The Toxicity of Coated Silver Nanoparticles and Their Stabilizers towards Paracentrotus lividus Sea Urchin Embryos
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
2.2. Characterization of AgNPs
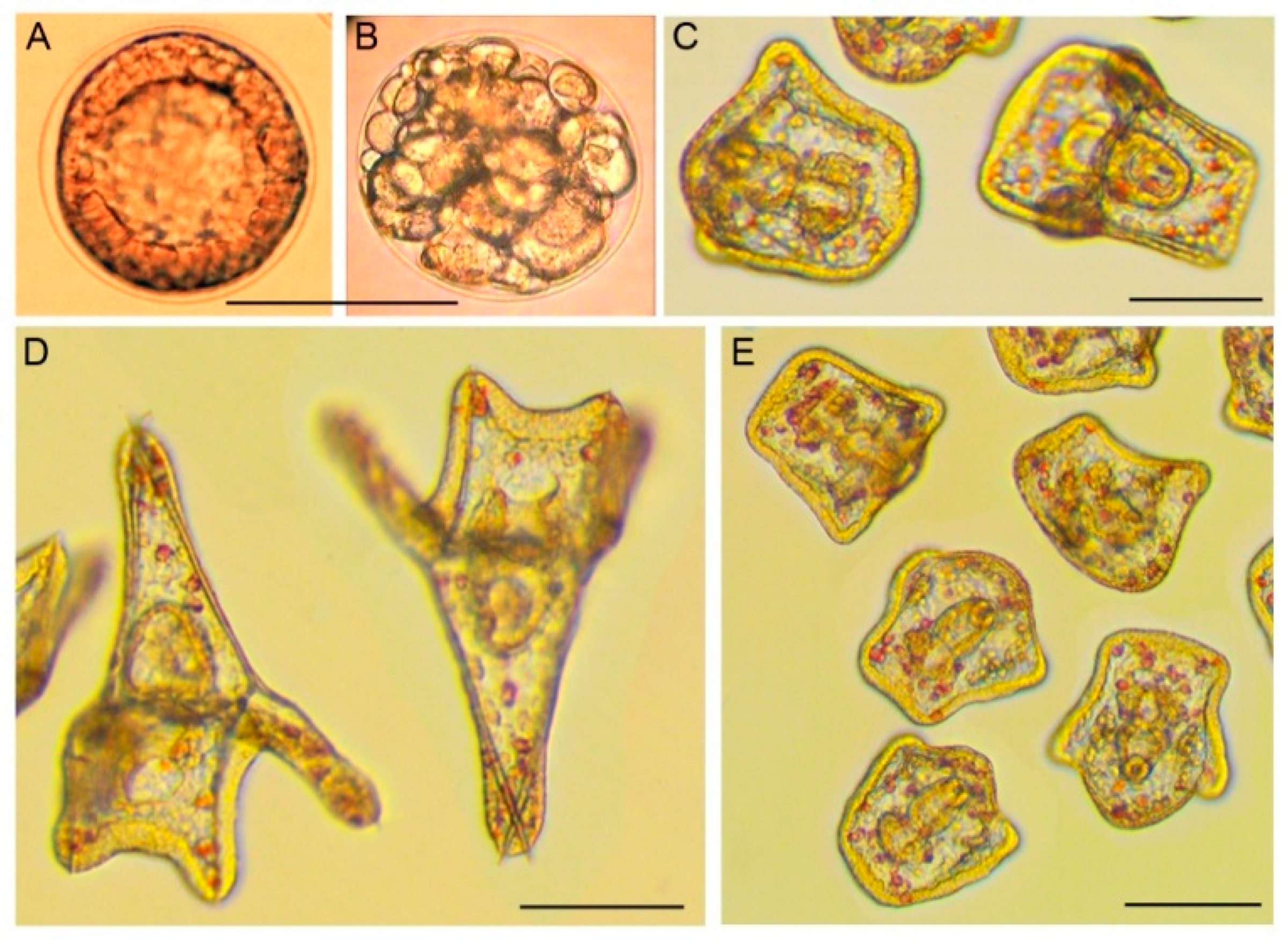
2.3. Phenotypic Sea Urchin Embryo Assay
3. Results and Discussion
3.1. Characterization of Nanosilver Samples
3.2. Environmental Fate
3.3. Toxicity of AgNPs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Usman, M.; Farooq, M.; Wakeel, A.; Nawaz, A.; Cheema, S.A.; ur Rehman, H.; Ashraf, I.; Sanaullah, M. Nanotechnology in agriculture: Current status, challenges and future opportunities. Sci. Total Environ. 2020, 721, 137778. [Google Scholar] [CrossRef] [PubMed]
- Hicks, A.L.; Temizel-Sekeryan, S. Understanding the potential environmental benefits of nanosilver enabled consumer products. NanoImpact 2019, 16, 100183. [Google Scholar] [CrossRef]
- Boughbina-Portolés, A.; Sanjuan-Navarro, L.; Moliner-Martínez, Y.; Campíns-Falcó, P. Study of the Stability of Citrate Capped AgNPs in Several Environmental Water Matrices by Asymmetrical Flow Field Flow Fractionation. Nanomater 2021, 11, 926. [Google Scholar] [CrossRef] [PubMed]
- Krutyakov, Y.A.; Kudrinskiy, A.A.; Zherebin, P.M. Method of stimulating plant growth and developmentTitle of Patent. EAPO Patent No. 037437, 26 March 2021. [Google Scholar]
- Chindera, K.; Mahato, M.; Kumar Sharma, A.; Horsley, H.; Kloc-Muniak, K.; Kamaruzzaman, N.F.; Kumar, S.; McFarlane, A.; Stach, J.; Bentin, T.; et al. The antimicrobial polymer PHMB enters cells and selectively condenses bacterial chromosomes. Sci. Rep. 2016, 6, 23121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krutyakov, Y.A.; Kudrinsky, A.A.; Gusev, A.A.; Zakharova, O.V.; Klimov, A.I.; Yapryntsev, A.D.; Zherebin, P.M.; Shapoval, O.A.; Lisichkin, G.V. Synthesis of positively charged hybrid PHMB-stabilized silver nanoparticles: The search for a new type of active substances used in plant protection products. Mater. Res. Express 2017, 4, 075018. [Google Scholar] [CrossRef]
- Krutyakov, Y.A.; Kudrinskiy, A.A.; Zherebin, P.M.; Yapryntsev, A.D.; Pobedinskaya, M.A.; Elansky, S.N.; Denisov, A.N.; Mikhaylov, D.M.; Lisichkin, G.V. Tallow amphopolycarboxyglycinate-stabilized silver nanoparticles: New frontiers in development of plant protection products with a broad spectrum of action against phytopathogens. Mater. Res. Express 2016, 3, 075403. [Google Scholar] [CrossRef] [Green Version]
- Krutyakov, Y.A.; Kudrinskiy, A.A.; Zherebin, P.M.; Lisichkin, G.V. Correlation between the rate of silver nanoparticle oxidation and their biological activity: The role of the capping agent. J. Nanopart. Res. 2019, 21, 69. [Google Scholar] [CrossRef]
- Abramenko, N.B.; Demidova, T.B.; Krutyakov, Y.A.; Zherebin, P.M.; Krysanov, E.Y.; Kustov, L.M.; Peijnenburg, W. The effect of capping agents on the toxicity of silver nanoparticles to Danio rerio embryos. Nanotoxicology 2019, 13, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Pagano, G.; Guida, M.; Trifuoggi, M.; Thomas, P.; Palumbo, A.; Romano, G.; Oral, R. Sea Urchin Bioassays in Toxicity Testing: I. Inorganics, Organics, Complex Mixtures and Natural Products. Expert Opin. Environ. Biol. 2017, 6, 1. [Google Scholar] [CrossRef] [Green Version]
- Ettensohn, C.A. Sea Urchins as a Model System for Studying Embryonic Development. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Agnello, M.; Zhadan, P. Sea Urchin—From Environment to Aquaculture and Biomedicine; InTech: Rijeka, Croatia, 2017. [Google Scholar] [CrossRef]
- Burić, P.; Jakšić, Ž.; Štajner, L.; Sikirić, M.D.; Jurašin, D.; Cascio, C.; Calzolai, L.; Lyons, D.M. Effect of silver nanoparticles on Mediterranean sea urchin embryonal development is species specific and depends on moment of first exposure. Mar. Environ. Resactions 2015, 111, 50–59. [Google Scholar] [CrossRef] [Green Version]
- Siller, L.; Lemloh, M.L.; Piticharoenphun, S.; Mendis, B.G.; Horrocks, B.R.; Brümmer, F.; Medaković, D. Silver nanoparticle toxicity in sea urchin Paracentrotus lividus. Environ. Pollut. 2013, 178, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Magesky, A.; Pelletier, É. Toxicity mechanisms of ionic silver and polymer-coated silver nanoparticles with interactions of functionalized carbon nanotubes on early development stages of sea urchin. Aquat. Toxicol. 2015, 167, 106–123. [Google Scholar] [CrossRef] [PubMed]
- Gusev, A.A.; Kudrinsky, A.A.; Zakharova, O.V.; Klimov, A.I.; Zherebin, P.M.; Lisichkin, G.V.; Vasyukova, I.A.; Denisov, A.N.; Krutyakov, Y.A. Versatile synthesis of PHMB-stabilized silver nanoparticles and their significant stimulating effect on fodder beet (Beta vulgaris L.). Mater. Sci. Eng. C 2016, 62, 152–159. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and zeta potential—What they are and what they are not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Semenova, M.N.; Demchuk, D.V.; Tsyganov, D.V.; Chernysheva, N.B.; Samet, A.V.; Silyanova, E.A.; Kislyi, V.P.; Maksimenko, A.S.; Varakutin, A.E.; Konyushkin, L.D.; et al. Sea Urchin Embryo Model as a Reliable in Vivo Phenotypic Screen to Characterize Selective Antimitotic Molecules. Comparative evaluation of Combretapyrazoles, -isoxazoles, -1,2,3-triazoles, and -pyrroles as Tubulin-Binding Agents. ACS Comb. Sci. 2018, 20, 700–721. [Google Scholar] [CrossRef] [PubMed]
- Semenova, M.N.; Tsyganov, D.V.; Yakubov, A.P.; Kiselyov, A.S.; Semenov, V.V. A Synthetic Derivative of Plant Allylpolyalkoxybenzenes Induces Selective Loss of Motile Cilia in Sea Urchin Embryos. ACS Chem. Biol. 2008, 3, 95–100. [Google Scholar] [CrossRef]
- Schaumann, G.; Philippe, A.; Bundschuh, M.; Metreveli, G.; Klitzke, S.; Rakcheev, D.; Grün, A.; Kumahor, S.; Kühn, M.; Baumann, T.; et al. Understanding the fate and biological effects of Ag- and TiO₂-nanoparticles in the environment: The quest for advanced analytics and interdisciplinary concepts. Sci. Total Environ. 2015, 535, 3–19. [Google Scholar] [CrossRef]
- Levard, C.; Hotze, E.; Lowry, G.; Brown, G. Environmental transformations of silver nanoparticles: Impact on stability and toxicity. Environ. Sci. Technol. 2012, 46, 6900–6914. [Google Scholar] [CrossRef]
- Li, P.; Su, M.; Wang, X.; Zou, X.; Sun, X.; Shi, J.; Zhang, H. Environmental fate and behavior of silver nanoparticles in natural estuarine systems. J. Environ. Sci. (China) 2020, 88, 248–259. [Google Scholar] [CrossRef]
- Gambardella, C.; Ferrando, S.; Morgana, S.; Gallus, L.; Ramoino, P.; Ravera, S.; Bramini, M.; Diaspro, A.; Faimali, M.; Falugi, C. Exposure of Paracentrotus lividus male gametes to engineered nanoparticles affects skeletal bio-mineralization processes and larval plasticity. Aquat. Toxicol. 2015, 158, 181–191. [Google Scholar] [CrossRef]
- Magesky, A.; Pelletier, É. Cytotoxicity and Physiological Effects of Silver Nanoparticles on Marine Invertebrates. Adv. Exp. Med. Biol. 2018, 1048, 285–309. [Google Scholar] [CrossRef] [PubMed]
- Piticharoenphun, S.; Šiller, L.; Lemloh, M.-L.; Salome, M.; Cotte, M.; Kaulich, B.; Gianoncelli, A.; Mendis, B.G.; Bangert, U.; Poolton, N.R.J.; et al. Agglomeration of Silver Nanoparticles in Sea Urchin. Int. J. Environ. Pollut. Remediat. 2012, 1, 46–52. [Google Scholar] [CrossRef] [Green Version]
- US EPA. Technical Overview of Ecological Risk Assessment—Analysis Phase: Ecological Effects Characterization. Available online: https://www.epa.gov/pesticide-science-and-assessing-pesticide-risks/technical-overview-ecological-risk-assessment-0 (accessed on 27 August 2022).
- Gambardella, C.; Aluigi, M.G.; Ferrando, S.; Gallus, L.; Ramoino, P.; Gatti, A.M.; Rottigni, M.; Falugi, C. Developmental abnormalities and changes in cholinesterase activity in sea urchin embryos and larvae from sperm exposed to engineered nanoparticles. Aquat. Toxicol. 2013, 130–131, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Manno, D.; Serra, A.; Buccolieri, A.; Panzarini, E.; Carata, E.; Tenuzzo, B.; Izzo, D.; Vergallo, C.; Rossi, M.; Dini, L. Silver and carbon nanoparticles toxicity in sea urchin Paracentrotus lividus embryos. BioNanoMaterials 2013, 14, 229–238. [Google Scholar] [CrossRef]
- Angel, B.; Batley, G.; Jarolimek, C.; Rogers, N. The impact of size on the fate and toxicity of nanoparticulate silver in aquatic systems. Chemosphere 2013, 93, 359–365. [Google Scholar] [CrossRef]
- Golovina, N.B.; Kustov, L.M. Toxicity of metal nanoparticles with a focus on silver. Mendeleev Commun. 2013, 23, 59–65. [Google Scholar] [CrossRef]
- Bundschuh, M.; Filser, J.; Lüderwald, S.; McKee, M.S.; Metreveli, G.; Schaumann, G.E.; Schulz, R.; Wagner, S. Nanoparticles in the environment: Where do we come from, where do we go to? Environ. Sci. Eur. 2018, 30, 6. [Google Scholar] [CrossRef] [Green Version]
- Kudrinskiy, A.; Zherebin, P.; Gusev, A.; Shapoval, O.; Pyee, J.; Lisichkin, G.; Krutyakov, Y. New Relevant Descriptor of Linear QNAR Models for Toxicity Assessment of Silver Nanoparticles. Nanomater 2020, 10, 1459. [Google Scholar] [CrossRef]




| Sample Ag/Stabilizer | CAg/Stabilizer, mg/L | D (TEM), nm | D, nm a | ζ-Potential, mV b | ||
|---|---|---|---|---|---|---|
| Distilled Water | Seawater | Distilled Water | Seawater | |||
| Ag/STAPCG | 3000/48,000 | 19.4 ± 9.4 | 52.5 ± 5.9 | 75.2 ± 13.1 | −52.9 | −75.9 |
| Ag/PHMB&SLES | 500/500/5200 | 14.9 ± 10.2 | 59.5 ± 3.1 | 33.4 ± 9.9 | −54.7 | −61.5 |
| Ag/SADG | 500/2000 | 12.8 ± 7.1 | 61.9 ± 3.8 | 70.7 ± 10.8 | −36.1 | −22.4 |
| Ag/AMA | 500/4000 | 8.0 ± 1.9 | 52.2 ± 9.6 | 200.3 ± 2.9 | −50.5 | −24.4 |
| Ag/PHMB | 500/500 | 6.6 ± 4.8 | 48.8 ± 0.5 | 1139.0 ± 57.8 | 42.8 | NA c |
| Ag/SLES | 100/1000 | 7.5 ± 3.6 | 45.8 ± 1.5 | 299.3 ± 20.5 | −46.8 | NA c |
| Stabilizer | MLC FE, mg/L | MLC HB, mg/L | MEC, mg/L |
|---|---|---|---|
| STAPCG | 2.4 | 4.8 | 2.4 |
| PHMB/PHMB&SLES a | 1 | 1 | 0.25 |
| SLES/PHMB&SLES b | 10.4 | 10.4 | 2.6 |
| SADG | 4 | 4 | 2 |
| AMA | 8 | 20 | 2 |
| PHMB | >5 | >5 | 2.5 |
| SLES | 5 | 10 | 2.5 |
| AgNPs | AgNPs MLC, mg/L | AgNPs MEC, mg/L | ||
|---|---|---|---|---|
| FE | HB | FE | HB | |
| Ag/STAPCG | 0.15 a | 0.30 a | 0.06 | 0.06 |
| Ag/PHMB&SLES | 0.50 | 1.00 a | 0.25 a | 0.25 a |
| Ag/SADG | 0.50 a | 0.50 a | 0.13 | 0.13 |
| Ag/AMA | 0.50 | 0.50 | 0.13 | 0.13 |
| Ag/PHMB b | 2.50 | >2.50 | 0.30 | 1.25 |
| Ag/SLES | 0.50 a | 0.50 a | 0.12 | 0.12 |
| Ag ions | 0.064 | >0.13 | 0.03 | 0.064 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abramenko, N.; Semenova, M.; Khina, A.; Zherebin, P.; Krutyakov, Y.; Krysanov, E.; Kustov, L. The Toxicity of Coated Silver Nanoparticles and Their Stabilizers towards Paracentrotus lividus Sea Urchin Embryos. Nanomaterials 2022, 12, 4003. https://doi.org/10.3390/nano12224003
Abramenko N, Semenova M, Khina A, Zherebin P, Krutyakov Y, Krysanov E, Kustov L. The Toxicity of Coated Silver Nanoparticles and Their Stabilizers towards Paracentrotus lividus Sea Urchin Embryos. Nanomaterials. 2022; 12(22):4003. https://doi.org/10.3390/nano12224003
Chicago/Turabian StyleAbramenko, Natalia, Marina Semenova, Alexander Khina, Pavel Zherebin, Yurii Krutyakov, Evgeny Krysanov, and Leonid Kustov. 2022. "The Toxicity of Coated Silver Nanoparticles and Their Stabilizers towards Paracentrotus lividus Sea Urchin Embryos" Nanomaterials 12, no. 22: 4003. https://doi.org/10.3390/nano12224003






