Kinetic Analysis of 4-Nitrophenol Reduction by “Water-Soluble” Palladium Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Dynamic Light Scattering (DLS) Analysis
2.3. Spectrophotometer (UV–vis) Analysis
2.4. Catalytic Reduction of 4-Nitrophenol
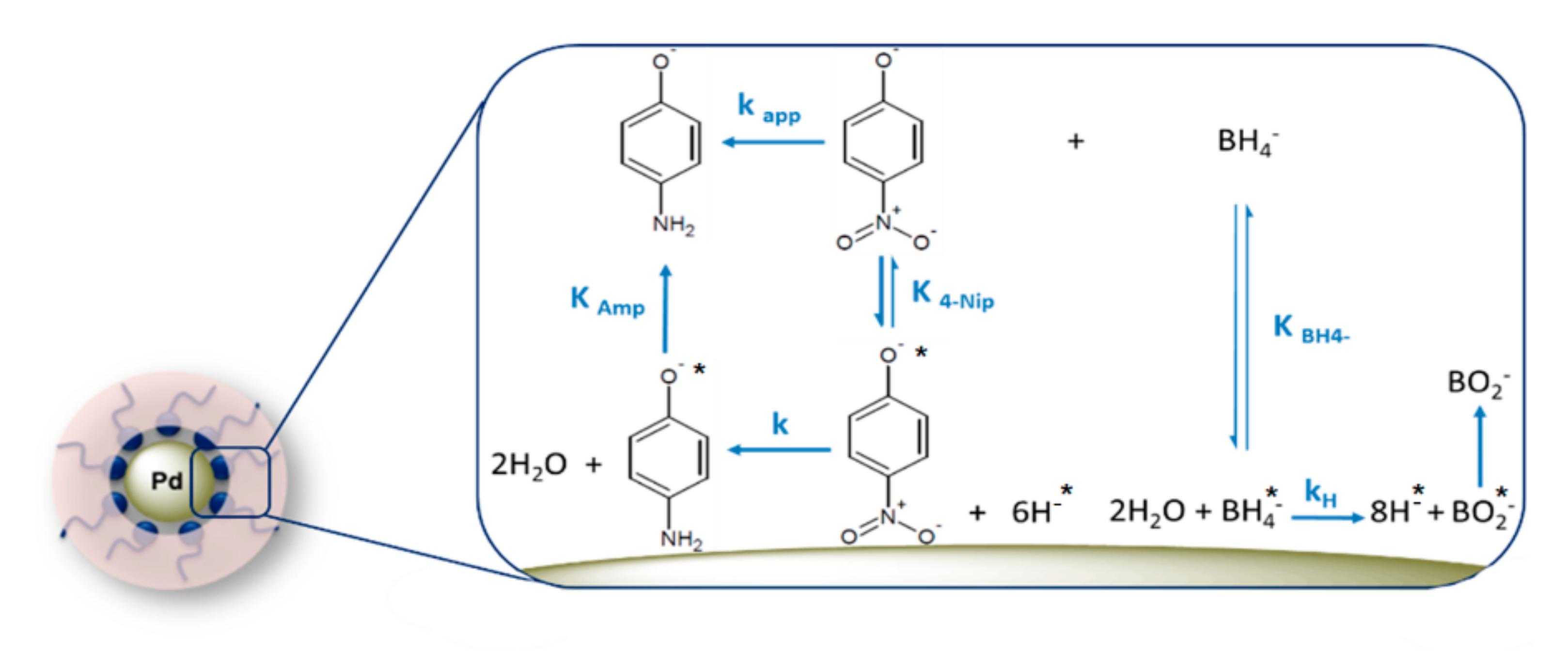
3. Kinetics Analysis
4. Results and Discussion
4.1. Catalytic Reduction of 4-Nitrophenol Using Pd NPs
4.2. Influence of Temperature (Derivation of Thermodynamic Parameters)
4.3. Effects of Surface Area and Concentration of Reagents
4.4. Influence of Reagent Concentration (Derivation of the Langmuir–Hinshelwood Parameters)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferrando, R.; Jellinek, J.; Johnston, R.L. Nanoalloys: From theory to applications of alloy clusters and nanoparticles. Chem. Rev. 2008, 108, 845–910. [Google Scholar] [CrossRef] [PubMed]
- Zahmakıran, M.; Özkar, S. Metal nanoparticles in liquid phase catalysis; from recent advances to future goals. Nanoscale 2011, 3, 3462. [Google Scholar] [CrossRef] [PubMed]
- Astruc, D. Transition-metal nanoparticles in catalysis: From historical background to the state-of-the art. Nanoparticles Catal. 2008, 16, 1–48. [Google Scholar]
- Sardar, R.; Funston, A.M.; Mulvaney, P.; Murray, R.W. Gold nanoparticles: Past, present, and future. Langmuir 2009, 25, 13840–13851. [Google Scholar] [CrossRef] [PubMed]
- Reiss, H. The Fermi level and the redox potential. J. Phys. Chem. 1985, 89, 3783–3791. [Google Scholar] [CrossRef]
- Khan, S.U.M.; Kainthla, R.C.; Bockris, J.O.M. The redox potential and the Fermi level in solution. J. Phys. Chem. 1987, 91, 5974–5977. [Google Scholar] [CrossRef]
- Pradhan, N.; Pal, A.; Pal, T. Silver nanoparticle catalyzed reduction of aromatic nitro compounds. Colloids Surf. A Physicochem. Eng. Asp. 2002, 196, 247–257. [Google Scholar] [CrossRef]
- Antonels, N.C.; Meijboom, R. Preparation of Well-De fi ned Dendrimer Encapsulated Ruthenium Nanoparticles and Their Evaluation in the Reduction of 4-Nitrophenol According to the Langmuir–Hinshelwood Approach. Langmuir 2013, 29, 13433–13442. [Google Scholar] [CrossRef]
- Chen, J.; Song, M.; Li, Y.; Zhang, Y.; Taya, K.; Li, C.M. The effect of phytosterol protects rats against 4-nitrophenol-induced liver damage. Environ. Toxicol. Pharmacol. 2016, 41, 266–271. [Google Scholar] [CrossRef]
- Narayanan, K.B.; Sakthivel, N. Synthesis and characterization of nano-gold composite using Cylindrocladium floridanum and its heterogeneous catalysis in the degradation of 4-nitrophenol. J. Hazard. Mater. 2011, 189, 519–525. [Google Scholar] [CrossRef]
- Hervés, P.; Pérez-Lorenzo, M.; Liz-Marzán, L.M.; Dzubiella, J.; Lu, Y.; Ballauff, M. Catalysis by metallic nanoparticles in aqueous solution: Model reactions. Chem. Soc. Rev. 2012, 41, 5577–5587. [Google Scholar] [CrossRef] [PubMed]
- Wunder, S.; Lu, Y.; Albrecht, M. Catalytic activity of facetted gold nanoparticles studied by a model reaction: Evidence for substrate-induced surface restructuring. ACS Catal. 2011, 1, 908–916. [Google Scholar] [CrossRef]
- Wunder, S.; Polzer, F.; Lu, Y.; Mei, Y.; Ballauff, M. Kinetic analysis of catalytic reduction of 4-nitrophenol by metallic nanoparticles immobilized in spherical polyelectrolyte brushes. J. Phys. Chem. C. 2010, 114, 8814–8820. [Google Scholar] [CrossRef]
- Mahmoud, M.A.; Garlyyev, B.; El-Sayed, M.A. Determining the mechanism of solution metallic nanocatalysis with solid and hollow nanoparticles: Homogeneous or heterogeneous. J. Phys. Chem. C 2013, 117, 21886–21893. [Google Scholar] [CrossRef]
- Iben Ayad, A.; Belda Marín, C.; Colaco, E.; Lefevre, C.; Méthivier, C.; Ould Driss, A.; Landoulsi, J.; Guénin, E. “water soluble” palladium nanoparticle engineering for C–C coupling, reduction and cyclization catalysis. Green Chem. 2019, 21, 6646–6657. [Google Scholar] [CrossRef]
- Gu, S.; Wunder, S.; Lu, Y.; Ballauff, M.; Fenger, R.; Rademann, K.; Jaquet, B.; Zaccone, A. Kinetic analysis of the catalytic reduction of 4-nitrophenol by metallic nanoparticles. J. Phys. Chem. C 2014, 118, 18618–18625. [Google Scholar] [CrossRef]
- Behrens, S.; Heyman, A.; Maul, R.; Essig, S.; Steigerwald, S.; Quintilla, A.; Wenzel, W.; Bürck, J.; Dgany, O.; Shoseyov, O. Constrained synthesis and organization of catalytically active metal nanoparticles by self-assem bled protein templates. Adv. Mater. 2009, 21, 3515–3519. [Google Scholar] [CrossRef]
- Pradhan, N.; Pal, A.; Pal, T. Catalytic reduction of aromatic nitro compounds by coinage metal nanoparticles. Langmuir 2001, 17, 1800–1802. [Google Scholar] [CrossRef]
- Bingwa, N.; Meijboom, R. Kinetic evaluation of dendrimer-encapsulated palladium nanoparticles in the 4-nitrophenol reduction reaction. J. Phys. Chem. C 2014, 118, 19849–19858. [Google Scholar] [CrossRef]
- Tang, S.; Vongehr, S.; Meng, X. Controllable incorporation of Ag and Ag-Au nanoparticles in carbon spheres for tunable optical and catalytic properties. J. Mater. Chem. 2010, 20, 5436–5445. [Google Scholar] [CrossRef]
- Zhang, Z.; Shao, C.; Sun, Y.; Mu, J.; Zhang, M.; Zhang, P.; Guo, Z.; Liang, P.; Wanga, C.; Liu, Y. Tubular nanocomposite catalysts based on size-controlled and highly dispersed silver nanoparticles assembled on electrospun silica nanotubes for catalytic reduction of 4-nitrophenol. J. Mater. Chem. 2012, 22, 1387–1395. [Google Scholar] [CrossRef]
- Huang, J.; Vongehr, S.; Tang, S.; Lu, H.; Meng, X. Highly Catalytic Pd–Ag Bimetallic Dendrites. J. Phys. Chem. C 2010, 114, 15005–15010. [Google Scholar] [CrossRef]
- Zhang, J.S.; Delgass, W.N.; Fisher, T.S.; Gore, J.P. Kinetics of Ru-catalyzed sodium borohydride hydrolysis. J. Power Sources 2007, 164, 772–781. [Google Scholar] [CrossRef]
- Kong, X.; Zhu, H.; Chen, C.L.; Huang, G.; Chen, Q. Insights into the reduction of 4-nitrophenol to 4-aminophenol on catalysts. Chem. Phys. Lett. 2017, 684, 148–152. [Google Scholar] [CrossRef]
- Tewari, B.B.; Boodhoo, M. Removal of p-aminophenol and p-nitrophenol from aqueous solution through adsorption on antimony, cadmium, and zirconium ferrocyanides. J. Colloid Interface Sci. 2005, 289, 328–332. [Google Scholar] [CrossRef]
- Guella, G.; Patton, B.; Miotello, A. Kinetic features of the platinum catalyzed hydrolysis of sodium borohydride from 11B NMR measurements. J. Phys. Chem. C 2007, 111, 18744–18750. [Google Scholar] [CrossRef]
- Liu, B.H.; Li, Z.P. A review: Hydrogen generation from borohydride hydrolysis reaction. J. Power Sources 2009, 187, 527–534. [Google Scholar] [CrossRef]
- Rill, C.; Kolar, Z.I.; Kickelbick, G.; Wolterbeek, H.T.; Peters, J.A. Kinetics and thermodynamics of adsorption on hydroxyapatite of the [160Tb]terbium complexes of the bone-targeting ligands DOTP and BPPED. Langmuir 2009, 25, 2294–2301. [Google Scholar] [CrossRef]
- László, K.; Podkościelny, P.; Da̧browski, A. Heterogeneity of polymer-based active carbons in adsorption of aqueous solutions of phenol and 2,3,4-trichlorophenol. Langmuir 2003, 19, 5287–5294. [Google Scholar] [CrossRef]
- Mei, Y.; Lu, Y.; Polzer, F.; Ballauff, M.; Drechsler, M. Catalytic Activity of Palladium Nanoparticles Encapsulated in Spherical Polyelectrolyte Brushes and Core-Shell Microgels. Chem. A Eur. J. 2007, 19, 1062–1069. [Google Scholar] [CrossRef]
- Menumerov, E.; Hughes, R.A.; Neretina, S. Catalytic Reduction of 4-Nitrophenol: A Quantitative Assessment of the Role of Dissolved Oxygen in Determining the Induction Time. Nano Lett. 2016, 16, 7791–7797. [Google Scholar] [CrossRef] [PubMed]
- Nemanashi, M.; Meijboom, R. Synthesis and characterization of Cu, Ag and Au dendrimer-encapsulated nanoparticles and their application in the reduction of 4-nitrophenol to 4-aminophenol. J. Colloid Interface Sci. 2013, 389, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Grzeschik, R.; Schäfer, D.; Holtum, T.; Küpper, S.; Hoffmann, A.; Schlücker, S. On the Overlooked Critical Role of the pH Value on the Kinetics of the 4-Nitrophenol NaBH4-Reduction Catalyzed by Noble-Metal Nanoparticles (Pt, Pd, and Au). J. Phys. Chem. C 2020, 124. [Google Scholar] [CrossRef]
- Mahmoud, M.A.; Saira, F.; El-Sayed, M.A. Experimental evidence for the nanocage effect in catalysis with hollow nanoparticles. Nano Lett. 2010, 10, 3764–3769. [Google Scholar] [CrossRef]
- Noh, J.H.; Meijboom, R. Catalytic evaluation of dendrimer-templated Pd nanoparticles in the reduction of 4-nitrophenol using Langmuir–Hinshelwood kinetics. Appl. Surf. Sci. 2014, 320, 400–413. [Google Scholar] [CrossRef]
- Petek, A.; Krajnc, M. The enthalpy and entropy of activation for ethyl acetate saponification. Int. J. Chem. Kinet. 2012, 44, 692–698. [Google Scholar] [CrossRef]
- Consorti, C.S.; Flores, F.R.; Dupont, J. Kinetics and mechanistic aspects of the Heck reaction promoted by a CN-palladacycle. J. Am. Chem. Soc. 2005, 127, 12054–12065. [Google Scholar] [CrossRef]
- Corma, S.W.A.; Llopis, F.; Monton, J.B. On the Compensation Effect in Acid-Base Catalyzed-Reactions on Zeolites-Reply. J. Catal. 1994, 148, 415–416. [Google Scholar] [CrossRef]
- Bond, G.C.; Keane, M.A.; Kral, H.; Lercher, J.A. Compensation Phenomena in Heterogeneous Catalysis: General Principles and a Possible Explanation. Catal. Rev. Sci. Eng. 2000, 42, 323–383. [Google Scholar] [CrossRef]
- Bond, G.C. Kinetics of alkane reactions on metal catalysts: Activation energies and the compensation effect. Catal. Today 1999, 49, 41–48. [Google Scholar] [CrossRef]
- Rooney, J.J. The extended Eyring kinetic equation and the compensation effect in catalysis. J. Mol. Catal. A Chem. 1998, 129, 131–134. [Google Scholar] [CrossRef]
- Kohantorabi, M.; Gholami, M.R. Kinetic Analysis of the Reduction of 4-Nitrophenol Catalyzed by CeO2 Nanorods-Supported CuNi Nanoparticles. Ind. Eng. Chem. Res. 2017, 56, 1159–1167. [Google Scholar] [CrossRef]
- Chen, L.; Hu, J.; Qi, Z.; Fang, Y.; Richards, R. Gold nanoparticles intercalated into the walls of mesoporous silica as a versatile redox catalyst. Ind. Eng. Chem. Res. 2011, 50, 13642–13649. [Google Scholar] [CrossRef]
- Vannice, M.A. Catalytic Reactions; Springer: New York, NY, USA, 2005. [Google Scholar]
- Bielejewska, A.; Bylina, A.; Duszczyk, K.; Fiałkowski, M.; Hołyst, R. Evaluation of Ligand-Selector Interaction from Effective Diffusion Coefficient. Anal. Chem. 2010, 82, 5463–5469. [Google Scholar] [CrossRef]
- Johnson, J.A.; Makis, J.J.; Marvin, K.A.; Rodenbusch, S.E.; Stevenson, K.J. Size-dependent hydrogenation of p-nitrophenol with Pd nanoparticles synthesized with poly(amido)amine dendrimer templates. J. Phys. Chem. C 2013, 117, 22644–22651. [Google Scholar] [CrossRef]
- Zhao, P.; Feng, X.; Huang, D.; Yang, G.; Astruc, D. Basic concepts and recent advances in nitrophenol reduction by gold- and other transition metal nanoparticles. Coord. Chem. Rev. 2015, 287, 114–136. [Google Scholar] [CrossRef]









| BH4− | Eaapp (kJ·mol−1) | A | ΔH‡ (kJ·mol−1) | ΔS‡ (J·mol−1·K−1) | ΔG‡ (kJ·mol−1) |
|---|---|---|---|---|---|
| 150 eq | 81.22 | 1.46 × 1011 | 78.68 | −39.57 | 90.47 |
| 200 eq | 112.77 | 7.13 × 1016 | 110.22 | 69.36 | 89.56 |
| a (m−1) | β (m/s) | δ (nm) | kappa (×10−3 s−1) | DaII a (×10−4) | kappb (×10−3 s−1) | DaII b (×10−4) |
|---|---|---|---|---|---|---|
| 4613.1 | 0.077 | 9.045 | 34.05 | 0.965 | 10.23 | 0.290 |
| 1858.5 | 0.077 | 9.045 | 24.50 | 1.723 | 4.22 | 0.297 |
| 929.3 | 0.077 | 9.045 | 17.62 | 2.478 | 0.95 | 0.134 |
| 371.7 | 0.077 | 9.045 | 0.55 | 5.453 | 0.02 | 0.221 |
| T (K) | k (mol/m2·s) | K4-Nip (L/mol) | KBH4− (L/mol) |
|---|---|---|---|
| 317 | 0.02 | 8900 | 2.45 |
| 307 | 0.0115 | 6300 | 2.34 |
| 303 | 0.0083 | 5200 | 2.3 |
| 301 | 0.007 | 4800 | 2.28 |
| 300 | 0.006 | 4600 | 2.27 |
| 297.8 | 0.005 | 4200 | 2.25 |
| 296.3 | 0.0037 | 3900 | 2.235 |
| K4-Nip | KBH4− | |
|---|---|---|
| ΔHad [kJ/mol] | 30.87 | 3.31 |
| ΔSad [J/mol K] | 173 | 17.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iben Ayad, A.; Luart, D.; Ould Dris, A.; Guénin, E. Kinetic Analysis of 4-Nitrophenol Reduction by “Water-Soluble” Palladium Nanoparticles. Nanomaterials 2020, 10, 1169. https://doi.org/10.3390/nano10061169
Iben Ayad A, Luart D, Ould Dris A, Guénin E. Kinetic Analysis of 4-Nitrophenol Reduction by “Water-Soluble” Palladium Nanoparticles. Nanomaterials. 2020; 10(6):1169. https://doi.org/10.3390/nano10061169
Chicago/Turabian StyleIben Ayad, Anas, Denis Luart, Aissa Ould Dris, and Erwann Guénin. 2020. "Kinetic Analysis of 4-Nitrophenol Reduction by “Water-Soluble” Palladium Nanoparticles" Nanomaterials 10, no. 6: 1169. https://doi.org/10.3390/nano10061169
APA StyleIben Ayad, A., Luart, D., Ould Dris, A., & Guénin, E. (2020). Kinetic Analysis of 4-Nitrophenol Reduction by “Water-Soluble” Palladium Nanoparticles. Nanomaterials, 10(6), 1169. https://doi.org/10.3390/nano10061169






