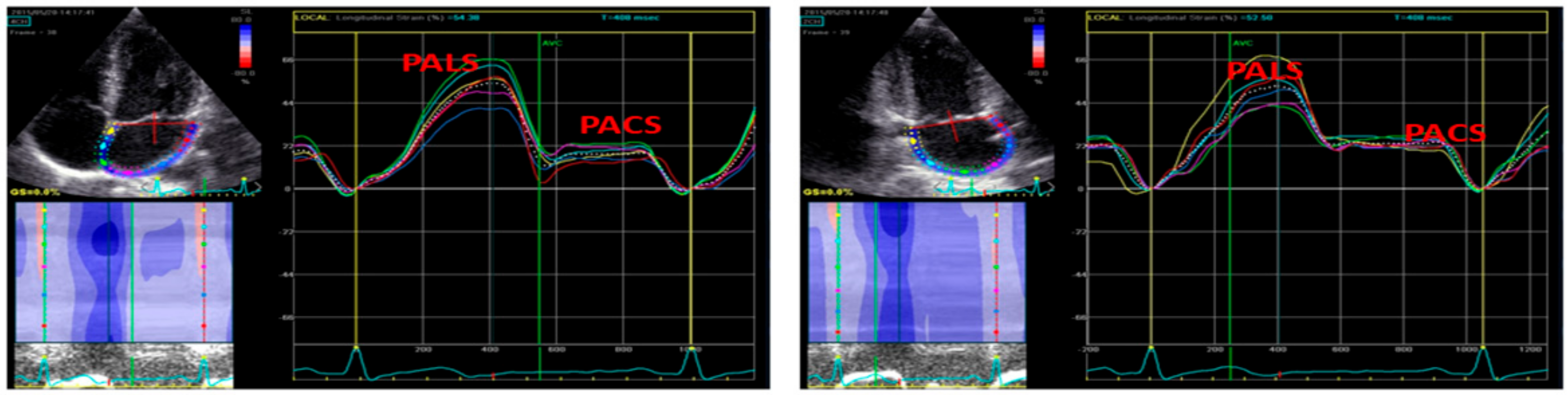
3.1. Standard and Speckle Tracking Echocardiography
3.2. Brain MRI
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Desnick, R.J.; Ioannou, Y.A.; Eng, C.M. α-Galactosidase A deficiency: Fabry disease. In The Metabolic and Molecular Bases of Inherited Disease, 8th ed.; Scriver, C.R., Beaudet, A.L., Sly, W.S., Valle, D., Eds.; McGraw Hill: New York, NY, USA, 2001; pp. 3733–3774. [Google Scholar]
- Weidemann, F.; Sanchez-Niño, M.D.; Politei, J.; Oliveira, J.P.; Wanner, C.; Warnock, D.G.; Ortiz, A. Fibrosis: A key feature of Fabry disease with potential therapeutic implications. Orphanet J. Rare Dis. 2013, 8, 116. [Google Scholar] [CrossRef] [Green Version]
- Patel, V.; O’Mahony, C.; Hughes, D.; Rahman, M.S.; Coats, C.; Murphy, E.; Lachmann, R.; Mehta, A.; Elliott, P.M. Clinical and genetic predictors of major cardiac events in patients with Anderson-Fabry disease. Heart 2015, 101, 961–966. [Google Scholar] [CrossRef]
- Rombach, S.M.; Smid, B.E.; Linthorst, G.E.; Dijkgraaf, M.G.; Hollak, C.E. Natural course of Fabry disease and the effectiveness of enzyme replacement therapy: A systematic review and meta-analysis: Effectiveness of ERT in different disease stages. J. Inherit. Metab. Dis. 2014, 37, 341–352. [Google Scholar] [CrossRef]
- Linhart, A.; Palecek, T.; Bultas, J.; Ferguson, J.J.; Hrudova, J.; Karetova, D.; Zeman, J.; Ledvinova, J.; Poupetova, H.; Elleder, M.; et al. New insights in cardiac structural changes in patients with Fabry’s disease. Am. Heart J. 2000, 139, 1101–1108. [Google Scholar] [CrossRef]
- Cocozza, S.; Russo, C.; Pontillo, G.; Pisani, A.; Brunetti, A. Neuroimaging in Fabry disease: Current knowledge and future directions. Insights Imaging 2018, 9, 1077–1088. [Google Scholar] [CrossRef] [Green Version]
- Wardlaw, J.M.; Smith, E.E.; Biessels, G.J.; Cordonnier, C.; Fazekas, F.; Frayne, R. Standards for Reporting Vascular changes on neuroimaging (STRIVE v1). Neuroimaging standards for research into small vessel disease and its contribution to ageing and neuro-degeneration. Lancet Neurol. 2013, 12, 822–838. [Google Scholar] [CrossRef] [Green Version]
- Kolodny, E.; Fellgiebel, A.; Hilz, M.J.; Sims, K.; Caruso, P.; Phan, T.G.; Politei, J.; Manara, R.; Burlina, A. Cerebrovascular involvement in Fabry disease: Current status of knowledge. Stroke 2015, 46, 302–313. [Google Scholar] [CrossRef]
- Fazekas, F.; Chawluk, J.B.; Alavi, A.; Hurtig, H.I.; Zimmerman, R.A. MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. Am. J. Roentgenol. 1987, 149, 351–356. [Google Scholar] [CrossRef] [Green Version]
- Lechner, H.; Schmidt, R.; Fazekas, F.; Koch, M.; Offenbacher, H.; Reinhart, B.; Grieshofer, P. White matter lesions on magnetic resonance imaging in a healthy elderly population: Correlations to vascular risk factors and carotid atherosclerosis. J. Stroke Cerebrovasc. Dis. 1994, 4, 224–228. [Google Scholar] [CrossRef]
- Fazekas, F.; Barkhof, F.; Wahlund, L.O.; Pantoni, L.; Erkinjuntti, T.; Scheltens, P.; Schmidt, R. CT and MRI rating of white matter lesions. Cerebrovasc. Dis. 2002, 13 (Suppl. S2), 31–36. [Google Scholar] [CrossRef]
- Vianna-Pinton, R.; Moreno, C.A.; Baxter, C.M.; Lee, K.S.; Tsang, T.S.; Appleton, C.P. Two-dimensional speckle-tracking echocardiography of the left atrium: Feasibility and regional contraction and relaxation differences in normal subjects. J. Am. Soc. Echocardiogr. 2009, 22, 299–305. [Google Scholar] [CrossRef]
- Sarvari, S.I.; Haugaa, K.H.; Stokke, T.M.; Ansari, H.Z.; Leren, I.S.; Hegbom, F.; Smiseth, O.A.; Edvardsen, T. Strain echocardiographic assessment of left atrial function predicts recurrence of atrial fibrillation. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 660–667. [Google Scholar] [CrossRef]
- Cameli, M.; Lisi, M.; Righini, F.M.; Massoni, A.; Natali, B.M.; Focardi, M.; Tacchini, D.; Geyer, A.; Curci, V.; Di Tommaso, C.; et al. Usefulness of atrial deformation analysis to predict left atrial fibrosis and endocardial thickness in patients undergoing mitral valve operations for severe mitral regurgitation secondary to mitral valve prolapse. Am. J. Cardiol. 2013, 111, 595–601. [Google Scholar] [CrossRef]
- Morris, D.A.; Blaschke, D.; Canaan-Kühl, S.; Krebs, A.; Knobloch, G.; Walter, T.C.; Haverkamp, W. Global cardiac alterations detected by speckle-tracking echocardiography in Fabry disease: Left ventricular, right ventricular, and left atrial dysfunction are common and linked to worse symptomatic status. Int. J. Cardiovasc. Imaging 2015, 31, 301–313. [Google Scholar] [CrossRef]
- Aerts, J.M.; Groener, J.E.; Kuiper, S.; Donker-Koopman, W.E.; Strijland, A.; Ottenhoff, R.; van Roomen, C.; Mirzaian, M.; Wijburg, F.A.; Linthorst, G.E.; et al. Elevated globotriaosylsphingosine is a hallmark of Fabry disease. Proc. Natl. Acad. Sci. USA 2008, 105, 2812–2817. [Google Scholar] [CrossRef] [Green Version]
- Galderisi, M.; Cosyns, B.; Edvardsen, T.; Cardim, N.; Delgado, V.; Di Salvo, G.; Donal, E.; Sade, L.E.; Ernande, L.; Garbi, M.; et al. Standardization of adult transthoracic echocardiography reporting in agreement with recent chamber quantification, diastolic function, and heart valve disease recommendations: An expert consensus document of the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2017, 18, 1301–1310. [Google Scholar] [CrossRef] [Green Version]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kusnetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 233–270. [Google Scholar] [CrossRef]
- De Simone, G.; Daniels, S.R.; Devereux, R.B.; Meyer, R.A.; Roman, M.J.; de Divitiis, O.; Alderman, M.H. Left ventricular mass and body size in normotensive children and adults: Assessment of allometric relations and impact of overweight. J. Am. Coll. Cardiol. 1992, 20, 251–260. [Google Scholar] [CrossRef] [Green Version]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F., III; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P.; et al. Recommendations for the evaluation of left ventricular diastolic function by Echocardiography: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2016, 17, 1321–1360. [Google Scholar] [CrossRef]
- Rudski, L.G.; Lai, W.W.; Afilalo, J.; Hua, L.; Handschumacher, M.D.; Chandrasekaran, K.; Solomon, S.D.; Louie, E.K.; Schiller, N.B. Guidelines for the echocardiographic assessment of the right heart in adults: A report from the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2010, 23, 685–713. [Google Scholar] [CrossRef]
- Lembo, M.; Esposito, R.; Lo Iudice, F.; Santoro, C.; Izzo, R.; De Luca, N.; Trimarco, B.; de Simone, G.; Galderisi, M. Impact of pulse pressure on left ventricular global longitudinal strain in normotensive and newly diagnosed, untreated hypertensive patients. J. Hypertens. 2016, 34, 1201–1207. [Google Scholar] [CrossRef]
- Alcidi, G.M.; Esposito, R.; Evola, V.; Santoro, C.; Lembo, M.; Sorrentino, R.; Lo ludice, F.; Borgia, F.; Novo, G.; Trimarco, B.; et al. Normal reference values of multilayer longitudinal strain according to age decades in a healthy population: A single-centre experience. Eur. Heart J. Cardiovasc. Imaging 2018, 19, 1390–1396. [Google Scholar] [CrossRef]
- Mor-Avi, V.; Lang, R.M.; Badano, L.P.; Belohlavek, M.; Cardim, N.M.; Derumeaux, G.; Galderisi, M.; Marwick, T.; Nagueh, S.F.; Sengupta, P.P.; et al. Current and evolving echocardiographic techniques for the quantitative evaluation of cardiac mechanics: ASE/EAE consensus statement on methodology and indications endorsed by the Japanese Society of Echocardiography. J. Am. Soc. Echocardiogr. 2011, 24, 277–313. [Google Scholar] [CrossRef]
- Cameli, M.; Caputo, M.; Mondillo, S.; Ballo, P.; Palmerini, E.; Lisi, M.; Marino, E.; Galderisi, M. Feasibility and reference values of left atrial longitudinal strain imaging by two-dimensional speckle tracking. Cardiovasc. Ultrasound 2009, 7, 6. [Google Scholar] [CrossRef] [Green Version]
- Cameli, M.; Lisi, M.; Mondillo, S.; Padeletti, M.; Ballo, P.; Tsioulpas, C.; Bernazzali, S.; Maccherini, M. Left atrial longitudinal strain by speckle tracking echocardiography correlates well with left ventricular filling pressures in patients with heart failure. Cardiovasc. Ultrasound 2010, 8, 14. [Google Scholar] [CrossRef] [Green Version]
- Fazekas, F.; Enzinger, C.; Schmidt, R.; Grittner, U.; Giese, A.K.; Hennerici, M.G.; Huber, R.; Jungehulsing, G.J.; Kaps, M.; Kessler, C.; et al. Brain magnetic resonance imaging findings fail to suspect Fabry disease in young patients with an acute cerebrovascular event. Stroke 2015, 46, 1548–1553. [Google Scholar] [CrossRef] [Green Version]
- Sugimoto, T.; Robinet, S.; Dulgheru, R.; Bernard, A.; Ilardi, F.; Contu, L. NORRE Study. Echocardiographic reference ranges for normal left atrial function parameters: Results from the EACVI NORRE study. Eur. Heart J. Cardiovasc. Imaging 2018, 19, 630–638. [Google Scholar] [CrossRef] [Green Version]
- Mondillo, S.; Cameli, M.; Caputo, M.L.; Lisi, M.; Palmerini, E.; Padeletti, M.; Ballo, P. Early detection of left atrial strain abnormalities by speckle-tracking in hypertensive and diabetic patients with normal left atrial size. J. Am. Soc. Echocardiogr. 2011, 24, 898–908. [Google Scholar] [CrossRef]
- Boyd, A.C.; Lo, Q.; Devine, K.; Tchan, M.C.; Sillence, D.O.; Sadick, N.; Richards, D.A.B.; Thomas, L. Left atrial enlargement and reduced atrial compliance occurs early in Fabry cardiomyopathy. J. Am. Soc. Echocardiogr. 2013, 26, 1415–1423. [Google Scholar] [CrossRef]
- Pichette, M.; Serri, K.; Pagè, M.; Zhao, L.; Bichet, D.G.; Poulin, F. Impaired left atrial function in Fabry disease: A longitudinal Speckle-Tracking Echocardiography study. J. Am. Soc. Echocardiogr. 2017, 30, 170–179. [Google Scholar] [CrossRef]
- Putko, B.N.; Yogasundaram, H.; Chow, K.; Pagano, J.; Khan, A.; Paterson, D.I.; Thompson, R.B.; Oudit, G.Y. Normal left-atrial structure and function despite concentric left-ventricular remodelling in a cohort of patients with Anderson-Fabry disease. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Kuppahally, S.S.; Akoum, N.; Burgon, N.S.; Badger, T.J.; Kholmovski, E.G.; Vijayakumar, S.; Rao, S.N.; Blauer, J.; Fish, E.N.; DiBella, E.V.R.; et al. Left atrial strain and strain rate in patients with paroxysmal and persistent atrial fibrillation: Relationship to left atrial structural remodeling detected by delayed-enhancement MRI. Circ. Cardiovasc. Imaging 2010, 3, 231–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chimenti, C.; Russo, M.A.; Frustaci, A. Atrial biopsy evidence of Fabry disease causing lone atrial fibrillation. Heart 2010, 96, 1782–1783. [Google Scholar] [CrossRef] [PubMed]
- Cocozza, S.; Olivo, G.; Riccio, E.; Russo, C.; Pontillo, G.; Ugga, L.; Migliaccio, S.; de Rosa, D.; Feriozzi, S.; Veroux, M.; et al. Corpus callosum involvement: A useful clue for differentiating Fabry Disease from Multiple Sclerosis. Neuroradiology 2017, 59, 563–570. [Google Scholar] [CrossRef]
- Ugga, L.; Cocozza, S.; Pontillo, G.; Russo, C.; Brescia Morra, V.; Lanzillo, R.; Riccio, E.; Pisani, A.; Brunetti, A. Absence of infratentorial lesions in Fabry disease contributes to differential diagnosis with multiple sclerosis. Brain Behav. 2018, 8, e01121. [Google Scholar] [CrossRef] [Green Version]
- Liu, D.; Hu, K.; Schmidt, M.; Müntze, J.; Maniuc, O.; Gensler, D.; Oder, D.; Salinger, T.; Weidemann, F.; Ertl, G.; et al. Value of the CHA(2)DS(2)-VASc score and Fabry-specific score for predicting new-onset or recurrent stroke/TIA in Fabry disease patients without atrial fibrillation. Clin. Res. Cardiol. 2018, 107, 1111–1121. [Google Scholar] [CrossRef]
- Segura, T.; Ayo-Martín, O.; Gómez-Fernandez, I.; Andrés, C.; Barba, M.A.; Vivancos, J. Cerebral hemodynamics and endothelial function in patients with Fabry disease. BMC Neurol. 2013, 13, 170. [Google Scholar] [CrossRef] [Green Version]
- Schiffmann, R.; Warnock, D.G.; Banikazemi, M.; Bultas, J.; Linthorst, G.E.; Packman, S.; Sorensen, S.A.; Wilcox, W.R.; Desnick, R.J. Fabry disease: Progression of nephropathy, and prevalence of cardiac and cerebrovascular events before enzyme replacement therapy. Nephrol. Dial. Transplant. 2009, 24, 2102–2111. [Google Scholar] [CrossRef]
- Kaffashian, S.; Tzourio, C.; Zhu, Y.C.; Mazoyer, B.; Debette, S. Differential effect of white-matter lesions and covert brain infarcts on the risk of ischemic stroke and intracerebral hemorrhage. Stroke 2016, 47, 1923–1925. [Google Scholar] [CrossRef] [Green Version]
- Ortiz, A.; Germain, D.P.; Desnick, R.J.; Politei, J.; Mauer, M.; Burlina, A.; Eng, C.; Hopkin, R.J.; Laney, D.; Linhart, A.; et al. Fabry disease revisited: Management and treatment recommendations for adult patients. Mol. Genet. Metab. 2018, 123, 416–427. [Google Scholar] [CrossRef]
- Biegstraaten, M.; Arngrimsson, R.; Barbey, F.; Boks, L.; Cecchi, F.; Deegan, P.B.; Feldt–Rasmussen, U.; Gaberhiwot, T.; Germain, D.P.; Hendriksz, C.; et al. Recommendations for initiation and cessation of enzyme replacement therapy in patients with Fabry disease: The European Fabry Working Group consensus document. Orphanet J. Rare Dis. 2015, 10, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fellgiebel, A.; Gartenschläger, M.; Wildberger, K.; Scheurich, A.; Desnick, R.J.; Sims, K. Enzyme replacement therapy stabilized white matter lesion progression in Fabry disease. Cerebrovasc. Dis. 2014, 38, 448–456. [Google Scholar] [CrossRef]



| Characteristics | AFD (n = 22) | Controls (n = 22) | p-Value |
|---|---|---|---|
| Gender (M/F) | 14/8 | 14/8 | 1.00 |
| Age (years) | 39.0 ± 14.6 | 39.1 ± 14.4 | 0.99 |
| BMI (Kg/m2) | 26.1 ± 4.3 | 23.6 ± 2.5 | <0.05 |
| Systolic BP (mmHg) | 124.7 ± 17.8 | 118.4 ± 8.8 | 0.14 |
| Diastolic BP (mmHg) | 76.7 ± 10.8 | 74.3 ± 6.9 | 0.37 |
| HR (bpm) | 71.4 ± 9.8 | 73.2 ± 7.6 | 0.56 |
| Characteristics | AFD (n = 22) | Controls (n = 22) | p Value |
|---|---|---|---|
| LVMi(g/m2.7) | 37.0 ± 12.5 | 27.0 ± 4.6 | <0.002 |
| RWT | 0.34 ± 0.6 | 0.29 ± 0.3 | <0.001 |
| LV EF (%) | 64.1 ± 3.8 | 62.6 ± 3.8 | 0.19 |
| GLS (%) | 21.9 ± 1.9 | 23.3 ± 1.7 | <0.01 |
| Transmitral E/A ratio | 1.4 ± 0.44 | 1.3 ± 0.32 | 0.21 |
| E velocity DT (msec) | 202.5 ± 44.5 | 202.1 ± 37.1 | 0.96 |
| E’ velocity (cm/sec) | 0.78 ± 0.21 | 0.80 ± 0.21 | 0.64 |
| E/e’ ratio | 6.4 ± 1.2 | 5.6 ± 1.1 | 0.22 |
| PASP (mmHg) | 28.4 ± 6.2 | 26.2± 4.5 | 0.20 |
| LAVi (ml/m2) | 29.3 ± 8.6 | 25.3 ± 5.3 | 0.07 |
| PALS | 32.6 ± 6.9 | 42.2 ± 6.6 | <0.0001 |
| PACS | 13.7 ± 3.7 | 16.3 ± 4.0 | 0.25 |
| Variables | FS ≥ 1 (n = 13) | FS < 1 (n = 9) | p-Value |
|---|---|---|---|
| Age (years) | 43.3 ± 13.1 | 32.7 ± 10.3 | 0.09 |
| LVMi (g/m2.7) | 40.5 ± 12.5 | 31.9 ± 4.6 | 0.12 |
| LV EF (%) | 64.3 ± 03.5 | 63.6 ± 4.5 | 0.69 |
| GLS (%) | 22.3 ± 2.1 | 21.1 ± 1.5 | 0.17 |
| Transmitral E/A ratio | 1.5 ± 0.21 | 1.3 ± 0.54 | 0.14 |
| E velocity DT (msec) | 187.3 ± 39.5 | 194.6 ± 55.5 | 0.70 |
| E’ velocity (cmsec) | 0.70 ± 1.5 | 0.78 ± 0.8 | 0.09 |
| E/e’ ratio | 5.9 ± 1.1 | 5.1 ± 0.9 | 0.08 |
| RWT | 0.36 ± 0.6 | 0.32 ± 0.5 | 0.23 |
| LAVi (mL/m2) | 31.4 ± 10.0 | 26.2 ± 5.2 | 0.17 |
| PALS | 29.4 ± 6.7 | 37.4 ± 3.9 | 0.0003 |
| PACS | 13.9 ± 2.8 | 13.5 ± 4.2 | 0.80 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esposito, R.; Russo, C.; Santoro, C.; Cocozza, S.; Riccio, E.; Sorrentino, R.; Pontillo, G.; Luciano, F.; Imbriaco, M.; Brunetti, A.; et al. Association between Left Atrial Deformation and Brain Involvement in Patients with Anderson-Fabry Disease at Diagnosis. J. Clin. Med. 2020, 9, 2741. https://doi.org/10.3390/jcm9092741
Esposito R, Russo C, Santoro C, Cocozza S, Riccio E, Sorrentino R, Pontillo G, Luciano F, Imbriaco M, Brunetti A, et al. Association between Left Atrial Deformation and Brain Involvement in Patients with Anderson-Fabry Disease at Diagnosis. Journal of Clinical Medicine. 2020; 9(9):2741. https://doi.org/10.3390/jcm9092741
Chicago/Turabian StyleEsposito, Roberta, Camilla Russo, Ciro Santoro, Sirio Cocozza, Eleonora Riccio, Regina Sorrentino, Giuseppe Pontillo, Federica Luciano, Massimo Imbriaco, Arturo Brunetti, and et al. 2020. "Association between Left Atrial Deformation and Brain Involvement in Patients with Anderson-Fabry Disease at Diagnosis" Journal of Clinical Medicine 9, no. 9: 2741. https://doi.org/10.3390/jcm9092741





