Non-Thermal Atmospheric Plasma Enhances Biological Effects of Fluoride on Oral Biofilms
Abstract
:1. Introduction
2. Materials and Methods
2.1. Non-Thermal Atmospheric Plasma
2.2. Biofilm Culture
2.3. Assessing Immediate Destruction of In Vitro Dual-Species Biofilms by FNTAP
2.4. Assessing Recovery of In Vitro Dual-Species Biofilms Exposed to FNTAP
2.5. PrestoBlue Assay
2.6. Colony Forming Units (CFU) Assay
2.7. Live/Dead Staining Assay
2.8. pH Analysis
2.9. Statistical Analysis
3. Results
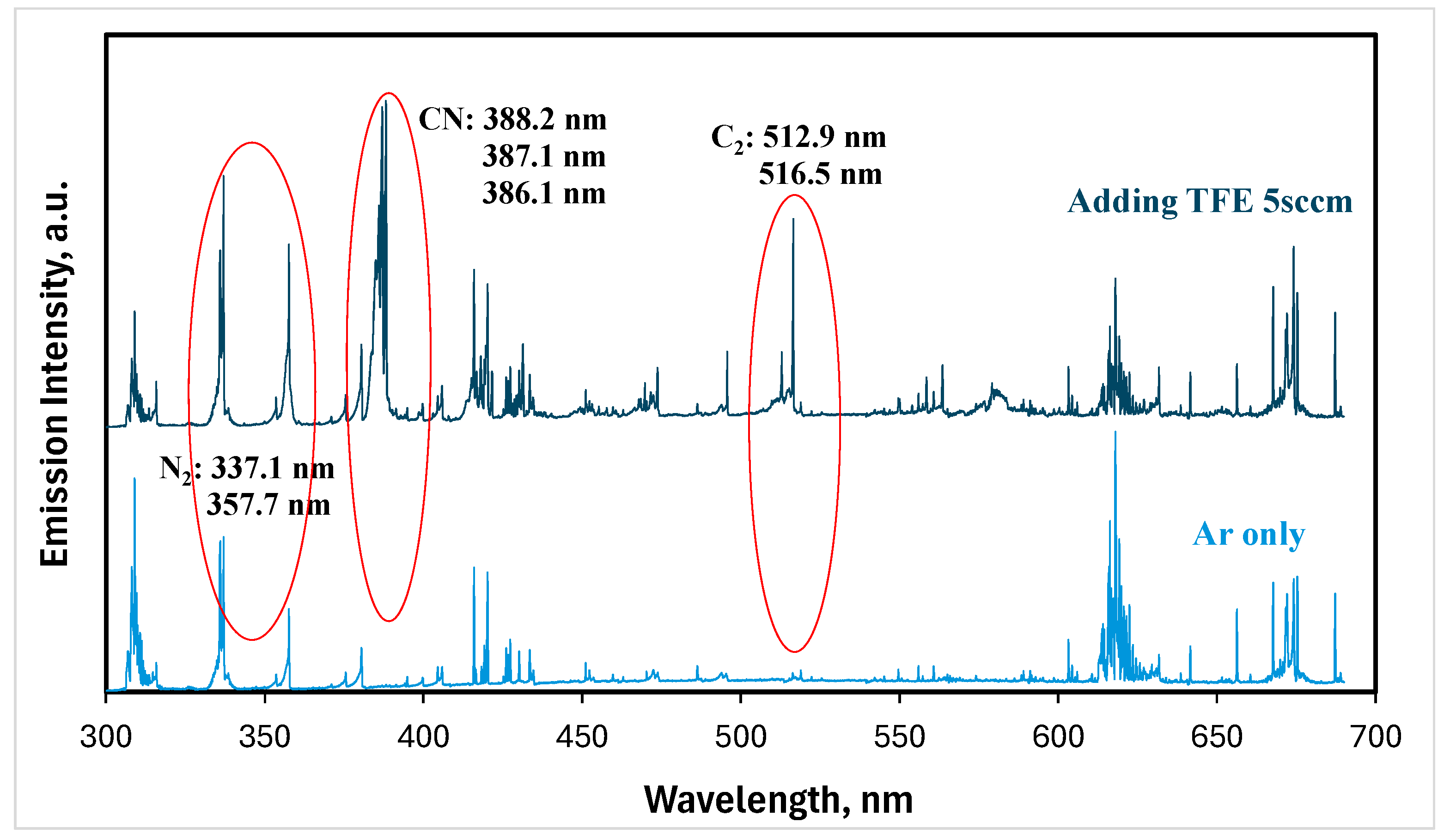
3.1. FNTAPs
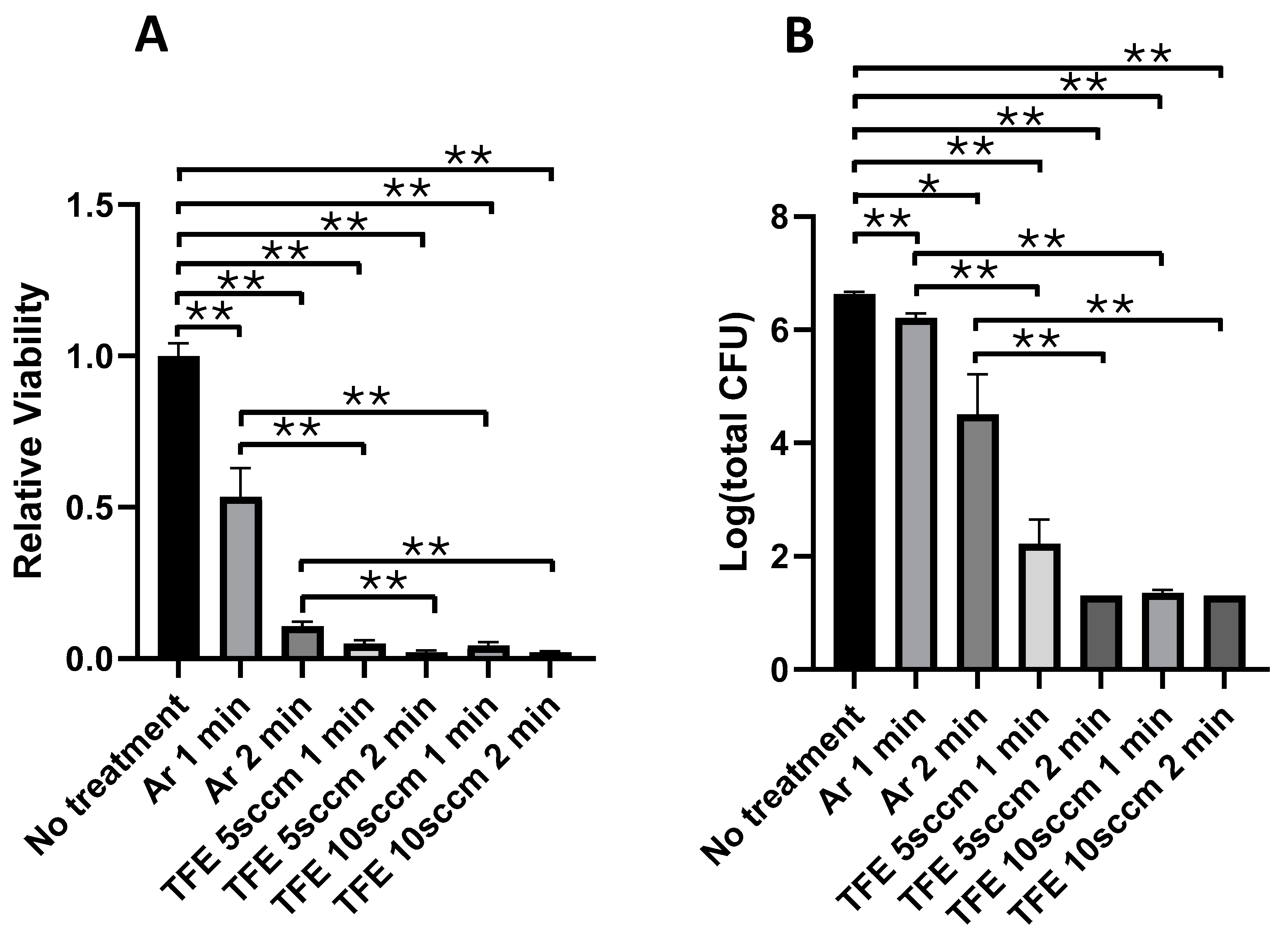
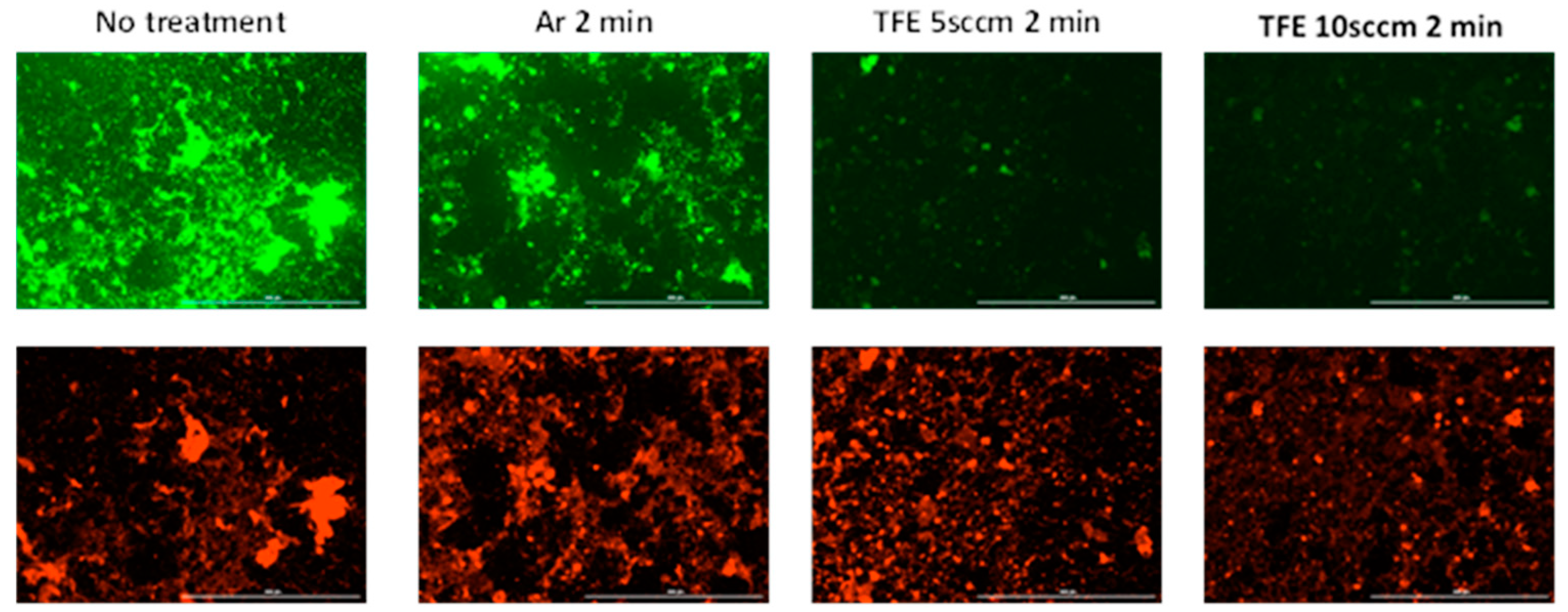
3.2. Immediate Effects of FNTAPs on S. mutans and S. sanguinis Dual-Species Biofilm
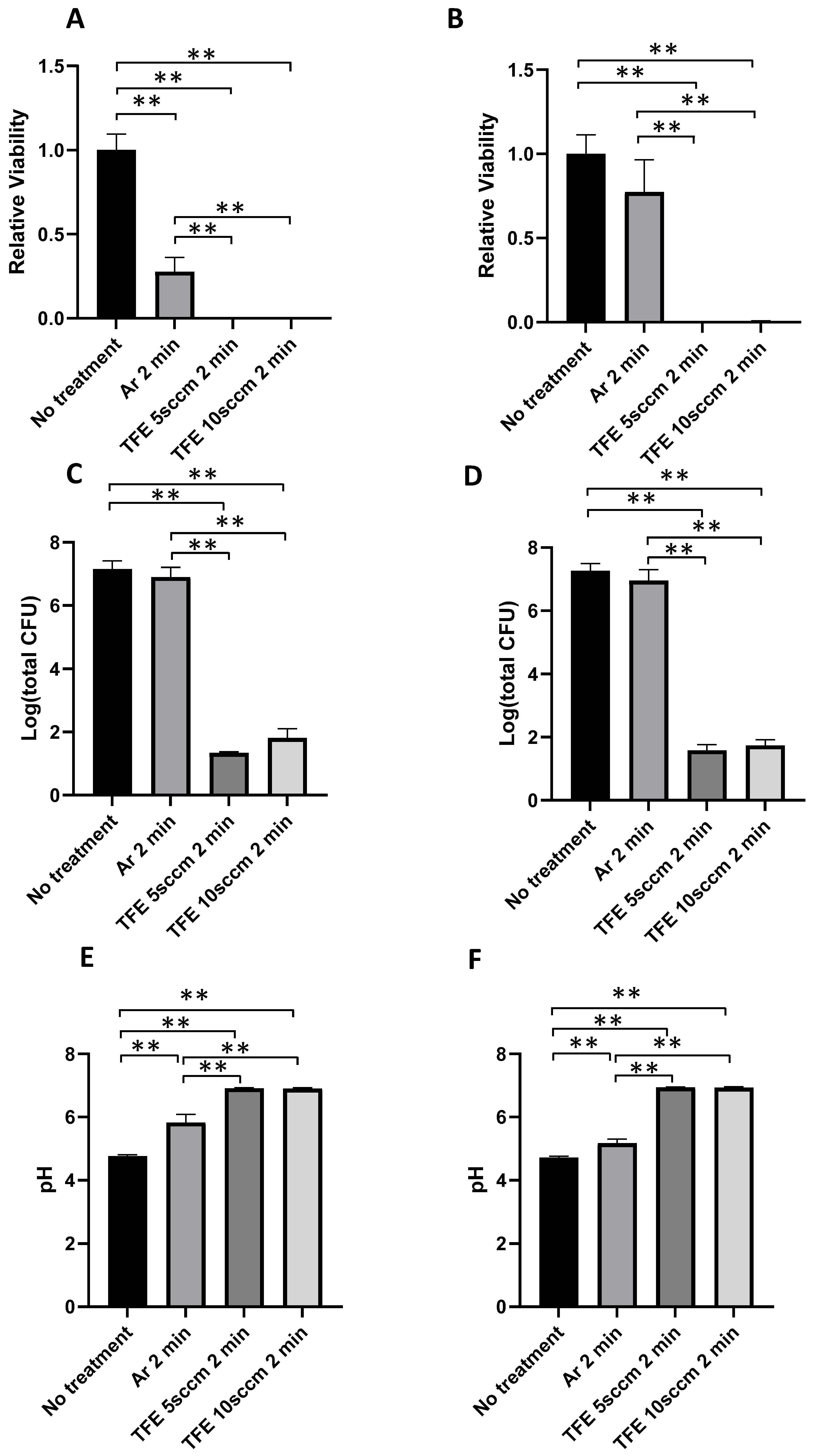
3.3. Recovery of S. mutans and S. sanguinis Dual-Species Biofilms Exposed to FNTAP
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BHI | Brain heart infusion |
| CFU | Colony forming units |
| DC | Direct current |
| DPBS | Dulbecco’s Phosphate-Buffered Saline |
| EPS | Extracellular polysaccharide |
| FNTAP | Fluoride non-thermal atmospheric plasma |
| MSA | Mitis Salivarius Agar |
| MTT | 3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide |
| NTAP | Non-thermal atmospheric plasma |
| OD600 | Optical density at 600 nm |
| OES | Optical emission spectroscopy |
| sccm | Standard cubic centimeters per minute |
| S. sanguinis | Streptococcus sanguinis |
| S. mutans | Streptococcus mutans |
| TFE | 1,1,1,2-tetrafluoroethane |
References
- Marcenes, W.; Kassebaum, N.J.; Bernabé, E.; Flaxman, A.; Naghavi, M.; Lopez, A.; Murray, C.J. Global burden of oral conditions in 1990-2010: A systematic analysis. J. Dent. Res. 2013, 92, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Flaxman, A.D.; Naghavi, M.; Lozano, R.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; Aboyans, V.; et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2163–2196. [Google Scholar] [CrossRef]
- Ushanthika, T.; Ganapathy, D. Awareness Among Dental Students On Strategies Over Early Childhood Caries. Int. J. Pharm. Res. 2020, 12, 1781–1788. [Google Scholar] [CrossRef]
- Bawden, J.W. Fluoride Varnish: A Useful New Tool for Public Health Dentistry. J. Public Health Dent. 1998, 58, 266–269. [Google Scholar] [CrossRef]
- Nayak, P. Topical Fluoride for Prevention of Dental Caries: A Review. Indian J. Forensic Med. Toxicol. 2020, 14, 9120–9123. [Google Scholar] [CrossRef]
- Chu, C.H.; Mei, M.L.; Lo, E.C. Use of fluorides in dental caries management. Gen. Dent. 2010, 58, 37–43; quiz 44–35, 79–80. [Google Scholar] [PubMed]
- Gao, S.S.; Zhang, S.; Mei, M.L.; Lo, E.C.; Chu, C.H. Caries remineralisation and arresting effect in children by professionally applied fluoride treatment—A systematic review. BMC Oral Health 2016, 16, 12. [Google Scholar] [CrossRef]
- ten Cate, J.M. Review on fluoride, with special emphasis on calcium fluoride mechanisms in caries prevention. Eur. J. Oral Sci. 1997, 105 Pt 2, 461–465. [Google Scholar] [CrossRef]
- Buzalaf, M.A.R.; Pessan, J.P.; Honório, H.M.; Ten Cate, J.M. Mechanisms of action of fluoride for caries control. Monogr. Oral Sci. 2011, 22, 97–114. [Google Scholar] [CrossRef]
- Koo, H. Strategies to enhance the biological effects of fluoride on dental biofilms. Adv. Dent. Res. 2008, 20, 17–21. [Google Scholar] [CrossRef]
- Bowden, G.H.W. Effects of Fluoride on the Microbial Ecology of Dental Plaque. J. Dent. Res. 1990, 69, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Hata, S.; Iwami, Y.; Kamiyama, K.; Yamada, T. Biochemical mechanisms of enhanced inhibition of fluoride on the anaerobic sugar metabolism by Streptococcus sanguis. J. Dent. Res. 1990, 69, 1244–1247. [Google Scholar] [CrossRef]
- Hamilton, I.R. Biochemical effects of fluoride on oral bacteria. J. Dent. Res. 1990, 69, 660–667. [Google Scholar] [CrossRef]
- Koo, H.; Sheng, J.; Nguyen, P.T.; Marquis, R.E. Co-operative inhibition by fluoride and zinc of glucosyl transferase production and polysaccharide synthesis by mutans streptococci in suspension cultures and biofilms. FEMS Microbiol. Lett. 2006, 254, 134–140. [Google Scholar] [CrossRef]
- Reilly, C.; Goettl, M.; Steinmetz, M.; Nikrad, J.; Jones, R.S. Short-term effects of povidone iodine and sodium fluoride therapy on plaque levels and microbiome diversity. Oral Dis. 2016, 22, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Koopman, J.E.; van der Kaaij, N.C.; Buijs, M.J.; Elyassi, Y.; van der Veen, M.H.; Crielaard, W.; Ten Cate, J.M.; Zaura, E. The Effect of Fixed Orthodontic Appliances and Fluoride Mouthwash on the Oral Microbiome of Adolescents—A Randomized Controlled Clinical Trial. PLoS ONE 2015, 10, e0137318. [Google Scholar] [CrossRef]
- Rošin-Grget, K.; Peroš, K.; Sutej, I.; Bašić, K. The cariostatic mechanisms of fluoride. Acta. Med. Acad. 2013, 42, 179–188. [Google Scholar] [CrossRef]
- Thurnheer, T.; Belibasakis, G.N. Effect of sodium fluoride on oral biofilm microbiota and enamel demineralization. Arch. Oral Biol. 2018, 89, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Jha, S.K.; Mishra, V.K.; Sharma, D.K.; Damodaran, T. Fluoride in the environment and its metabolism in humans. Rev. Environ. Contam. Toxicol. 2011, 211, 121–142. [Google Scholar] [CrossRef]
- Shen, Y.; Yu, F.; Qiu, L.; Gao, M.; Xu, P.; Zhang, L.; Liao, X.; Wang, M.; Hu, X.; Sun, Y.; et al. Ecological influence by colonization of fluoride-resistant Streptococcus mutans in oral biofilm. Front. Cell Infect. Microbiol. 2022, 12, 1106392. [Google Scholar] [CrossRef]
- Srivastava, S.; Flora, S.J.S. Fluoride in Drinking Water and Skeletal Fluorosis: A Review of the Global Impact. Curr. Environ. Health Rep. 2020, 7, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Streckfuss, J.L.; Perkins, D.; Horton, I.M.; Brown, L.R.; Dreizen, S.; Graves, L. Fluoride resistance and adherence of selected strains of Streptococcus mutans to smooth surfaces after exposure to fluoride. J. Dent. Res. 1980, 59, 151–158. [Google Scholar] [CrossRef]
- Brown, L.R.; White, J.O.; Horton, I.M.; Dreizen, S.; Streckfuss, J.L. Effect of continuous fluoride gel use on plaque fluoride retention and microbial activity. J. Dent. Res. 1983, 62, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Van Loveren, C.; Spitz, L.M.; Buijs, J.F.; Ten Cate, J.M.; Eisenberg, A.D. In vitro demineralization of enamel by F-sensitive and F-resistant mutans streptococci in the presence of 0, 0.05, or 0.5 mmol/L NaF. J. Dent. Res. 1991, 70, 1491–1496. [Google Scholar] [CrossRef] [PubMed]
- Men, X.; Shibata, Y.; Takeshita, T.; Yamashita, Y. Identification of Anion Channels Responsible for Fluoride Resistance in Oral Streptococci. PLoS ONE 2016, 11, e0165900. [Google Scholar] [CrossRef]
- Murata, T.; Hanada, N. Contribution of chloride channel permease to fluoride resistance in Streptococcus mutans. FEMS Microbiol. Lett. 2016, 363. [Google Scholar] [CrossRef]
- Tang, B.; Gong, T.; Zhou, X.; Lu, M.; Zeng, J.; Peng, X.; Wang, S.; Li, Y. Deletion of cas3 gene in Streptococcus mutans affects biofilm formation and increases fluoride sensitivity. Arch. Oral Biol. 2019, 99, 190–197. [Google Scholar] [CrossRef]
- Lu, M.; Xiang, Z.; Gong, T.; Zhou, X.; Zhang, Z.; Tang, B.; Zeng, J.; Wang, L.; Cui, T.; Li, Y. Intrinsic Fluoride Tolerance Regulated by a Transcription Factor. J. Dent. Res. 2020, 99, 1270–1278. [Google Scholar] [CrossRef]
- Yu, J.; Wang, Y.; Han, D.; Cao, W.; Zheng, L.; Xie, Z.; Liu, H. Identification of Streptococcus mutans genes involved in fluoride resistance by screening of a transposon mutant library. Mol. Oral Microbiol. 2020, 35, 260–270. [Google Scholar] [CrossRef]
- Pandit, S.; Kim, H.J.; Park, S.H.; Jeon, J.G. Enhancement of fluoride activity against Streptococcus mutans biofilms by a substance separated from Polygonum cuspidatum. Biofouling 2012, 28, 279–287. [Google Scholar] [CrossRef]
- Han, S.; Washio, J.; Abiko, Y.; Zhang, L.; Takahashi, N. Green Tea-Derived Catechins Suppress the Acid Productions of Streptococcus mutans and Enhance the Efficiency of Fluoride. Caries Res. 2023, 57, 255–264. [Google Scholar] [CrossRef]
- Liu, J.; Weng, Q.; Da, D.; Yao, S.; Zhang, Y.; Wu, Y. Enhancement of Fluoride’s Antibacterial and Antibiofilm Effects against Oral Staphylococcus aureus by the Urea Derivative BPU. Antibiotics 2024, 13, 930. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Cheng, X.; Wang, L.; Qiu, W.; Wang, S.; Zhou, Y.; Li, M.; Li, Y.; Cheng, L.; Li, J.; et al. Combinatorial effects of arginine and fluoride on oral bacteria. J. Dent. Res. 2015, 94, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Moreau, M.; Orange, N.; Feuilloley, M.G.J. Non-thermal plasma technologies: New tools for bio-decontamination. Biotechnol. Adv. 2008, 26, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Laroussi, M. Nonthermal decontamination of biological media by atmospheric-pressure plasmas: Review, analysis, and prospects. IEEE Trans. Plasma Sci. 2002, 30, 1409–1415. [Google Scholar] [CrossRef]
- Gaunt, L.F.; Beggs, C.B.; Georghiou, G.E. Bactericidal Action of the Reactive Species Produced by Gas-Discharge Nonthermal Plasma at Atmospheric Pressure: A Review. IEEE Trans. Plasma Sci. 2006, 34, 1257–1269. [Google Scholar] [CrossRef]
- Graves, D.B. Reactive Species from Cold Atmospheric Plasma: Implications for Cancer Therapy. Plasma Process. Polym. 2014, 11, 1120–1127. [Google Scholar] [CrossRef]
- Ratovitski, E.A.; Cheng, X.; Yan, D.; Sherman, J.H.; Canady, J.; Trink, B.; Keidar, M. Anti-Cancer Therapies of 21st Century: Novel Approach to Treat Human Cancers Using Cold Atmospheric Plasma. Plasma Process. Polym. 2014, 11, 1128–1137. [Google Scholar] [CrossRef]
- Dubey, S.K.; Parab, S.; Alexander, A.; Agrawal, M.; Achalla, V.P.K.; Pal, U.N.; Pandey, M.M.; Kesharwani, P. Cold atmospheric plasma therapy in wound healing. Process Biochem. 2022, 112, 112–123. [Google Scholar] [CrossRef]
- Haertel, B.; von Woedtke, T.; Weltmann, K.-D.; Lindequist, U. Non-thermal atmospheric-pressure plasma possible application in wound healing. Biomol. Ther. 2014, 22, 477–490. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.J.; Wang, S.B.; Choi, E.H.; Han, I. Non-Thermal Bio-Compatible Plasma Induces Osteogenic Differentiation of Human Mesenchymal Stem/Stromal Cells With ROS-Induced Activation of MAPK. IEEE Access 2020, 8, 36652–36663. [Google Scholar] [CrossRef]
- Choi, B.-B.; Choi, J.-H.; Kang, T.-H.; Lee, S.-J.; Kim, G.-C. Enhancement of Osteoblast Differentiation Using No-Ozone Cold Plasma on Human Periodontal Ligament Cells. Biomedicines 2021, 9, 1542. [Google Scholar] [CrossRef]
- Xiong, Z.; Zhao, S.; Yan, X. Nerve Stem Cell Differentiation by a One-step Cold Atmospheric Plasma Treatment In Vitro. J. Vis. Exp. 2019, 143, e58663. [Google Scholar] [CrossRef]
- Wen, X.; Xin, Y.; Hamblin, M.R.; Jiang, X. Applications of cold atmospheric plasma for transdermal drug delivery: A review. Drug Deliv. Transl. Res. 2020, 11, 741–747. [Google Scholar] [CrossRef]
- Hong, Q.; Dong, X.; Chen, M.; Sun, H.; Hong, L.; Wang, Y.; Li, H.; Yu, Q. An in vitro and in vivo study of plasma treatment effects on oral biofilms. J. Oral Microbiol. 2019, 11, 1603524. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Yu, Q.; Hsieh, F.-h.; Duan, Y. Bacterial Deactivation Using a Low Temperature Argon Atmospheric Plasma Brush with Oxygen Addition. Plasma Process. Polym. 2007, 4, 77–87. [Google Scholar] [CrossRef]
- Yang, B.; Chen, J.; Yu, Q.; Li, H.; Lin, M.; Mustapha, A.; Hong, L.; Wang, Y. Oral bacterial deactivation using a low-temperature atmospheric argon plasma brush. J. Dent. 2011, 39, 48–56. [Google Scholar] [CrossRef]
- Hong, Q.; Dong, X.; Chen, M.; Xu, Y.; Sun, H.; Hong, L.; Wang, Y.; Yu, Q. Disinfection of Streptococcus mutans biofilm by a non-thermal atmospheric plasma brush. Jpn. J. Appl. Phys. 2016, 55, 07LG02. [Google Scholar] [CrossRef]
- Blumhagen, A.; Singh, P.; Mustapha, A.; Chen, M.; Wang, Y.; Yu, Q. Plasma deactivation of oral bacteria seeded on hydroxyapatite disks as tooth enamel analogue. Am. J. Dent. 2014, 27, 84–90. [Google Scholar]
- Dong, X.; Wu, L.; Wang, Y.; Yu, Q.; Hong, L. Effects of Atmospheric Fluorine-Containing Nonthermal Plasmas on Demineralization and Remineralization of Tooth Enamel. IEEE Trans. Plasma Sci. 2022, 50, 2910–2916. [Google Scholar] [CrossRef]
- Ritts, A.C.; Li, H.; Yu, Q.; Xu, C.; Yao, X.; Hong, L.; Wang, Y. Dentin surface treatment using a non-thermal argon plasma brush for interfacial bonding improvement in composite restoration. Eur. J. Oral Sci. 2010, 118, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Hong, Q.; Sun, H.; Chen, M.; Zhang, S.; Yu, Q. Plasma treatment effects on destruction and recovery of Porphyromonas gingivalis biofilms. PLoS ONE 2022, 17, e0274523. [Google Scholar] [CrossRef] [PubMed]
- Ivanoff, C.S.; Swami, N.S.; Hottel, T.L.; Garcia-Godoy, F. Enhanced penetration of fluoride particles into bovine enamel by combining dielectrophoresis with AC electroosmosis. Electrophoresis 2013, 34, 2945–2955. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; de Sousa, F.B.; Gan, H.Y.; Kwon, H.J.; Park, S.; Kilpatrick-Liverman, L.; Wang, W.; Lavender, S.; Pilch, S.; Han, J. Enhanced Delivery of F(-), Ca(2+), K(+), and Na(+) Ions into Enamel by Electrokinetic Flows. J. Dent. Res. 2019, 98, 430–436. [Google Scholar] [CrossRef]
- Yu, Q.; Huang, C.; Hsieh, F.H.; Huff, H.; Duan, Y. Bacterial inactivation using a low-temperature atmospheric plasma brush sustained with argon gas. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 80, 211–219. [Google Scholar] [CrossRef]
- López-López, A.; Mira, A. Shifts in Composition and Activity of Oral Biofilms After Fluoride Exposure. Microb. Ecol. 2020, 80, 729–738. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warang, A.; Deol, I.; Fakher, S.; Wu, L.; Hong, L.; Zhang, S.; Yu, Q.; Sun, H. Non-Thermal Atmospheric Plasma Enhances Biological Effects of Fluoride on Oral Biofilms. J. Funct. Biomater. 2025, 16, 132. https://doi.org/10.3390/jfb16040132
Warang A, Deol I, Fakher S, Wu L, Hong L, Zhang S, Yu Q, Sun H. Non-Thermal Atmospheric Plasma Enhances Biological Effects of Fluoride on Oral Biofilms. Journal of Functional Biomaterials. 2025; 16(4):132. https://doi.org/10.3390/jfb16040132
Chicago/Turabian StyleWarang, Anushri, Isha Deol, Sarah Fakher, Linfeng Wu, Liang Hong, Shaoping Zhang, Qingsong Yu, and Hongmin Sun. 2025. "Non-Thermal Atmospheric Plasma Enhances Biological Effects of Fluoride on Oral Biofilms" Journal of Functional Biomaterials 16, no. 4: 132. https://doi.org/10.3390/jfb16040132
APA StyleWarang, A., Deol, I., Fakher, S., Wu, L., Hong, L., Zhang, S., Yu, Q., & Sun, H. (2025). Non-Thermal Atmospheric Plasma Enhances Biological Effects of Fluoride on Oral Biofilms. Journal of Functional Biomaterials, 16(4), 132. https://doi.org/10.3390/jfb16040132






