Advances in Biomimetic Nerve Guidance Conduits for Peripheral Nerve Regeneration
Abstract
:1. Introduction
2. Physicochemical Properties and Regeneration Capacity of Peripheral Nerves
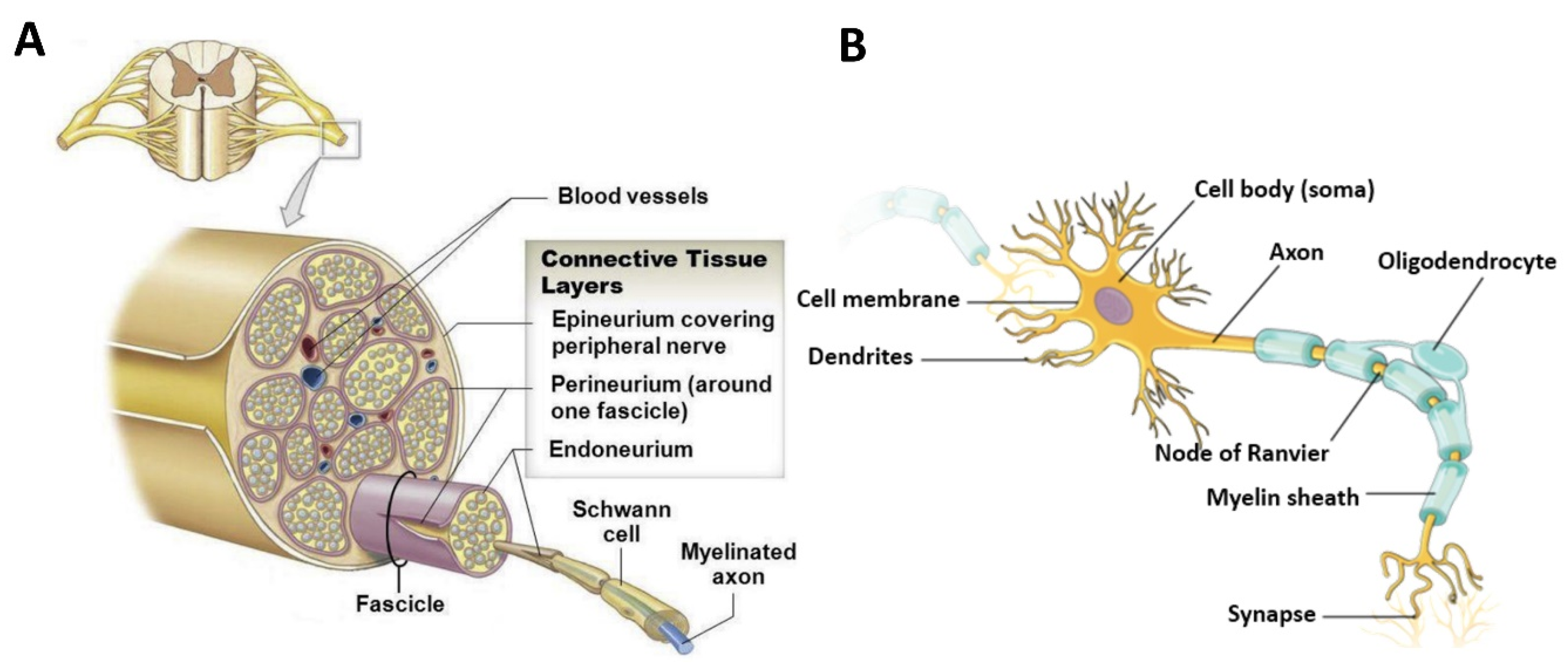
2.1. Structural, Compositional, and Physical Properties of Peripheral Nerves
2.2. Regeneration of Peripheral Nerve Injuries
3. Design Considerations for Biomimetic Nerve Guidance Conduits
3.1. Compositional Considerations
3.1.1. Material Choice
Biomimetic Synthetic Materials
Biomimetic Natural Materials
Biomimetic Hybrid Materials

3.1.2. Cellular and Biomolecular Choices
Cellular Choices
Biomolecular Choices
3.2. Structural Considerations
3.2.1. Biomimetic Architectures
Biomimetic Topographical Features of NGCs
Biomimetic Fillers for NGCs
Multi-Channeled NGCs
3.2.2. Fabrication of Biomimetic Features
| Fabricating Topographical Features | |||
| Technologies | Advantages | Disadvantages | Ref. |
| Electro- spinning |
|
| [13,14,49,105,123,135] |
| Inkjet printing |
|
| [138] |
| Micropatterning |
|
| [102,125,142,144] |
| Fabricating Porous Features | |||
| Technologies | Advantages | Disadvantages | Ref. |
| Freeze-drying |
|
| [49,62] |
| Solvent casting and salt-leaching |
|
| [15,150,151] |
| Fabricating Multichannel | |||
| Technologies | Advantages | Disadvantages | Ref. |
| Molding |
|
| [15,56,120] |
| 3D printing |
|
| [9,152] |
3.2.3. Mechanical Properties of Biomimetic NGCs
4. Discussion and Future Outlooks
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lopes, B.; Sousa, P.; Alvites, R.; Branquinho, M.; Sousa, A.C.; Mendonça, C.; Atayde, L.M.; Luís, A.L.; Varejão, A.S.P.; Maurício, A.C. Peripheral Nerve Injury Treatments and Advances: One Health Perspective. Int. J. Mol. Sci. 2022, 23, 918. [Google Scholar] [CrossRef]
- Yan, Y.; Yao, R.; Zhao, J.; Chen, K.; Duan, L.; Wang, T.; Zhang, S.; Guan, J.; Zheng, Z.; Wang, X.; et al. Implantable nerve guidance conduits: Material combinations, multi-functional strategies and advanced engineering innovations. Bioact. Mater. 2022, 11, 57–76. [Google Scholar] [CrossRef]
- Bhandari, P.S. Management of peripheral nerve injury. J. Clin. Orthop. Trauma 2019, 10, 862–866. [Google Scholar] [CrossRef]
- Zhang, F.; Fischer, K.A. End-to-side neurorrhaphy. Microsurgery 2002, 22, 122–127. [Google Scholar] [CrossRef]
- Yan, Z.; Qian, Y.; Fan, C. Biomimicry in 3D printing design: Implications for peripheral nerve regeneration. Regen. Med. 2021, 16, 683–701. [Google Scholar] [CrossRef]
- Hussain, G.; Wang, J.; Rasul, A.; Anwar, H.; Qasim, M.; Zafar, S.; Aziz, N.; Razzaq, A.; Hussain, R.; de Aguilar, J.L.G.; et al. Current status of therapeutic approaches against peripheral nerve injuries: A detailed story from injury to recovery. Int. J. Biol. Sci. 2020, 16, 116–134. [Google Scholar] [CrossRef]
- Faroni, A.; Mobasseri, S.A.; Kingham, P.J.; Reid, A.J. Peripheral nerve regeneration: Experimental strategies and future perspectives. Adv. Drug Deliv. Rev. 2015, 82, 160–167. [Google Scholar] [CrossRef]
- Behtaj, S.; Ekberg, J.A.K.; St John, J.A. Advances in Electrospun Nerve Guidance Conduits for Engineering Neural Regeneration. Pharmaceutics 2022, 14, 219. [Google Scholar] [CrossRef]
- Joung, D.; Lavoie, N.S.; Guo, S.Z.; Park, S.H.; Parr, A.M.; McAlpine, M.C. 3D Printed Neural Regeneration Devices. Adv. Funct. Mater. 2020, 30, 1906237. [Google Scholar] [CrossRef]
- Wofford, K.L.; Shultz, R.B.; Burrell, J.C.; Cullen, D.K. Neuroimmune interactions and immunoengineering strategies in peripheral nerve repair. Prog. Neurobiol. 2022, 208, 102172. [Google Scholar] [CrossRef]
- Rodríguez-Sánchez, D.N.; Pinto, G.B.A.; Cartarozzi, L.P.; de Oliveira, A.L.R.; Bovolato, A.L.C.; de Carvalho, M.; da Silva, J.V.L.; de Dernowsek, J.A.; Golim, M.; Barraviera, B.; et al. 3D-printed nerve guidance conduits multi-functionalized with canine multipotent mesenchymal stromal cells promote neuroregeneration after sciatic nerve injury in rats. Stem Cell Res. Ther. 2021, 12, 303. [Google Scholar] [CrossRef]
- Sabongi, R.G.; Fernandes, M.; Dos Gomes Santos, J.B. Peripheral nerve regeneration with conduits: Use of vein tubes. Neural Regen. Res. 2015, 10, 529–533. [Google Scholar] [CrossRef]
- Zargar Kharazi, A.; Dini, G.; Naser, R. Fabrication and evaluation of a nerve guidance conduit capable of Ca2+ ion release to accelerate axon extension in peripheral nerve regeneration. J. Biomed. Mater. Res. Part A 2018, 106, 2181–2189. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Zhao, X.; Han, Q.; Chen, W.; Li, H.; Yuan, W. An integrated multi-layer 3D-fabrication of PDA/RGD coated graphene loaded PCL nanoscaffold for peripheral nerve restoration. Nat. Commun. 2018, 9, 323. [Google Scholar] [CrossRef] [PubMed]
- Kang, N.U.; Lee, S.J.; Gwak, S.J. Fabrication Techniques of Nerve Guidance Conduits for Nerve Regeneration. Yonsei Med. J. 2022, 63, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, X.; Xiao, C.; Liu, B. Engineered hydrogels for peripheral nerve repair. Mater. Today Bio 2023, 20, 100668. [Google Scholar] [CrossRef]
- Nadeau, S.; Filali, M.; Zhang, J.; Kerr, B.J.; Rivest, S.; Soulet, D.; Iwakura, Y.; de Vaccari, J.P.R.; Keane, R.W.; Lacroix, S. Functional recovery after peripheral nerve injury is dependent on the pro-inflammatory cytokines IL-1β and TNF: Implications for neuropathic pain. J. Neurosci. 2011, 31, 12533–12542. [Google Scholar] [CrossRef] [PubMed]
- Topp, K.S.; Boyd, B.S. Structure and biomechanics of peripheral nerves: Nerve responses to physical stresses and implications for physical therapist practice. Phys. Ther. 2006, 86, 92–109. [Google Scholar] [CrossRef]
- Dalton, P.D.; O’Neill, K.L.; Pêgo, A.P.; Plant, G.W.; Nisbet, D.R.; Oudega, M.; Brook, G.A.; Harvey, A.R. Tissue engineering of the nervous system. In Tissue Engineering; Elsevier: Amsterdam, The Netherlands, 2023; pp. 585–627. [Google Scholar]
- Dubový, P.; Jančálek, R.; Kubek, T. Role of Inflammation and Cytokines in Peripheral Nerve Regeneration. Int. Rev. Neurobiol. 2013, 108, 173–206. [Google Scholar]
- Stoll, G.; Müller, H.W. Nerve injury, axonal degeneration and neural regeneration: Basic insights. Brain Pathol. 1999, 9, 313–325. [Google Scholar] [CrossRef]
- Cashman, C.R.; Höke, A. Mechanisms of distal axonal degeneration in peripheral neuropathies. Neurosci. Lett. 2015, 596, 33–50. [Google Scholar] [CrossRef]
- Martini, F.H.; Tallitsch, R.B.; Judi, L.; Nath, P.D. Human Anatomy; Pearson: London, UK, 2016; ISBN 9780134320762. [Google Scholar]
- Gordon Betts, J.B. Anatomy and Physiology; OpenStax: Houston, TX, USA, 2012. [Google Scholar]
- Gao, X.; Wang, Y.; Chen, J.; Peng, J. The role of peripheral nerve ECM components in the tissue engineering nerve construction. Rev. Neurosci. 2013, 24, 443–453. [Google Scholar] [CrossRef]
- Estrada, J.B.; Cramer, H.C.; Scimone, M.T.; Buyukozturk, S.; Franck, C. Neural cell injury pathology due to high-rate mechanical loading. Brain Multiphysics 2021, 2, 100034. [Google Scholar] [CrossRef]
- Hasan, F.; Mahmud, K.A.H.A.; Khan, M.I.; Adnan, A. Viscoelastic damage evaluation of the axon. Front. Bioeng. Biotechnol. 2022, 10, 904818. [Google Scholar] [CrossRef]
- Kwan, M.K.; Wall, E.J.; Massie, J.; Garfin, S.R. Strain, stress and stretch of peripheral nerve rabbit experiments in vitro and in vivo. Acta Orthop. 1992, 63, 267–272. [Google Scholar] [CrossRef]
- Arslantunali, D.; Dursun, T.; Yucel, D.; Hasirci, N.; Hasirci, V. Peripheral nerve conduits: Technology update. Med. Devices Evid. Res. 2014, 7, 405–424. [Google Scholar] [CrossRef]
- Du, J.; Liu, J.; Yao, S.; Mao, H.; Peng, J.; Sun, X.; Cao, Z.; Yang, Y.; Xiao, B.; Wang, Y.; et al. Prompt peripheral nerve regeneration induced by a hierarchically aligned fibrin nanofiber hydrogel. Acta Biomater. 2017, 55, 296–309. [Google Scholar] [CrossRef]
- Gaudin, R.; Knipfer, C.; Henningsen, A.; Smeets, R.; Heiland, M.; Hadlock, T. Approaches to peripheral nerve repair: Generations of biomaterial conduits yielding to replacing autologous nerve grafts in craniomaxillofacial surgery. Biomed Res. Int. 2016, 2016, 3856262. [Google Scholar] [CrossRef]
- Kaplan, B.; Levenberg, S. The Role of Biomaterials in Peripheral Nerve and Spinal Cord Injury: A Review. Int. J. Mol. Sci. 2022, 23, 1244. [Google Scholar] [CrossRef]
- Ma, Y.; Gao, H.; Wang, H.; Cao, X. Engineering topography: Effects on nerve cell behaviors and applications in peripheral nerve repair. J. Mater. Chem. B 2021, 9, 6310–6325. [Google Scholar] [CrossRef]
- Lee, D.J.; Fontaine, A.; Meng, X.; Park, D. Biomimetic Nerve Guidance Conduit Containing Intraluminal Microchannels with Aligned Nanofibers Markedly Facilitates in Nerve Regeneration. ACS Biomater. Sci. Eng. 2016, 2, 1403–1410. [Google Scholar] [CrossRef]
- Du, J.; Chen, H.; Qing, L.; Yang, X.; Jia, X. Biomimetic neural scaffolds: A crucial step towards optimal peripheral nerve regeneration. Biomater. Sci. 2018, 6, 1299–1311. [Google Scholar] [CrossRef] [PubMed]
- Ciardelli, G.; Chiono, V. Materials for peripheral nerve regeneration. Macromol. Biosci. 2006, 6, 13–26. [Google Scholar] [CrossRef]
- Gan, L.; Zhao, L.; Zhao, Y.; Li, K.; Tong, Z.; Yi, L.; Wang, X.; Li, Y.; Tian, W.; He, X.; et al. Cellulose/soy protein composite-based nerve guidance conduits with designed microstructure for peripheral nerve regeneration. J. Neural Eng. 2016, 13, 056019. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Yang, W.; Chen, J.; Zhang, J.; Lu, X.; Zhao, X.; Huang, K.; Li, H.; Chang, P.; Wang, Z.; et al. A Silk Sericin/Silicone Nerve Guidance Conduit Promotes Regeneration of a Transected Sciatic Nerve. Adv. Healthc. Mater. 2015, 4, 2195–2205. [Google Scholar] [CrossRef] [PubMed]
- Parker, B.J.; Rhodes, D.I.; O’Brien, C.M.; Rodda, A.E.; Cameron, N.R. Nerve guidance conduit development for primary treatment of peripheral nerve transection injuries: A commercial perspective. Acta Biomater. 2021, 135, 64–86. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Mo, X.; Wang, H. Reduced Graphene Oxide-Encapsulated Microfiber Patterns Enable Controllable Formation of Neuronal-Like Networks. Adv. Mater. 2020, 32, 2004555. [Google Scholar] [CrossRef]
- Pan, X.; Sun, B.; Mo, X. Electrospun polypyrrole-coated polycaprolactone nanoyarn nerve guidance conduits for nerve tissue engineering. Front. Mater. Sci. 2018, 12, 438–446. [Google Scholar] [CrossRef]
- Sun, B.; Zhou, Z.; Li, D.; Wu, T.; Zheng, H.; Liu, J.; Wang, G.; Yu, Y.; Mo, X. Polypyrrole-coated poly(l-lactic acid-co-ε-caprolactone)/silk fibroin nanofibrous nerve guidance conduit induced nerve regeneration in rat. Mater. Sci. Eng. C 2019, 94, 190–199. [Google Scholar] [CrossRef]
- Wang, G.; Wu, W.; Yang, H.; Zhang, P.; Wang, J.Y. Intact polyaniline coating as a conductive guidance is beneficial to repairing sciatic nerve injury. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 128–142. [Google Scholar] [CrossRef]
- Xu, B.; Bai, T.; Sinclair, A.; Wang, W.; Wu, Q.; Gao, F.; Jia, H.; Jiang, S.; Liu, W. Directed neural stem cell differentiation on polyaniline-coated high strength hydrogels. Mater. Today Chem. 2016, 1–2, 15–22. [Google Scholar] [CrossRef]
- Kayser, L.V.; Lipomi, D.J. Stretchable Conductive Polymers and Composites Based on PEDOT and PEDOT:PSS. Adv. Mater. 2019, 31, 1806133. [Google Scholar] [CrossRef]
- Choi, J.-S.; Kim, H.; An, H.-Y.; Shim, B.S.; Lim, J.-Y. Regeneration of Recurrent Laryngeal Nerve using Polycaprolactone (PCL) Nerve Guide Conduit Coated with Conductive Materials. J. Korean Thyroid Assoc. 2015, 8, 88. [Google Scholar] [CrossRef]
- Lee, S.J.; Zhu, W.; Nowicki, M.; Lee, G.; Heo, D.N.; Kim, J.; Zuo, Y.Y.; Zhang, L.G. 3D printing nano conductive multi-walled carbon nanotube scaffolds for nerve regeneration. J. Neural Eng. 2018, 15, 016018. [Google Scholar] [CrossRef]
- Arslantunali, D.; Budak, G.; Hasirci, V. Multiwalled CNT-pHEMA composite conduit for peripheral nerve repair. J. Biomed. Mater. Res. Part A 2014, 102, 828–841. [Google Scholar] [CrossRef] [PubMed]
- Hibbitts, A.J.; Kočí, Z.; Kneafsey, S.; Matsiko, A.; Žilić, L.; Dervan, A.; Hinton, P.; Chen, G.; Cavanagh, B.; Dowling, J.K.; et al. Multi-factorial nerve guidance conduit engineering improves outcomes in inflammation, angiogenesis and large defect nerve repair. Matrix Biol. 2022, 106, 34–57. [Google Scholar] [CrossRef]
- Itai, S.; Suzuki, K.; Kurashina, Y.; Kimura, H.; Amemiya, T.; Sato, K.; Nakamura, M.; Onoe, H. Cell-encapsulating chitosan-collagen hybrid hydrogel conduit for peripheral nerve regeneration. In Proceedings of the 23rd International Conference on Miniaturized Systems for Chemistry and Life Sciences, MicroTAS 2019, Basel, Switzerland, 27–31 October 2019; pp. 880–881. [Google Scholar]
- Zhang, Z.; Zhang, C.; Li, Z.; Zhang, S.; Liu, J.; Bai, Y.; Pan, J.; Zhang, C. Collagen/β-TCP nerve guidance conduits promote facial nerve regeneration in mini-swine and the underlying biological mechanism: A pilot in vivo study. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Pawar, K.; Mueller, R.; Caioni, M.; Prang, P.; Bogdahn, U.; Kunz, W.; Weidner, N. Increasing capillary diameter and the incorporation of gelatin enhance axon outgrowth in alginate-based anisotropic hydrogels. Acta Biomater. 2011, 7, 2826–2834. [Google Scholar] [CrossRef]
- Zhan, X. Effect of matrix stiffness and adhesion ligand density on chondrogenic differentiation of mesenchymal stem cells. J. Biomed. Mater. Res. Part A 2020, 108, 675–683. [Google Scholar] [CrossRef]
- Antman-Passig, M.; Giron, J.; Karni, M.; Motiei, M.; Schori, H.; Shefi, O. Magnetic Assembly of a Multifunctional Guidance Conduit for Peripheral Nerve Repair. Adv. Funct. Mater. 2021, 31, 2010837. [Google Scholar] [CrossRef]
- Razavi, S.; Jahromi, M.; Vatankhah, E.; Seyedebrahimi, R. Differential effects of rat ADSCs encapsulation in fibrin matrix and combination delivery of BDNF and Gold nanoparticles on peripheral nerve regeneration. BMC Neurosci. 2021, 22, 50. [Google Scholar] [CrossRef]
- Liu, S.; Sun, L.; Zhang, H.; Hu, Q.; Wang, Y.; Ramalingam, M. High-resolution combinatorial 3D printing of gelatin-based biomimetic triple-layered conduits for nerve tissue engineering. Int. J. Biol. Macromol. 2021, 166, 1280–1291. [Google Scholar] [CrossRef]
- Ye, W.; Li, H.; Yu, K.; Xie, C.; Wang, P.; Zheng, Y.; Zhang, P.; Xiu, J.; Yang, Y.; Zhang, F.; et al. 3D printing of gelatin methacrylate-based nerve guidance conduits with multiple channels. Mater. Des. 2020, 192, 108757. [Google Scholar] [CrossRef]
- Vardar, E.; Larsson, H.M.; Engelhardt, E.M.; Pinnagoda, K.; Briquez, P.S.; Hubbell, J.A.; Frey, P. IGF-1-containing multi-layered collagen-fibrin hybrid scaffolds for bladder tissue engineering. Acta Biomater. 2016, 41, 75–85. [Google Scholar] [CrossRef]
- Zhou, Q.; Li, B.; Zhao, J.; Pan, W.; Xu, J.; Chen, S. IGF-I induces adipose derived mesenchymal cell chondrogenic differentiation in vitro and enhances chondrogenesis in vivo. Vitr. Cell. Dev. Biol. Anim. 2016, 52, 356–364. [Google Scholar] [CrossRef]
- Yang, J.; Hsu, C.C.; Cao, T.T.; Ye, H.; Chen, J.; Li, Y.Q. A hyaluronic acid granular hydrogel nerve guidance conduit promotes regeneration and functional recovery of injured sciatic nerves in rats. Neural Regen. Res. 2023, 18, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Vasile, C.; Pieptu, D.; Dumitriu, R.P.; Pânzariu, A.; Profire, L. Chitosan/hyaluronic acid polyelectrolyte complex hydrogels in the management of burn wounds. Rev. Med. Chir. Soc. Med. Nat. IASI 2013, 117, 565–571. [Google Scholar] [PubMed]
- Wang, J.; Cheng, Y.; Wang, H.; Wang, Y.; Zhang, K.; Fan, C.; Wang, H.; Mo, X. Biomimetic and hierarchical nerve conduits from multifunctional nanofibers for guided peripheral nerve regeneration. Acta Biomater. 2020, 117, 180–191. [Google Scholar] [CrossRef]
- Yang, Y.; Ding, F.; Wu, J.; Hu, W.; Liu, W.; Liu, J.; Gu, X. Development and evaluation of silk fibroin-based nerve grafts used for peripheral nerve regeneration. Biomaterials 2007, 28, 5526–5535. [Google Scholar] [CrossRef]
- Suhar, R.A.; Marquardt, L.M.; Song, S.; Buabbas, H.; Doulames, V.M.; Johansson, P.K.; Klett, K.C.; Dewi, R.E.; Enejder, A.M.K.; Plant, G.W.; et al. Elastin-like Proteins to Support Peripheral Nerve Regeneration in Guidance Conduits. ACS Biomater. Sci. Eng. 2021, 7, 4209–4220. [Google Scholar] [CrossRef]
- Farokhi, M.; Mottaghitalab, F.; Shokrgozar, M.A.; Kaplan, D.L.; Kim, H.W.; Kundu, S.C. Prospects of peripheral nerve tissue engineering using nerve guide conduits based on silk fibroin protein and other biopolymers. Int. Mater. Rev. 2017, 62, 367–391. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, W.; Jiang, Y.; Guo, C. Gradient degradable nerve guidance conduit with multilayer structure prepared by electrospinning. Mater. Lett. 2020, 276, 128238. [Google Scholar] [CrossRef]
- Niu, Y.; Stadler, F.J.; Fu, M. Biomimetic electrospun tubular PLLA/gelatin nanofiber scaffold promoting regeneration of sciatic nerve transection in SD rat. Mater. Sci. Eng. C 2021, 121, 111858. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, Z.; Wang, H.; Guo, J.; Cai, J.; Chen, X.; Wei, H.; Qi, J.; Wang, Q.; Liu, H.; et al. Conductive Nerve Guidance Conduits Based on Morpho Butterfly Wings for Peripheral Nerve Repair. ACS Nano 2022, 16, 1868–1879. [Google Scholar] [CrossRef]
- Farzan, A.; Borandeh, S.; Seppälä, J. Conductive polyurethane/PEGylated graphene oxide composite for 3D-printed nerve guidance conduits. Eur. Polym. J. 2022, 167, 111068. [Google Scholar] [CrossRef]
- Fabbro, A.; Prato, M.; Ballerini, L. Carbon nanotubes in neuroregeneration and repair. Adv. Drug Deliv. Rev. 2013, 65, 2034–2044. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, C.; Pfattner, R.; Yan, H.; Jin, L.; Chen, S.; Molina-Lopez, F.; Lissel, F.; Liu, J.; Rabiah, N.I.; et al. A highly stretchable, transparent, and conductive polymer. Sci. Adv. 2017, 3, e1602076. [Google Scholar] [CrossRef]
- Nazeer, M.A.; Yilgor, E.; Yilgor, I. Electrospun polycaprolactone/silk fibroin nanofibrous bioactive scaffolds for tissue engineering applications. Polymer 2019, 168, 86–94. [Google Scholar] [CrossRef]
- Zhou, G.; Chen, Y.; Dai, F.; Yu, X. Chitosan-based nerve guidance conduit with microchannels and nanofibers promotes schwann cells migration and neurite growth. Colloids Surf. B Biointerfaces 2023, 221, 112929. [Google Scholar] [CrossRef]
- Itai, S.; Suzuki, K.; Kurashina, Y.; Kimura, H.; Amemiya, T.; Sato, K.; Nakamura, M.; Onoe, H. Cell-encapsulated chitosan-collagen hydrogel hybrid nerve guidance conduit for peripheral nerve regeneration. Biomed. Microdevices 2020, 22, 81. [Google Scholar] [CrossRef]
- Cunha, C.; Panseri, S.; Antonini, S. Emerging nanotechnology approaches in tissue engineering for peripheral nerve regeneration. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 50–59. [Google Scholar] [CrossRef]
- Furlani, F.; Montanari, M.; Sangiorgi, N.; Saracino, E.; Campodoni, E.; Sanson, A.; Benfenati, V.; Tampieri, A.; Panseri, S.; Sandri, M. Electroconductive and injectable hydrogels based on gelatin and PEDOT:PSS for a minimally invasive approach in nervous tissue regeneration. Biomater. Sci. 2022, 10, 2040–2053. [Google Scholar] [CrossRef]
- Phamornnak, C.; Han, B.; Spencer, B.F.; Ashton, M.D.; Blanford, C.F.; Hardy, J.G.; Blaker, J.J.; Cartmell, S.H. Instructive electroactive electrospun silk fibroin-based biomaterials for peripheral nerve tissue engineering. Biomater. Adv. 2022, 141, 213094. [Google Scholar] [CrossRef]
- Srivastava, E.; Qayoom, I.; Singh, A.; Kumar, A. A reduced graphene oxide functionalized electrospun nerve wrap: Amalgamating electrical and biochemical cues to enhance nerve regeneration in median nerve injury model. Carbon N. Y. 2023, 213, 118226. [Google Scholar] [CrossRef]
- Wang, J.; Cheng, Y.; Chen, L.; Zhu, T.; Ye, K.; Jia, C.; Wang, H.; Zhu, M.; Fan, C.; Mo, X. In vitro and in vivo studies of electroactive reduced graphene oxide-modified nanofiber scaffolds for peripheral nerve regeneration. Acta Biomater. 2019, 84, 98–113. [Google Scholar] [CrossRef]
- Zhang, W.; Fang, X.X.; Li, Q.C.; Pi, W.; Han, N. Reduced graphene oxide-embedded nerve conduits loaded with bone marrow mesenchymal stem cell-derived extracellular vesicles promote peripheral nerve regeneration. Neural Regen. Res. 2023, 18, 200–206. [Google Scholar] [CrossRef]
- Pillai, M.M.; Sathishkumar, G.; Houshyar, S.; Senthilkumar, R.; Quigley, A.; Shanthakumari, S.; Padhye, R.; Bhattacharyya, A. Nanocomposite-Coated Silk-Based Artificial Conduits: The Influence of Structures on Regeneration of the Peripheral Nerve. ACS Appl. Bio Mater. 2020, 3, 4454–4464. [Google Scholar] [CrossRef]
- Vallejo, F.A.; Diaz, A.; Errante, E.L.; Smartz, T.; Khan, A.; Silvera, R.; Brooks, A.E.; Lee, Y.S.; Burks, S.S.; Levi, A.D. Systematic review of the therapeutic use of Schwann cells in the repair of peripheral nerve injuries: Advancements from animal studies to clinical trials. Front. Cell. Neurosci. 2022, 16, 929593. [Google Scholar] [CrossRef]
- Liu, S.; Xie, Y.Y.; Di Wang, L.; Tai, C.X.; Chen, D.; Mu, D.; Cui, Y.Y.; Wang, B. A multi-channel collagen scaffold loaded with neural stem cells for the repair of spinal cord injury. Neural Regen. Res. 2021, 16, 2284–2292. [Google Scholar] [CrossRef]
- Ye, J.; Chen, Y.; Yang, H.; Chen, Q.; Huang, Y.; Zhao, J. Human IL12p80 and Neural Stem Cells Enhance Sciatic Nerve Regeneration. Lancent Pschch 2020, 23, 7002. [Google Scholar] [CrossRef]
- Li, Y.; Men, Y.; Wang, B.; Chen, X.; Yu, Z. Co-transplantation of Schwann cells and neural stem cells in the laminin-chitosan-PLGA nerve conduit to repair the injured recurrent laryngeal nerve in SD rats. J. Mater. Sci. Mater. Med. 2020, 31, 99. [Google Scholar] [CrossRef]
- Shalaby, S.M.; El-Shal, A.S.; Ahmed, F.E.; Shaban, S.F.; Wahdan, R.A.; Kandel, W.A.; Senger, M.S. Combined Wharton’s jelly derived mesenchymal stem cells and nerve guidance conduit: A potential promising therapy for peripheral nerve injuries. Int. J. Biochem. Cell Biol. 2017, 86, 67–76. [Google Scholar] [CrossRef]
- Jahromi, M.; Razavi, S.; Seyedebrahimi, R.; Reisi, P.; Kazemi, M. Regeneration of Rat Sciatic Nerve Using PLGA Conduit Containing Rat ADSCs with Controlled Release of BDNF and Gold Nanoparticles. J. Mol. Neurosci. 2021, 71, 746–760. [Google Scholar] [CrossRef]
- Wu, P.; Tong, Z.; Luo, L.; Zhao, Y.; Chen, F.; Li, Y.; Huselstein, C.; Ye, Q.; Ye, Q.; Chen, Y. Comprehensive strategy of conduit guidance combined with VEGF producing Schwann cells accelerates peripheral nerve repair. Bioact. Mater. 2021, 6, 3515–3527. [Google Scholar] [CrossRef]
- Huang, Z.; Powell, R.; Kankowski, S.; Phillips, J.B.; Haastert-Talini, K. Culture Conditions for Human Induced Pluripotent Stem Cell-Derived Schwann Cells: A Two-Centre Study. Int. J. Mol. Sci. 2023, 24, 5366. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Wu, J.Y.; Kennedy, K.M.; Yeager, K.; Bernhard, J.C.; Ng, J.J.; Zimmerman, B.K.; Robinson, S.; Durney, K.M.; Shaeffer, C.; et al. Tissue engineered autologous cartilage-bone grafts for temporomandibular joint regeneration. Sci. Transl. Med. 2020, 12, eabb6683. [Google Scholar] [CrossRef]
- Lu, Q.; Zhang, F.; Cheng, W.; Gao, X.; Ding, Z.; Zhang, X.; Lu, Q.; Kaplan, D.L. Nerve Guidance Conduits with Hierarchical Anisotropic Architecture for Peripheral Nerve Regeneration. Adv. Healthc. Mater. 2021, 10, 2100427. [Google Scholar] [CrossRef]
- Ramburun, P.; Kumar, P.; Choonara, Y.E.; Bijukumar, D.; du Toit, L.C.; Pilay, V. A Review of Bioactive Release from Nerve Conduits as a Neurotherapeutic Strategy for Neuronal Growth in Peripheral Nerve Injury. BioMed Res. Int. 2014, 2014, 132350. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.C.; Chen, M.H.; Liao, S.Y.; Wu, H.C.; Kuan, C.H.; Sun, J.S.; Wang, T.W. Multichanneled Nerve Guidance Conduit with Spatial Gradients of Neurotrophic Factors and Oriented Nanotopography for Repairing the Peripheral Nervous System. ACS Appl. Mater. Interfaces 2017, 9, 37623–37636. [Google Scholar] [CrossRef]
- Chen, Y.S.; Ng, H.Y.; Chen, Y.W.; Cho, D.Y.; Ho, C.C.; Chen, C.Y.; Chiu, S.C.; Jhong, Y.R.; Shie, M.Y. Additive manufacturing of Schwann cell-laden collagen/alginate nerve guidance conduits by freeform reversible embedding regulate neurogenesis via exosomes secretion towards peripheral nerve regeneration. Biomater. Adv. 2023, 146, 213276. [Google Scholar] [CrossRef]
- Hu, T.; Chang, S.; Qi, F.; Zhang, Z.; Chen, J.; Jiang, L.; Wang, D.; Deng, C.; Nie, K.; Xu, G.; et al. Neural grafts containing exosomes derived from Schwann cell-like cells promote peripheral nerve regeneration in rats. Burn. Trauma 2023, 11, tkad013. [Google Scholar] [CrossRef]
- Namini, M.S.; Ebrahimi-Barough, S.; Ai, J.; Jahromi, H.K.; Mikaeiliagah, E.; Azami, M.; Bahrami, N.; Lotfibakhshaiesh, N.; Saremi, J.; Shirian, S. Tissue-Engineered Core-Shell Silk-Fibroin/Poly-l-Lactic Acid Nerve Guidance Conduit Containing Encapsulated Exosomes of Human Endometrial Stem Cells Promotes Peripheral Nerve Regeneration. ACS Biomater. Sci. Eng. 2023, 9, 3496–3511. [Google Scholar] [CrossRef]
- Schmidt, C.E.; Leach, J.B. Neural tissue engineering: Strategies for repair and regeneration. Annu. Rev. Biomed. Eng. 2003, 5, 293–347. [Google Scholar] [CrossRef]
- Letourneau, P.C.; Condic, M.L.; Snow, D.M. Interactions of developing neurons with the extracellular matrix. J. Neurosci. 1994, 14, 915–928. [Google Scholar] [CrossRef]
- Yan, L.; Yao, Z.; Lin, T.; Zhu, Q.; Qi, J.; Gu, L.; Fang, J.; Zhou, X.; Liu, X. The role of precisely matching fascicles in the quick recovery of nerve function in long peripheral nerve defects. Neuroreport 2017, 28, 1008–1015. [Google Scholar] [CrossRef]
- Ni, H.C.; Tseng, T.C.; Chen, J.R.; Hsu, S.H.; Chiu, I.M. Fabrication of bioactive conduits containing the fibroblast growth factor 1 and neural stem cells for peripheral nerve regeneration across a 15 mm critical gap. Biofabrication 2013, 5, 035010. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S.; Zhang, S.; Thaharah, S.; Sriram, G.; Lu, W.F.; Fuh, J.Y.H. Electrohydrodynamic Jet 3D Printed Nerve Guide Conduits (NGCs) for peripheral Nerve Injury Repair. Polymers 2018, 10, 753. [Google Scholar] [CrossRef]
- Carvalho, C.R.; Oliveira, J.M.; Reis, R.L. Modern Trends for Peripheral Nerve Repair and Regeneration: Beyond the Hollow Nerve Guidance Conduit. Front. Bioeng. Biotechnol. 2019, 7, 337. [Google Scholar] [CrossRef]
- Song, S.; Wang, X.; Wang, T.; Yu, Q.; Hou, Z.; Zhu, Z.; Li, R. Additive Manufacturing of Nerve Guidance Conduits for Regeneration of Injured Peripheral Nerves. Front. Bioeng. Biotechnol. 2020, 8, 590596. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, D.; Bushman, J.S.; Murthy, N.S.; Merolli, A.; Kaplan, H.M.; Kohn, J. Fibrin glue as a stabilization strategy in peripheral nerve repair when using porous nerve guidance conduits. J. Mater. Sci. Mater. Med. 2017, 28, 79. [Google Scholar] [CrossRef]
- Sun, B.; Zhou, Z.; Wu, T.; Chen, W.; Li, D.; Zheng, H.; El-Hamshary, H.; Al-Deyab, S.S.; Mo, X.; Yu, Y. Development of Nanofiber Sponges-Containing Nerve Guidance Conduit for Peripheral Nerve Regeneration in Vivo. ACS Appl. Mater. Interfaces 2017, 9, 26684–26696. [Google Scholar] [CrossRef]
- Li, X.; Yang, W.; Xie, H.; Wang, J.; Zhang, L.; Wang, Z.; Wang, L. CNT/Sericin Conductive Nerve Guidance Conduit Promotes Functional Recovery of Transected Peripheral Nerve Injury in a Rat Model. ACS Appl. Mater. Interfaces 2020, 12, 36860–36872. [Google Scholar] [CrossRef]
- Mohan, S.; Hernández, I.C.; Wang, W.; Yin, K.; Sundback, C.A.; Wegst, U.G.K.; Jowett, N. Fluorescent Reporter Mice for Nerve Guidance Conduit Assessment: A High-Throughput in vivo Model. Laryngoscope 2018, 128, E386–E392. [Google Scholar] [CrossRef]
- Jenkins, P.M.; Laughter, M.R.; Lee, D.J.; Lee, Y.M.; Freed, C.R.; Park, D. A nerve guidance conduit with topographical and biochemical cues: Potential application using human neural stem cells. Nanoscale Res. Lett. 2015, 10, 264. [Google Scholar] [CrossRef]
- Singh, A.; Asikainen, S.; Teotia, A.K.; Shiekh, P.A.; Huotilainen, E.; Kumar, A. Biomimetic Photocurable Three-Dimensional Printed Nerve Guidance Channels with Aligned Cryomatrix Lumen for Peripheral Nerve Regeneration. ACS Appl. Mater. Interfaces 2018, 10, 43327–43342. [Google Scholar] [CrossRef]
- Pawar, K.; Welzel, G.; Haynl, C.; Schuster, S.; Scheibel, T. Recombinant Spider Silk and Collagen-Based Nerve Guidance Conduits Support Neuronal Cell Differentiation and Functionality in Vitro. ACS Appl. Bio Mater. 2019, 2, 4872–4880. [Google Scholar] [CrossRef]
- Shah, M.B.; Chang, W.; Zhou, G.; Glavy, J.S.; Cattabiani, T.M.; Yu, X. Novel spiral structured nerve guidance conduits with multichannels and inner longitudinally aligned nano fi bers for peripheral nerve regeneration. J. Biomed. Mater. Res. 2018, 107, 1410–1419. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, S.; Duan, L.; Yao, R.; Yan, Y.; Wang, T.; Wang, J.; Zheng, Z.; Wang, X.; Li, G. Preparation and mechanical optimization of a two-layer silk/magnesium wires braided porous artificial nerve guidance conduit. J. Biomed. Mater. Res. Part A 2022, 110, 1801–1812. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Lee, M.S.; Jeon, J.; Lee, D.H.; Yang, K.; Cho, S.W.; Han, I.; Yang, H.S. Biodegradable Nerve Guidance Conduit with Microporous and Micropatterned Poly(lactic-co-glycolic acid)-Accelerated Sciatic Nerve Regeneration. Macromol. Biosci. 2018, 18, 1800290. [Google Scholar] [CrossRef]
- Huang, C.; Ouyang, Y.; Niu, H.; He, N.; Ke, Q.; Jin, X.; Li, D.; Fang, J.; Liu, W.; Fan, C.; et al. Nerve guidance conduits from aligned nanofibers: Improvement of nerve regeneration through longitudinal nanogrooves on a fiber surface. ACS Appl. Mater. Interfaces 2015, 7, 7189–7196. [Google Scholar] [CrossRef]
- Manoukian, O.S.; Rudraiah, S.; Arul, M.R.; Bartley, J.M.; Baker, J.T.; Yu, X.; Kumbar, S.G. Biopolymer-nanotube nerve guidance conduit drug delivery for peripheral nerve regeneration: In vivo structural and functional assessment. Bioact. Mater. 2021, 6, 2881–2893. [Google Scholar] [CrossRef]
- Mendibil, X.; González-Pérez, F.; Bazan, X.; Díez-Ahedo, R.; Quintana, I.; Rodríguez, F.J.; Basnett, P.; Nigmatullin, R.; Lukasiewicz, B.; Roy, I.; et al. Bioresorbable and Mechanically Optimized Nerve Guidance Conduit Based on a Naturally Derived Medium Chain Length Polyhydroxyalkanoate and Poly(ϵ-Caprolactone) Blend. ACS Biomater. Sci. Eng. 2021, 7, 672–689. [Google Scholar] [CrossRef] [PubMed]
- Stocco, E.; Barbon, S.; Emmi, A.; Tiengo, C.; Macchi, V.; De Caro, R.; Porzionato, A. Bridging Gaps in Peripheral Nerves: From Current Strategies to Future Perspectives in Conduit Design. Int. J. Mol. Sci. 2023, 24, 9170. [Google Scholar] [CrossRef]
- Xu, H.; Yu, Y.; Zhang, L.; Zheng, F.; Yin, Y.; Gao, Y.; Li, K.; Xu, J.; Wen, J.; Chen, H.; et al. Sustainable release of nerve growth factor for peripheral nerve regeneration using nerve conduits laden with Bioconjugated hyaluronic acid-chitosan hydrogel. Compos. Part B Eng. 2022, 230, 109509. [Google Scholar] [CrossRef]
- Tagandurdyyeva, N.A.; Trube, M.A.; Shemyakin, I.O.; Solomitskiy, D.N.; Medvedev, G.V.; Dresvyanina, E.N.; Nashchekina, Y.A.; Ivan’kova, E.M.; Dobrovol’skaya, I.P.; Kamalov, A.M.; et al. Properties of Resorbable Conduits Based on Poly(L-Lactide) Nanofibers and Chitosan Fibers for Peripheral Nerve Regeneration. Polymers 2023, 15, 3323. [Google Scholar] [CrossRef]
- Kokai, L.E.; Lin, Y.C.; Oyster, N.M.; Marra, K.G. Diffusion of soluble factors through degradable polymer nerve guides: Controlling manufacturing parameters. Acta Biomater. 2009, 5, 2540–2550. [Google Scholar] [CrossRef]
- Rao, F.; Wang, Y.; Zhang, D.; Lu, C.; Cao, Z.; Sui, J.; Wu, M.; Zhang, Y.; Pi, W.; Wang, B.; et al. Aligned chitosan nanofiber hydrogel grafted with peptides mimicking bioactive brain-derived neurotrophic factor and vascular endothelial growth factor repair long-distance sciatic nerve defects in rats. Theranostics 2020, 10, 1590–1603. [Google Scholar] [CrossRef]
- Amagat, J.; Su, Y.; Svejsø, F.H.; Le Friec, A.; Sønderskov, S.M.; Dong, M.; Fang, Y.; Chen, M. Self-snapping hydrogel-based electroactive microchannels as nerve guidance conduits. Mater. Today Bio 2022, 16, 100437. [Google Scholar] [CrossRef] [PubMed]
- Aigner, T.B.; Haynl, C.; Salehi, S.; O’Connor, A.; Scheibel, T. Nerve guidance conduit design based on self-rolling tubes. Mater. Today Biol. 2020, 5, 100042. [Google Scholar] [CrossRef]
- Teuschl, A.H.; Schuh, C.; Halbweis, R.; Pajer, K.; Márton, G.; Hopf, R.; Mosia, S.; Rünzler, D.; Redl, H.; Nógrádi, A.; et al. A New Preparation Method for Anisotropic Silk Fibroin Nerve Guidance Conduits and Its Evaluation in Vitro and in a Rat Sciatic Nerve Defect Model. Tissue Eng. Part C Methods 2015, 21, 945–957. [Google Scholar] [CrossRef]
- Lee, Y.B.; Polio, S.; Lee, W.; Dai, G.; Menon, L.; Carroll, R.S.; Yoo, S.S. Bio-printing of collagen and VEGF-releasing fibrin gel scaffolds for neural stem cell culture. Exp. Neurol. 2010, 223, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Fan, C.; Wang, J.; Xiong, H.; Zhu, T.; Liu, Y.; Pan, H.; Weijia Lu, W. Bioinspired multichannel nerve guidance conduit based on shape memory nanofibers for potential application in peripheral nerve repair. ACS Nano 2020, 14, 12579–12595. [Google Scholar] [CrossRef]
- Belanger, K.; Schlatter, G.; Hébraud, A.; Marin, F.; Testelin, S.; Dakpé, S.; Devauchelle, B.; Egles, C. A multi-layered nerve guidance conduit design adapted to facilitate surgical implantation. Health Sci. Rep. 2018, 1, e86. [Google Scholar] [CrossRef] [PubMed]
- Grinberg, Y.; Schiefer, M.A.; Tyler, D.J.; Gustafson, K.J. Fascicular perineurium thickness, size, and position affect model predictions of neural excitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2008, 16, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.D. Peripheral nerve fascicles: Anatomy and clinical relevance. Muscle Nerve 2003, 28, 525–541. [Google Scholar] [CrossRef] [PubMed]
- Winter, C.C.; Katiyar, K.S.; Hernandez, N.S.; Song, Y.J.; Struzyna, L.A.; Harris, J.P.; Kacy Cullen, D. Transplantable living scaffolds comprised of micro-tissue engineered aligned astrocyte networks to facilitate central nervous system regeneration. Acta Biomater. 2016, 38, 44–58. [Google Scholar] [CrossRef]
- Pawelec, K.M.; Koffler, J.; Shahriari, D.; Galvan, A.; Tuszynski, M.H.; Sakamoto, J. Microstructure and in vivo characterization of multi-channel nerve guidance scaffolds. Biomed. Mater. 2018, 13, 044104. [Google Scholar] [CrossRef]
- Krych, A.J.; Rooney, G.E.; Chen, B.; Schermerhorn, T.C.; Ameenuddin, S.; Gross, L.A.; Moore, M.J.; Currier, B.L.; Spinner, R.J.; Friedman, J.A.; et al. Relationship between scaffold channel diameter and number of regenerating axons in the transected rat spinal cord. Acta Biomater. 2009, 5, 2551–2559. [Google Scholar] [CrossRef]
- Zhao, R.; Jiang, L.; Du, J.; Xu, B.; Li, A.; Wang, W.; Zhao, S.; Li, X. Fluffy sponge-reinforced electrospun conduits with biomimetic structures for peripheral nerve repair. Compos. Part B Eng. 2022, 230, 109482. [Google Scholar] [CrossRef]
- Wang, H.; Wan, H.; Wang, Q.; Ma, Y.; Su, G.; Cao, X.; Gao, H. Engineered multifunctional silk fibroin/gelatin hydrogel conduit loaded with miR-29a@ZIF-8 nanoparticles for peripheral nerve regeneration. Smart Mater. Med. 2023, 4, 480–492. [Google Scholar] [CrossRef]
- Anastasiou, E.; Lorentz, K.O.; Stein, G.J.; Mitchell, P.D. Prehistoric schistosomiasis parasite found in the Middle East. Lancet Infect. Dis. 2014, 14, 553–554. [Google Scholar] [CrossRef] [PubMed]
- Gul, J.Z.; Sajid, M.; Rehman, M.M.; Uddin, G. 3D printing for soft robotics–a review. Sci. Technol. Adv. Mater. 2018, 19, 243–262. [Google Scholar] [CrossRef]
- Lee, S.; Patel, M.; Patel, R. Electrospun nanofiber nerve guidance conduits for peripheral nerve regeneration: A review. Eur. Polym. J. 2022, 181, 111663. [Google Scholar] [CrossRef]
- Lin, H.H.; Hsieh, F.Y.; Tseng, C.S.; Hsu, S.H. Preparation and characterization of a biodegradable polyurethane hydrogel and the hybrid gel with soy protein for 3D cell-laden bioprinting. J. Mater. Chem. B 2016, 4, 6694–6705. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Wang, C.; Liu, Z.; Ko, J.; Chen, L.; Zhang, T.; Xiong, Z.; Zhang, L.; Sun, W. 3D Printed Conductive Multiscale Nerve Guidance Conduit with Hierarchical Fibers for Peripheral Nerve Regeneration. Adv. Sci. 2023, 10, 2205744. [Google Scholar] [CrossRef] [PubMed]
- Das, S.R.; Lentner, M.T.; Hondred, J.A. Electrical Differentiation of Mesenchymal Stem Cells into Schwann-Cell-Like Phenotypes Using Inkjet-Printed Graphene Circuits Electrical Differentiation of Mesenchymal Stem Cells into Schwann-Cell-Like Phenotypes Using Inkjet-Printed Graphene. Adv. Healthc. Mater. 2017, 6, 1601087. [Google Scholar] [CrossRef] [PubMed]
- Ilkhanizadeh, S.; Teixeira, A.I.; Hermanson, O. Inkjet printing of macromolecules on hydrogels to steer neural stem cell differentiation. Biomaterials 2007, 28, 3936–3943. [Google Scholar] [CrossRef]
- Huang, W.C.; Lin, C.C.; Chiu, T.W.; Chen, S.Y. 3D Gradient and Linearly Aligned Magnetic Microcapsules in Nerve Guidance Conduits with Remotely Spatiotemporally Controlled Release to Enhance Peripheral Nerve Repair. ACS Appl. Mater. Interfaces 2022, 14, 46188–46200. [Google Scholar] [CrossRef]
- Yucel, D.; Kose, G.T.; Hasirci, V. Polyester based nerve guidance conduit design. Biomaterials 2010, 31, 1596–1603. [Google Scholar] [CrossRef]
- Zhang, D.; Yao, Y.; Duan, Y.; Yu, X.; Shi, H.; Nakkala, J.R.; Zuo, X.; Hong, L.; Mao, Z.; Gao, C. Surface-Anchored Graphene Oxide Nanosheets on Cell-Scale Micropatterned Poly(d, l-lactide-co-caprolactone) Conduits Promote Peripheral Nerve Regeneration. ACS Appl. Mater. Interfaces 2020, 12, 7915–7930. [Google Scholar] [CrossRef]
- Taylor, C.S.; Haycock, J.W. Biomaterials and Scaffolds for Repair of the Peripheral Nervous System. In Peripheral Nerve Tissue Engineering and Regeneration; Springer: Cham, Switzerland, 2022; pp. 245–279. [Google Scholar]
- Chiu, Y.C.; Larson, J.C.; Isom, A.; Brey, E.M. Generation of porous poly(Ethylene Glycol) hydrogels by salt leaching. Tissue Eng. Part C Methods 2010, 16, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Suamte, L.; Tirkey, A.; Barman, J.; Jayasekhar Babu, P. Various manufacturing methods and ideal properties of scaffolds for tissue engineering applications. Smart Mater. Manuf. 2023, 1, 100011. [Google Scholar] [CrossRef]
- Takeya, H.; Itai, S.; Kimura, H.; Kurashina, Y.; Amemiya, T.; Nagoshi, N.; Iwamoto, T.; Sato, K.; Shibata, S.; Matsumoto, M.; et al. Schwann cell-encapsulated chitosan-collagen hydrogel nerve conduit promotes peripheral nerve regeneration in rodent sciatic nerve defect models. Sci. Rep. 2023, 13, 11932. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.F.; O’Shea, H.; Kehoe, S.; Boyd, D. Time-dependent evaluation of mechanical properties and in vitro cytocompatibility of experimental composite-based nerve guidance conduits. J. Mech. Behav. Biomed. Mater. 2011, 4, 1266–1274. [Google Scholar] [CrossRef]
- Park, D.Y.; Kim, D.; Park, S.J.; Choi, J.H.; Seo, Y.; Kim, D.H.; Lee, S.H.; Hyun, J.K.; Yoo, J.; Jung, Y.; et al. Micropattern-based nerve guidance conduit with hundreds of microchannels and stem cell recruitment for nerve regeneration. npj Regen. Med. 2022, 7, 62. [Google Scholar] [CrossRef] [PubMed]
- Talebi, A.; Labbaf, S.; Rahmati, S. Biofabrication of a flexible and conductive 3D polymeric scaffold for neural tissue engineering applications; physical, chemical, mechanical, and biological evaluations. Polym. Adv. Technol. 2022, 34, 134–144. [Google Scholar] [CrossRef]
- Koffler, J.; Zhu, W.; Qu, X.; Platoshyn, O.; Dulin, J.N.; Brock, J.; Graham, L.; Lu, P.; Sakamoto, J.; Marsala, M.; et al. Biomimetic 3D-printed scaffolds for spinal cord injury repair. Nat. Med. 2019, 25, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Lackington, W.A.; Raftery, R.M.; O’Brien, F.J. In vitro efficacy of a gene-activated nerve guidance conduit incorporating non-viral PEI-pDNA nanoparticles carrying genes encoding for NGF, GDNF and c-Jun. Acta Biomater. 2018, 75, 115–128. [Google Scholar] [CrossRef]
- Zhu, W.; Tringale, K.R.; Woller, S.A.; You, S.; Johnson, S.; Shen, H.; Schimelman, J.; Whitney, M.; Steinauer, J.; Xu, W.; et al. Rapid continuous 3D printing of customizable peripheral nerve guidance conduits. Mater. Today 2018, 21, 951–959. [Google Scholar] [CrossRef]
- Vijayavenkataraman, S. Nerve guide conduits for peripheral nerve injury repair: A review on design, materials and fabrication methods. Acta Biomater. 2020, 106, 54–69. [Google Scholar] [CrossRef]
- Yang, Z.; Yang, Y.; Xu, Y.; Jiang, W.; Shao, Y.; Xing, J.; Chen, Y.; Han, Y. Biomimetic nerve guidance conduit containing engineered exosomes of adipose-derived stem cells promotes peripheral nerve regeneration. Stem Cell Res. Ther. 2021, 12, 442. [Google Scholar] [CrossRef] [PubMed]
- Barrozo, R.M.; Hansen, L.M.; Lam, A.M.; Skoog, E.C.; Martin, M.E.; Cai, L.P.; Lin, Y.; Latoscha, A.; Suerbaum, S.; Canfield, D.R.; et al. CagY is an Immune-Sensitive Regulator of the Helicobacter pylori Type IV Secretion System. Physiol. Behav. 2017, 176, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, Y.; Lv, M.; Zhao, X.; So, K.F.; Li, H.; EL-Newehy, M.; EL-Hamshary, H.; Morsi, Y.; Mo, X. Regulation of nerve cells using conductive nanofibrous scaffolds for controlled release of Lycium barbarum polysaccharides and nerve growth factor. Regen. Biomater. 2023, 10, rbad038. [Google Scholar] [CrossRef]
- Gordon, T.; Amirjani, N.; Edwards, D.C.; Chan, K.M. Brief post-surgical electrical stimulation accelerates axon regeneration and muscle reinnervation without affecting the functional measures in carpal tunnel syndrome patients. Exp. Neurol. 2010, 223, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Luo, B.; Huang, J.; Lu, L.; Hu, X.; Luo, Z.; Li, M. Electrically induced brain-derived neurotrophic factor release from schwann cells. J. Neurosci. Res. 2014, 92, 893–903. [Google Scholar] [CrossRef]
- Lu, S.; Chen, W.; Wang, J.; Guo, Z.; Xiao, L.; Wei, L.; Yu, J.; Yuan, Y.; Chen, W.; Bian, M.; et al. Polydopamine-Decorated PLCL Conduit to Induce Synergetic Effect of Electrical Stimulation and Topological Morphology for Peripheral Nerve Regeneration. Small Methods 2023, 7, 2200883. [Google Scholar] [CrossRef]
- Lee, H.S.; Jeon, E.Y.; Nam, J.J.; Park, J.H.; Choi, I.C.; Kim, S.H.; Chung, J.J.; Lee, K.; Park, J.W.; Jung, Y. Development of a regenerative porous PLCL nerve guidance conduit with swellable hydrogel-based microgrooved surface pattern via 3D printing. Acta Biomater. 2022, 141, 219–232. [Google Scholar] [CrossRef]
- Hobson, M.I.; Green, C.J.; Terenghi, G. VEGF enhances intraneural angiogenesis and improves nerve regeneration after axotomy. J. Anat. 2000, 197, 591–605. [Google Scholar] [CrossRef]





| Synthetic Biomaterials | |||
| Biomaterials | Advantages | Disadvantages | Ref. |
| PLA |
|
| [14,35,36] |
| PLGA |
|
| [15,37,38] |
| PLCL |
|
| [40] |
| PPy |
|
| [41,42] |
| PANI |
|
| [43,44] |
| PEDOT: PSS |
|
| [41,45] |
| CNTs |
|
| [41,42,46] |
| rGO |
|
| [46] |
| Natural Biomaterials | |||
| Biomaterials | Advantages | Disadvantages | Ref. |
| Collagen |
|
| [45,46,47,48,49,50,51] |
| Gelatin/GelMA |
|
| [44,46,52,53] |
| Fibrin |
|
| [52,54,55] |
| Hyaluronic acid |
|
| [56,57,58,59,60,61] |
| Silk fibroin |
|
| [62,63] |
| Chitosan |
|
| [52,63] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mankavi, F.; Ibrahim, R.; Wang, H. Advances in Biomimetic Nerve Guidance Conduits for Peripheral Nerve Regeneration. Nanomaterials 2023, 13, 2528. https://doi.org/10.3390/nano13182528
Mankavi F, Ibrahim R, Wang H. Advances in Biomimetic Nerve Guidance Conduits for Peripheral Nerve Regeneration. Nanomaterials. 2023; 13(18):2528. https://doi.org/10.3390/nano13182528
Chicago/Turabian StyleMankavi, Faranak, Rana Ibrahim, and Hongjun Wang. 2023. "Advances in Biomimetic Nerve Guidance Conduits for Peripheral Nerve Regeneration" Nanomaterials 13, no. 18: 2528. https://doi.org/10.3390/nano13182528
APA StyleMankavi, F., Ibrahim, R., & Wang, H. (2023). Advances in Biomimetic Nerve Guidance Conduits for Peripheral Nerve Regeneration. Nanomaterials, 13(18), 2528. https://doi.org/10.3390/nano13182528







