Progress and Prospect of Breeding Utilization of Green Revolution Gene SD1 in Rice
Abstract
:1. Introduction
2. History of sd1 Utilization in Rice Semi-Dwarf Breeding
3. The Main Variation Types of SD1 Allele Used in Rice Semi-Dwarf Breeding
4. Advantages of Semi-Dwarf Gene sd1 in Rice Breeding
5. Contribution of SD1 to Biological Studies in Rice
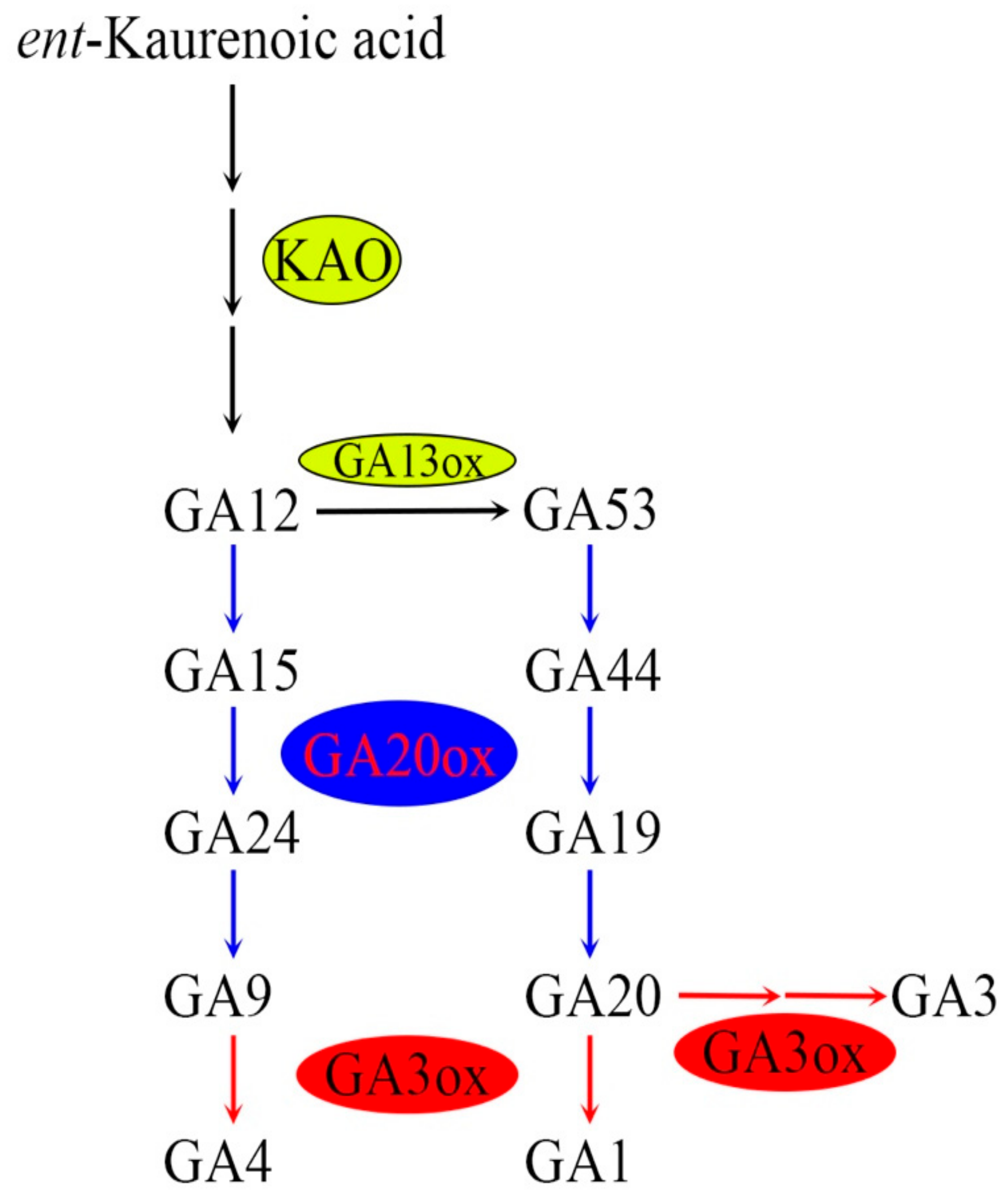
5.1. Main Biological Functions of SD1 in Gibberellin Biosynthesis
5.2. Molecular Regulation Mechanism of SD1 in Gibberellin Biosynthesis
5.3. Molecular Regulated Mechanism of SD1 in Rice Breeding
6. Prospects of Utilization of sd1 in Rice Breeding
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khan, M.H.; Dar, Z.A.; Dar, S.A. Breeding Strategies for Improving Rice Yield—A Review. Agric. Sci. 2015, 6, 467–478. [Google Scholar] [CrossRef] [Green Version]
- Briney, A. History and Overview of the Green Revolution. ThoughtCo. Available online: https://www.thoughtco.com/green-revolution-overview-1434948 (accessed on 27 August 2020).
- Gaud, W.S. The Green Revolution: Accomplishments and Apprehensions. AgBioWorld. Available online: http://www.agbioworld.org/biotech-info/topics/borlaug/borlaug-green.html (accessed on 8 March 2021).
- Khush, G.S. Modern varieties—Their real contribution to food supply and equity. GeoJournal 1995, 35, 275–284. [Google Scholar] [CrossRef]
- Peng, J.; Richards, D.E.; Hartley, N.M.; Murphy, G.P.; Devos, K.M.; Flintham, J.E.; Beales, J.; Fish, L.J.; Worland, A.J.; Pelica, F.; et al. ‘Green revolution’ genes encode mutant gibberellin response modulators. Nature 1999, 400, 256–261. [Google Scholar] [CrossRef]
- Peng, S.; Cassman, K.G.; Virmani, S.S.; Sheehy, J.; Khush, G.S. Yield Potential Trends of Tropical Rice since the Release of IR8 and the Challenge of Increasing Rice Yield Potential. Crop Sci. 1999, 39, 1552–1559. [Google Scholar] [CrossRef] [Green Version]
- Monna, L.; Kitazawa, N.; Yoshino, R.; Suzuki, J.; Masuda, H.; Maehara, Y.; Tanji, M.; Sato, M.; Nasu, S.; Minobe, Y. Positional cloning of rice semidwarfing gene, sd-1: Rice “green revolution gene” encodes a mutant enzyme involved in gibberellin synthesis. DNA Res. 2002, 9, 11–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sasaki, A.; Ashikari, M.; Ueguchi-Tanaka, M.; Itoh, H.; Nishimura, A.; Swapan, D.; Ishiyama, K.; Saito, T.; Kobayashi, M.; Khush, G.S.; et al. Green revolution: A mutant gibberellin-synthesis gene in rice. Nature 2002, 416, 701–702. [Google Scholar] [CrossRef]
- Spielmeyer, W.; Ellis, M.H.; Chandler, P.M. Semidwarf (sd-1), “green revolution” rice, contains a defective gibberellin 20-oxidase gene. Proc. Natl. Acad. Sci. USA 2002, 99, 9043–9048. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Wu, Y.; Zeng, X.; Yuan, L. Present Situation of Utilization on Rice Dwarf Gene Resources and Its Research Advances in Molecular Biology. Hunan Agric. Sci. 2007, 20–24. [Google Scholar]
- Xie, W.; Wang, G.; Yuan, M.; Yao, W.; Lyu, K.; Zhao, H.; Yang, M.; Li, P.; Zhang, X.; Yuan, J.; et al. Breeding signatures of rice improvement revealed by a genomic variation map from a large germplasm collection. Proc. Natl. Acad. Sci. USA 2015, 112, E5411–E5419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khush, G.S. Green revolution: The way forward. Nat. Rev. Genet. 2001, 2, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Aquino, R.C.; Jennings, P.R. Inheritance and Significance of Dwarfism in an Indica Rice Variety. Crop Sci. 1966, 6, 551–554. [Google Scholar] [CrossRef]
- Tang, S.; Wang, X.; Liu, X. Study on the Renewed Tendency and Key Backbone-Parents of Inbred Rice Varieties (O. sativa L.) in China. Sci. Agric. Sin. 2012, 45, 1455–1464. [Google Scholar] [CrossRef]
- Gu, M. Dwarf source and its utilization in rice breeding. J. Jiangsu Agric. Univ. 1980, 40–44. [Google Scholar]
- Hargrove, T.R.; Cabanilla, V.L. The Impact of Semidwarf Varieties on Asian Rice-Breeding Programs. Bioscience 1979, 29, 731–735. [Google Scholar] [CrossRef]
- Futsuhara, Y.; Toriyama, K.; Tsunoda, K. Breeding of a new rice variety Reimei by gamma-ray irradiation. Gamma Field Symp. 1967, 17, 85–90. [Google Scholar] [CrossRef] [Green Version]
- Dalrymple, D.G. Development and Spread of High-Yielding Rice Varieties in Developing Countries; Oxford University Press: Oxford, UK, 1986; Volume 67, p. 117. [Google Scholar]
- Khush, G.S. Green revolution: Preparing for the 21st century. Genome 1999, 42, 646–655. [Google Scholar] [CrossRef]
- Angira, B.; Addison, C.K.; Cerioli, T.; Rebong, D.B.; Wang, D.R.; Pumplin, N.; Ham, J.H.; Oard, J.H.; Linscombe, S.D.; Famoso, A.N. Haplotype Characterization of the sd1 Semidwarf Gene in United States Rice. Plant Genome 2019, 12, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Asano, K.; Takashi, T.; Miura, K.; Qian, Q.; Kitano, H.; Matsuoka, M.; Ashikari, M. Genetic and Molecular Analysis of Utility of sd1 Alleles in Rice Breeding. Breed. Sci. 2007, 57, 53–58. [Google Scholar] [CrossRef] [Green Version]
- Asano, K.; Yamasaki, M.; Takuno, S.; Miura, K.; Katagiri, S.; Ito, T.; Doi, K.; Wu, J.; Ebana, K.; Matsumoto, T.; et al. Artificial selection for a green revolution gene during japonica rice domestication. Proc. Natl. Acad. Sci. USA 2011, 108, 11034–11039. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Hu, X.; Zhu, Y.; Mao, D. Re-evaluation of the rice ‘Green Revolution’ gene: The weak allele SD1-EQ from japonica rice may be beneficial for super indica rice breeding in the post-Green Revolution era. Mol. Breed. 2020, 40, 84. [Google Scholar] [CrossRef]
- Tomita, M.; Ishii, K. Genetic Performance of the Semidwarfing Allele sd1 Derived from a Japonica Rice Cultivar and Minimum Requirements to Detect Its Single-Nucleotide Polymorphism by MiSeq Whole-Genome Sequencing. BioMed Res. Int. 2018, 2018, 4241725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuroha, T.; Nagai, K.; Gamuyao, R.; Wang, D.R.; Furuta, T.; Nakamori, M.; Kitaoka, T.; Adachi, K.; Minami, A.; Mori, Y.; et al. Ethylene-gibberellin signaling underlies adaptation of rice to periodic flooding. Science 2018, 361, 181–186. [Google Scholar] [CrossRef] [Green Version]
- Bhuvaneswari, S.; Gopala Krishnan, S.; Ellur, R.K.; Vinod, K.K.; Bollinedi, H.; Bhowmick, P.K.; Bansal, V.P.; Nagarajan, M.; Singh, A.K. Discovery of a Novel Induced Polymorphism in SD1 Gene Governing Semi-Dwarfism in Rice and Development of a Functional Marker for Marker-Assisted Selection. Plants 2020, 9, 1198. [Google Scholar] [CrossRef]
- Biswas, S.; Tian, J.; Li, R.; Chen, X.; Luo, Z.; Chen, M.; Zhao, X.; Zhang, D.; Persson, S.; Yuan, Z.; et al. Investigation of CRISPR/Cas9-induced SD1 rice mutants highlights the importance of molecular characterization in plant molecular breeding. J. Genet. Genom. 2020, 47, 273–280. [Google Scholar] [CrossRef]
- Hu, X.; Cui, Y.; Dong, G.; Feng, A.; Wang, D.; Zhao, C.; Zhang, Y.; Hu, J.; Zeng, D.; Guo, L.; et al. Using CRISPR-Cas9 to generate semi-dwarf rice lines in elite landraces. Sci. Rep. 2019, 9, 19096. [Google Scholar] [CrossRef] [Green Version]
- Ashikari, M.; Sasaki, A.; Ueguchi-Tanaka, M.; Itoh, H.; Nishimura, A.; Datta, S.; Ishiyama, K.; Saito, T.; Kobayashi, M.; Khush, G.S.; et al. Loss-of-function of a Rice Gibberellin Biosynthetic Gene, GA20 oxidase (GA20ox-2), Led to the Rice “Green Revolution”. Breed. Sci. 2002, 52, 143–150. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Wei, H.; Zhang, H.; Zhang, W.-H. Enhanced accumulation of gibberellins rendered rice seedlings sensitive to ammonium toxicity. J. Exp. Bot. 2020, 71, 1514–1526. [Google Scholar] [CrossRef]
- Li, S.; Tian, Y.; Wu, K.; Ye, Y.; Yu, J.; Zhang, J.; Liu, Q.; Hu, M.; Li, H.; Tong, Y.; et al. Modulating plant growth–metabolism coordination for sustainable agriculture. Nature 2018, 560, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Huang, J.; Wang, Y.; Xu, R.; Yang, Z.; Zhao, Z.; Liu, S.; Tian, Y.; Zheng, X.; Li, F.; et al. Identification and genetic analysis of qCL1.2, a novel allele of the “green revolution” gene SD1 from wild rice (Oryza rufipogon) that enhances plant height. BMC Genet. 2020, 21, 62. [Google Scholar] [CrossRef]
- Gu, M.; Pan, X.; Li, X. Genetic Analysis of the Pedigrees of the Improved Cultivars in Oryza sativa L. subsp. hsien in South China. Sci. Agric. Sin. 1986, 19, 41–48. Available online: https://www.chinaagrisci.com/CN/Y1986/V19/I01/41 (accessed on 8 March 2021).
- Shi, C.; Shen, Z. Effects of semidwarf gene sd1 on agronomic traits in rice (Oryza sativa subsp. indica). Chin. J. Rice Sci. 1996, 10, 13–18. Available online: http://www.ricesci.cn/EN/abstract/abstract1394.shtml (accessed on 8 March 2021).
- Chang, T.T. Semidwarf in rice germplasm collections and their potentials in rice improvement. Phytobreedon 1985, 1, 1–9. [Google Scholar]
- Mackill, D.J.; Rutger, J.N. The inheritance of induced-mutant semidwarfing genes in rice. J. Hered. 1979, 70, 335–341. [Google Scholar] [CrossRef]
- Lin, H.; Xiong, Z.; Min, S.; Wu, M.; Yu, G. The Location of the Dwarf Gene in Rice Variety Xueheaizao. Chin. J. Rice Sci. 1993, 7, 17–20. [Google Scholar]
- Qian, Q.; Guo, L.; Smith, S.M.; Li, J. Breeding high-yield superior quality hybrid super rice by rational design. Natl. Sci. Rev. 2016, 3, 283–294. [Google Scholar] [CrossRef] [Green Version]
- Zeng, D.; Tian, Z.; Rao, Y.; Dong, G.; Yang, Y.; Huang, L.; Leng, Y.; Xu, J.; Sun, C.; Zhang, G.; et al. Rational design of high-yield and superior-quality rice. Nat. Plants 2017, 3, 17031. [Google Scholar] [CrossRef]
- Qian, Q.; Cheng, S. Rice Genetics and Functional Genomics; Science Press: Beijing, China, 2006. [Google Scholar]
- Yamaguchi, S. Gibberellin metabolism and its regulation. Annu. Rev. Plant Biol. 2008, 59, 225–251. [Google Scholar] [CrossRef]
- Magome, H.; Nomura, T.; Hanada, A.; Takeda-Kamiya, N.; Ohnishi, T.; Shinma, Y.; Katsumata, T.; Kawaide, H.; Kamiya, Y.; Yamaguchi, S. CYP714B1 and CYP714B2 encode gibberellin 13-oxidases that reduce gibberellin activity in rice. Proc. Natl. Acad. Sci. USA 2013, 110, 1947–1952. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, F.; Zhu, B. Evolutionary analysis of three gibberellin oxidase genesin rice, Arabidopsis, and soybean. Gene 2011, 473, 23–35. [Google Scholar] [CrossRef]
- Oikawa, T.; Koshioka, M.; Kojima, K.; Yoshida, H.; Kawata, M. A role of OsGA20ox1, encoding an isoform of gibberellin 20-oxidase, for regulation of plant stature in rice. Plant Mol. Biol. 2004, 55, 687–700. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Liu, J.H.; Zhao, W.S.; Chen, X.J.; Guo, Z.J.; Peng, Y.L. Gibberellin 20-oxidase gene OsGA20ox3 regulates plant stature and disease development in rice. Mol. Plant Microbe Interact. MPMI 2013, 26, 227–239. [Google Scholar] [CrossRef] [Green Version]
- Sakamoto, T.; Miura, K.; Itoh, H.; Tatsumi, T.; Ueguchi-Tanaka, M.; Ishiyama, K.; Kobayashi, M.; Agrawal, G.K.; Takeda, S.; Abe, K.; et al. An overview of gibberellin metabolism enzyme genes and their related mutants in rice. Plant Physiol. 2004, 134, 1642–1653. [Google Scholar] [CrossRef] [Green Version]
- Ma, Q.; Hedden, P.; Zhang, Q. Heterosis in rice seedlings: Its relationship to gibberellin content and expression of gibberellin metabolism and signaling genes. Plant Physiol. 2011, 156, 1905–1920. [Google Scholar] [CrossRef] [Green Version]
- Ye, H.; Feng, J.; Zhang, L.; Zhang, J.; Mispan, M.S.; Cao, Z.; Beighley, D.H.; Yang, J.; Gu, X.Y. Map-Based Cloning of Seed Dormancy1-2 Identified a Gibberellin Synthesis Gene Regulating the Development of Endosperm-Imposed Dormancy in Rice. Plant Physiol. 2015, 169, 2152–2165. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Wang, Y.; Mi, X.F.; Shan, J.X.; Li, X.M.; Xu, J.L.; Lin, H.X. The QTL GNP1 Encodes GA20ox1, Which Increases Grain Number and Yield by Increasing Cytokinin Activity in Rice Panicle Meristems. PLoS Genet. 2016, 12, e1006386. [Google Scholar] [CrossRef]
- Yano, K.; Takashi, T.; Nagamatsu, S.; Kojima, M.; Sakakibara, H.; Kitano, H.; Matsuoka, M.; Aya, K. Efficacy of microarray profiling data combined with QTL mapping for the identification of a QTL gene controlling the initial growth rate in rice. Plant Cell Physiol. 2012, 53, 729–739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakata, T.; Oda, S.; Tsunaga, Y.; Shomura, H.; KawagishiKobayashi, M.; Aya, K.; Saeki, K.; Endo, T.; Nagano, K.; Kojima, M. Reduction of gibberellin by low temperature disrupts pollen development in rice. Plant Physiol. 2014, 164, 2011. [Google Scholar] [CrossRef] [Green Version]
- Fukazawa, J.; Mori, M.; Watanabe, S.; Miyamoto, C.; Ito, T.; Takahashi, Y. DELLA-GAF1 Complex Is a Main Component in Gibberellin Feedback Regulation of GA20 Oxidase 2. Plant Physiol. 2017, 175, 1395–1406. [Google Scholar] [CrossRef] [Green Version]
- Kaneko, M.; Itoh, H.; Inukai, Y.; Sakamoto, T.; Ueguchi-Tanaka, M.; Ashikari, M.; Matsuoka, M. Where do gibberellin biosynthesis and gibberellin signaling occur in rice plants? Plant J. Cell Mol. Biol. 2003, 35, 104–115. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.L.; Li, L.; Wu, K.; Peeters, A.J.; Gage, D.A.; Zeevaart, J.A. The GA5 locus of Arabidopsis thaliana encodes a multifunctional gibberellin 20-oxidase: Molecular cloning and functional expression. Proc. Natl. Acad. Sci. USA 1995, 92, 6640–6644. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Y.L.; Gage, D.A.; Zeevaart, J.A. Gibberellins and stem growth in Arabidopsis thaliana. Effects of photoperiod on expression of the GA4 and GA5 loci. Plant Physiol. 1997, 114, 1471–1476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, D.J.; Zeevaart, J.A. Regulation of gibberellin 20-oxidase1 expression in spinach by photoperiod. Planta 2007, 226, 35–44. [Google Scholar] [CrossRef]
- Wu, K.; Li, L.; Gage, D.A.; Zeevaart, J.A. Molecular cloning and photoperiod-regulated expression of gibberellin 20-oxidase from the long-day plant spinach. Plant Physiol. 1996, 110, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Duan, M.; Ke, X.-J.; Lan, H.-X.; Yuan, X.; Huang, P.; Xu, E.-S.; Gao, X.-Y.; Wang, R.-Q.; Tang, H.-J.; Zhang, H.-S.; et al. A Cys2/His2 Zinc Finger Protein Acts as a Repressor of the Green Revolution Gene SD1/OsGA20ox2 in Rice (Oryza sativa L.). Plant Cell Physiol. 2021, 61, 2055–2066. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shang, L.; Yu, H.; Zeng, L.; Hu, J.; Ni, S.; Rao, Y.; Li, S.; Chu, J.; Meng, X.; et al. A Strigolactone Biosynthesis Gene Contributed to the Green Revolution in Rice. Mol. Plant 2020, 13, 923–932. [Google Scholar] [CrossRef]
- Huang, L.J.; Luo, J.; Wang, Y.; Li, N. From Green Revolution to Green Balance: The Nitrogen and Gibberellin Mediated Rice Tiller Growth. Plant Signal. Behav. 2021, 16, 1917838. [Google Scholar] [CrossRef] [PubMed]
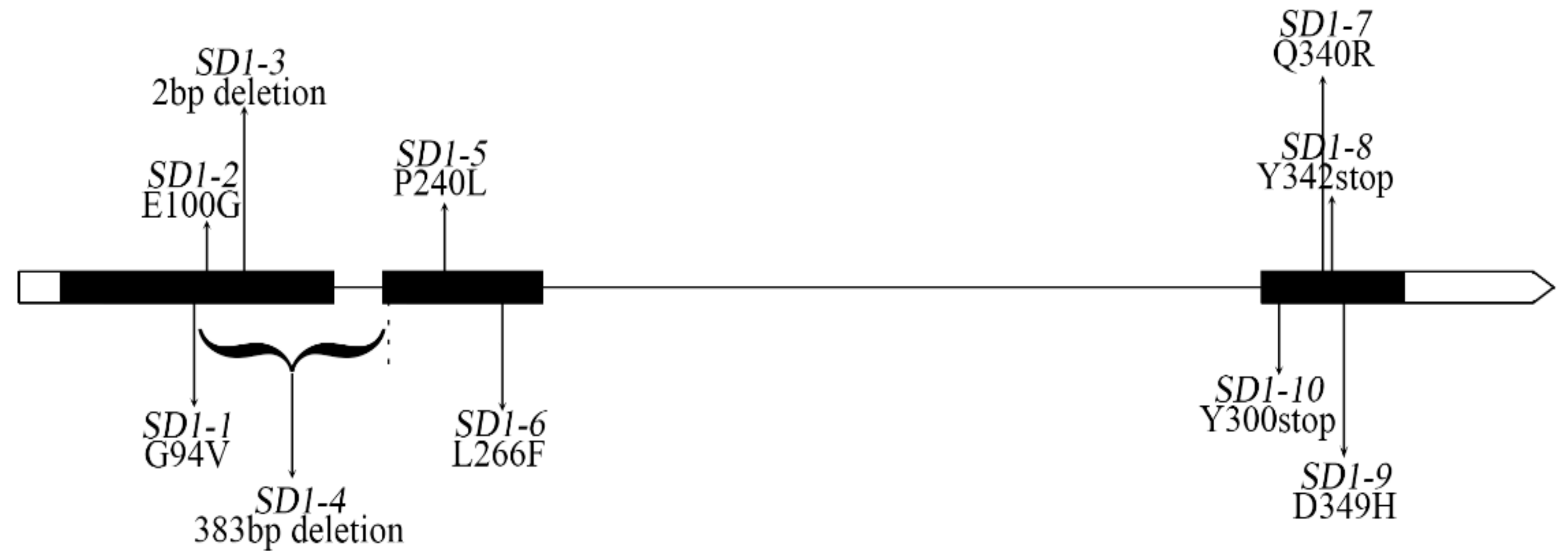
| SD1 Allele | Mutation Site in cDNA | Amino Acid Change | Represent Variety | Ref |
|---|---|---|---|---|
| SD1-1 | G281T | G94V | Jikkoku | [21,29] |
| SD1-2 | G99A | G100E | Nipponbare, Pusa1652 | [22,26] |
| SD1-3 | C382 and G383 deletion | premature stop | Ai-Jiao-Nan-Te | [21] |
| SD1-4 | deletion of 381bp in exon1 and 2bp in exon2 | premature stop | Deo-geo-woo-gen, IR8, Habataki, Minghui 63, PA64s | [9,21,30,31] |
| SD1-5 | C719T | P240L | Zhai-ye-qing 8 | [21] |
| SD1-6 | C796T | L266F | Calrose76 | [21,29] |
| SD1-7 | G1019A | R340Q | Nipponbare | [22] |
| SD1-8 | C1026G | Y342stop | 93-11 | [21,32] |
| SD1-9 | G1045C | D349H | Reimei | [21,29] |
| SD1-10 | T900A | Y300stop | Pusa1652 | [26] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, Y.; Hu, Y.; Qian, Q.; Ren, D. Progress and Prospect of Breeding Utilization of Green Revolution Gene SD1 in Rice. Agriculture 2021, 11, 611. https://doi.org/10.3390/agriculture11070611
Peng Y, Hu Y, Qian Q, Ren D. Progress and Prospect of Breeding Utilization of Green Revolution Gene SD1 in Rice. Agriculture. 2021; 11(7):611. https://doi.org/10.3390/agriculture11070611
Chicago/Turabian StylePeng, Youlin, Yungao Hu, Qian Qian, and Deyong Ren. 2021. "Progress and Prospect of Breeding Utilization of Green Revolution Gene SD1 in Rice" Agriculture 11, no. 7: 611. https://doi.org/10.3390/agriculture11070611






