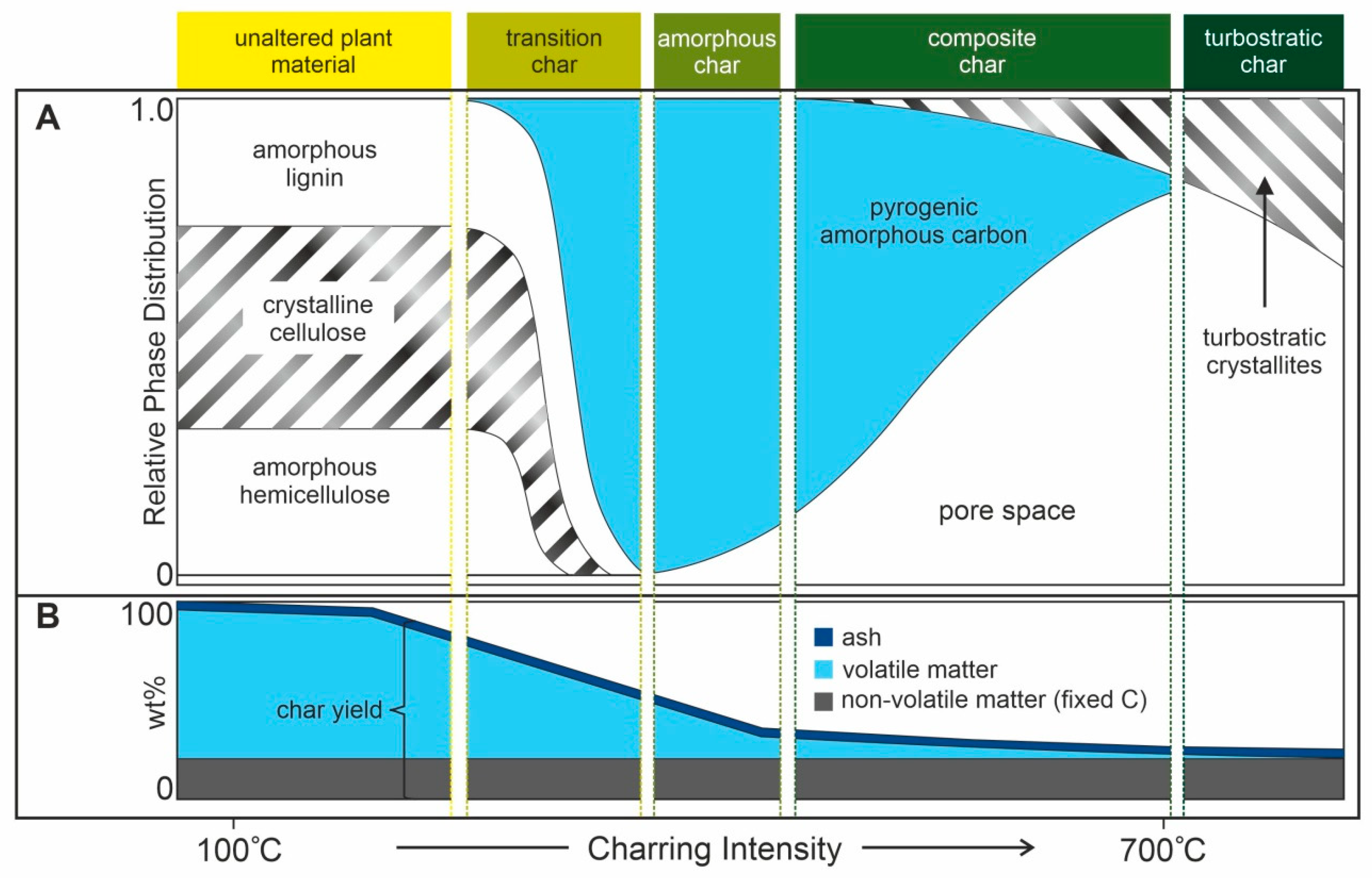
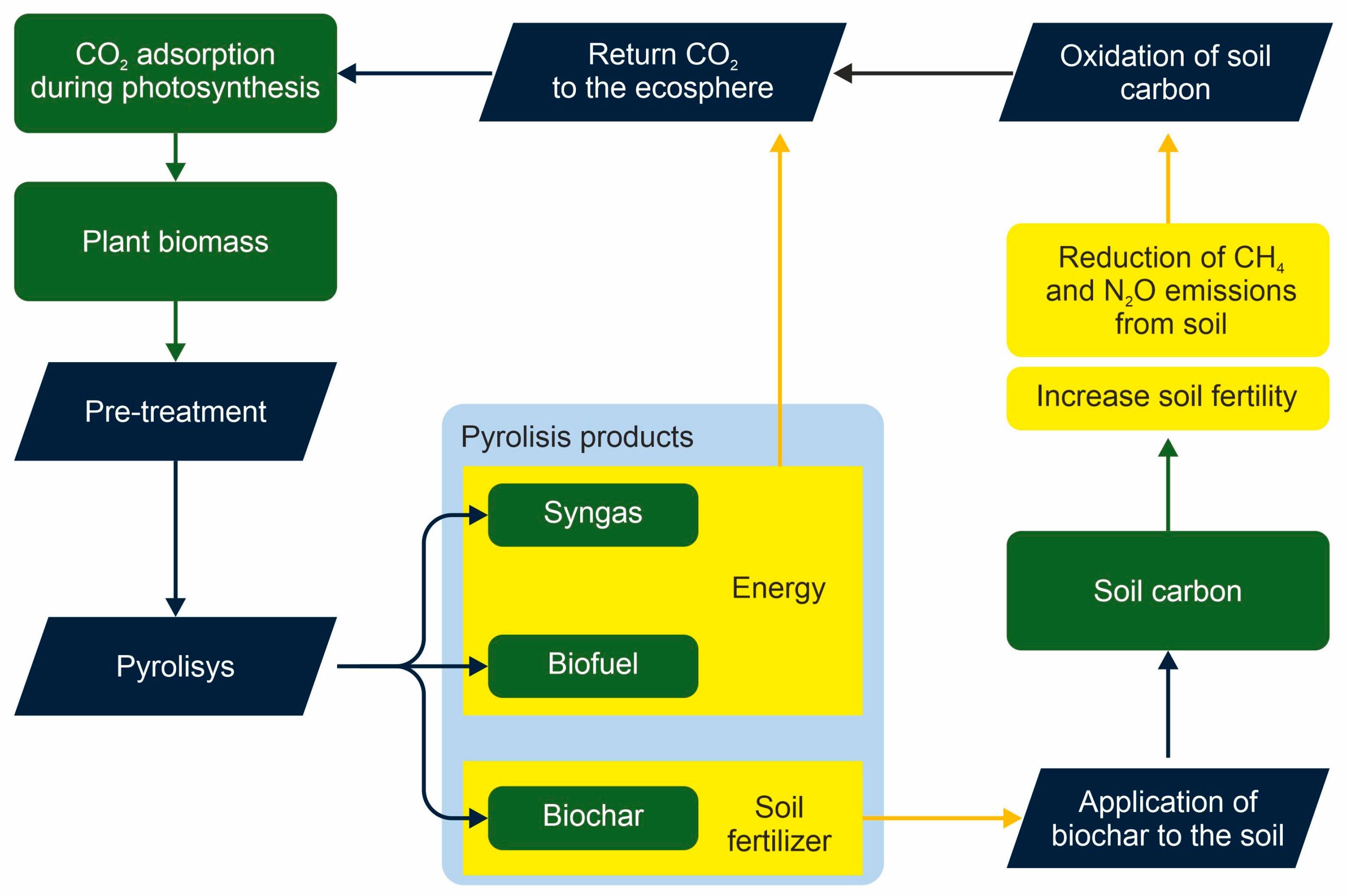
3.2. Effects on Soil Chemical Properties
Great attention has been paid to studying the use of biochar for the remediation and restoration of polluted soils [
157,
158,
159,
160]. Its beneficial effects are most often attributed to immobilizing toxicants and reducing the bioavailability of toxins to soil organisms. An increase in the pyrolysis temperature increases the carbonization degree of biomasses and the surface of biochar particles, which contributes to an increase in their sorption capacity for organic pollutants and hence a decrease in their potential hazard to soils. In particular, a decrease in the bioaccumulation of polychlorinated biphenyls by soil organisms was found when activated carbons were introduced into soils, supported by an adequate change in the equilibrium concentration of the toxicant in the study of their extraction from water systems [
161,
162]. A similar effect of biochar on the equilibrium concentrations of organic toxicants was established for DDT [
163] and polycyclic aromatic compounds [
164]. Sorption of organic toxicants does not preclude their subsequent release from the bound form over time. Biochar also binds heavy metals in a carbon matrix. In particular, a relationship has been registered between a decrease in the mobility of copper, cadmium and nickel during the period of decomposition of biochar introduced into the soil and the content of organic matter in it [
165]. Therefore, its production and application to soils can be considered an effective strategy in limiting the circulation of such elements in the environment. A fairly large number of examples of biochar use with a view to suppressing the phytotoxic properties of heavy metals have been described. For example, a regular decrease in the phytotoxic concentrations of cadmium and zinc was registered in a 60-day experiment with biochars introduced into silt masses [
166,
167]. The amounts of retained heavy metals correlate with the cation-exchange capacity of biochar, which provides evidence of the mechanism of their retention on the carboxylate groups of biochars. At the same time, an inverse relationship was found for copper, associated with an increase in soluble organic carbon in soil moisture when biochar from hardwood was introduced. This proves that the specific effects of biochar application on the bioavailability of toxic soil elements require verification for actual application conditions. There is a necessity to certify the maximum allowable levels of toxic elements in biochar [
168]. The ability of biochar to remove arsenic from wastewater has been found [
169]. Unlike metals, arsenic is present in soil in the form of oxo anions. Accordingly, the mobility of arsenic in soils does not decrease with pH increase, as in the case of copper, zinc, lead and cadmium, but increases. Its retention in soils is due to the presence of aluminum, iron and manganese oxides in them, which react with arsenic forms. Thus, there is a possibility of increasing the bioavailability of this element due to the fact that the introduction of biochar increases the pH and soluble carbon of soils. In laboratory tests aimed at assessing the influence of biochar on the arsenic content in wastewater moderately polluted with heavy metals, the effect of biochar introduced in an amount of 30 vol.% was not reliably detected [
170]. Additional benefits in the removal of arsenic can be expected from biochar enriched with iron oxides, and if used in conjunction with compost, a simultaneous decrease in the bioavailability of other common heavy metals can be achieved [
171]. Similar patterns were revealed for the binding of chromium (VI) compounds.
There are works devoted to the use of biochar produced from chicken manure with the aim of removing metals from the environment. Thus, its high efficiency with respect to copper (II) ions has been proven. The share of the removed metal increases with biochar mineralization and varies from 1.3 to 26 mg/g of the sorbent. The use of chicken manure as a raw material provided a higher capacity of biochars compared to coals produced from corn waste, pine wood, and eucalyptus [
172]. A decrease in copper bioavailability was also observed when chicken manure biochar was applied to copper-contaminated sandy loamy soils. Biochar was prepared by slow pyrolysis at 500 °C for 2 h [
173]. It was characterized by a high content of phosphorus (19.4 g/kg) and potassium (17.2 g/kg), pH 9.1 (water suspension 1:5) and a specific surface area of 11.5 m
2/g. The interaction of biochars with copper was also controlled by changing the growth rate of
O. picenis; the indicator species is a metallophile. In model experiments in seed germination in a greenhouse, biochar was applied in the amount of 5 and 10%. In addition to the effect on plants, a decrease in the concentration of copper in soil moisture and an increase in soil microbial activity are described. The manifestation of the phytotoxic properties of biochar with a high content of a number of other trace elements (phosphorus, boron, zinc, manganese) in relation to plants sensitive to their presence in the soil was registered [
174,
175]. There are other risks associated with the agronomic use of biochar. Thus, it was found that the introduction of 1% biochar made of eucalyptus chips halved the bioavailability of two insecticides (chlorpyrifos and carbofuran) [
176]. Sorption of pesticides can help in reducing crop contamination, but it reduces the efficacy of their application against insect pests. In addition, the biochar itself may contain phytotoxic compounds [
177]. The influence of biochar on the content of polycyclic aromatic hydrocarbons (PAHs) is the focus, as they are partially formed in the pyrolysis of organic matter, the precursor of biochar. Adsorption or other protection against microbiological degradation of PAHs limits their removal from polluted soils; saturation of biochar pores with other adsorbed substances reduces its effect on the removal of those compounds into groundwater. It is not entirely clear how the influence of biochar on the microbiological community and its protective effect on PAHs correlate, as both adsorption and microbiological activity depend in a complex way on soil pH after the application of biochars. Examples of agrochemical and ecotoxicological consequences of biochar application to soils are given in
Table 6 [
178].
In general, an increase in the pyrolysis temperature during the production of biochar increases the sorption efficiency in relation to organic pollutants. Presumably, this occurs as a result of an increase in the specific surface area and microporosity of coals.
3.3. Effects on Soil Biological Properties
Although the positive effect of biochar on soil fertility and agriculture ecosystems mainly refers to the pH changes in acidic soils and improved nutrient retention and aeration, remarkable influence of biochar amendments on microbial communities and soil animals has been reported [
186,
187,
188]. Changes in microbiota composition and abundance also follow soil structure improvement and nutrient availability caused by biochar application (indirect biochar effect). All of these factors influence plant growth and crop quality and consequently, more input of rhizodeposits into the soil. The positive influence of biochar amendments can be counterbalanced by toxic metals contained in the pyrolysis products, especially those derived from sewage sludge or wastewater sediments. The consideration of the biochar effects on soil microorganisms reported in the literature mainly depends on what kind of effect is expected, i.e., biodiversity, abundance, or secondary effects on soil biota on crops and soil fertility. Thus, soil biota diversity is often believed to be a key factor for soil functions [
189] and organic amendments derived from biochar are key factors affecting trophic relations in soil [
190]. The second approach assumes indirect influence of biochar on nutrient availability and possible effects of various species adsorbed in biochar particles and released in the environment during biochar aging and mineralization. Microbial abundance always increases with biochar amendments with low dependence on the feedstock and soil type. The mycorrhizal response estimated by root colonization increased twice for a two-year exposition of 0.6–6 t/ha of biochar to
Eucalyptus wood [
191]. The protective effect of biochar particles, sorption of signaling compounds, detoxification of allelochemicals, and improvement of physical properties of soil were mentioned as possible reasons for the positive effect of biochar on extraradical mycelium [
3]. In addition, biochar was reported to be able to stimulate spore germination [
192]. An opposite effect of biochar amendment was attributed to symbiotic relations in conditions of high nutrient availability, undesirable pH shifts and toxic effects of metal cations or high salt content [
193,
194]. Some other effects of biochar on the microbial abundance affected by its application are summarized in
Table 7.
Fertilizers commonly reduce the influence of biochar application on microbial abundance. The effect is mostly pronounced for P, N-containing fertilizers that inhibit acceleration of root colonization observed with biochar added alone [
195]. The plants did not rely on biological N fixation in the presence of fertilizers as much as in the presence of biochar. Non-symbiotic organisms can slightly increase their abundance in the same conditions due to the higher availability of nutrients from fertilizer. Biological nitrogen fixation can also be stimulated by introduction with soybean straw-derived biochar [
196]. Microbial biomass increases with pH values caused by biochar in acidic soils. The effect is more pronounced for bacteria, whereas fungi are more tolerant to this factor and can even reduce their growth at high pH [
197]. It should be taken into account that changes in the soil pH observed after biochar application can reflect secondary effects on microbial activity [
192]. The pH effect significantly depends on the soil properties. Biochars added to soil protect microorganisms from the inhibitory effect of some species (catechols, flavonoids, phenolics) that are adsorbed on the pores and hence excluded from the microorganism metabolism. Such an effect was described for biochar obtained from corn stover [
198]. Fast-pyrolysis biochar from wood promoted arbuscular colonization of asparagus due to limitations in access to some aromatic acids exerting an allelopathic effect.
The microbial response to the biochar addition is also sensitive to the adhesion of the cells on the particle surface. This formation of biofilms mostly affects bacterial but not fungal abundance. The attachment of the cells to the biochar particles can occur via hydrophobic attraction and electrostatic forces. The mineralization of the biochar, as well as higher salt content, accelerate adhesion. Inclusion of the cells into the pores is limited by their size, which should be at least 2–5 times bigger than the cell size. For
Bacillus mucilaginous and
Acinetobacter sp., it was estimated of 204 μm [
150]. In addition to abundance, microbial community composition significantly differs with biochar addition against that of unmodified soils of the same mineralogy [
151]. Viscosity decreases stimulate the development of plant roots and rhizobia because of the higher availability of nutrients. Additionally, invertebrates move easier through the amended soil, making pores and influencing predator/prey ratio. A similar influence on plant roots and fungi can be attributed to the changes in the soil bulk density caused by the rather small value of this parameter of biochar particles (typically between 0.09 and 0.5 g/cm
3 against 1.5–2.1 g/cm
3 for true density).
Transformation of soil nutrients. It has been established that nitrification is accelerated when biochar is added to forest soils, which is explained by the sorption of phenols that inhibit the process and an increase in the mass of ammonium-oxidizing bacteria [
199]. In addition, an increase in the activity of alkaline phosphatase, aminopeptidase and
N-acetylglucosamine oxidase was revealed upon the introduction of biochar [
200], which may be associated with an increase in the production of organic nitrogen and phosphorus due to the accelerated growth of plant roots in the pores of biochar.
Bradyrhizobiaceae (
Rhodoblastus,
Rhodopseudomonas,
Bradyrhizobium and
Nitrobacter) may take part in the process, as well as nitrogen utilizing
Hyphomicrobiaceae (
Rhodoplanes,
Starkeya), nitrates and ammonia in nitrogen fixation or denitrification [
201]. The same microorganisms can produce ethylene from fresh biochar, which in turn leads to a reduction in greenhouse gas (N
2O and CO
2) emissions [
202]. To study nitrogen fixation by legumes (
Phaseolus vulgaris L.) in soils treated with 0–90 g/kg biochar soil, the isotope dilution method (
15NH
4)
2SO
4 was used [
203]. At the maximum dose of biochar, the proportion of fixed nitrogen increased from 50% in the control to 72%. The increase in total nitrogen supplied from the atmosphere was 49% for the dose of 30 g/kg of biochar and 78% for 60 g/kg, a decrease was registered at the maximum dose of application. A possible reason for the increase in microbial nitrogen fixation was an increase in the availability of boron and molybdenum and, to a lesser extent, potassium, calcium and phosphate, an increase in pH, a decrease in the availability of nitrogen, and saturation of aluminum. At the same time, the input of soil nitrogen decreased by 14 and 17% at an application rate of 30 and 60 g/kg of biochar, respectively. Biochar has a positive effect on phosphorus-mobilizing mycorrhiza, as it protects mycelium, indirectly influencing the changes in the physicochemical characteristics of soils, and detoxifying allelopathic secretions [
204,
205]. There are also opposite observations, indicating the absence of the influence of biochar or its negative effect on mycelium. One explanation for this contradiction is the indirect effect of biochar due to changes in pH, saturation of the soil with oxygen, and changes in its porosity.
Microbial diversity in soils enriched with biochar has been studied using various methods, including total DNA analysis of the soil microbial community [
206], colony culture and counting [
207], substrate-induced respiration [
208,
209], microbial biomass [
210,
211], extraction of phospholipid fatty acids [
212,
213,
214,
215], contrasting, and direct examination of individual biochar particles [
3]. The introduction of biochar increases microbial diversity in different ways for different groups of microorganisms. The two most common types of mycorrhizal fungi (arbuscular and ectomycorrhizal fungi) most often respond positively to the application of biochar (see review [
3]). The response of mycorrhiza is usually assessed by root colonization. Thus, the rate of formation and the number of processes of ectomycorrhizae on larch seedlings in the presence of biochar in the soil increased by 20–160% [
216]. Similar results were obtained on wheat in a two-year experiment with the introduction of biochar from eucalyptus wood (0.6–6.0 t/ha)—the acceleration of colonization was 5–20% relative to the control without the introduction of biochar [
191]. It is less clear how the part of the mycelium located in the soil interacts with the biochar. The direct interaction of biochar and mycelium may be important. For example, the internal pores of the biochar particles can protect the extraradial mycelium from external influences, for example, from grazing animals or from soil overconsolidation [
188].
The introduction of biochar at doses of 5 and 25 t/ha was studied when cultivating wheat for 10 weeks while simultaneously varying the dose of applied nitrogen [
217]. It was shown that Cmic decreased when biochar was introduced, while Nmic remained practically unchanged. The experiment cannot be explained by the sorption of inorganic nitrogen on coals, since carbon dioxide emissions decreased in the presence of 5 t/ha of biochar, but not in the presence of 25 t/ha. The structure of the ammonium-oxidizing microbial community changed only when biochar was introduced together with a nitrogen source. The authors concluded that the introduction of biochar reduced the activity of the microbial community as a whole. An increase in the dose of mineral fertilizers reduces the positive effect of biochar on the rate of reproduction of microorganisms [
218], depending on the nature of the fertilizer and the group of microorganisms. Thus, mycorrhizal infection is suppressed by phosphorus-containing fertilizers, regardless of the application of biochar, but it does not depend on the application of nitrogen fertilizers. The opposite situation was registered for
Rhizobium [
219]. The reason may be the different influences of external conditions on the symbiosis of microorganisms. Therefore, when nitrogen fertilizers are added, the plant may not need biological nitrogen fixation to the same extent as in the absence of top dressing. A similar reason may explain the different effects of increased carbon load in the rhizosphere during exudation. The microbial diversity of non-symbiotic microorganisms can increase with increased availability of nutrients, either as a result of their longer retention in soil enriched with biochar or due to their entry into soils together with biochar. In most cases, a similar improvement can be achieved by the direct application of nitrogen and phosphorus fertilizers without biochar.
An increase in the concentration of microelements, such as molybdenum or boron, can also cause an increase in biological nitrogen fixation by
Rhizobium colonizing biochar [
220]. This phenomenon was discovered during the study of the immobilization of toxic elements by biochar. Similarly, sorption reduces the toxic effect of antimicrobial compounds and elements by reducing the time of their action, especially when they first enter the soil [
221].
An example of the indirect effect of biochar on soil microorganisms is a change in pH. An increase in microbial biomass (Cmic) from 20 to 180 μg/g and in ninhydrin nitrogen from 0.5 to 4.5 μg/g was registered with an increase in soil pH from 3.7 to 8.3 [
222]. At the same time, bacteria and fungi react differently to changes in pH. Bacteria increase diversity with an increase in pH to 7 or more, while fungi do not change their biomass in the specified pH range and decrease biomass with further increase [
197,
223]. Therefore, biochar with pH varying within the specified range can affect soil biota differently depending on soil pH. In addition, even with the introduction of acidic samples, for example, hydrochar with pH 4, an increase in the pH of fertilized soils was observed, apparently due to secondary processes of microbial reduction of unidentified organic substrates and electron transfer mediators [
192]. The said work indicates the importance of secondary processes in fertilized soils, which, in turn, are determined by the pH value of the soil before its cultivation, the direction and magnitude of the change in pH when biochar is added, etc. In addition, it should be taken into account that the pH on the surface of biochar particles, where a bacterial film is formed, may differ from that in the bulk of the soil [
188].
In addition to changing the microbial biomass, the introduction of biochar and related changes in the conditions of microbiological community development change its structure up to the change of dominants. Such changes have been repeatedly recorded for Terra preta (artificial soil based on low-grade activated carbon, also called Brazilian black soil), soils enriched with coal from natural fires and soils with biochar and for soil fungi, bacteria and archaea [
207,
224,
225,
226]. As a rule, the diversity of the bacterial community increased up to 25% when biochar was applied to the soils of Terra preta but decreased with a similar tillage in soils after natural fires. Simultaneously, in these cases, the diversity of archaea and fungi decreased, which indicates the unequal effect of biochar on various components of soil communities. However, it should be noted that these studies were carried out at different times, from 0.5 and 2.5 years for soils after fires and with the introduction of biochar and up to hundreds and thousands of years for Terra preta soils.
The bacterial community in soils with a high content of biochar differs from that in soils possessing the same mineralogy without biochar in them [
207,
227]. This was also shown with the help of genetic fingerprinting of Terra Preta soils and unmodified forest soil in the Amazonian region. The first ones showed a larger number of unique taxonomic units. At the same time, all taxa present in the forest soil were found in Terra preta, while the latter contained unique units that were absent in the forest soil. The greatest difference between the microbiological communities of both soils was established for an evolutionary distance of 5%, which indicates the presence of differences at the genome and species levels. Studies [
207] revealed a high taxonomic diversity in biochar-fertilized Terra Preta soils in four areas of the central Amazonian region compared to unmodified soils. The maximum difference is fixed at the family level. Similar discrepancy values of 80% were observed for soils enriched with biochar and agricultural soils differing in cultivation time, past and present crops, and other conditions [
206]. For uncultivated soils, the diversity did not exceed 40%. Up to the present, the biochar of Terra preta soils, introduced hundreds and thousands of years ago, remains the main factor determining the development of the microbiological community, despite the dissimilarity of other factors, such as agricultural use, soil texture, mineralogy, nutrient content and pH. Although the application of fertilizers, especially for podzolic soils, leads to differences in the results of genetic fingerprinting, it is incomparably small with the effect of biochar additions, which at times drastically changed the microbiological community. An increase in the application of biochar to the soils of the temperate zone increased the divergence of the bacterial community composition in the rhizosphere and soil. Rhizospheric soils with the application of 12 and 30 g/ha of biochar were characterized by the greatest dissimilarity from the soil, with minimal or no additional fertilization (0 or 1 g/ha) [
188]. It was concluded that the introduction of biochar into soils leads to the development of communities similar to rhizospheric ones, which were formed without the introduction of biochar.
Similar studies were carried out at the level of taxa. Thus, two new taxa of
Acidobacteria [
227], and later two potentially new taxa of α-
Proteobacteria were identified in the soils of Terra preta. It was shown that
Acidobacteria are widely represented in all studied soils and differ in their genetic profile from analogs included in databases by at least 2% of the code. Some genomes isolated from Brazilian soils enriched with biochar were grouped with 93% similarity with
Verrucomicrobia, whose genome is mainly found in tropical rice husks, but is characterized by a gradual spread to other soils. Genomes assigned to
Pseudomonas,
Acidobacteria and
Flexibacter sp. were found both in Terra preta soils and in control soils. In moderately rich soils with and without biochar additions, 70% of the isolated sequences are classified as
Ascomycota,
Basidiomycota or
Zygomycota. However, the occurrence frequency of the main phylotype genes differed for soils enriched and not enriched with biochar. In the presence of biochar, the communities were characterized by less genetic diversity. Similarly, less diversity was found in the Archaean community in Terra Preta soils, particularly in the ammonium-oxidizing
Chrenarcheota [
225]. Probably, this effect indirectly reflects changes in soil pH caused by the introduction of biochar [
228]. Soils enriched with biochar show multiple
Zygomycota involved in the degradation of glucose and cellulose and forming
Glomeromycota mycorrhiza. At the same time, the diversity of
Basidiomycota decreases by a third compared to unenriched soils. Some
Ascomycota are known to degrade lignin but can also use simpler organic compounds as a substrate. In this regard, the lack of available carbon in biochar can inhibit the colonization of this fungal species, while soluble carbon adsorbed on the surface of biochar particles will increase the proportion of
Zygomycota that apparently find sufficient resources of oxidizable carbon.
Similarly, under the influence of biochar, the bacterial community also changes. In response to the introduction of biochar obtained in high temperature pyrolysis (pyrolysis products of oak wood and a mixture of herbs at 650 °C), bacterial diversity increases, including taxonomy, in contrast to the effect of biochar obtained at 250 °C [
226]. An increase in the diversity of
Actinobacteria and
Gemmatimonadetes was found, in agreement with similar observations of Terra preta soils and forest soils naturally fertilized with fire coal [
229]. Separately, the effect of biochar on pathogenic microorganisms should be mentioned. The application of biochar to soils can increase
E.
coli contamination of groundwater, especially since current approaches do not consider reducing microbiological diversity in soil moisture as a goal. The transfer of
E. coli to soil and ground moisture depends on the biochar application rate and the pyrolysis temperature. Thus, high temperature biochar (700 °C) from poultry litter at a dose of 2% applied to sandy soils does not reduce the number of
E. coli in the leachate; increasing the dose to 10% (not realistic in real land use for economic reasons) reduces the number of microorganisms by several orders of magnitude [
230]. Soil treatment with the same biochar, produced at 350 °C, increases the abundance of
E.
coli itself, which should be taken into account when assessing the consequences of its use in agriculture.
Along with the conservation of carbon and the reduction of the greenhouse effect in general, the introduction of biochar into soils has a general soil-improving effect, mainly associated with agronomic and environmental factors. There are various assumptions about the nature of such an effect, but most of them associate biochar with improved storage of soil moisture and nutrients, improved soil structure and drainage [
231]. There is evidence of a connection between the application of biochar and the state of the soil microbiological community. These factors also affect the yield of agricultural plants, sometimes indirectly, for example, through the acceleration of nitrogen fixation by free and symbiotic diazotrophs [
232]. The ability of biochars to absorb pollutants should also be taken into account, thereby reducing their availability for biota during the restoration of damaged soils and the neutralization of production wastes [
203]. At the same time, such processes can lead to secondary contamination of soils with the same pollutants in the future during the decomposition of biochars.
Biochar stability in soil, i.e., its resistance to degradation, leaching and chemical oxidation, depends on the aromatic structure, surface functional groups, and sorption characteristics for minerals and organic compounds [
233]. Biochar destruction proceeds under the influence of biotic factors (microbial community) due to photooxidation and dispersion [
234]. Observations of freshly prepared biochar during the year showed that a decrease in its mass is accompanied by an increase in the number of surface oxygen atoms in the composition of carboxyl and phenolic groups, a decrease in the positive charge of the surface and the acquisition of a negative charge by it [
138]. The O/C ratio determines the stability of a biochar, as expressed by its half-life, from >1000 years (O/C < 0.2) to <100 years at O/C > 0.6 [
181]. It is difficult to accurately determine the lifetime of biochar in the soil, since the inhomogeneity of samples and the tendency of biochar to modify depending on environmental conditions increase during exposure.
3.4. Mineralization of Biochar
Biochar has a high sequestration potential due to the conversion of easily accessible C from plant biomass, organic waste, etc., into a hard-to-reach form, due to the absence of a priming effect when used as a soil improver, due to non-energy-intensive production (the resulting pyrolysis fuel and pyrolysis gas ensures the maintenance of the process, without the use of additional fuel sources), as well as by increasing the frequency of application (every 3–10 years) compared to other organic fertilizers, such as compost or digestate [
22,
235,
236,
237,
238,
239,
240,
241,
242]. Evaluation of retention time of biochar carbon in soils varies from hundreds to thousands of years, while biomass carbon is retained for several decades [
243]. The introduction of biochar derived from biomass reduces the return of carbon to the atmosphere in the form of carbon dioxide. Thus, provided that the gases released in parallel, as well as the products released into the ecosphere during the production, transportation and storage of biochar, do not compensate for the positive effect, the introduction of biochar contributes to a decrease in the greenhouse effect [
244]. The manifestation of such an effect depends on the degree of carbon conservation in the biochar, i.e., on the ratio of carbon content in biomass and the same value in biochar obtained from the specified amount of biomass. Slow pyrolysis gives a carbon conservation value of about 50%; higher values are typical for a less stable biochar with a stable existence time in soils of 4 to 29 years [
245].
The rate of natural mineralization of coals under natural conditions is usually low. Its evaluation is difficult because the changes over a reasonable period of observations are too small and because of the difficulty of unambiguous identification of the chemical nature of the resulting products. One of the methods of such an assessment is the use of biomass enriched in the
14C carbon isotope in the production of biochar. In particular, it was experimentally possible to prove the mineralization of 6% biochar, obtained in this way from a chaff, to CO
2, during an 8.5-year experiment. This corresponds to the processing of 0.3% of the carbon introduced per year [
246]. In another laboratory experiment with the carbonization products of
14C-labeled Pyrenean chaff, the rate of mineralization was estimated from the rate of
14CO
2 release after the introduction of coals into loess soils [
247]. It amounted to no more than 0.5% of the introduced carbon per year, which, in terms of real conditions, gives a lifetime of coals in soils of at least 2000 years. At the same time, the joint application of glucose to intensify the co-metabolism of soil bacteria reduces this time by more than 10 times.
The literature provides a fairly wide range of data on the carbon footprint of biochar, from 0.04 tCO
2-eq to a net reduction of 3.9 tCO
2-eq per ton of raw material [
153,
248,
249,
250,
251,
252]. This wide range is obtained due to different pyrolysis feedstocks, different pyrolysis plants, and different system boundaries established during the biochar life cycle assessment. A similarly wide range is set for the price of biochar. According to calculations by Nematian [
253] and co-authors, the cost of producing 1 ton of biochar in the USA is USD 449–1847, in European countries EUR 300–2000 [
254].Calculations made for Russia showed that the cost of production of 1 ton of biochar is USD 40; at a cost of USD 110/t, biochar production becomes profitable, which is comparable to other soil fertilizers and immobilization agents. It is worth noting that the growing demand for biochar, according to the European Biochar Market Report 2022/2023 biochar production capacity continues to grow in 2022 by 52% to 53,000 t biochar [
252]. Indeed, the cost of compost in European countries and the USA is about 10 times lower than that of biochar and amounts to USD20–50/t. However, it is important to consider that when producing 1 ton of biochar in European countries, up to 4 tons of carbon credits worth EUR 70–370/t can be returned [
153,
254,
255]. In addition, despite the same application doses, the frequency of applying biochar is once every 2–5 years, in contrast to the annual application of compost. In connection with the above, the most rational thing is to calculate the carbon footprint of crop products grown using biochar, as well as to change its cost taking into account the carbon units obtained.
3.5. Biochar Influence on Soil Microorganisms
Biochar influences many soil processes, such as denitrification, methane oxidation [
256,
257], carbon mineralization [
210,
247] and the transformation of nutrients. The reasons for this are numerous. They include switching to other carbon sources, changing nutrient availability, sorption of inorganic and organic components, including enzymes, to biochar, soil moisture retention, and changes in infiltration or pore structure. Later, those that are directly or indirectly related to the microbial community of soils will be considered.
As mentioned above, the microbial community is able to actively respond to the introduction of biochar. Higher microbial diversity could potentially lead to greater mineralization or oxidation of the biochar itself, as shown for organic carbons of non-pyrolysis origin. Usually, these processes are stimulated by an increase in microbial biomass. However, some reports have indicated the opposite effect or a combination of decreased diversity and absolute respiration carbon (carbon turnover) [
210,
258]. This may be the consequence of the lower amount of available carbon, the high stability of biochar, or the adsorption of organic carbon on it, which could contribute to its slower degradation. Vice versa, the introduction of freshly prepared biochar from waste products usually increases both respiratory activity and community metabolism [
208]. It is explained, inter alia, by a high content of nutrients, such as nitrogen and phosphorus, as well as a significant proportion of organic biochar, which, in particular, follows from a slight activation of respiration with increasing biochar application rates [
259,
260]. In the same work, a positive connection was described between the amount of volatile organic components in the biochar and carbon dioxide released during incubation. Both of these factors, i.e., an increase in the nutrient component and labile carbon, directly follow the introduction of biochar into soils, and their overall effect on biochar mineralization depends on the ratio of labile carbon and nutrient components both in the biochar itself and in the soil inorganic matter.
From the same standpoint, it would be logical to expect a multidirectional effect of biochar on the structure of the microbial community. The shift toward a greater variety of fungi after the introduction of biochar into the soil potentially indicates a greater mineralization of the biochar itself. Root fungi are known to promote the degradation of lignin in woody biomasses and coal [
261,
262]. Interestingly, among fungi, there is a shift in diversity toward taxa that prefer glucose as a source of carbon, and among bacteria, in the opposite direction. It is not entirely clear how the introduction of a much more oxidatively tolerant biochar could contribute to the development of such preferences, especially given the wide variety of other carbon sources available in soils (own organic matter, litter, etc.). It is possible that mineralization of biochar does not increase access to labile carbon but promotes the mineralization of available non-pyrolysis carbon. This statement is consistent with observations showing a connection between an increase in microbial biomass and a higher rate of soil carbon decomposition (so-called priming) in the presence of biochar. The fact that this increase usually remains below the initial higher salinity when freshly prepared biochar is added [
209] suggests that there are other mechanisms leading to carbon loss, such as physical carbon removal, changes in nutrient content or pH. In addition, volatile carbon compounds present in biochar, along with similar smoke components, can stimulate microbial activity immediately after the application of biochar to soils, but then quickly mineralize [
263,
264]. Longer-term (more than a year) observations show that biochar reduces the rate of soil carbon mineralization [
137,
210,
265]. A similar situation (more biomass but less soil respiration) was also registered in liquid waste treatment, when the biofilm on irrigated fields showed a higher rate of mineralization of soluble aromatic carbon than the same film on activated carbons with a larger specific surface area of the latter [
266]. It is possible that carbon dioxide forms carbonates on the surface of the biochars due to the higher pH value of their surface. This explains the decrease in the amount of recorded CO
2 with a simultaneous increase in microbial biomass.
Changes in the composition and enzymatic activity of the microbial community upon the introduction of biochar into soils may be responsible for the lower mineralization of soil carbon. The activity of glycosidase and cellobiosidase decreased with the introduction of more than 12 t/ha of biochar. In addition to soil analysis, a similar change was recorded for the purified enzyme and biochar obtained by rapid pyrolysis from
Panicum virgatum [
200]. In such experiments, it is also necessary to take into account the localization of microorganism colonies on biochar particles. Their proximity to a carbon source increases the efficiency of carbon mineralization, even without additional enzyme production. An alternative explanation associated with the assumption of adsorption of enzymes on biochar seems less probable; for example, lipases form stable and highly effective adducts with activated carbons [
267]. Biochar particles can generate regions of local enrichment in available carbon and thereby promote the growth of microbial colonies, as occurs in biological wastewater treatment [
268]. In the latter case, immobilization of biochars also reduces the possible toxic effect of organic waste on microorganisms, which contributes to their higher metabolic activity [
269]. Biochar can adsorb large amounts of soil organic carbon, which was demonstrated by studying microbial cultures and the processes of substance leaching from soil horizons [
270]. Numerous evidence of strong adsorption of aromatic hydrocarbons on various kinds of coals and soot also proves this statement [
271]. Although such processes, with the inclusion of microbial biomass and litter, are slower than the sorption of PAHs, they can significantly determine the influence of coals on the mineralization of soil carbon. The influence of biochar on multicellular soil organisms is of great interest from the point of view of monitoring the state of the microbiological community based on the following considerations: soil organisms are included in the general flow of soil matter and energy, they are present at the upper levels of the food chain, and their reactions are derived from the reactions of microorganisms to the introduction of biochar. Then, geophages, for example, earthworms, are important participants in the processes of biochar modification and its transfer to other soil horizons, which cannot but affect microorganisms. Finally, the reactions of soil organisms to toxic elements may reflect corresponding changes in the structure and abundance of the microbiological community.
The interaction of biochars with earthworms has been the most studied. They are able to absorb particles of coal, crushing them along the way and mixing them with the soil. In a number of experiments with microcosms, it was shown that worms prefer mixtures of soils with biochar over pure soils [
180]. Perhaps they use coal particles to ground soil organic matter, or they are attracted to microorganisms and their metabolic products that accumulate in the pores of the biochar. Long-term plot experiments have shown that worms also contribute to the transportation of biochar particles within the soil layer but most likely not beyond it. At the same time, in similar experiments with PAH-contaminated biochar, a decrease in the mass of worms was observed in comparison with the inhabitants of the same soils without biochar [
272]. The joint enrichment of soils with biochar and worms increased the concentrations of inorganic nitrogen, as well as the productivity of agricultural crops (for example, rice [
273]). Data on the influence of biochar on nematodes are scarce. Soils treated with smoke from the production of activated carbons show a higher number of nematodes, which may be due not to the direct action of the components contained in the pyrolysis products but to their effect on competitive interaction in soils. The same can be said about arthropods, the change in the abundance of which in soils enriched with coals was established from the products of their vital activity.
3.6. Effect of Biochar on Crop Yields
Biochar application usually improves crop productivity by increasing the availability of nutrients, increasing the activity of microorganisms that determine the mobility of soil nutrients [
274,
275,
276,
277], and accelerating the development of the plant root system because of colonization of arbuscular mycorrhizal fungi and improved soil physical properties [
278]. There is also a decrease in the loss of nutrients due to their reduced leaching from soils and an improvement in the physical characteristics of soils that contribute to water retention and oxygenation [
279,
280]. There is also data on the absence of the biochar effect or even its negative effect on crop yields. In
Table 8, there is an example of generalization of the biochar effect on crop yields.
Table 8 also contains information on the application rate and statistical parameters of the assessment [
281,
282].
When evaluating the contribution of biochar to soil fertility, it is necessary to take into account all aspects of a complex interrelated system that includes, in addition to biochar and soil, a specific agricultural crop, inorganic and organic fertilizers used in parallel, biochar application volumes, climatic and other environmental conditions (
Table 8).
In
Table 9 the results of testing various types of biochar in a field experiment are given. The effect of biochar on crop yield depends on soil type. For example, corn showed a 2–3-fold increase in yield when applied at 4 t/ha in acid sandy soils and only 30% to 40% when biochars were used in sandy-clay acid soils. In neutral clayey soils, no effect of biochar application was found.
In the study [
295], a 50% increase in the yield of chaff was established on slightly acidic fatty loams and 44% on calcareous sandy soils. In Australian calcareous soils, a negative effect of biochar on the yield of lettuce and hay grass was registered [
296]. Technological aspects complicate the dependence of yield on the type of soil, cultivated crops and the method of biochar preparation. Thus, in a greenhouse experiment, a decrease in the yield of corn in the first year of cultivation was registered when biochar from eucalyptus wood obtained in fast pyrolysis at 800 °C was introduced into acidic marl soils [
297]. At the same time, biochar did not affect this value when applied to weakly acid silty loams. When using biochar obtained in slow pyrolysis at 350 °C, no increase in yield was observed for either of the two soil types. Different results were obtained in the second year of the experiment: after the introduction of biochar obtained in slow pyrolysis, the yield increased by 500% on slightly acidic loams and by 150% on acidic marl soils. The authors attributed the reasons for the decrease in the efficiency of using the fast pyrolysis product to the possible presence of PAHs and the antagonistic effect of excess potassium on calcium and magnesium, although reliable results for measuring these chemical compounds are not given in the work. However, an increase in yield in the second year of biochar application is recorded quite often due to an increase in the cation exchange capacity of soils, the availability of nutrients, the ability to retain water and a decrease in the mobility and toxicity of aluminum and manganese [
296,
298].
The direct or indirect effect of biochar on the growth of the root system and phytotoxic compounds indirectly affects crop yields. The acceleration of root culture development is due to the general improving effect of biochar on the soil structure, including its aeration and moisture saturation. Moreover, more than a hundred years ago, it was already believed that root development was accelerated due to the sorption removal of allelopathic compounds by activated carbons and similar products. At the same time, it should be taken into account that, most likely, the sorption removal of allelopaths and other phytotoxic compounds, including those released from litter and other plant residues, is complicated by the complex effect of biochar on the availability of nutrients, which leads to contradictory results in laboratory experiments [
299]. In a number of studies, biochar was used as a substitute for peat in a nutrient medium for growing plants (soilless method). At a content of less than 30 wt%, biochar had a positive effect on plants, which was manifested in improved seed germination and seedling survival. In a number of studies, biochar was used as a substitute for peat in a substrate for growing plants (soilless method). Peat moss is conventionally used as a container substrate; however, this use causes several environmental concerns, such as peatland ecosystem destruction when peat is obtained, over-use of fertilizers since peat itself does not provide plants with essential nutrients, and carbon release since peat organic matter is quickly decomposed by plant-associated microbes. Biochar seems to have the potential to alleviate these concerns; however, the number of publications on peat substitution by biochar is still quite low as compared with the total number of publications on biochar used as a soil conditioner, suggesting that this study direction will be developed in the future. In most cases (depending on the initial substrate and pyrolysis process characteristics), biochar has a larger surface area, better pore size distribution, better rewetting characteristics, and higher content of N, P and other essential elements [
300]. Examples of assessing the impact of biochar in the soilless method of cultivating a number of crops are shown in
Table 10.
It is necessary to discuss separately the effect of biochar on plant diseases [
308]. Even 170 years ago, there was a decrease in rust damage in wheat and powdery mildew in other crops. Since then, the effect of biochar has been tested on 13 pathogens, of which 85% have responded to biochar application by reducing plant infection, 12% have not, and 3% have increased plant disease. Later [
309], the list of potential pathogens was expanded to 15 (30 plant/pathogen pathosystems). In 60% of the cases (70% of pathosystems), the maximum biochar concentration of 0.5–1% did not have any effect or had a negative effect on plant disease. Moreover, increasing the dose of biochar to 3% did not affect or accelerate the course of the disease. The study covered 12 different environments and 5 different prepared biochars, differing in salinity, carbon content, alkalinity and other parameters. The negative effect of biochar was less pronounced for foliar infections compared to soil pathogens. Other examples of assessing the impact of biochar on pathogens and plant infections are given in the review [
310]. Examples of the acceleration of pathogen effects on plants are given in
Table 11.
Strengthening the influence of pathogens under the effect of biochar often coincides with its suppressive effect on the development of the plants themselves, which manifests itself at high application rates. Moreover, even in the case of a positive effect on the growth rate of plants, the risk of irreversible damage to plants in the event of infection is potentially high. The contrary is also true—infected plants are more sensitive than healthy ones to an excess of applied biochar. The reasons for this phenomenon are not fully understood, but similar dose-dependent effects were observed for other agrochemicals, for example, glyphosate, which acts as a growth regulator at low concentrations, and as a herbicide at high concentrations. Biochar may contain certain organic components that, individually or in combination, have hormone-like or phytotoxic effects. Ethylene released from some biochars can serve as an example of such an indirect effect. In small doses, it has an effect on plants as a growth promoter and increases resistance to diseases, and in large doses, it has an opposite effect.