Silencing Bcl-2 Expression in Epithelial Cancer Cells Using “Smart” Particles
Abstract
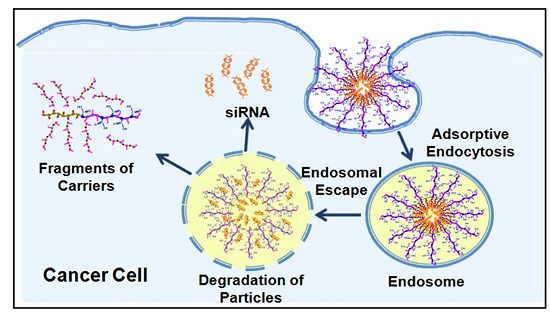
:1. Introduction

2. Results and Discussion
2.1. Formulation and Characterization of “Smart” Particles

2.2. Uptake of “Smart” Particles by HeLa and UM-SCC-17B Cells

2.3. Effect of “Smart” Particles on GAPDH Expression

2.4. Effect of “Smart” Particles on Bcl-2 Expression


3. Experimental Section
3.1. Materials
3.2. Synthesis of pH-Sensitive Comb-Like Polymer

3.3. Formulation and Characterization of “Smart” Particles
3.4. Culture of HeLa and UM-SCC-17B Cells
3.5. Cellular Uptake of “Smart” Particles
3.6. In Vitro Evaluation of GAPDH Knockdown in HeLa and UM-SCC-17B Cells
3.7. In Vitro Evaluation of Bcl-2 Knockdown in HeLa and UM-SCC-17B Cells
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Adams, J.M.; Cory, S. The Bcl-2 protein family: Arbiters of cell survival. Science 1998, 281, 1322–1326. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.M.; Cory, S. The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene 2007, 26, 1324–1337. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, L.V.; Carvalho, A.L. Bcl-2 targeted-therapy for the treatment of head and neck squamous cell carcinoma. Recent Pat. Anticancer Drug Discov. 2011, 6, 45–57. [Google Scholar]
- Reed, J.C. Mechanisms of apoptosis avoidance in cancer. Curr. Opin. Oncol. 1999, 11, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.C. Dysregulation of apoptosis in cancer. J. Clin. Oncol. 1999, 17, 2941–2953. [Google Scholar] [PubMed]
- Kaufmann, S.H.; Gores, G.J. Apoptosis in cancer: Cause and cure. Bioessays 2000, 22, 1007–1017. [Google Scholar] [CrossRef] [PubMed]
- Gallo, O.; Boddi, V.; Calzolari, A.; Simonetti, L.; Trovati, M.; Bianchi, S. Bcl-2 protein expression correlates with recurrence and survival in early stage head and neck cancer treated by radiotherapy. Clin. Cancer Res. 1996, 2, 261–267. [Google Scholar]
- Sharma, H.; Sen, S.; Mathur, M.; Bahadur, S.; Singh, N. Combined evaluation of expression of telomerase, survivin, and anti-apoptotic Bcl-2 family members in relation to loss of differentiation and apoptosis in human head and neck cancers. Head Neck 2004, 26, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Zhang, Z.; Mantellini, M.G.; Karl, E.; Zeitlin, B.; Verhaegen, M.; Soengas, M.S.; Lingen, M.; Strieter, R.M.; Nunez, G.; et al. Bcl-2 orchestrates a cross-talk between endothelial and tumor cells that promotes tumor growth. Cancer Res. 2007, 67, 9685–9693. [Google Scholar] [CrossRef] [PubMed]
- Sharma, H.; Sen, S.; Lo Muzio, L.; Mariggio, A.; Singh, N. Antisense-mediated downregulation of anti-apoptotic proteins induces apoptosis and sensitizes head and neck squamous cell carcinoma cells to chemotherapy. Cancer Biol. Ther. 2005, 4, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-L.; Jiang, G.; Birrell, L.K.; El-Sayed, M.E.H. Degradable, ph-sensitive, membrane-destabilizing, comb-like polymers for intracellular delivery of nucleic acids. Biomaterials 2010, 31, 7150–7166. [Google Scholar] [CrossRef] [PubMed]
- Bulmus, V.; Woodward, M.; Lin, L.; Murthy, N.; Stayton, P.S.; Hoffman, A.S. A new pH-responsive and glutathione-reactive, endosomal membrane-disruptive polymeric carrier for intracellular delivery of biomolecular drugs. J. Control. Release 2003, 93, 105–120. [Google Scholar] [PubMed]
- El-Sayed, M.E.H.; Hoffman, A.S.; Stayton, P.S. Rational design of composition and activity correlations for pH-sensitive and glutathione-reactive polymer therapeutics. J. Control. Release 2005, 101, 47–58. [Google Scholar]
- Oskuee, R.K.; Dehshahri, A.; Shier, W.T.; Ramezani, M. Alkylcarboxylate grafting to polyethylenimine: A simple approach to producing a DNA nanocarrier with low toxicity. J. Gene Med. 2009, 11, 921–932. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Pan, S.; Luo, X.; Zhang, X.; Zhang, W.; Feng, M. A biodegradable low molecular weight polyethylenimine derivative as low toxicity and efficient gene vector. Bioconjug. Chem. 2009, 20, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Dellian, M.; Fukumura, D.; Leunig, M.; Berk, D.A.; Torchilin, V.P.; Jain, R.K. Vascular permeability in a human tumor xenograft: Molecular size dependence and cutoff size. Cancer Res. 1995, 55, 3752–3756. [Google Scholar] [PubMed]
- Lampela, P.; Soininen, P.; Puttonen, K.A.; Ruponen, M.; Urtti, A.; Mannisto, P.T.; Raasmaja, A. Effect of cell-surface glycosaminoglycans on cationic carrier combined with low-MW PEI-mediated gene transfection. Int. J. Pharm. 2004, 284, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Brownlie, A.; Uchegbu, I.F.; Schätzlein, A.G. PEI-based vesicle-polymer hybrid gene delivery system with improved biocompatibility. Int. J. Pharm. 2004, 274, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Deng, R.; Yue, Y.; Jin, F.; Chen, Y.; Kung, H.F.; Lin, M.C.; Wu, C. Revisit the complexation of PEI and DNA—How to make low cytotoxic and highly efficient PEI gene transfection non-viral vectors with a controllable chain length and structure? J. Control. Release 2009, 140, 40–46. [Google Scholar] [CrossRef]
- Gebhart, C.L.; Kabanov, A.V. Evaluation of polyplexes as gene transfer agents. J. Control. Release 2001, 73, 401–416. [Google Scholar] [CrossRef] [PubMed]
- Rybak, S.L.; Lanni, F.; Murphy, R.F. Theoretical considerations on the role of membrane potential in the regulation of endosomal pH. Biophys. J. 1997, 73, 674–687. [Google Scholar] [CrossRef] [PubMed]
- Cain, C.C.; Sipe, D.M.; Murphy, R.F. Regulation of endocytic ph by the Na+,K+-ATPase in living cells. Proc. Natl. Acad. Sci. USA 1989, 86, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Simon, S.M. Role of organelle pH in tumor cell biology and drug resistance. Drug Discov. Today 1999, 4, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Duvvuri, M.; Konkar, S.; Hong, K.H.; Blagg, B.S.; Krise, J.P. A new approach for enhancing differential selectivity of drugs to cancer cells. ACS Chem. Biol. 2006, 1, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Weisz, O.A. Organelle acidification and disease. Traffic 2003, 4, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Mallapragada, S. The mechanism of selective transfection mediated by pentablock copolymers; Part I: Investigation of cellular uptake. Acta Biomater. 2011, 7, 1570–1579. [Google Scholar] [CrossRef] [PubMed]
- Buck, A.C.; Shen, C.X.; Schirrmeister, H.; Schmid-Kotsas, A.; Munzert, G.; Guhlmann, A.; Mehrke, G.; Klug, N.; Gross, H.J.; Bachem, M.; et al. Liposomal delivery of antisense oligonucleotides for efficient downregulation of Bcl-2 and induction of apoptosis. Cancer Biother. Radiopharm. 2002, 17, 281–289. [Google Scholar]
- Ferritto, M.S.; Tirrell, D.A. Poly(2-ethylacrylic acid). Macromol. Synth. 1992, 11, 59–62. [Google Scholar]
- Lai, J.T.; Filla, D.; Shea, R. Functional polymers from novel carboxyl-terminated trithiocarbonates as highly efficient raft agents. Macromolecules 2002, 35, 6754–6756. [Google Scholar] [CrossRef]
- Neiva, K.G.; Zhang, Z.; Miyazawa, M.; Warner, K.A.; Karl, E.; Nor, J.E. Cross talk initiated by endothelial cells enhances migration and inhibits anoikis of squamous cell carcinoma cells through STAT3/Akt/ERK signaling. Neoplasia 2009, 11, 583–593. [Google Scholar] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, Y.-L.; Jiang, G.; Zhang, Z.; Nör, J.E.; ElSayed, M.E.H. Silencing Bcl-2 Expression in Epithelial Cancer Cells Using “Smart” Particles. J. Funct. Biomater. 2014, 5, 167-182. https://doi.org/10.3390/jfb5030167
Lin Y-L, Jiang G, Zhang Z, Nör JE, ElSayed MEH. Silencing Bcl-2 Expression in Epithelial Cancer Cells Using “Smart” Particles. Journal of Functional Biomaterials. 2014; 5(3):167-182. https://doi.org/10.3390/jfb5030167
Chicago/Turabian StyleLin, Yen-Ling, Guohua Jiang, Zhaocheng Zhang, Jacques E. Nör, and Mohamed E. H. ElSayed. 2014. "Silencing Bcl-2 Expression in Epithelial Cancer Cells Using “Smart” Particles" Journal of Functional Biomaterials 5, no. 3: 167-182. https://doi.org/10.3390/jfb5030167
APA StyleLin, Y.-L., Jiang, G., Zhang, Z., Nör, J. E., & ElSayed, M. E. H. (2014). Silencing Bcl-2 Expression in Epithelial Cancer Cells Using “Smart” Particles. Journal of Functional Biomaterials, 5(3), 167-182. https://doi.org/10.3390/jfb5030167