Integrating Fly Ash-Controlled Surface Morphology and Candle Grease Coating: Access to Highly Hydrophobic Poly (L-lactic Acid) Composite for Anti-Icing Application
Abstract
:1. Introduction
2. Materials and Experimental Methods
2.1. Materials
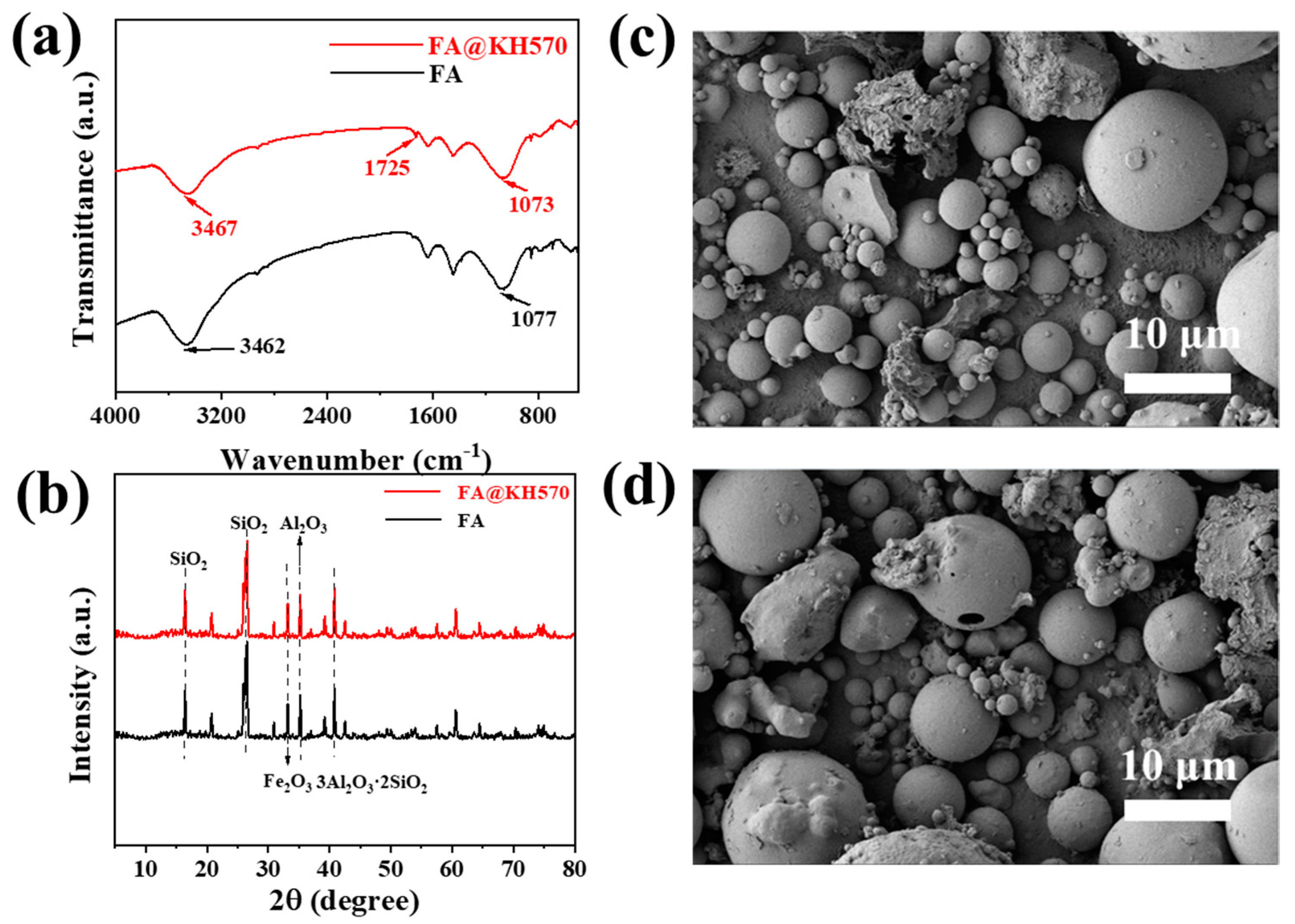
2.2. Modification of FA
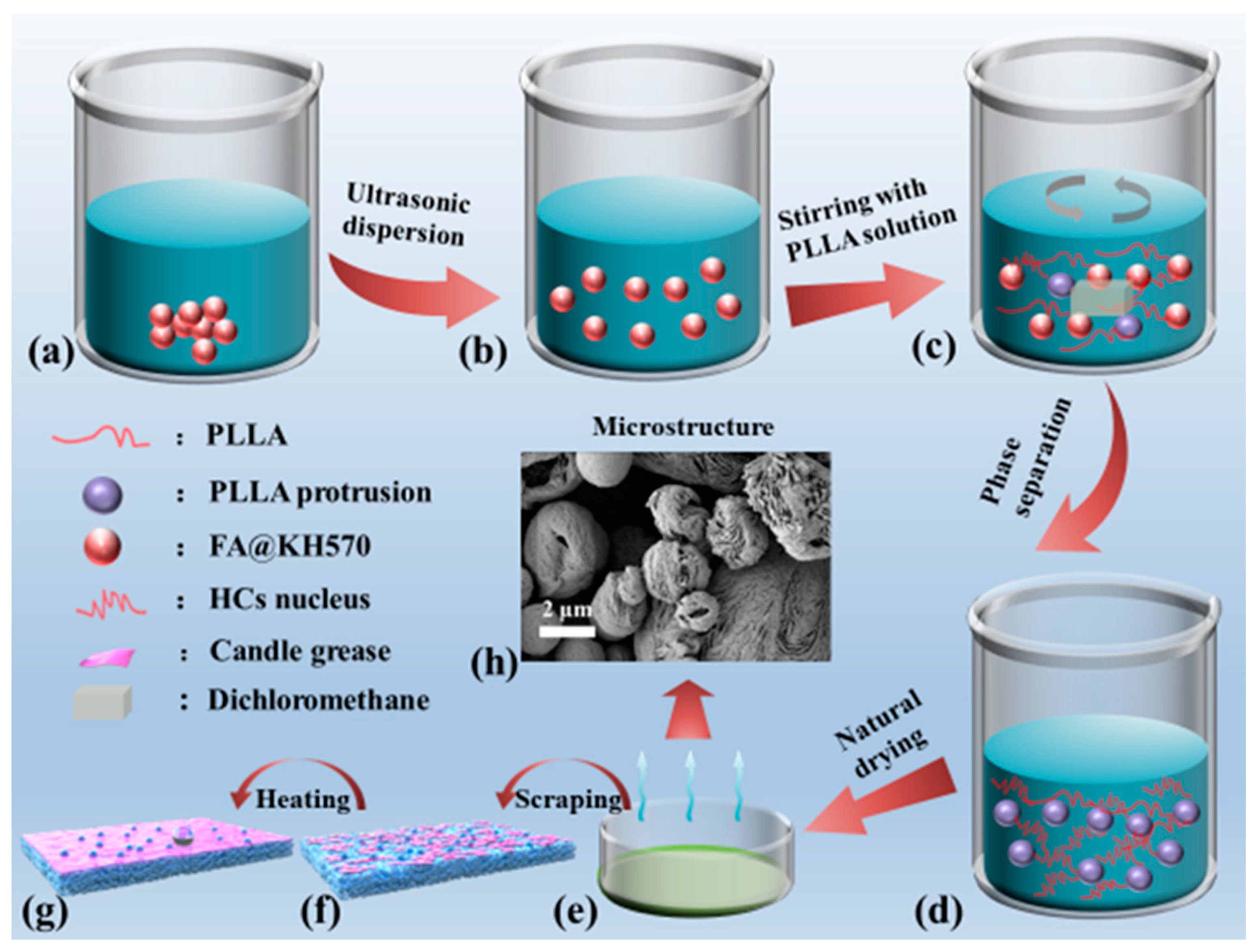
2.3. Preparation of PLLA/FA@KH570 Composites
2.4. Characterizations
3. Results and Discussion
3.1. Dispersibility and Morphology of FA@KH570
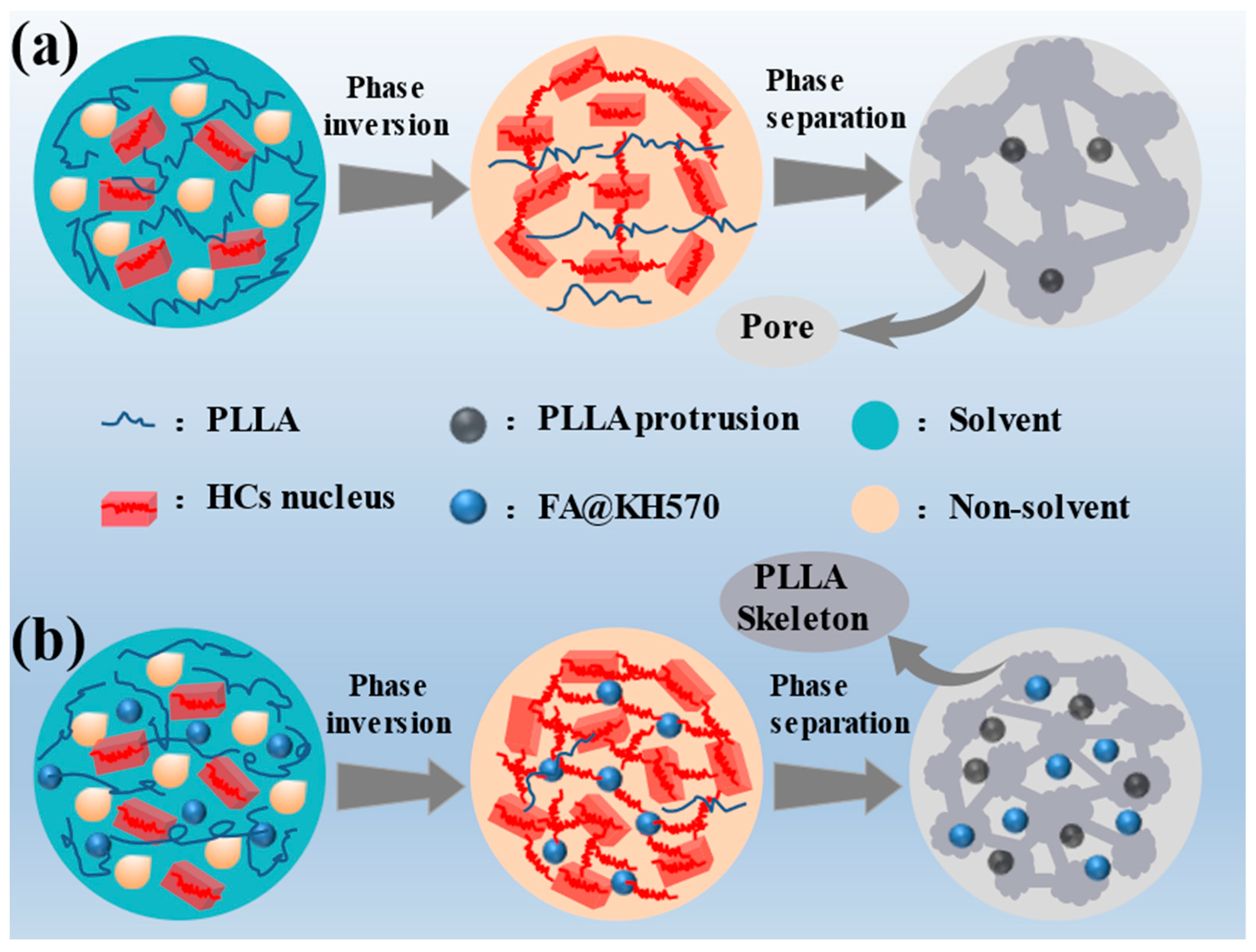
3.2. Morphology of PLLA/FA Composites
3.3. Crystallization Behaviors of PLLA/FA@KH570 Composites
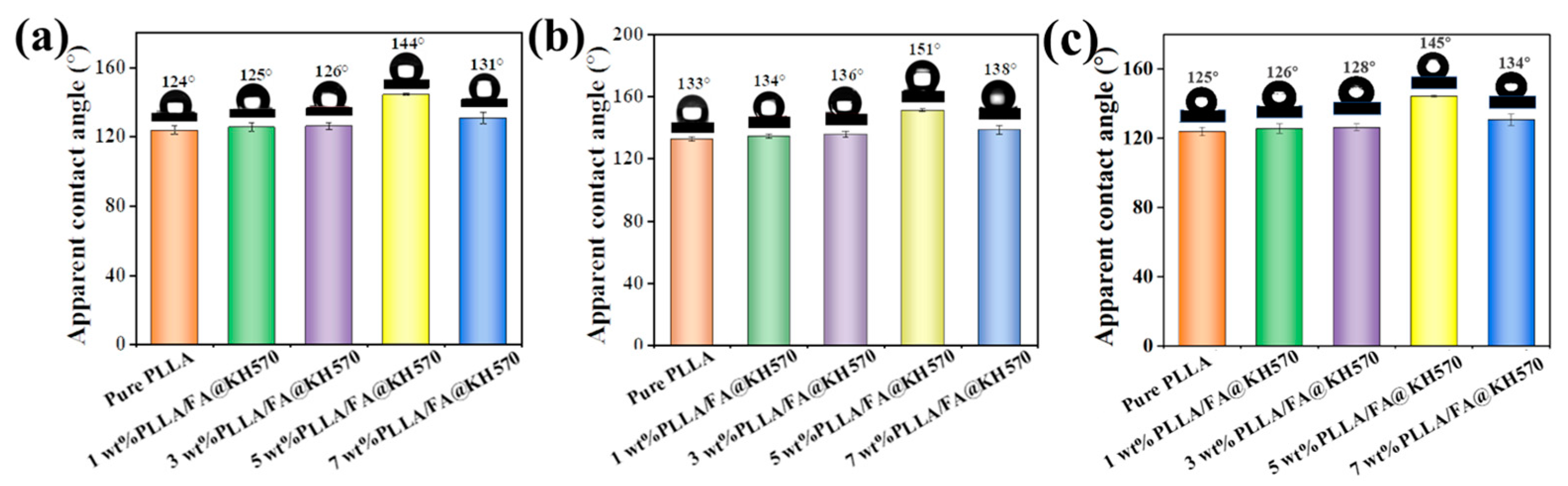
3.4. Surface Wettability of PLLA/FA@KH570 Composites
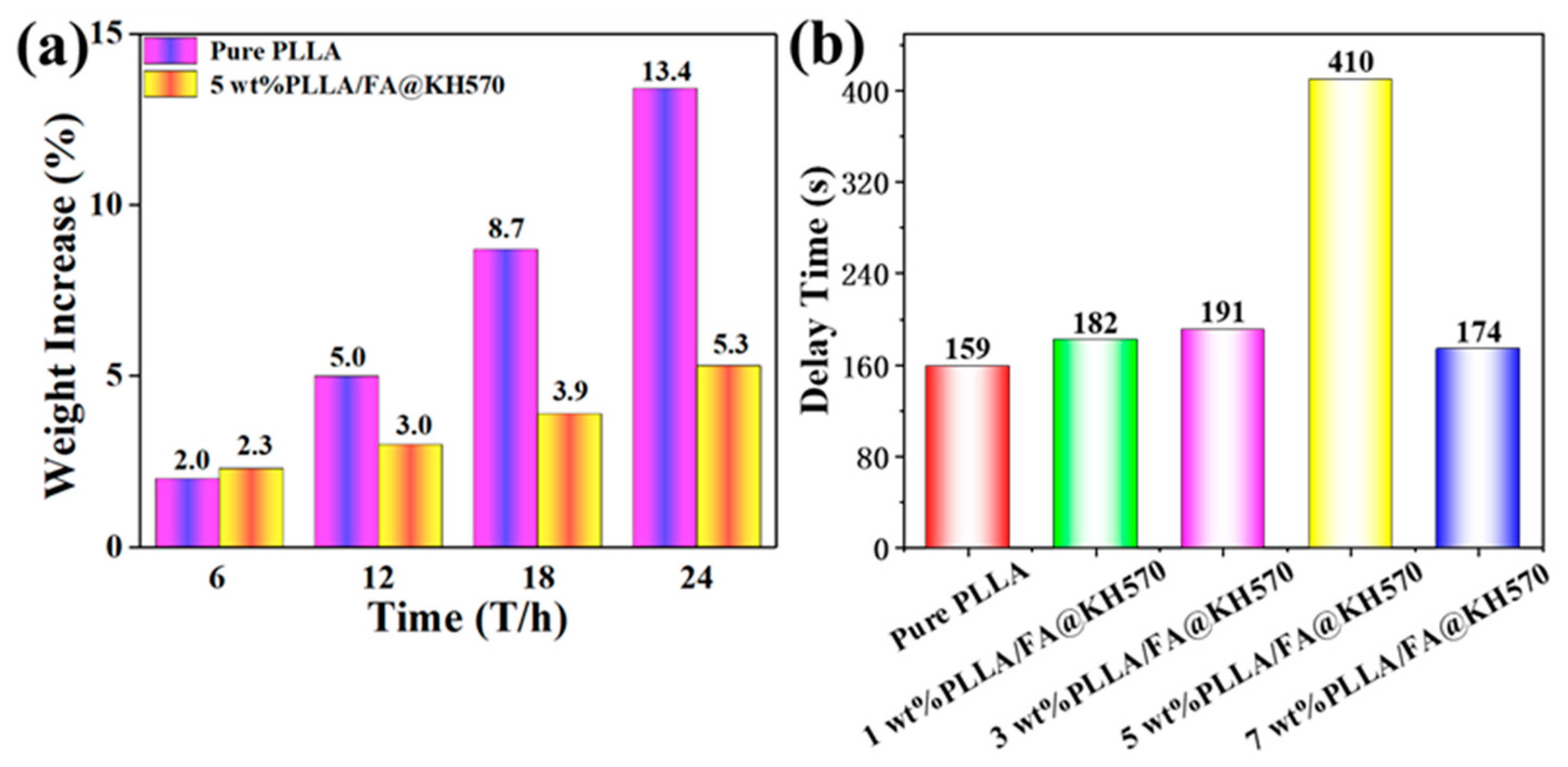
3.5. Anti-Icing Property
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Campardelli, R.; Oleandro, E.; Reverchon, E. Supercritical assisted injection in a liquid antisolvent for PLGA and PLA microparticle production. Powder Technol. 2016, 287, 12–19. [Google Scholar] [CrossRef]
- Xie, Y.; Lan, X.R.; Bao, R.Y. High-performance porous polylactide stereocomplex crystallite scaffolds prepared by solution blending and salt leaching. Mater. Sci. Eng. C 2018, 90, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Gonc, C.; Pinto, A.; Machado, A.V.; Moreira, O. Biocompatible Reinforcement of Poly (Lactic Acid) With Graphene Nanoplatelets. Polym. Compos. 2018, 39, E308–E320. [Google Scholar]
- Zhou, S.Q.; Cheng, X.C.; Jin, Y.L. Molecular dynamics simulation on interacting and mechanical properties of polylactic acid and attapulgite. J. Appl. Polym. Sci. 2013, 128, 3043–3049. [Google Scholar] [CrossRef]
- Wang, X.C.; Song, Y.L.; Wang, Y.M.; Huang, C.P.; Li, Y.X.; Chen, B.H. Preparation of Lactic Acid by Polymer-Catalyzed Conversion of Maltose in Aqueous Alkaline Media. Adv. Mater. Res. 2014, 3381, 947–953. [Google Scholar] [CrossRef]
- Li, H.G.; Guo, X.J.; Ding, Y.R.; Liu, B.Y.; An, Q.F. Superhydrophobic anti-icing coatings with self-deicing property using melanin nanoparticles from cuttlefish juice. Chem. Eng. J. 2021, 424, 130553. [Google Scholar]
- Sun, X.; Yang, S.D.; Xue, B.; Xie, L.; Zheng, Q. Super-hydrophobic poly (lactic acid) by controlling the hierarchical structure and polymorphic transformation. Chem. Eng. J. 2020, 397, 12529. [Google Scholar] [CrossRef]
- Roca, F.G.; Santos, L.G.; Roig, M.M.; Medina, L.M.; Martínez-Ramos, C.; Pradas, M.M. Novel Tissue-Engineered Multimodular Hyaluronic Acid-Polylactic Acid Conduits for the Regeneration of Sciatic Nerve Defect. Biomedicines 2022, 10, 963. [Google Scholar] [CrossRef]
- Butt, M.S.; Bai, J.; Wan, X.F.; Chu, C.L.; Xue, F.; Ding, H.Y.; Zhou, G.H. Mg alloy rod reinforced biodegradable poly-lactic acid composite for load bearing bone replacement. Surf. Coat. Tech. 2017, 309, 471–479. [Google Scholar] [CrossRef]
- Chhajed, M.; Verma, C.; Sathawane, M.; Singh, S.; Maji, P.K. Mechanically durable green aerogel composite based on agricultural lignocellulosic residue for organic liquids/oil sorption. Mar. Pollut. Bull. 2022, 180, 113790. [Google Scholar] [CrossRef] [PubMed]
- Peng, P.P.; Ke, Q.P. Fabrication of microcavity-array superhydrophobic surfaces using an improved template method. J. Colloid Interf. Sci. 2013, 395, 326–328. [Google Scholar] [CrossRef] [PubMed]
- Aghaee, M.M.; Hassan, K.; Mohammad, T. Investigation of wettability of copper plate with nickel-graphene oxide coating produced by electroplating method. Appl. Phys. A Mater. 2022, 143, 128–143. [Google Scholar]
- Makowskia, T.; Svyntkivska, M.; Piorkowskaa, M.; Kregiel, D. Multifunctional polylactide nonwovens with 3D network of multiwall carbon nanotubes. Appl. Surf. Sci. 2020, 527, 146898. [Google Scholar] [CrossRef]
- Liu, W.L.; Huang, N.L.; Yang, J.J. Characterization and application of porous polylactic acid films prepared by nonsolvent-induced phase separation method. Food. Chem. 2021, 373, 131525. [Google Scholar] [CrossRef] [PubMed]
- Bae, G.Y.; Jang, J.; Jeong, Y.G.; Lyoo, W.S.; Min, B.G. Superhydrophobic PLA fabrics prepared by UV photo-grafting of hydrophobic silica particles possessing vinyl groups. J. Colloid Interface Sci. 2010, 344, 584–587. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.Y.; Wang, S.H.; Zhang, J.h.; Liu, Y. Chemical grafting of the superhydrophobic surface on copper with hierarchical microstructure and its formation mechanism. Appl. Surf. Sci. 2018, 436, 950–956. [Google Scholar] [CrossRef]
- Liu, X.H.; Zhang, X.d.; Chen, Q. A simple superhydrophobic/superhydrophilic Janus-paper with enhanced biocompatibility by PDMS and candle soot coating for actuator. Chem. Eng. J. 2021, 406, 126532. [Google Scholar] [CrossRef]
- Li, E.; Pan, Y.M.; Liu, X.H. Asymmetric Superhydrophobic Textiles for Electromagnetic Interference Shielding, Photothermal Conversion, and Solar Water Evaporation. ACS. Appl. Mater. Inter. 2021, 13, 28996–29007. [Google Scholar] [CrossRef]
- Sun, X.; Guo, Y.F.; Wang, R.; Qin, S.H. Flexure-resistant and additive-free poly (L-lactic acid) hydrophobic membranes fabricated by slow phase separation. Int. J. Biol. Macromol. 2022, 209, 1605–1612. [Google Scholar] [CrossRef]
- Rezabeigi, E.; Adams, P.M.W.; Drew, R.A.L. Isothermal ternary phase diagram of the polylactic acid-dichloromethane-hexane system. Polymer 2014, 55, 3100–3106. [Google Scholar] [CrossRef]
- Liu, Z.J.; Ren, L.N.; Jing, J.; Wang, C.J. Fabrication of robust superhydrophobic organic-inorganic hybrid coating through a novel two-step phase separation method. Prog. Org. Coat. 2021, 157, 106320. [Google Scholar] [CrossRef]
- Wang, X.L.; Pan, Y.M.; Liu, X.H. Facile Fabrication of Superhydrophobic and Eco-Friendly Poly (lactic acid) Foam for Oil−Water Separation via Skin Peeling. ACS. Appl. Mater. Inter. 2019, 11, 14362–14367. [Google Scholar] [CrossRef]
- Zhao, Y.Q.; Yang, Q.; Fu, Y.Y. Facile preparation of patterned petal-like PLA surfaces with tunable water micro-droplet adhesion properties based on stereo-complex co-crystallization from non-solvent induced phase separation processes. J. Mater. Chem. A 2016, 4, 12058. [Google Scholar]
- Su, Y.Z.; Zhao, Y.Q.; Zheng, W.G. Asymmetric Sc-PLA Membrane with Multi-scale Microstructures: Wettability, Antifouling, and Oil−Water Separation. ACS Appl. Mater. Inter. 2020, 12, 55520–55526. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.Y.; Zhao, X.L.; Li, Y.D.; Weng, Y.X.; Zeng, J.B. Robust and nanoparticle-free superhydrophobic cotton fabric fabricated from all biological resources for oil/water separation. Int. J. Biol. Macromol. 2019, 140, 1175–1182. [Google Scholar] [CrossRef]
- Sethi, S.K.; Singh, M.; Manik, G. A multi-scale modeling and simulation study to investigate the effect of roughness of a surface on its self-cleaning performance. Mol. Syst. Des. Eng. 2020, 5, 1277. [Google Scholar] [CrossRef]
- Sun, X.; Xue, B.; Yang, S.D.; Huo, K.W.; Liao, X.Y. Structural conversion of PLLA/ZnO composites facilitated by interfacial crystallization to potential application in oil-water separation. Appl. Surf. Sci. 2020, 517, 14613. [Google Scholar] [CrossRef]
- Zhang, D.; Jin, X.Z.; Huang, T.; Zhang, N. Electrospun Fibrous Membranes with Dual-Scaled Porous Structure: Super Hydrophobicity, Super Lipophilicity, Excellent Water Adhesion, and Anti-Icing for Highly Efficient Oil Adsorption/Separation. ACS Appl. Mater. Inter. 2019, 11, 5073–5083. [Google Scholar] [CrossRef]
- Wu, X.N.; Zhao, B.; Wang, L.; Zhang, Z.H. Superhydrophobic PVDF membrane induced by hydrophobic SiO2 nanoparticles and its use for CO2 absorption. Sep. Purif. Technol. 2018, 190, 108–115. [Google Scholar] [CrossRef]
- Rani, R.; Manish, K. Effect of bottom ash at different ratios on hydraulic transportation of fly ash during mine fill. Powder Technol. 2017, 315, 309–317. [Google Scholar] [CrossRef]
- Shu, J.C.; Lei, T.Y. Metal mobility and toxicity of reclaimed copper smelting fly ash and smelting slag. RSC. Adv. 2021, 11, 6877–6884. [Google Scholar] [CrossRef]
- Sulaiman, A.; Ibrahim, A. Treatment of fly ash from power plants using thermal plasma. Beilstein. J. Nanotech. 2017, 8, 1043–1048. [Google Scholar]
- Venkatanarayanan, H.K.; Rangaraju, P.R. Decoupling the effects of chemical composition and fineness of fly ash in mitigating alkali-silica reaction. Cem. Concr. Compos. 2013, 43, 54–68. [Google Scholar] [CrossRef]
- Ming, X.C.; Zhou, K.; Liu, J. Effect of Fly ash Content on Mechanical Properties of Cement-Fly ash Stabilized crushed stones. Appl. Mech. Mater. 2014, 548, 228–232. [Google Scholar]
- Li, C.Y.; Geng, H.B.; Zhou, S.Y. Experimental Study on Preparation and Performance of Concrete with Large Content of Fly-ash. Front. Mater. 2022, 8, 764820. [Google Scholar] [CrossRef]
- Krishnaraj, L.; Ravichandran, P.T. Characterisation of ultra-fine fly ash as sustainable cementitious material for masonry construction. Ain. Shams. Eng. J. 2021, 12, 259–269. [Google Scholar] [CrossRef]
- Rayzman, V.L.; Shcherban, S.A.; Dworkin, R.S. Technology for Chemical-Metallurgical Coal Ash Utilization. Energy Fuels 1997, 11, 761–773. [Google Scholar] [CrossRef]
- Flores, C.G.; Schneider, H.; Marcilio, N.R.; Ferret, L. Potassic zeolites from Brazilian coal ash for use as a fertilizer in agriculture. Waste Manag. Res. 2017, 70, 263–271. [Google Scholar] [CrossRef]
- Ge, X.L.; Zhai, J.W.; Feng, Y.L. The comprehensive utilization of fly ash. Adv. Mater. Res. 2011, 347, 1362–1365. [Google Scholar] [CrossRef]
- Verma, C.; Verma, R. Leaching Behaviour of Fly Ash: A Review, Nature Environment & Polution. Technology 2019, 18, 2. [Google Scholar]
- Xiao, P.; Liang, Y.; He, J.; Zhang, L.; Chen, T. Hydrophilic/Hydrophobic Interphase Mediated Bubble-like Stretchable Janus Ultrathin Films toward Self-Adaptive and Pneumatic Multifunctional Electronics. ACS Nano 2019, 13, 4368–4378. [Google Scholar] [CrossRef]
- Yuan, Q.X.; Yang, G.; Zhang, Y.S. Supercritical CO2 coupled with mechanical force to enhance carbonation of fly ash and heavy metal solidification. Fuel 2022, 315, 123–154. [Google Scholar] [CrossRef]
- Abdellaoui, Y.; Ibrahimi, B.E.; Oualid, H.A.; Kassab, Z.; Quintal-Franco, C. Iron-zirconium microwave-assisted modification of small-pore zeolite W and its alginate composites for enhanced aqueous removal of As(V) ions: Experimental and theoretical studies. Chem. Eng. J. 2021, 421, 129909. [Google Scholar] [CrossRef]
- Pan, Y.; Liu, G.; Chai, B.B. Control of Endogenous Phosphorus Release at the Sediment–Water Interface by Lanthanum-Modified Fly Ash. Coatings 2022, 12, 719. [Google Scholar] [CrossRef]
- Xu, K.; Deng, T.; Liu, J.T. Study on the phosphate removal from aqueous solution using modified fly ash. Fuel 2010, 89, 3668–3674. [Google Scholar] [CrossRef]
- Rashad, A.M.; Ouda, A.S. An investigation on alkali-activated fly ash pastes modified with quartz powder subjected to elevated temperatures. Constr. Build. Mater. 2016, 122, 417–425. [Google Scholar] [CrossRef]
- Xie, Q.; Lin, Y.; Wu, D.Y. Performance of surfactant modified zeolite/hydrous zirconium oxide as a multi-functional adsorbent. Fuel 2017, 203, 411–418. [Google Scholar] [CrossRef]
- Liu, D.J.; Yang, Y.L.; Zhao, F.Q. Adsorption Effect of Cetyltrimethyl-ammonium Bromide Modified Fly Ash on Methyl Orange Waste Water. Mater. Sci. Eng. 2018, 409, 6–12. [Google Scholar] [CrossRef]
- Seo, K.; Kim, M.; Kim, D.H. Candle-based process for creating a stable superhydrophobic surface. Carbon 2014, 68, 583–596. [Google Scholar] [CrossRef]
- Pang, J.F.; Li, Q.; Wang, B.; Tao, D.J. Preparation and characterization of electroless Ni-Fe-P alloy films on fly ash cenospheres. Powder Technol. 2012, 226, 246–252. [Google Scholar] [CrossRef]
- Munief, W.M.; Heib, F.; Hempel, F.; Lu, X.; Schwartz, M.; Pachauri, V.; Hempelmann, R.; Schmitt, M.; Ingebrandt, S. Silane Deposition via Gas-Phase Evaporation and High-Resolution Surface Characterization of the Ultrathin Siloxane Coatings. Langmuir 2018, 34, 10217–10229. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, M.; Weibel, J.A.; Schaber, J.A.; Garimella, S.V. The Wetting State of Water on a Rose Petal. Adv. Mater. Interfaces 2019, 6, 1900652. [Google Scholar] [CrossRef]
- Ghosh, U.U.; Naira, S.; Das, A.; Mukherjee, R.; DasGupta, S. Replicating and resolving wetting and adhesion characteristics of a Rose petal. Colloid Surface A. 2019, 561, 9–17. [Google Scholar] [CrossRef]
- Sun, X.; Xue, B.; Tian, Y.Z.; Xie, L. 3D Porous Poly (L-lactic Acid) Materials with Controllable Multi-scale Microstructures and Their Potential Application in Oil-Water Separation. Appl. Surf. Sci. 2018, 4332, 32256–32261. [Google Scholar] [CrossRef]
- Li, Y.D.; Fu, Q.Q.; Wang, M.; Zeng, J.B. Morphology, crystallization and rheological behavior in poly (butylene succin-ate)/cellulose nanocrystal nanocomposites fabricated by solution coagulation. Carbohydr. Polym. 2017, 164, 75–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szustakiewicz, K.; Kryszak, B.; Dzienny, P.; Pozniak, B.; Tikhomirov, M.; Hoppe, V.; Ziolkowska, P.; Tylus, W.; Grzymajlo, M.; Gajadhur, A.; et al. Cytotoxicity Study of UV-Laser-Irradiated PLLA Surfaces Subjected to Bio-ceramisation: A New Way towards Implant Surface Modification. Int. J. Mol. Sci. 2021, 22, 8436. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, Z.; Xue, B.; Mai, X.; Wu, C.; Zeng, L.; Xie, L.; Zheng, Q. Integrating Fly Ash-Controlled Surface Morphology and Candle Grease Coating: Access to Highly Hydrophobic Poly (L-lactic Acid) Composite for Anti-Icing Application. Nanomaterials 2023, 13, 1230. https://doi.org/10.3390/nano13071230
Jiang Z, Xue B, Mai X, Wu C, Zeng L, Xie L, Zheng Q. Integrating Fly Ash-Controlled Surface Morphology and Candle Grease Coating: Access to Highly Hydrophobic Poly (L-lactic Acid) Composite for Anti-Icing Application. Nanomaterials. 2023; 13(7):1230. https://doi.org/10.3390/nano13071230
Chicago/Turabian StyleJiang, Zhiqiang, Bai Xue, Xiaoping Mai, Changmei Wu, Lingjun Zeng, Lan Xie, and Qiang Zheng. 2023. "Integrating Fly Ash-Controlled Surface Morphology and Candle Grease Coating: Access to Highly Hydrophobic Poly (L-lactic Acid) Composite for Anti-Icing Application" Nanomaterials 13, no. 7: 1230. https://doi.org/10.3390/nano13071230
APA StyleJiang, Z., Xue, B., Mai, X., Wu, C., Zeng, L., Xie, L., & Zheng, Q. (2023). Integrating Fly Ash-Controlled Surface Morphology and Candle Grease Coating: Access to Highly Hydrophobic Poly (L-lactic Acid) Composite for Anti-Icing Application. Nanomaterials, 13(7), 1230. https://doi.org/10.3390/nano13071230





