4.1. Effect of Temperature on Feeding of M. aeruginosa by M. mongolica
In the investigation, temperature was found to have a significant effect on the feeding of
M. aeruginosa by
M. mongolica. According to relevant observations, 28–32 °C is generally reported as the most appropriate temperature range for the population growth of
M. mongolica [
11,
12]. In the experiment, the most significant intake of
M. aeruginosa by the two stages of
M. mongolica occurred at 30 °C. This study also revealed that the most suitable temperature range for
M. aeruginosa to be consumed by
M. mongolica was 25–30 °C. With the increase in temperature, time, and life activity, the feeding behavior of
M. mongolica was correspondingly promoted, which was reflected in the increase in feeding rate in the later period. A previous investigation reported that when the temperature of
M. mongolica was 15 °C or lower, part of its reproductive mode could be changed from parthenogenesis to hermaphroditism, and its filtering ability was also decreased [
16]. In the experiment, the feeding rate below 25 °C was low, which verified the above assertion.
The feeding rate of
M. mongolica in different periods exhibited a decreasing trend, which may be associated with the starvation treatment before the experiment. The feeding rate of Pacific
Spinoza was much higher than that of regular feeding after it was placed in a starving environment for a period of time [
17]. Additionally, the feeding ability of
Daphnia magna to
Chlorellapyrenoidosa under starvation also increased significantly [
18]. Moreover, Ferrão–Filho and Kozlowsky–Suzuki [
19] demonstrated that some cladhorn species that are highly sensitive to starvation are also highly sensitive to
M. aeruginosa. Their work indicated that
M. mongolica might not be strong enough to restrict the growth of toxic cyanobacterial blooms like
Daphnia magna.
M. mongolica is a small cladoceran, and its feeding ability is slightly less than that of
Daphnia magna. However,
M. mongolica is a saltwater species and can survive in salt waters and low-salt coasts. Now, only
M. mongolica can be used to restrict cyanobacteria blooming occurring in saltwaters and low-salt coasts, and we can select good strains from
M. mongolica species and increase the efficiency for restricting toxic
M. aeruginosa in the future.
4.3. Effect of Salinity on the Feeding of M. aeruginosa by M. mongolica
Although salinity significantly differed in
M. mongolica feeding on salt-tolerant
M. aeruginosa, no clear trend was found. This may be attributable to the broad salt characteristics of
M. aeruginosa on the one hand and the response of
M. aeruginosa to salt stress on the other hand. When the salinity was high, the feeding rate of
M. mongolica was low, which may be associated with the effect of salt stress on the growth of
M. aeruginosa [
24]. It was reported that approximately 7‰ may be the salt tolerance range of most
M. aeruginosa [
25]. Overall, the higher the salt level, the stronger the growth stress. Furthermore, the higher the level of salt, the more
M. aeruginosa cells would die, which is not conducive to feeding
M. mongolica. Research also demonstrated that the feeding rate of
M. mongolica was high at low salinity (5–10‰) [
12].
M. mongolica is characterized as being of saltwater origin and possessing the ability of two-way hypertonic and hypotonic regulation. The hyperosmotic capability of
M. mongolica in low salinity water can also promote its appetite. It was determined that the feeding rate of juvenile and adult
M. mongolica changed significantly with salinity, indicating that
M. mongolica was sensitive to salinity changes. Some studies also examined the effects of pH and salinity on the feeding of
Daphnia magna in different growth stages and found that
Daphnia magna at different ages exhibited a trend of low promotion and high inhibition in response to changes in the external environment. In addition, juvenile
Daphnia magna were more sensitive to changes in salinity [
26], which is in accordance with the results of this study.
4.4. Molecular Mechanism of the Decrease of Survival Rate of M. mongolica under Toxic Microcystis Stress
Most
Microcystis spp. can produce the hepatotoxin microcystin (MC) [
6,
27,
28], and MC would inhibit protein phosphatases [
29,
30] and damage DNA through the promotion of oxidative stress [
31]. Lyu et al. [
6] studied the transcriptional analysis of
Daphnia clonal variation in tolerance to toxic
Microcystis and reported that major MoAs (modes of action), such as glutathione metabolism, protein processing in endoplasmic reticulum, amino sugar/nucleotide sugar metabolism, and arachidonic acid metabolism were linked to tolerance fitness in
Daphnia similoides. Most
Daphnia would be negatively affected by toxic
Microcystis, as toxic
Microcystis could contribute to enhanced mortality, abnormal development, and lower reproduction [
32,
33].
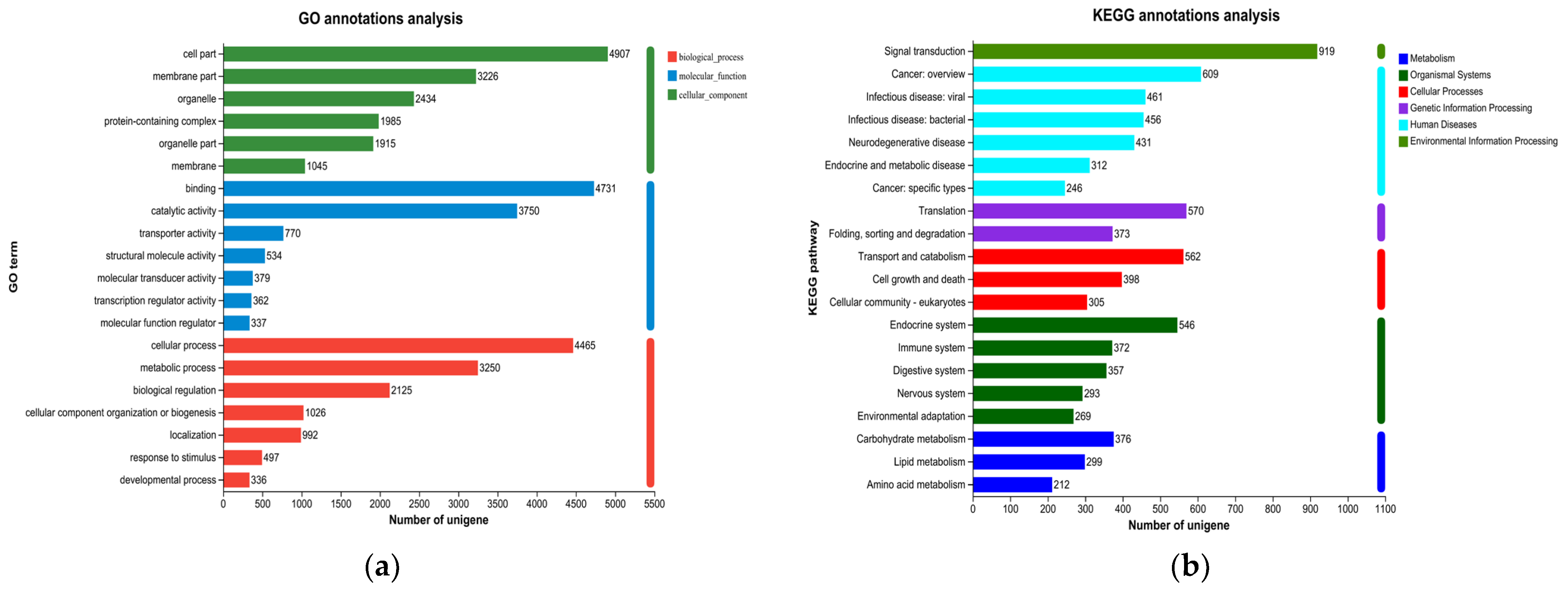
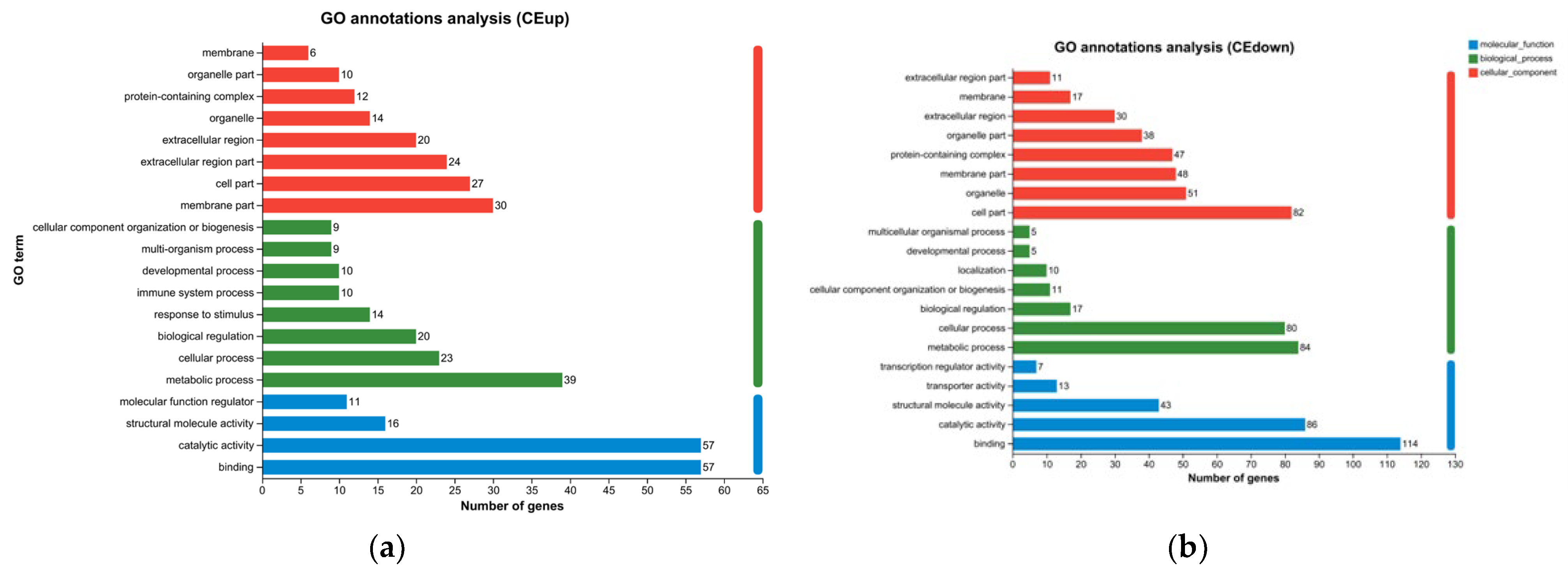
The study systematically explained the molecular mechanism of the decline in the survival rate of M. mongolica under the stress of toxic microcystins at the transcriptome level. Through transcriptome data, it was determined that in the GO library classification, with the toxic microcystins FACHB-905 as the bait, the differential gene changes in M. mongolica mainly belonged to three categories. Combined with KEGG significant enrichment analysis, it primarily belonged to six categories (except drug development). The above results indicate that the inhibition of the population growth of M. mongolica was closely related to the internal gene and protein pathway, revealing the molecular mechanism of M. mongolica in response to the stress of M. aeruginosa from the molecular level. These findings provide helpful information support for the research of cladoceran ecotoxicology, further achieving the early warning function for the monitoring of microcystin blooms.
As the test organism of potential water environment pollution monitoring, the paper also demonstrates the feasibility of
M. mongolica as a model organism. The available literature has shown that the toxic effects of
M. aeruginosa on cladoceran mainly lie in the destruction of ribosomal protein synthesis, interference with the digestion pathway, and inhibition of the protein phosphorylation pathway [
34]. Liu et al. [
35] observed that polystyrene nanomaterials function to induce functional genes of
Daphniapulex to change in oxidative stress, immune defense, glucose metabolism, etc., depending on RNA sequencing. In this experiment, with the assistance of high-throughput sequencing technology, we investigated the molecular mechanism of
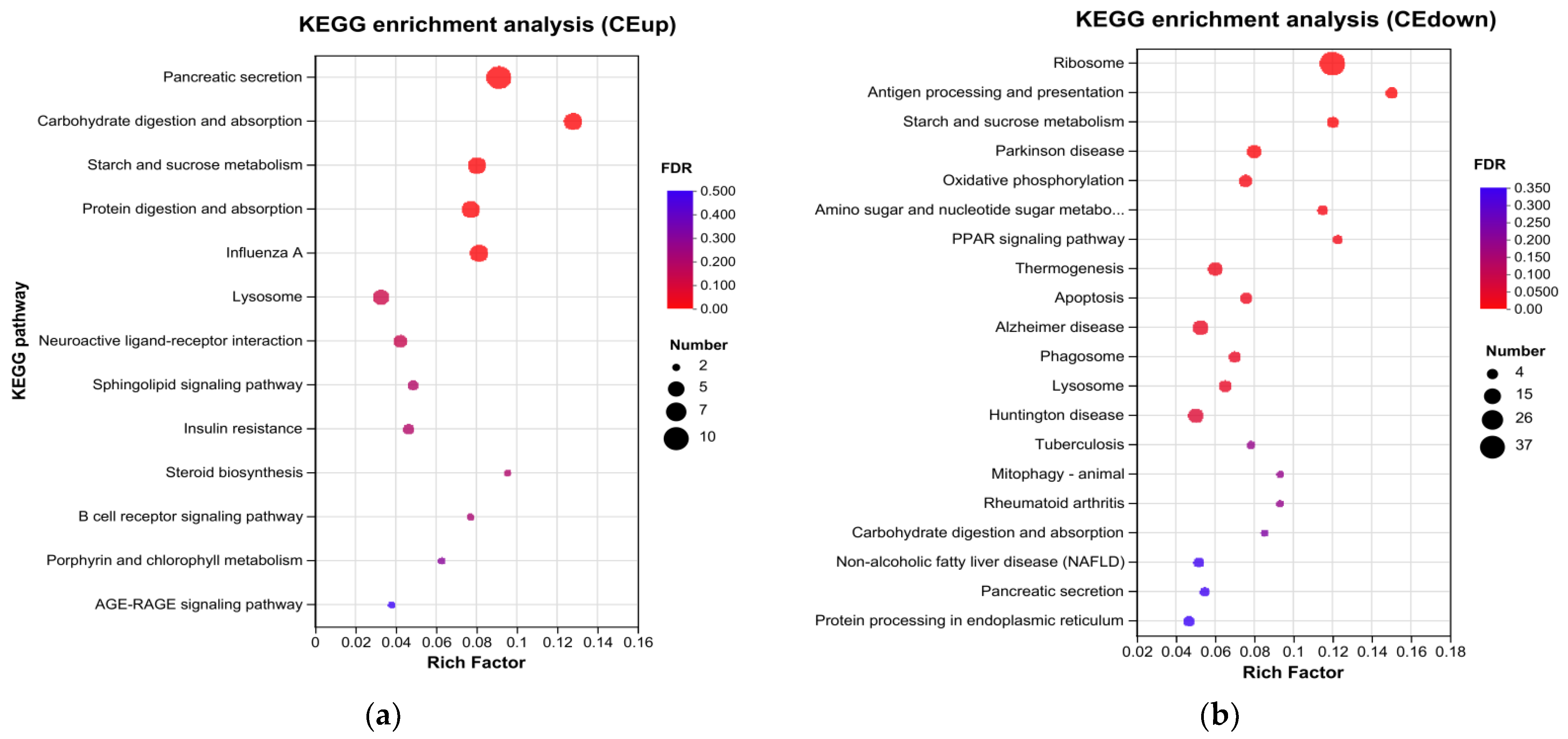
M. mongolica in response to the stress of the microcystins toxin. A total of five pathways were found to be significantly upregulated after enrichment. They mainly comprised protein digestion and absorption, pancreatic secretion, starch and sucrose metabolism, and lysosome and carbohydrate absorption. Among them, in the carbohydrate digestion and metabolic absorption pathway, Alpha-amylase (α-amylase) was significantly upregulated under the stress of toxic microcystins. In carbohydrate metabolism, the tricarboxylic acid (TCA) cycle is the final oxidative decomposition pathway of carbohydrates, lipids, and amino acids.In the protein digestion and absorption pathway, to cope with stress,
M. mongolica sustained its growth by absorbing protein and other nutrients, as shown in the significant upregulation of trypsin, which speeds up the operation of the digestive system of
M. mongolica. Klumpen et al. [
36] even reported that the white matter biosynthesis gene was downregulated and the carbohydrate metabolism gene was upregulated in
Daphniapulex under starvation conditions.
Relevant studies have confirmed that toxic microcystins have low nutritional value and are not a high-quality food, and, thus, that it is necessary to strengthen carbohydrate absorption [
37,
38]. A-amylase can synthesize high-energy substances in numerous ways, commonly known as ATP. Therefore, the polysaccharide enzymes and starch-related proteins were significantly upregulated, which may be related to the stimulation of microcystin.
M. mongolica needs to consume more energy and absorb nutrients to resist interference of the external environment. Therefore, it needs to obtain more carbohydrates and protein, which can be converted into nutrients to promote its growth to resist external stress. In the lipid metabolism pathway, lysosomal aspartic protease was significantly downregulated, while sphingomyelin phosphodiesterase was significantly upregulated [
39]. Aspartase is an acid protein enzyme that can degrade proteins, promote antigens, and activate enzymes. It is mainly reflected in the pathological immune process [
40]. Sphingolipid phosphodiesterase is a very important hydrolase in animal metabolism. It can induce apoptosis and cell differentiation under external stimuli and plays an essential role in preventing animal poisoning. On the basis of the enriched KEGG pathway, we can find that ribosomes are significantly downregulated genes. Ribosomes serve an essential function in cells, and their primary function is translating into proteins. Their downregulation will inhibit the protein synthesis of
M. mongolica. In addition, some studies have found that the decline of ribosomal function may be an important way for the body to cope with the lack of energy supply due to mitochondrial damage during aging, which may realize healthy aging through the redistribution of energy [
41]. Therefore, the significant downregulation of ribosome pathway-related genes may constitute one of the stress modes of
M. mongolica in response to the toxicity of
M. aeruginosa. Since the endoplasmic reticulum plays a role in transporting proteins, it can effectively prevent proteins from being misfolded [
42]. Protein disulfide isomerase is mainly used to secrete and catalyze proteins, promoting disulfide bond enzyme folding to the correct position. Heat shock proteins and their partners can also repair some affected misfolding proteins on this basis. However, it was found that protein disulfide isomerase and heat shock protein (Hsp70) were significantly downregulated in the toxic
M. aeruginosa group, resulting in the aggregation of misfolded proteins in the cell body. According to the latest literature, ROS is an early (pre-injury) stress signal, and protein defect is a late (post-injury) stress signal, which can trigger the heat stress response [
43]. ROS is induced in large quantities that are far beyond the scavenging capacity of the body’s antioxidant system [
44]. The oxidative phosphorylation pathway was also downregulated, which may be caused by microcystin. MC toxin is water-soluble and stable in molecular structure. Once it enters the organism, it will combine with protein phosphatase and inhibit protein phosphorylation. ROS is induced by MC in large quantities, causing lipids to be in a state of oxidation, resulting in redox damage and then interfering with the cell signal pathway.
Moreover, the KEGG pathway shows that the genes related to porphyrin and chlorophyll metabolism were significantly upregulated. Although chlorophyll does not exist in
M. mongolica, the porphyrin metabolism pathway may be activated under the action of
M. aeruginosa. The porphyrin molecule can be used to synthesize heme, whereas heme generally exists in cladoceran, which plays a role in regulating oxygen content in the body. The upregulation of related genes may be an adaptive mechanism of
M. mongolica to cope with oxidative stress. Nucleotides and other energy synthesis come directly from ATP. The downregulation of ATP lipid-binding protein is unfavorable to nucleotide synthesis and DNA and RNA synthesis [
45].
The amino sugar and nucleoside sugar metabolism pathway is also a significantly enriched down-regulation pathway, mainly related to the catabolism of sugars. The downregulation of this pathway indicates that
M. mongolica cannot perform standard sugar decomposition when feeding on toxic
M. aeruginosa, and thus there are few ways to obtain energy, damaging the growth and metabolism of animals. In the process of amino sugar and nucleoside sugar metabolism, chitinase directly affects animal molting behavior, and its downregulation will also influence animal development and growth [
46]. Furthermore, the down-regulation pathway includes apoptosis, phagosome, antigen processing and display, PPAR signal pathway, etc. These pathways are primarily related to the cellular processes of
M. mongolica, in which phagocytosis is directly related to the animal immune system. First, phagosomes are formed in the body, and their phagocytic response is to resist the invasion of toxic substances from the outside world. However, the downregulation of the phagosome pathway directly affects the body’s immune system [
47]. The PPAR-signaling pathway is mainly used to protect and immunize adipocytes, avoid lipid peroxidation, and protect the body from damage [
48]. Therefore, the blockage of the PPAR-signaling pathway will inhibit the natural immunity and growth ability of
M. mongolica. These down-regulation pathways will have various genetic information, and the long-term growth of the population will also be affected.