Advances in Crop Genetic Improvement to Overcome Drought Stress: Bibliometric and Meta-Analysis
Abstract
:1. Introduction
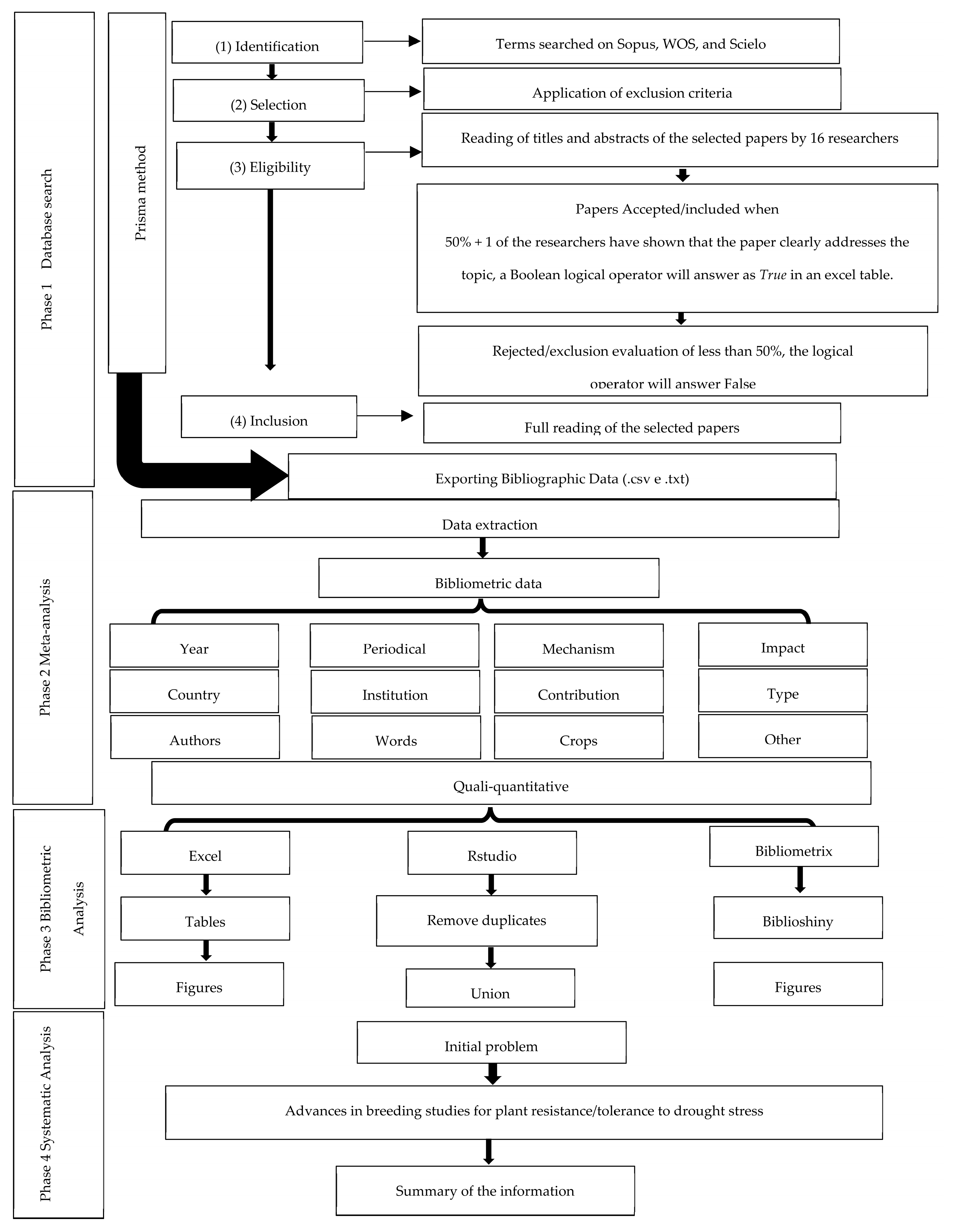
2. Materials and Methods
2.1. Study Phases
2.2. Search Bases and Strings
2.3. Meta-Analysis
2.4. Bibliometric Analysis
2.5. Systematic Analysis
3. Results
3.1. Bibliometric Analysis
Scientific Production per Country and Affiliation
3.2. Systematic Review
Keywords of Highest Occurrence and Co-Occurrence Network
4. Discussion
4.1. Bibliometric Analysis
Scientific Production per Country and Affiliation
4.2. Systematic Review
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yu, K.; Wei, L.; Yuan, H.; Zhang, W.; Zeng, X.; Wang, B.; Wang, Y. Genetic architecture of inducible and constitutive metabolic profile related to drought resistance in qingke (Tibetan hulless barley). Front. Plant Sci. 2022, 13, 1076000. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, D.; Bhardwaj, S.; Landi, M.; Sharma, A.; Ramakrishnan, M.; Sharma, A. The impact of drought in plant metabolism: How to exploit tolerance mechanisms to increase crop production. Appl. Sci. 2020, 10, 5692. [Google Scholar] [CrossRef]
- Praxedes, S.S.C.; Ferreira Neto, M.; Loiola, A.T.; Santos, F.J.Q.; Umbelino, B.F.; Silva, L.D.A.; Moreira, R.C.L.; Melo, A.S.D.; Lacerda, C.F.D.; Fernandes, P.D. Photosynthetic Responses, Growth, Production, and Tolerance of Traditional Varieties of Cowpea under Salt Stress. Plants 2022, 11, 1863. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yin, Y.; Wang, L.F.; Wang, M.; Zhao, M.; Tian, Y.; Li, Y.F. Transcriptome profiling of the elongating internode of cotton (Gossypium hirsutum L.) seedlings in response to mepiquat chloride. Front. Plant Sci. 2020, 10, 1751. [Google Scholar] [CrossRef]
- Wang, B.; Li, Z.; Ran, Q.; Li, P.; Peng, Z.; Zhang, J. ZmNF-YB16 Overexpression Improves Drought Resistance and Yield by Enhancing Photosynthesis and the Antioxidant Capacity of Maize Plants. Front. Plant Sci. 2018, 9, 709. [Google Scholar] [CrossRef]
- Messmer, R.; Stamp, P. Trends in drought research. Kasetsart J. Nat. Sci. 2010, 44, 507–516. [Google Scholar]
- Cao, L.; Lu, X.; Wang, G.; Zhang, Q.; Zhang, X.; Fan, Z.; Cao, Y.; Wei, L.; Wang, T.; Wang, Z. Maize ZmbZIP33 Is Involved in Drought Resistance and Recovery Ability Through an Abscisic Acid-Dependent Signaling Pathway. Front. Plant Sci. 2021, 12, 629903. [Google Scholar] [CrossRef] [PubMed]
- Gillani, S.F.A.; Zhuang, Z.; Rasheed, A.; Haq, I.U.; Abbasi, A.; Ahmed, S.; Wang, Y.; Khan, M.T.; Sardar, R.; Peng, Y. Brassinosteroids induced drought resistance of contrasting drought-responsive genotypes of maize at physiological and transcriptomic levels. Front. Plant Sci. 2022, 13, 961680. [Google Scholar] [CrossRef]
- Priatama, R.A.; Heo, J.; Kim, S.H.; Rajendran, S.; Yoon, S.; Jeong, D.H.; Choo, Y.K.; Bae, J.H.; Kim, C.M.; Lee, Y.H.; et al. Narrow lpa1 Metaxylems Enhance Drought Tolerance and Optimize Water Use for Grain Filling in Dwarf Rice. Front. Plant Sci. 2022, 13, 894545. [Google Scholar] [CrossRef]
- Tang, X.; Fei, X.; Sun, Y.; Shao, H.; Zhu, J.; He, X.; Wang, X.; Yong, B.; Tao, X. Abscisic acid-polyacrylamide (ABA-PAM) treatment enhances forage grass growth and soil microbial diversity under drought stress. Front. Plant Sci. 2022, 13, 973665. [Google Scholar] [CrossRef]
- Tiwari, P.; Srivastava, D.; Chauhan, A.S.; Indoliya, Y.; Singh, P.K.; Tiwari, S. Root system architecture, physiological analysis and dynamic transcriptomics unravel the drought-responsive traits in rice genotypes. Ecotoxicol. Environ. Saf. 2021, 207, 111252. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, A.B. Caracterização dos padrões de estresse hídrico para a cultura do arroz (ciclo curto e médio) no estado de Goiás e suas conseqüências para o melhoramento genético. Ciência E Agrotecnologia 2010, 34, 29–36. [Google Scholar] [CrossRef]
- Huang, X.; Chen, M.H.; Yang, L.T.; Li, Y.R.; Wu, J.M. Effects of exogenous abscisic acid on cell membrane and endogenous hormone contents in leaves of sugarcane seedlings under cold stress. Sugar Tech 2015, 17, 59–64. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, X.; Jiang, J.; Zhao, T.; Xu, X.; Yang, H.; Li, J. Virus-induced gene silencing of SlPYL4 decreases the drought tolerance of tomato. Hortic. Plant J. 2022, 8, 361–368. [Google Scholar] [CrossRef]
- Yang, S.; Chu, N.; Feng, N.; Zhou, B.; Zhou, H.; Deng, Z.; Shen, X.; Zheng, D. Global Responses of Autopolyploid Sugarcane Badila (Saccharum officinarum L.) to Drought Stress Based on Comparative Transcriptome and Metabolome Profiling. Int. J. Mol. Sci. 2023, 24, 3856. [Google Scholar] [CrossRef]
- Araújo, E.D.; Melo, A.S.; Rocha, M.S.; Silva, P.C.C.; Ferraz, R.L.S.; Melo, Y.L.; Alencar, R.S.; Sá, F.V.S.; Lacerda, C.F. Improvement of Silicon-Induced Tolerance to Water Stress Is Dependent on Genotype Sensitivity and Phenological Stage. J. Soil Sci. Plant Nutr. 2023, 23, 1648–1659. [Google Scholar] [CrossRef]
- Santos, A.R.; Melo, Y.L.; Oliveira, L.F.; Cavalcante, I.E.; Ferraz, R.L.S.; Sá, F.V.S.; Lacerda, C.F.; Melo, A.S. Exogenous Silicon and Proline Modulate Osmoprotection and Antioxidant Activity in Cowpea Under Drought Stress. J. Soil Sci. Plant Nutr. 2023, 22, 1692–1699. [Google Scholar] [CrossRef]
- Galvão, T.F.; Pereira, M.G. Revisões sistemáticas da literatura: Passos para sua elaboração. Rev. Epidemiol. Serviços Saúde 2014, 23, 183–184. [Google Scholar] [CrossRef]
- Pritchard, A. Statistical Bibliography or Bibliometrics. J. Doc. 1969, 25, 348–349. [Google Scholar]
- Donthu, N.; Kumar, S.; Pattnaik, D.; Lim, W.M. A bibliometric retrospection of marketing from the lens of psychology: Insights from psychology & marketing. Psychol. Mark. 2021, 38, 834–865. [Google Scholar] [CrossRef]
- Fang, H.C.; Kellermanns, F.W.; Eddleston, K.A.; Memili, E.; Dibrell, C. Empirical modeling in testing for family firm heter-ogeneity. In The Palgrave Handbook of Heterogeneity among Family Firms; Springer: Berlin/Heidelberg, Germany, 2019; Volume 6, pp. 69–85. [Google Scholar]
- Pluye, P.; Hong, Q.N. Combining the Power of Stories and the Power of Numbers: Mixed Methods Research and Mixed Studies Reviews. Annu. Rev. Public Health 2014, 35, 29–45. [Google Scholar] [CrossRef]
- Aragão Júnior, W.R.; Oliveira Júnior, A.I. Internet das coisas na gestão de resíduos sólidos: Revisão sistemática com análise bibliométrica da literatura. J. Environ. Anal. Prog. 2021, 6, 194–209. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef]
- Najmi, A.; Rashidi, T.H.; Abbasi, A.; Travis Waller, S. Reviewing the transport domain: An evolutionary bibliometrics and network analysis. Scientometrics 2017, 110, 843–865. [Google Scholar] [CrossRef]
- Liu, W.; Wang, J.; Li, C.; Chen, B.; Sun, Y. Using Bibliometric Analysis to Understand the Recent Progress in Agroecosystem Services Research. Ecol. Econ. 2019, 156, 293–305. [Google Scholar] [CrossRef]
- Pantaleão, P.F.; Veiga, H.M.S. Bem-estar no trabalho: Revisão sistemática da literatura nacional na última década. Holos 2019, 35, 1–24. [Google Scholar] [CrossRef]
- Fapesp. Publicações Científicas por Países: Contagem por Autoria e por Artigo. Revista Pesquisa Fapesp. 2020, Volume 288. Available online: https://revistapesquisa.fapesp.br/publicacoes-cientificas-por-paises-contagem-por-autoria-e-por-artigo/ (accessed on 25 April 2023).
- McHugh, M.L. Descriptive statistics. Part II: Most commonly used descriptive statistics. J. Spec. Pediatr. Nurs. 2003, 8, 111–116. [Google Scholar] [CrossRef]
- Ma, J.; Liu, Y.; Zhang, P.; Chen, T.; Tian, T.; Wang, P.; Che, Z.; Shahinnia, F.; Yang, D. Identification of quantitative trait loci (QTL) and meta-QTL analysis for kernel size-related traits in wheat (Triticum aestivum L.). BMC Plant Biol. 2022, 22, 607. [Google Scholar] [CrossRef]
- Merchuk-Ovnat, L.; Barak, V.; Fahima, T.; Ordon, F.; Lidzbarsky, G.A.; Krugman, T.; Saranga, Y. Ancestral QTL Alleles from Wild Emmer Wheat Improve Drought Resistance and Productivity in Modern Wheat Cultivars. Front. Plant Sci. 2016, 7, 1–14. [Google Scholar] [CrossRef]
- Jacob, F.; Mahatma, M.; Deshmukh, Y.; Kandoliya, U.K.; Maraviya, G.V.; Joshi, M.; Vala, A. Altered expression levels of transcripts of GNAC TFs during drought stress in susceptible and tolerant cultivars of groundnut. Plant Stress 2022, 3, 100062. [Google Scholar] [CrossRef]
- Cheng, Y.J.; Deng, X.P.; SooKwak, S.; Chen, W.; EEneji, A. Enhanced tolerance of transgenic potato plants expressing choline oxidase in chloroplasts against water stress. Bot. Stud. 2013, 54, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Macena, T.N.S.; Santos, N.O.; Gonçalves, M.A.S.; Alves, J.V.A. Utilização do sistema crispr/cas-9 no melhoramento vegetal revisão sistemática de literatura. Rev. Mosaicum 2021, 33, 1–25. [Google Scholar]
- Bernini, C.S.; Guimarães, P.S.; Gallo, P.B.; Paterniani, M.E.A.G.Z. Parâmetros genéticos de progênies interpopulacionais de milho para caracteres relacionados com tolerância à seca. Sci. Electron. Arch. 2021, 14, 25–31. [Google Scholar] [CrossRef]
- Teixeira, R.K.C.; Silveira, T.S.; Botelho, N.M.; Tcbc-Mg, P. Citação de artigos nacionais: A (des)valorização dos periódicos brasileiros. Rev. Do Colégio Bras. De Cir. 2012, 39, 421–425. [Google Scholar] [CrossRef]
- Jales Filho, R.C.; Melo, Y.L.; Viegas, P.R.A.; Oliveira, A.P.; Almeida Neto, V.E.; Ferraz, R.L.S.; Gheyi, H.R.; Carol, P.; Lacerda, C.F.; Melo, A.S. Salicylic acid and proline modulate water stress tolerance in a traditional variety of cowpea. Rev. Bras. Eng. Agrícola Ambient. 2023, 27, 18–25. [Google Scholar] [CrossRef]
- Egghe, L. The Hirsch index and related impact measures. Annu. Rev. Inf. Sci. Technol. 2010, 44, 65–114. [Google Scholar] [CrossRef]
- Silva, D.D.; Almeida, C.C.; Grácio, M.C.C. Associação do Fator de Impacto e do Índice h para a avaliação de periódicos científicos: Uma aplicação no campo da Ciência da Informação. Rev. Em Questão 2018, 24, 132–151. [Google Scholar] [CrossRef]
- Moraes, P.F.; Laat, D.M.; Santos, M.E.A.H.P.; Colombo, C.A.; Kiih, T. Expressão gênica diferencial em genótipos de mamona (Ricinus communis L.) submetidos a déficit hídrico induzido por PEG. Bragantia 2015, 74, 25–32. [Google Scholar] [CrossRef]
- Veroneze-Júniora, V.; Martinsa, M.; Leodb, L.M.; Souza, K.R.D.; Santos-Filho, P.R.; Magalhães, P.C.; Carvalho, D.T.; Santos, M.H.; Souza, T.C. Leaf application of chitosan and physiological evaluation of maize hybrids contrasting for drought tolerance under water restriction. Braz. J. Biol. 2020, 80, 631–640. [Google Scholar] [CrossRef]
- Elliott, J.; Glotter, M.; Best, N.; Boote, K.J.; Jones, J.W.; Hatfield, J.L.; Rosenzweig, C.; Smith, L.; Foster, I. Predicting Agricultural Impacts of Large-Scale Drought: 2012 and the Case for Better Modeling; Center for Robust Decision Making on Climate & Energy Policy (RDCEP) Working Paper Series: Chicago, IL, USA, 2013; pp. 1–8. [Google Scholar] [CrossRef]
- Ghaffar, A.; Hussain, N.; Ajaj, R.; Shahin, S.M.; Bano, H.; Javed, M.; Khalid, A.; Yasmin, M.; Shah, K.H.; Zaheer, M.; et al. Photosynthetic activity and metabolic profiling of bread wheat cultivars contrasting in drought tolerance. Front. Plant Sci. 2023, 14, 1123080. [Google Scholar] [CrossRef]
- Prado, S.A.; Cabrera-Bosquet, L.; Grau, A.; Coupelledru, A.; Millet, E.J.; Welcker, C.; Tardieu, F. Phenomics allows identification of genomic regions affecting maize stomatal conductance with conditional effects of water deficit and evaporative demand. Plant Cell Environ. 2018, 41, 314–326. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Zheng, Y.; Wang, J.; Chen, W.; Yang, Z.; Chen, Y.; Yang, Y.; Lu, G.; Sun, B. SbNAC9 Improves Drought Tolerance by Enhancing Scavenging Ability of Reactive Oxygen Species and Activating Stress-Responsive Genes of Sorghum. Int. J. Mol. Sci. 2023, 24, 2401. [Google Scholar] [CrossRef] [PubMed]
- Pichyangkura, R.; Chadchawan, S. Biostimulant activity of chitosan in horticulture. Sci. Hortic. 2015, 196, 49–65. [Google Scholar] [CrossRef]
- Shafiq, S.; Akram, N.A.; Ashraf, M. Does exogenously-applied trehalose alter oxidative defense system in the edible part of radish (Raphanus sativus L.) under water-deficit conditions. Sci. Hortic. 2015, 185, 68–75. [Google Scholar] [CrossRef]
- Guo, Y.L.; Huang, G.M.; Guo, Q.; Peng, C.X.; Liu, Y.R.; Zhang, M.C.; Li, Z.H.; Zhou, Y.Y.; Duan, L.S. Increase in root density induced by coronatine improves maize drought resistance in North China. Crop J. 2023, 11, 278–290. [Google Scholar] [CrossRef]
- Guan, P.; Shen, X.; Mu, Q.; Wang, Y.; Wang, X.; Chen, Y.; Zhao, Y.; Chen, X.; Zhao, A.; Mao, W.; et al. Dissection and validation of a QTL cluster linked to Rht-B1 locus controlling grain weight in common wheat (Triticum aestivum L.) using near-isogenic lines. Theor. Appl. Genet. 2020, 133, 2639–2653. [Google Scholar] [CrossRef]
- Souza Guimarães, P.; Sarto Rocha, D.; Paterniani, M.E.A.G.Z. Conteúdo de carboidrato foliar em híbridos de milho submetidos à restrição hídrica. Evidência 2019, 19, 93–112. [Google Scholar] [CrossRef]
- Koornneef, M.; Meinke, D. The development of Arabidopsis as a model plant. Plant J. 2010, 61, 909–921. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, S.; Sun, M.; Guo, Y.; Zhao, S.; Zhou, X.; Bai, X.; Dai, K.; Li, H.; Yuan, X.; et al. SiMYBS3, Encoding a Setaria italica Heterosis-Related MYB Transcription Factor, Confers Drought Tolerance in Arabidopsis. Int. J. Mol. Sci. 2023, 24, 5418. [Google Scholar] [CrossRef]
- Pandian, B.A.; Sathishraj, R.; Djanaguiraman, M.; Prasad, P.V.V.; Jugulam, M. Role of cytochrome P450 enzymes in plant stress response. Antioxidants 2020, 9, 454. [Google Scholar] [CrossRef]
- Liu, H.; Song, S.; Liu, M.; Mu, Y.; Li, Y.; Xuan, Y.; Niu, L.; Zhang, H.; Wang, W. Transcription Factor ZmNAC20 Improves Drought Resistance by Promoting Stomatal Closure and Activating Expression of Stress-Responsive Genes in Maize. Int. J. Mol. Sci. 2023, 24, 4712. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.G.; Lv, W.T.; Li, M.J.; Wang, B.; Sun, D.M.; Deng, X. Maize and Arabidopsis membrane-bound transcription factor Zmbzip17 is a key regulator in the cross-talk of ER quality control and ABA signaling. Plant Cell Physiol. 2013, 54, 2020–2033. [Google Scholar] [CrossRef] [PubMed]
- Solanki, J.K.; Sarangi, S.K. Effect of drought stress on proline accumulation in peanut genotypes. Int. J. Adv. Res. 2014, 2, 301–309. [Google Scholar]
- Pecetti, L.; Annicchiarico, P.; Crosta, M.; Notário, T.; Ferrari, B.; Nazzicari, N. Tolerância à seca do tremoço branco: Variação genética, arquitetura genética de características e previsão habilitada pelo genoma. Int. J. Mol. Sci. 2023, 24, 2351. [Google Scholar] [CrossRef]
- Yi, Y.; Hassan, M.A.; Cheng, X.; Li, Y.; Liu, H.; Fang, W.; Zhu, Q.; Wang, S. QTL mapping and analysis for drought tolerance in rice by genome-wide association study. Front. Plant Sci. 2023, 14, 1223782. [Google Scholar] [CrossRef]
- Zhao, J.; Sun, L.; Gao, H.; Hu, M.; Mu, L.; Cheng, X.; Wang, J.; Zhao, Y.; Li, Q.; Wang, P.; et al. Genome-wide association study of yield-related traits in common wheat (Triticum aestivum L.) under normal and drought treatment conditions. Front. Plant Sci. 2023, 13, 1098560. [Google Scholar] [CrossRef]
- Salvi, S.; Tuberosa, R. A colheita QTLome atinge a maioridade. Curr. Opin. Biotechnol. 2015, 32, 179–185. [Google Scholar] [CrossRef]
- Wang, R.S.; Pandey, S.; Li, S.; Gookin, T.E.; Zhao, Z.X.; Albert, R. Common and unique elements of the ABA-regulated transcriptome of Arabidopsis guard cells. BMC Genom. 2011, 12, 216. [Google Scholar] [CrossRef]
- Anjum, S.A.; Ashraf, U.; Tanveer, M.; Khan, I.; Hussain, S.; Shahzad, B. Drought induced changes in growth, osmolyte accumulation and antioxidant metabolism of three maize hybrids. Front. Plant Sci. 2017, 8, 69. [Google Scholar] [CrossRef]
- Perveen, K.; Alfagham, A.T.; Debnath, S.; Bukhari, N.A.; Wei, D.Q.; Alshaikh, N.A.; Alwadai, A.S. Enriching drought resistance in Solanum lycopersicum using Abscisic acid as drought enhancer derived from Lygodium japonicum: A new-fangled computational approach. Front. Plant Sci. 2023, 14, 1106857. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, Y.; Shi, Z.; Jin, Y.; Sun, H.; Xie, F. Biosynthesis and signal transduction of ABA, JA, and BRs in response to drought stress of Kentucky bluegrass. Int. J. Mol. Sci. 2019, 20, 1289. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Wang, Q.; Lu, L.; Wang, S.D.; Chen, X.; Khan, M.H.U.; Zhang, Y.H.; Yang, S.X. Identification of the soybean small auxin upregulated RNA (SAUR) gene family and specific haplotype for drought tolerance. Biologia 2022, 77, 1197–1217. [Google Scholar] [CrossRef]







| Reference | Number of Times Cited | Mechanism | Response | |
|---|---|---|---|---|
| 1. | [1] | 214 | Gene expression | Identification of the CYP450 gene family, by highlighting the sensitivity and broadly targeted metabolites during drought stress. |
| 2. | [30] | 158 | Molecular markers | Mepiquat chloride (MCD) increases water resistance in plants and inhibits growth. Molecular markers contribute to identifying some genes and metabolites and expressing differences in the biosynthesis pathways of isoflavonoids and flavonoids in soybean crops. |
| 3. | [4] | 133 | Gene expression: Zm gene family | Drought resistance is significantly improved when plants overexpress genes from the ZmNF-YB16 family since these regulate photosynthetic and enzymatic activities. |
| 4. | [31] | 131 | Molecular markers | QTL alleles introduced through marker-assisted selection can increase drought resistance, productivity, and yield stability across multiple environments in wheat. |
| 5. | [10] | 119 | Abscisic acid (ABA) | The drought resistance of plants can be significantly improved with the application of phytohormones, especially abscisic acid (ABA), due to the proliferation of beneficial bacteria and fungi near the root zone of plants. |
| 6. | [7] | 117 | Gene expression: Zm gene family and ABA | Overexpression of genes from the ZmbZIP33 family contributes to positive increases in chlorophyll levels and the roots of plants when subjected to water stress. The presence of ZmbZIP33 genes promotes a defense mechanism that helps in the production and accumulation of abscisic acid (ABA). |
| 7. | [8] | 114 | Brassinosteroids | Plants synthesizing plant hormones such as brassinosteroids (BRs) are highly resistant to water stress. |
| 8. | [9] | 108 | Genetic combination: GMOs | Genetically modified organisms (GMOs) have made a clear and objective contribution to developing species resistant to water stress. In these cases, the genetic combination seeks to optimize the morphological and physiological structure of the plants so that they can withstand the most frequent adverse climatic conditions in the future without reducing productivity. |
| 9. | [32] | 107 | Gene expression | In drought-tolerant cultivars, nine transcription factors corresponded to overexpressed GNAC genes, predominant in drought-resistant cultivars. |
| 10. | [33] | 87 | Gene expression: CodA gene family and GMOs | Genetically modified potato plants show overexpression of the CodA gene, which confers enhanced drought resistance and recovery capacity. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva, P.F.; Cassa, N.; de Melo, A.S.; Dantas Neto, J.; Meneghetti, L.A.M.; Custódio, A.S.C.; de Oliveira, N.P.R.; da Silva, T.J.A.; Bonfim-Silva, E.M.; Andrade, S.P.; et al. Advances in Crop Genetic Improvement to Overcome Drought Stress: Bibliometric and Meta-Analysis. Agriculture 2023, 13, 1860. https://doi.org/10.3390/agriculture13101860
da Silva PF, Cassa N, de Melo AS, Dantas Neto J, Meneghetti LAM, Custódio ASC, de Oliveira NPR, da Silva TJA, Bonfim-Silva EM, Andrade SP, et al. Advances in Crop Genetic Improvement to Overcome Drought Stress: Bibliometric and Meta-Analysis. Agriculture. 2023; 13(10):1860. https://doi.org/10.3390/agriculture13101860
Chicago/Turabian Styleda Silva, Patrícia Ferreira, Natália Cassa, Alberto Soares de Melo, José Dantas Neto, Luana Aparecida Menegaz Meneghetti, Alisson Silva Costa Custódio, Niclene Ponce Rodrigues de Oliveira, Tonny José Araújo da Silva, Edna Maria Bonfim-Silva, Sérgio Plens Andrade, and et al. 2023. "Advances in Crop Genetic Improvement to Overcome Drought Stress: Bibliometric and Meta-Analysis" Agriculture 13, no. 10: 1860. https://doi.org/10.3390/agriculture13101860
APA Styleda Silva, P. F., Cassa, N., de Melo, A. S., Dantas Neto, J., Meneghetti, L. A. M., Custódio, A. S. C., de Oliveira, N. P. R., da Silva, T. J. A., Bonfim-Silva, E. M., Andrade, S. P., Duarte, T. F., da Silva Berilli, S., Souza, M. N., de Oliveira, A. d. F. M., Moulin, M. M., & Berilli, A. P. C. G. (2023). Advances in Crop Genetic Improvement to Overcome Drought Stress: Bibliometric and Meta-Analysis. Agriculture, 13(10), 1860. https://doi.org/10.3390/agriculture13101860








