1. Introduction
The American lobsters,
Homarus americanus (Milne-Edwards, 1837), is found in the Northwest Atlantic Ocean, between Newfoundland (Canada) and North Carolina (USA). This species is one of the most important exploited crustaceans in the Northwest Atlantic and its fishery represents the major local economy for many North Atlantic coastal communities. The total catch reported for this species by the FAO (Food and Agriculture Organization) in 2016 was 162,547 t. In 2017, the countries with the highest catches were Canada (97,452 t) [
1] and the United States (62,006 t) [
2]. American lobster is the most valuable species for Canadian fisheries (
$1.4 B), contributing to 45% of the whole commercial value of all fisheries in Atlantic Canada in 2018 [
3].
However, this economy has become threatened by another important sector in Canada. The oil industry. Indeed, Canada is one of the world’s largest oil producers, benefiting from the third-largest oil reserves at the global scale, mostly in the form of unconventional oil (including diluted bitumen). In 2019, the proven crude oil reserves in Canada accumulated 168.5 billion barrels, of which 164.1 and 4.4 billion barrels were extracted from oil sands and conventional sources, respectively [
4]. Most of the crude oil produced in Canada is exported to international markets for further refining. In 2018, 1.7 million barrels of oil (96 billion liters) were shipped daily to domestic refineries [
4]. Domestic scales of petroleum products were 1.9 million barrels per day (110 billion liters), with 30% represented by diesel [
5]. The exploitation and transport of this resource are still increasing, concomitantly with the demand for petroleum products [
6]. Although the frequency of large oil spills from tankers has been decreasing worldwide [
7], inshore fisheries remain vulnerable to such a risk. Coastal areas are more likely to be exposed to small and medium petroleum product spills (<1000 m
3), due to increased human activities [
8], with small diesel spills (10–100 m
3) being the most common in Canada, following an annual frequency of 0.6 [
7]. Two small spill events occurring in the Gulf of St. Lawrence (eastern Canada) illustrate that for lobster fishery, the risk of exposure to hydrocarbons is real. In September 2013, 5 m
3 of heavy fuel were released into the marine waters of the Baie de Sept-Îles from an inland spill of 450 m
3, caused by human error at an industrial site. In September 2014, 100 m
3 of diesel leaked in Cap-aux-Meules Harbour (Îles-de-la-Madeleine), due to a failure in a pipeline that annually pipes 40 million liters of diesel from this harbor to a local power plant. Globally, there is an increased probability of small to medium spill events of various petroleum products, from diesel to diluted bitumen, due to the ongoing expansion of routine shipping operations and coastal activities.
Oil spills contaminate sediments [
9] and the accumulated compounds degrade slowly [
10]. Benthic invertebrates can take up hydrocarbons by feeding on contaminated material, or through direct absorption from sediments or water [
11,
12]. For lobsters, this results in high levels of PAHs, which can then persist over years: up to six and seven years after for the Braer [
13] and Amoco Cadiz [
14] oil spills, respectively. Significantly elevated concentrations of total aromatics were found in the hepatopancreas of American lobster and their decrease was measured only after 11 days of depuration [
15]. Few reviews have reported the impact of hydrocarbons on lobsters during oil spills or laboratory experiments, highlighting higher mortalities or lower abundances as major impacts [
16,
17]. For example, a loss of 9 million lobsters was estimated after the North Cape oil spill in 1996 (23,000 barrels of home heating oil), based on observed abundances in the affected area compared to reference sites [
18,
19,
20], in addition to the 2.9 million lobsters stranded on the beaches [
21]. In crustaceans, integument damage may appear following an oil spill, thus compromising the integrity of their epicuticle, which serves as a physical barrier between the environment and the more permeable inner layers of the exoskeleton [
19,
22]. Behavioral effects have also been observed in lobsters, such as an increase in the delay between noticing food and pursuing it [
23,
24,
25,
26], disorientated behavior, and uncoordinated movements [
27]. Sublethal effects of heavy fuel oil have been observed in red rock lobster (
Jasus edwardsii), such as a change in circulating immune cell populations, which recovered by the conclusion of a 96 h exposure period [
28]. The depuration of PAHs appeared to be particularly slow in red rock lobster and significantly elevated PAH levels were still evident after 10 days of depuration [
28]. Furthermore, exposed lobsters may be unfit for human consumption due to gill-tissue damage [
29], as well as deterioration in meat color and smell [
30]. As a matter of fact, tainting occurs when the organism accumulates enough hydrocarbons to cause an “off” flavor or odor in the seafood [
11]. These organisms are unsuitable for consumption until this tainting disappears, where associated delays can take from days to up to a year after exposure, depending on the species [
17]. A study of Williams et al. [
15] observed that the odor of raw treated lobsters differed significantly from that of the control, even after 21 days of depuration; however, cooking appeared to remove the oily odor from the raw lobster. Despite the risk of diluted bitumen (dilbit) or diesel marine spills, to the best of our knowledge, there have been no studies dealing with the impacts of a dilbit spill on American lobsters and no studies on the sub-lethal impact of a marine diesel or dilbit spill. In addition, due to the bioavailability of soluble compounds and persistent polycyclic aromatic, organic sulfur, and heterocyclic compounds, it can be assumed that light petroleum products (e.g., marine diesel) are more toxic than heavy petroleum products (e.g., dilbit), despite their greater tendency to bind receiving organisms and environments.
Sub-lethal levels of contamination are usually quantified using biomarkers as exhibited observable or measurable changes at the molecular, biochemical, cellular, or physiological level. Biomarkers can reveal the exposure of a living organism to a present or past chemical substance and its associated effects [
31]. The monitoring of such markers can facilitate the early detection of pathogens or anthropomorphic stressors. For example, McDonald et al. [
32] measured a significant induction of enzymatic activity in gray shrimp (
Penaeus aztecus), for individuals sampled near (<100 m) an oil platform. Among the major physiological functions, the immune system is a significant mechanism that reflects the individual’s health status. A deterioration in immune functions can quickly lead to morbidity or mortality [
33]. The efficiency of an individual’s immune system can be compromised when specific tolerance thresholds of environmental conditions are exceeded, thus increasing their susceptibility to pathogens [
34]. In crustaceans, immunity is supported by hemocytes, circulating cells that are involved in multiple physiological functions and homeostasis processes. Therefore, the study of hemocyte functions is a prime target for use in the determination of health status.
In the lobster industry, the Brix index is routinely measured from the hemolymph, to evaluate the global lobster health. Simon et al. [
35] identified the Brix index as the most accurate and practical biomarker among eighteen hemolymph parameters, for reflecting the state of energetic reserves in spiny lobsters. Indeed, the Brix index value is highly correlated with the hemolymph total protein [
35,
36,
37,
38], triglycerides, cholesterol, calcium, and phosphorus concentrations as well as with lipase activity [
35]. To measure the lobster health status, various physiological changes can be assessed by assaying total blood proteins with the Brix index [
39,
40]. As Battison [
41] pointed out, other applications could include the evaluation of lobsters subjected to different suspected exposure conditions to noxious or infectious agent(s). However, the effectiveness of the Brix index as an indicator of lobster quality following a petroleum product spill not yet known.
In this context, our study aims to test three biomarkers in addition to the Brix index, in punctured lobster hemolymph which is routinely monitored in industrial practices after contamination by two petroleum products: CLB or marine diesel. These are cell viability, lysosomal membrane stability, and activity of a phase I detoxification enzyme (ethoxyresorufin O-deethylase; EROD). An initial sub-lethal effect is easily measured using Trypan blue (TB) dye exclusion to assess cell viability, which is one of the most common methods. It is based on the ability of the membrane of viable cells to exclude the dye. Destabilization of the lysosomal membrane is another recommended biomarker for monitoring the impact of petroleum hydrocarbon pollution [
42] as the excessive lysosome activity induced by dissolved hydrocarbons leads to an increase of enzymatic activity and can cause the destabilization of their membranes. The measurement of this destabilization by the neutral red retention (NRR) assay provides a sensitive index of the cell condition that can be related to the grade of contamination or recovery. Finally, the ethoxyresorufin-O-deethylase (EROD) biomarker is often used to assess exposure of marine organisms to PAHs. The EROD activity is measured to observe the induction of phase I P450 enzymes [
43,
44,
45], which are responsible for xenobiotic biotransformation and are involved in reactive oxygen species production and accumulation. This enzyme family exhibits the highest levels in cell tissues of organs involved in food processing [
45,
46,
47], explaining why EROD activity is classically measured in the hepatopancreas for crustaceans. A few studies have measured this activity induction in hemolymph [
48], but not in lobsters. These four biomarkers were tested at the end of exposure, as well as over 3 weeks following contamination. We also intended to explore the influence of water temperature on lobster recovery, as it may modulate the efficiency of this operation [
49]. In order to check the quality of lobsters in the context of industrial sales, the olfactory, visual, and taste quality were tested after the recovery phase. In addition, this study provides new data on the sublethal impacts of unconventional oil and marine diesel spills on American lobster and their recovery capacity. For exploratory purposes, chemical analyses of lobster tissue were carried out after several months in a fishpond.
2. Materials and Methods
2.1. Conditions of Oil Spill Simulation
2.1.1. Oil Products
Two types of petroleum products were tested: diluted bitumen (dilbit) from Alberta Cold Lake Blend (CLB) and a classical marine diesel from New Brunswick (
Table 1).
2.1.2. Water Accommodated Fraction
In this study, lobsters were exposed to oil physically dispersed by the technique of water accommodated fraction (WAF). The WAF was prepared according to the standardized protocol of Singer et al. [
51], modified by Barron and Ka’aihue [
52] and Payne et al. [
53] for CLB and marine diesel, respectively. The WAF was prepared before each exposure by adding the tested oil products (1:10) to filtered natural seawater (0.2 µm), collected in Cap-aux-Meules, in a fluorinated polyethylene carboy with 20% headspace. The solution was gently mixed for 18 h then left to settle for 6 h after the cessation of stirring. The contaminated seawater was then recovered and diluted with filtered natural seawater for the exposure. The dilutions used were 0% and 60% of WAF for Control and CLB/marine diesel treatments, respectively.
2.1.3. Monitoring of Dissolved Aromatic Hydrocarbons by Fluorescence
During lobster exposure, the concentration of the aromatic hydrocarbon dissolved fraction of crude oil was estimated using the Cyclops-7 submersible fluorometric sensor, equipped with the “O” sensor (Turner Designs, San José, CA, USA) for the detection of crude oil. It is one of the five UV submersible fluorometers that are commercially available for in situ measurements of PAHs, and one of the most commonly used in delineating oil plumes in the field [
54,
55]. Its fluorescence optical specifications are excitation wavelengths at 325/120 nm and emission wavelengths at 410–600 nm. The Cyclops-7 sensor was controlled by a DataBank module, which was connected to a computer by proprietary software (Turner Designs, San José, CA, USA). The sensor was calibrated with an aqueous solution of 100 μg∙L
−1 of tetrasodium 1,3,6,8-pyretetetrasulfonate (PTSA), a highly water-soluble pyrene derivative (CAS 6528-53-6; Sigma-Aldrich, Darmstadt, Germany). The fluorescence measurements were used as an indicator of hydrocarbon concentration and, thus, were expressed in equivalent μg PTSA∙L
−1. The sensor response was linear within the PTSA concentration range used, from 5 to 550 μg∙L
−1. The measurements were made in situ in 15-liters exposure tanks.
2.2. Animals
One batch of lobsters (Homarus americanus; sex ratio 1:1), captured during the 2018 fishing season in Lobster Fishing Area 22 (LFA 22, Quebec City, Canada), was kept in a Merinov containment unit (without feeding) in Cap-aux-Meules, and maintained at 2 °C to induce natural winter dormancy. A total of fifty-four (54) lobsters were used for the experiment, including 30 females and 24 males (88.3 ± 0.5 mm mean length and 559.5 ± 9.5 g).
2.3. Exposure and Recovery
Two series of assays were performed, with similar exposure conditions, but two different thermal regimes for recovery. For each series, twenty-seven (27) lobsters were randomly assigned to nine (3 × 3) independent 15 L tanks (
Figure 1) of oxygenated natural seawater, in order to carry out the following treatments in triplicate: Control (uncontaminated natural seawater), Cold Lake Blend (WAF-CLB) and marine diesel (WAF-Diesel). Three lobsters were used per replicate per treatment per series. Exposure was carried out for 96 h at 4 °C with filtered natural seawater for Control and 60% diluted WAF for CLB and marine diesel (no feeding and water renewal). During the exposure, the physicochemical characteristics of the water were monitored daily (i.e., temperature, salinity, dissolved oxygen and ammonia).
At the end of the exposure, the lobsters were removed and placed in two series of new tanks filled with uncontaminated and unfiltered natural seawater, thus starting the depuration period. The recovery period is defined as the time required for an organism to return to its pre-exposure health state. This recovery time depends on the type of pollutant and the amount accumulated in tissues. At this stage, two thermal conditions were applied during the three weeks: series A, with a constant water temperature at 4 °C, and series B, with a water temperature gradually increasing from 4 to 9 °C. The tanks were cleaned, the water was renewed daily, and the physicochemical characteristics of the water were monitored daily (i.e., temperature, salinity, dissolved oxygen, and ammonia). The seawater physicochemical characteristics did not change during the assay, with a level of ammonia remaining lower than the toxic levels for lobster (5.2 ppm for adult lobsters at 5 °C [
56]). Finally, the dissolved aromatic compounds concentration of the crude oils was also measured daily, as described earlier (by fluorescence).
At the end of the three weeks of recovery, the lobsters were returned to open live holding tanks dedicated to long-term contention at a constant temperature of 2 °C. Three months later, the organoleptic conditions (odor and color, before and after cooking) and PAH concentrations of tissues were checked.
Throughout the study (exposure and recovery) the lobsters were not fed in order to approximate industrial conditions, where no feed is provided to the lobster holding tanks. In addition, as benthic invertebrates can uptake hydrocarbons through the consumption of contaminated prey, the absence of food avoids the input of contamination from food as well as an additional variable in the analysis of results.
The results presented for the exposure impacts combined the two series values for each condition. As the environmental conditions were the same for series A and B, the data of both were grouped together for each treatment (n = 2 × 3). Then, the results of the recovery were presented by differentiating the two temperature templates.
2.4. Hemolymph Sampling
The Hemolymph puncture allows for rapid and non-lethal sampling. For each experimental condition, each lobster was punctured during the exposure period at the following times: 0 h (before exposure) and 96 h (end of exposure), and at: 24, 72, 168, 336 and 504 h in the recovery period (i.e., 1, 2, and 3 weeks of recovery). The 0 h time point during the exposure period corresponds to the puncturing of lobsters just before placing them in their tanks with associated samples, representing the initial health status.
The hemolymph was punctured under the abdomen (after the first pair of pleopods) with a 3 mL syringe and a 23 G needle. Two 0.5 mL hemolymph punctures were performed: the first for the Brix index test and a second one, using a preloaded syringe of a PBS_EDTA mixture (0.5 mL; 15 mM), for the three other biotests (TB, NRR, and EROD).
2.5. Biotests
2.5.1. Brix Index
The Brix index (%) value was obtained by reading a 0.5 mL hemolymph volume in a refractometer (PAL-1; Atago), which had been calibrated beforehand using deionized water (0). The Brix index can be converted into a refractive index (RI), as follows [
57]:
The total protein (TP; mg/mL) in the hemolymph can then be determined, using the refractive index RI, as follows:
2.5.2. Cellular Viability
The viability of hemocytes was assessed by a membrane integrity test, the Trypan blue dye exclusion method. After the hemolymph puncture, as described above, 40 µL of cell suspension was added to 20 µL of Trypan blue dye solution (CAS 72-57-1). After 15 min, 20 μL of the mixture was deposited on a glass slide and observed using an optical microscope. Fifty cells were classified according to two categories: (1) Living cells, uncolored; and (2) dead cells, blue-colored.
2.5.3. Lysosomal Membrane Destabilization Index
The lysosomal membrane destabilization index was determined using NRR assay, performed according to the method described by Song et al. [
58] modified by Small et al. [
59]. A stock solution of neutral red dye (3-amino-7-dimethylamino-2-methylphenazine hydrochloride, C
15H
17ClN
4, Sigma) was prepared by dissolving 2.22 mg in 1 mL of dimethyl sulfoxide (DMSO, C
2H
6OS, Sigma-Aldrich). The dye stock solution was kept at 4 °C until required. A working solution was made daily by dissolving 8.5 µL of the dye stock solution in 500 µL of physiological saline solution (NaCl 19.31 g∙L
−1; KCl 0.65 g∙L
−1; CaCl
2 1.38 g∙L
−1; MgSO
4 1.73 g∙L
−1; Na
2SO
4 0.38 g∙L
−1; HEPES 0.82 g∙L
−1 adjusted to the pH of natural seawater). The hemolymph (0.2 mL) was transferred to an Eppendorf tube with physiological saline solution (0.2 mL). An aliquot of 40 µL was transferred to a positively charged microscope slide and incubated in a dark humidity chamber at ambient temperature for 15 min to allow cells to attach to the slide. Then, the working solution (40 µL) was put on the slide. After 15 min in the same humidity chamber, a cover slip was placed on the slide. After 45 min, normal and destabilized cells were counted using a light microscope. In healthy cells, neutral red is retained in the lysosomes, turning them red, while the cytosol is colorless. For damaged cells, the efflux of red dye into the cytosol results in redness of the cytosol. Increased cellular retention of neutral red dye, therefore, corresponds to a healthier lobster. For each sample, at least 50 hemocytes were analyzed. The lysosomal membrane destabilization index (LDI; %) was calculated as the number of hemocytes with destabilized lysosomes (DH), as follows:
2.5.4. EROD
The EROD activity test used for this study was based on the method described by Burgeot and Ménard [
60] and modified according to Monari et al. [
48] to adapt it to the hemolymph. The resorufin formation was followed by fluorimetry at 585 nm using a microplate reader (BioTek Epoch2), with an excitation wavelength of 530 nm. The fluorescence of the samples indicates the resorufin concentration by a standard curve. EROD activity is related to the protein concentration determined according to the Bradford assay [
61]. The activities are expressed in picomoles per minute and per milligram of protein:
2.6. Tainting Assay
An oil spill can affect the olfactory, visual, and taste quality of crustaceans, precluding their commercial value. Tainted seafood presents abnormal odor or flavor, atypical of the seafood itself [
62]. This degradation may persist beyond the recovery period [
63]. Three months after the experiments, one lobster per treatment and per series (for a total of 6) was sacrificed to perform olfactory and visual evaluations in fresh and cooked (after 15 min in boiling water) states. However, due to this low number of specimens, these qualitative results are only indicative.
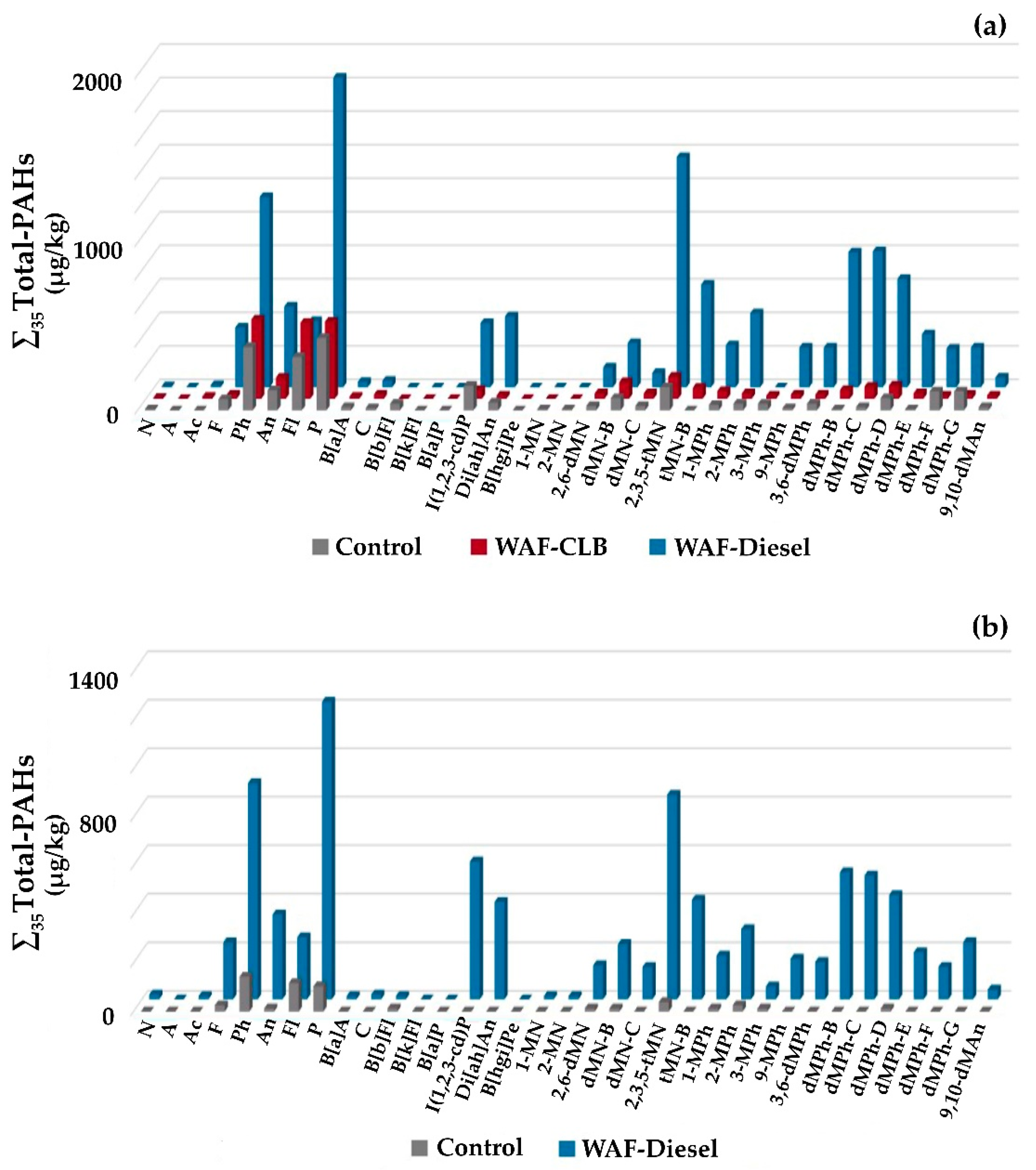
2.7. Tissues Analysis for Polycyclic Aromatic Hydrocarbons (PAHs)
After 3 months of live holding, 2 lobsters per treatment and per series (for a total of 12) were sacrificed and tissues were analyzed for their PAH contents. Lobsters were boiled (15 min) and several tissues were recovered for chemical analysis: abdomen, claw, and hepatopancreas. In addition, three females of the sampled lobsters (1 in Control and 2 for marine diesel treatment) had spawned during this stage, thus permitting us to also assess PAHs in the eggs. Tissues were kept at −20 °C until further analysis.
2.7.1. PAHs Extraction
Lobster tissues were freeze-dried (FreeZone, Labconco, USA) and homogenized into a fine powder using a Virtis homogenizer. A sub-sample of about 300 mg was suspended in 5 mL of tetramethylammonium hydroxide (TMAH, 25% water; Sigma Aldrich, Darmstadt, Germany) in a 12 mL glass tube with a Teflon-lined cap and vortexed for 1 min. The mixture was then heated (60 °C) for one h with manual stirring every 15 min, in order to complete the alkaline digestion. After cooling the mixture to room temperature, 1.0 mL of deionized water, 1.0 g of NaCl, and 4.0 mL of hexane/toluene mix (1:1) were added before stirring for one h (Wrist Action Shaker; Burrell Scientific, Pittsburgh, PA, USA). The mixture was centrifuged (at 3000 g), and the upper organic layer was recovered. A second extraction step was performed with another 4 mL of hexane/toluene mix (1:1) and this recovered organic layer was added to the first one. The whole organic extract was then cleaned on a silica column topped with sodium sulfate. The volume of the cleaned extract was reduced under a gentle stream of nitrogen, at room temperature, to a final volume of 1000 µL. A volume of 150 µL of the concentrated extract was pipetted and transferred to a GC vial equipped with a glass insert and a volume of 50 µL of a solution of deuterated PAHs was added as an internal standard.
2.7.2. PAHs Detection and Quantification
PAH analyses were performed using a gas chromatograph (GC, Agilent Technologies 6850 series II; Santa Clara, CA, USA) coupled to a mass spectrometer (MS, Agilent Technologies 5975B VL MSD). Injection (1µL) was performed using an Agilent Technologies Auto sampler 6850 series, at a temperature of 250 °C with a splitless injection mode. The capillary column was an Rxi
®-5ms (30 m × 0.25 mm ID × 0.25 µm FT, 5% diphenyl and 95% polysiloxane from RESTEK), with helium as the carrier gas at a flow rate of 1 mL/min. The oven temperature program was set as follows: 50 °C for 2 min, 15 °C/min until 275 °C and 2 min hold, 15 °C/min until 325 °C and 15 min hold, and a post-run of 2 min at 300 °C. The detection of PAHs was performed in scan mode between 50 to 500 amu, with positive ion detection, where quantification of each PAH was based on the ratio of the signal of their molecular ion relative to the signal of the appropriate internal standard. Sample blanks were processed and analyzed. Thirty-five PAHs (∑
35Total-PAHs) were quantified. Among them, sixteen PAHs classified as priority pollutants by the United States Environmental Protection Agency [
64] were quantified (∑
16PAHs-EPA), along with 19 alkylated PAHs (∑
19Alkylated-PAHs).
2.8. Statistical Treatment of Biomarker Responses
At first, the assumptions of normality and the homoscedasticity were verified using the Shapiro and Bartlett tests, respectively. Exposure data were analyzed by performing two-way repeated measures analysis of variance (ANOVA) to determine the significance of the effects of treatment and time. The post hoc Tukey’s multiple comparison test was performed for comparison among the 2 times (0 and 96 h) and 3 treatments. Recovery data were first analyzed using one-way ANOVA followed by Tukey’s test to compare the effects of petroleum treatments (CLB and marine diesel) to Control at each time. Secondly, each treatment was analyzed by performing a two-way repeated measures ANOVA, followed by Tukey’s test, to determine the significance of the effects of time and series.
All tests were regarded as statistically significant when p < 0.05. All data were statistically analyzed using R software version 3.5.2 [
65].
The chemical analysis was not assessed statistically, due to the low number of replicates.
5. Conclusions
The objective of the study was to monitor four biomarkers on lobster hemolymph to assess impact and recovery capacity to diluted bitumen (CLB) and marine diesel. Our results show that the Brix index used by the lobster fishery is not relevant in the case of CLB or marine diesel exposure, so it is necessary to work with other biomarkers. Selected biomarkers tested in complement to the measurement of the Brix index allowed for monitoring the health status of hemocytes as a proxy to visualize the impacts of CLB and marine diesel exposure on the American lobster, as well as to follow the recovery efficiency. Despite a higher measured PTSA equivalent concentration in WAF-Diesel compared to WAF-CLB, the biomarkers did not show any difference between the two petroleum products, except in terms of cell viability, which was more impacted by CLB. However, the persistent odor and the PAH levels induced by a marine diesel exposure would be more damageable for the fisheries than lobsters exposed to CLB.
Lobster recovery following WAF exposure (to CLB and marine diesel) was observed over 3 weeks using 2 biomarkers—NRR and EROD—for assessment as well as one more for CLB exposure—cellular viability. According to these biomarkers, lobsters were able to recover in less than 3 weeks in clean water. However, even after 3 months of contention, lobsters still had high concentrations of PAHs, especially for those exposed to marine diesel. The effectiveness of an increase in temperature in improving recovery could not be confirmed by our study. Indeed, the NRR values reached the Control values more quickly with an increase in temperature, while the reverse was observed for the cellular viability.
In order to improve the monitoring of PAH concentrations in lobsters, it would be interesting to attempt to quantify these contaminants directly in the hemolymph, as proposed in the study of Turnbull [
67] for paralytic shellfish toxins. Furthermore, monitoring of PAHs in lobsters in parallel with the biomarkers may represent a better strategy for monitoring recovery efficiency.
Further, to better assess the long-term impacts of a spill, it would be interesting to measure DNA damage; for example, by comet assay [
86]. Finally, the PAH concentrations observed in lobster eggs suggest serious potential transgenerational effects which could severely impair the health of subsequent generations.