1. Introduction
Nitrate and phosphate are necessary nutrients for the growth of plants, wildlife, and humans. However, elevated concentrations of nitrate and phosphate in surface and groundwater is a severe global concern leading to devastating impacts on ecosystems [
1,
2]. General sources of nitrate and phosphate contaminants in water bodies usually arise from the waste products of human activities such as discharges from industrialized practices, agricultural uses like inorganic fertilizers, compost, and wastewater treatment effluents [
3]. A phosphate generally arises from the element named phosphorous, and affects water quality by the disproportionate development of algae. Phosphates ions present in the water nourish algae, which further destroy other forms of life and yield unsafe contaminants. Excessive concentration of phosphorous results in the eutrophication process, which decreases the amount of dissolved oxygen in the aquatic streams of watercourses and ponds [
4]. Likewise, excessive levels of nitrates are considered the main harmful waste for ground and surface waters, and pose a severe threat to the survival of aquatic life. According to the World Health Organization, the acceptable limits of nitrate and phosphate ions in drinking water are 40 mg/L and nearly 0.1 mg/L, respectively [
5]. Therefore, there is an urgent need to propose sustainable and cost-effective engineering technologies to remove excess phosphate and nitrate ions in wastewater streams effectively before discharging them to the receiving bodies or their reuse.
Various physicochemical techniques, including chemical precipitation [
6], a membrane process [
7], oxidation [
8], electrodialysis [
9,
10], constructed wetlands [
11], and adsorption [
12,
13] have been employed as the route to eliminate nitrates and phosphates from aqueous systems. Among all of them, adsorption proved to be one of the most popular techniques because of its low cost, ease of operation, higher removal efficiency, and excellent reusability performance. In the adsorption process, the selection of proper adsorbent material is the key challenge; therefore, researchers and scientists are exerting enormous efforts to produce sustainable, economical, and less toxic adsorbents for the improved remediation performance of contaminants from water streams [
14,
15].
Biochar is a carbon-rich product obtained from pyrolysis of biomass that has attracted remarkable interest for application in soil improvement and removal of toxic pollutants from the aqueous medium. The porous surface morphology of biochar is highly favorable for holding water and water-soluble nutrients [
16]. Previous studies demonstrated biochar as a promising, eco-effective adsorbent due to its excellent physical characteristics, abundant surface functionalities, and superb ion-exchange capability [
17,
18]. The porous structure with a relatively high specific surface area of the different produced biochars showed enhanced uptake of various toxins [
19].
Xiaoning Liu et al. [
20] investigated calcium-activated biochar for the remediation of phosphate ions from phosphorus-rich waste streams, and revealed excellent removal efficiency with maximum sorption capacity of 197 mg/g. Ren Jing et al. [
21] demonstrated biochar derived from the cotton stalk as an effective phosphate removal adsorbent from water. Chemically modified biochar [
22] showed improved removal of nitrate from water solution and 28.21 mg/g of adsorption capacity.
Layered double hydroxides (LDH) are classified as a clay consisting of metal hydroxides layers sandwiched with anions. These materials are represented generally as [M
2+(1−x)M
3+x(OH)
2]
x+ [A
n−]
x/n·
mH
2O, where M
2+ and M
3+ represent the double- and triple-charged positive metal ions, A
n− is the intermediate negative ions, respectively. The versatile composites with outstanding surface and structure characteristics and excellent ion exchange capabilities attracted considerable attention for catalysis [
23], purification [
24], optics [
25], and energy applications [
26]. Ronghua Li et al. [
27] developed Mg/Al-layered double hydroxide at different mole ratios and revealed improved phosphate adsorption with increasing Mg/Al ratio in the Mg/Al–LDHs biochar composites capacity. Yuki Kamimoto et al. [
28] concluded excellent adsorption performance of nitrate onto magnetic calcined Mg–Fe LDH. Muayad Al Jaberi et al. [
29] prepared CaFe-layered double hydroxide by co-simple co-precipitation technique and reported a maximum removal capacity of 130 mg/g.
In recent years, substantial research has directed interest in developing biochar-based derivatives as super adsorbents for enhanced remediation of various toxic contaminants from an aqueous medium [
30,
31]. In particular, coupling biochar with LDH showed a promising and sustainable approach to significantly improve adsorbent characteristics and removal efficiency of pollutants due to the synergetic effect of biochar, and LDHs led to an improvement in surface active sites and thus resulted in enhanced sorption performance.
In 2019, Qing Liang Cui et al. 2019 [
32] investigated magnetic biochar/Mg–Al layered double hydroxide composite and reported relatively higher adsorption capacity as compared to other magnetic adsorbents, with moderate 51.43% removal efficiency. Yan-Hong Jiang et al. [
33] loaded ZnAl–LDH on to modified banana straw biochar via a hydrothermal process and demonstrated significant improvement in phosphorus removal. These studies indicated that the intercalation of LDHs into biochar could significantly improve the uptake of phosphate. Therefore, it is expected that the incorporation of MgAl LDHs into date palm-derived biochar could be a sustainable and promising approach to improve the adsorption performance of date palm derived biochar. In addition, the aforementioned studies produced a biochar-LDHs composite via a hydrothermal technique and investigated the removal of phosphate ions only. Therefore, this study investigated the production of biochar derived from date palm from waste intercalated with a MgAl-layered double hydroxides composite via simple co-precipitation technique and explored it for the recovery of both phosphate and nitrate from wastewater streams.
The main objective of this work is to enhance phosphate and nitrate recovery from the water phase using date palm waste-derived biochar augmented with MgAl layered double hydroxides. The synthesized nanocomposites characterization using Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), X-ray diffraction (XRD), and Brunauer–Emmett–Teller (BET) measurement techniques to examine the surface and structure properties after the inclusion of LDH layers into biochar. The study focused on the effects of several influencing parameters such as contact time, adsorbent dosage, pH, anions concentration, and temperature on the percentage removal of phosphate and nitrate ions. The adsorption capacity and interaction mechanism of the synthesized composite with phosphate and nitrate anions were evaluated in depth by considering isotherms, kinetics, and thermodynamic models.
2. Materials and Methods
2.1. Chemicals
The production of the biochars used in this study was done at slow pyrolysis (700 °C and four h at a heating rate of 3 °C/min) using date palm waste fronds collected from Al-Hasa city, Saudi Arabia. The pure salts of aluminum (III) nitrate nonahydrate and magnesium (II) nitrate hexahydrate, were obtained from Sigma Aldrich (Darmstadt, Germany) and used without any pretreatment. Potassium dihydrogen phosphate (KH2PO4), sodium nitrate (NaNO3), and potassium sulfate (K2SO4) were from Sigma Aldrich (Darmstadt, Germany). Hydrochloric acid (HCl) and sodium hydroxide (NaOH) were from Merck (Darmstadt, Germany). The salts of KH2PO4 and NaNO3 were the sources of phosphate and nitrate solution used in the study. Preparation of a stock solution of 1000 mg/L for the experimental work used deionized water. Phosphate and nitrate concentrations were measured using an ultraviolet–visible (UV–visible) spectrophotometer (DR 6000, Hach Lange, Mena, Dubai, UAE). The initial desired value of pH was adjusted using HCl and NaOH.
2.2. Synthesis of Date Palm-Derived Biochar-Decorated MgAl Composites
The production of biochar-modified MgAl-layered double hydroxide composites were from different masses of biochar (5 and 10 g) with a 3:1 mole ratio of magnesium (7.69 g) and aluminum (3.75 g) nitrate salts (M
2+:M
3+) via co-precipitation method. In detail, first, we dissolved a measured amount of salts in a reaction vessel containing 100 mL of double-distilled water. Simultaneously, a precisely known quantity of biochar derived date palm fronds (5 and 10 g) was also added in 100 mL deionized water, sonicated at 60 amplitude for 30–60 minutes, respectively and transmitted to the reaction flask. The mixture of salts and biochar was stirred vigorously at 800–1000 rpm for 10 min at 90 °C. Afterward, the pH of the reaction mixture was maintained at 10 ± 0.5 by adding 1 M of NaOH dropwise. When the required pH value was achieved, the reactor was kept for nearly 24 h reflux at 90 °C. After the reaction, the resultant precipitates were centrifuged and washed 3–4 times with double-distilled water, and finally by ethanol to remove all unreacted salts and impurities. The final product biochar decorated MgAl composite was kept at 60 °C in the oven for 48 h. The resultant composites were then stored in a desiccator for future experiments. The actual compositions of biochar–MgAl composites are listed in
Table 1.
2.3. Characterization of Biochar-Decorated MgAl Composites
The biochar decorated MgAl composites characterized by Fourier transform infrared spectroscopy (FTIR, Nicolet 6700, Thermo Fisher Scientific, Waltham, MA, USA, resolution 4 cm−1), X-ray diffraction (XRD, D8 advance X-ray instrument, Waltham, MA, USA, wavelength = 0.1542 nm, and 2θ = 10° to 80°), scanning electron microscopy (SEM, SM-6460LV (Jeol), Peabody, MA, USA), transmission electron microscopy (FEI, Morgagni 268, Berno, Czech Republic), and Brunauer–Emmett–Teller measurement (BET Tristar II series, Micromeritics, Norcross, GA, USA).
2.4. Adsorption of Phosphate and Nitrate onto Biochar MgAl Composites
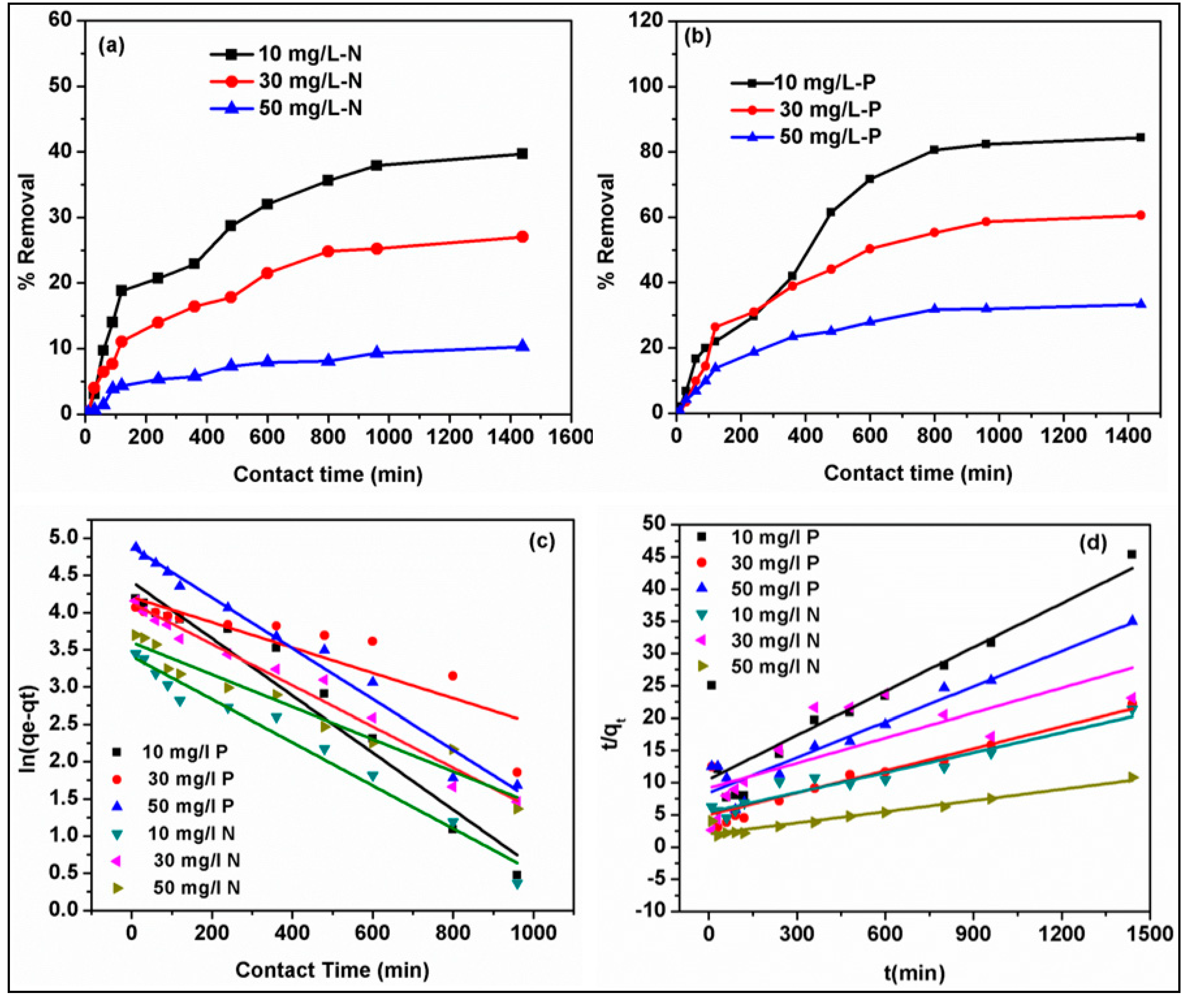
A preliminary adsorption experiment was performed using biochar and biochar-decorated MgAl composites to evaluate the removal performance of nitrate and phosphate and select the adsorbent showing better adsorptive performance for detailed adsorption studies. Batch mode adsorption experiments were performed on the adsorbent chosen to investigate the effect of adsorption parameters including initial solution pH 2–12, adsorbent amounts of 5–100 mg, anion concentration between 10–50 mg/L, contact time within 10–840 min, and temperature (298–318 K) in temperature-controlled agitator at fixed rpm of 275 containing 40 mL of solution in 50 mL plastic vials. All the experiments were performed in duplicate and the average values reported herein.
The removal efficiencies and adsorption capacities were determined by using the following Equations (1) and (2):
where
Co and
Ce are the initial and the equilibrium concentrations (mg/L) of PO
43− and NO
3−, respectively,
qe is the equilibrium amount of PO
43− and NO
3− adsorbed (mg) per unit mass of adsorbent (mg/g),
V is the volume of the solution (L), and m is the mass of the adsorbent in the solution (g).
2.5. Co-Existing Ions Experiment
There are a lot of anions present in the water or groundwater such as carbonate, bicarbonate, nitrate, sulfates, chlorides, and phosphates, etc. which might prevent the removal of nitrate or phosphate during a sorption process. Therefore, the influence of these anions (Na2CO3, NaCl, Na2SO4, NaHCO3, and NaNO3) was investigated at varied concentration from 0.001 to 0.1 M. The co-existing anions sorption experiment was conducted by using 40 mL of reaction mixture containing specified concentration of co-existing anions and 10 mg/L of phosphate and nitrate ions agitated with 5 mg of B5MgAl composite at 200 rpm for 24 h.
2.6. Adsorption Isotherm and Kinetic Modeling
The isotherm and kinetic experiments were performed at various phosphate and nitrate concentrations ranging between 10–50 mg/L, contact time between 10–840 minutes, and variable temperature values (298–318 K) at constant parameters including composite dosage of 5 mg, pH 6, and agitation speed of 275 rpm. The final concentrations of the samples were estimated using a barcoded test kits on the UV–visible spectrophotometer. The adsorption mechanism of phosphate and nitrate onto biochar MgAl composite was demonstrated by utilizing kinetics, isotherm models as well as characterization of the spent composites. Thus, in this study, three kinetic models (pseudo-first, second-order) were used while the linear and non-linear form of Langmuir and Freundlich’s models were employed for the equilibrium study. These models equations and the respective estimated models’ parameters are listed in
Section 3.5 “Adsorption Rate and Kinetic Modeling” and
Section 3.6 “Isotherm Modeling”, respectively.
2.7. Sequential Adsorption-Regeneration Cycles
The adsorption-regeneration experiment was conducted by shaking 200 mL of known concentration of phosphate and nitrate (10.0 mg/L) with 50 mg B
5MgAl composite at 200 rpm and 24 h. After the adsorption experiment, the supernatant was separated from the spent sorbent with the help of filtration followed by centrifugation (5000 rpm and 5 min). The final concentration of the supernatant was measured, as indicated in
Section 2.1. The spent B
5MgAl composite was regenerated with two different levels of NaOH (0.1 and 1.0 M). The recyclability of the B
5MgAl composites was assessed by accomplishing the five consecutive adsorption-regeneration sequences. The successive adsorption/regeneration experiments repeated, and the average values indicated in results.
4. Conclusions
This paper reports the enhanced phosphate and nitrate recovery from the wastewater phase using date palm waste-derived biochar decorated with MgAl-layered double hydroxides. The synthesized composites were characterized by FTIR, SEM, TEM, XRD, and BET techniques to examine the surface and structure properties after the inclusion of LDH layers into biochar. The performance of the new nanocomposite was investigated under several influencing parameters such as contact time, dosage, pH, anions concentration, and temperature. The adsorption capacity and interaction mechanism of the synthesized composite with phosphate and nitrate anions were evaluated in depth by considering isotherms, kinetics, and thermodynamic models. The presence of coexisting anions (Cl−, SO42−, NO3−, CO32−, and HCO3−) negligibly affected the removal of phosphate attributed to its stronger bond on the composites, while the presence of Cl− and PO43− reduced the nitrate removal as a result of these ions participating in the active adsorption sites on the surface of biochar–MgAl with nitrate ions. The composite had a superior selectivity and adsorption affinity towards SO42− and CO32− as compared to Cl− and PO43− ions. The pseudo-first-order model explained better the adsorption of both phosphate and nitrate onto the biochar–MgAl–LDH with a faster removal rate probably with equilibrium attainable quickly, especially at lower initial phosphate–nitrate concentration. The maximum monolayer adsorption capacities of phosphate and nitrate by the non-linear Langmuir model were 177.97 mg/g and 28.06 mg/g at 308 and 298 K, respectively. The regeneration test confirmed that the biochar–MgAl composite can be restored effortlessly and has the potential to be recycled after several treatment sequences. The biochar–MgAl–LDH demonstrated capabilities for higher removal of phosphate and nitrate from real wastewater.