Maximizing N-Nitrosamine Rejection via RO Membrane Plugging with Hexylamine and Hexamethylenediamine
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
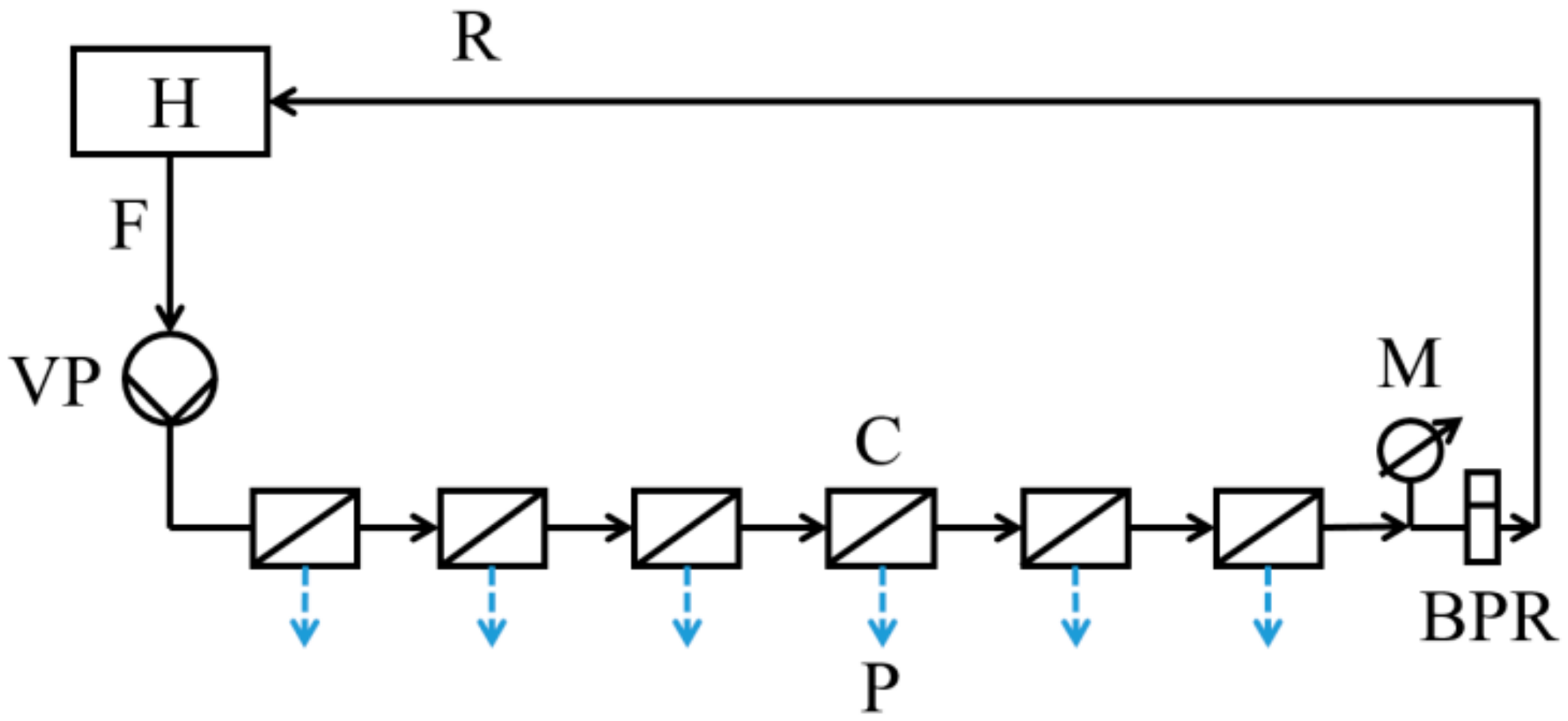
2.2. Membrane Plugging Procedure
2.3. Characterization of TFC-PA Membranes
2.3.1. RO Performance
2.3.2. ATR-IR Spectral Analysis
2.3.3. Contact Angle
2.3.4. Scanning Electron Microscopy
2.3.5. Solid Phase Extraction Procedure (SPE)
2.3.6. High-Performance Liquid Chromatography Analysis
2.3.7. Pore Size Distribution
3. Results
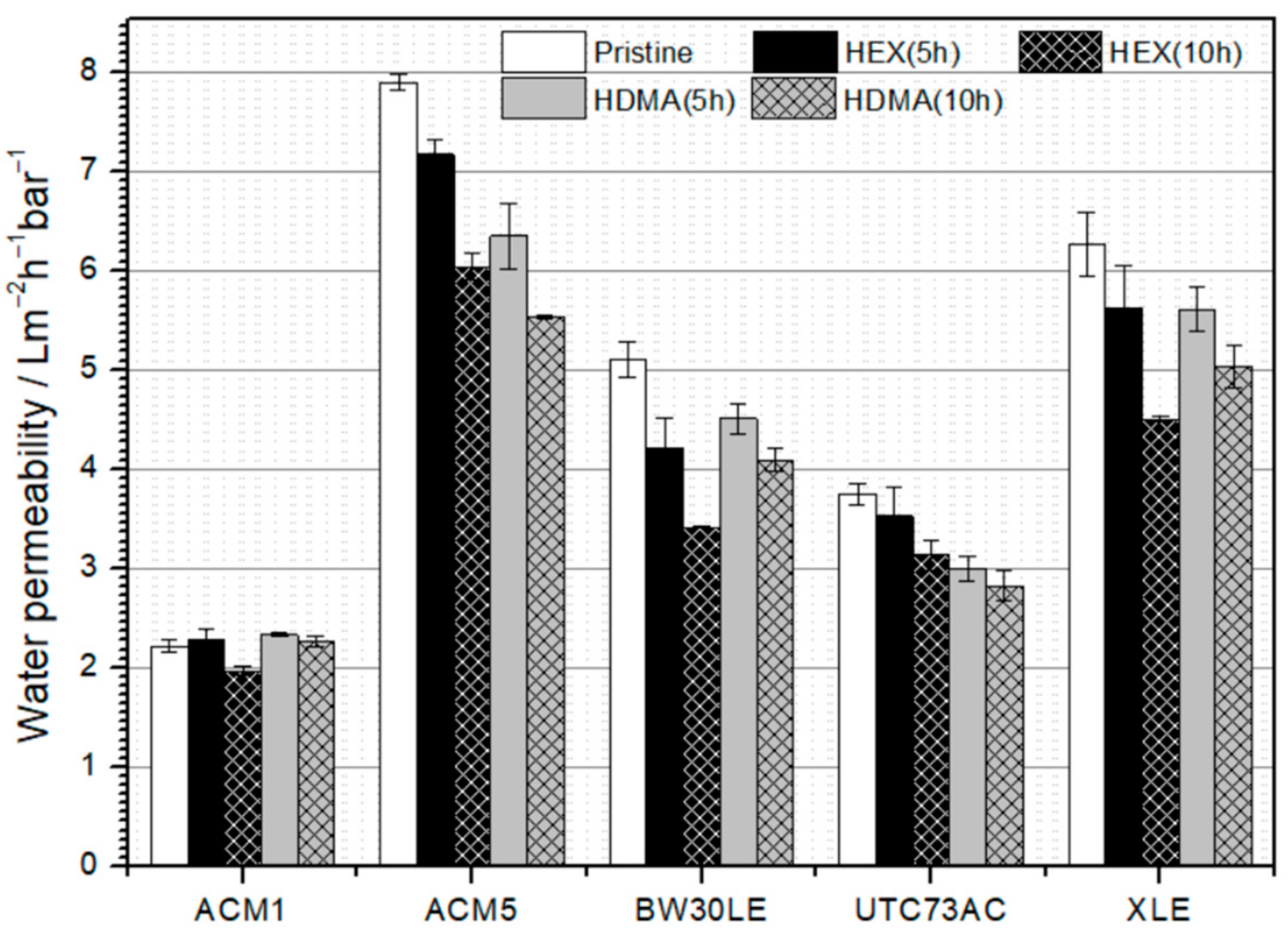
3.1. Permeability
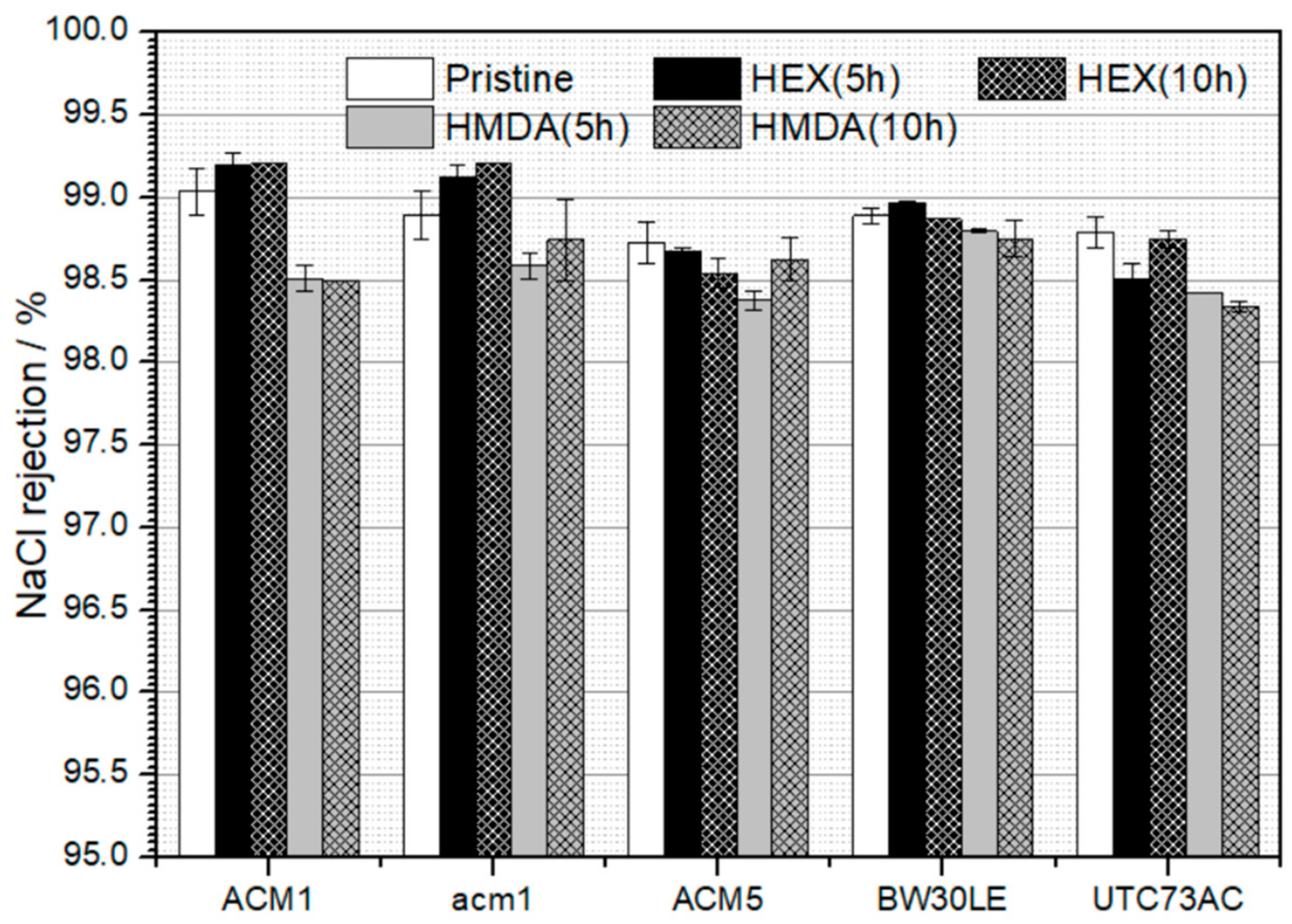
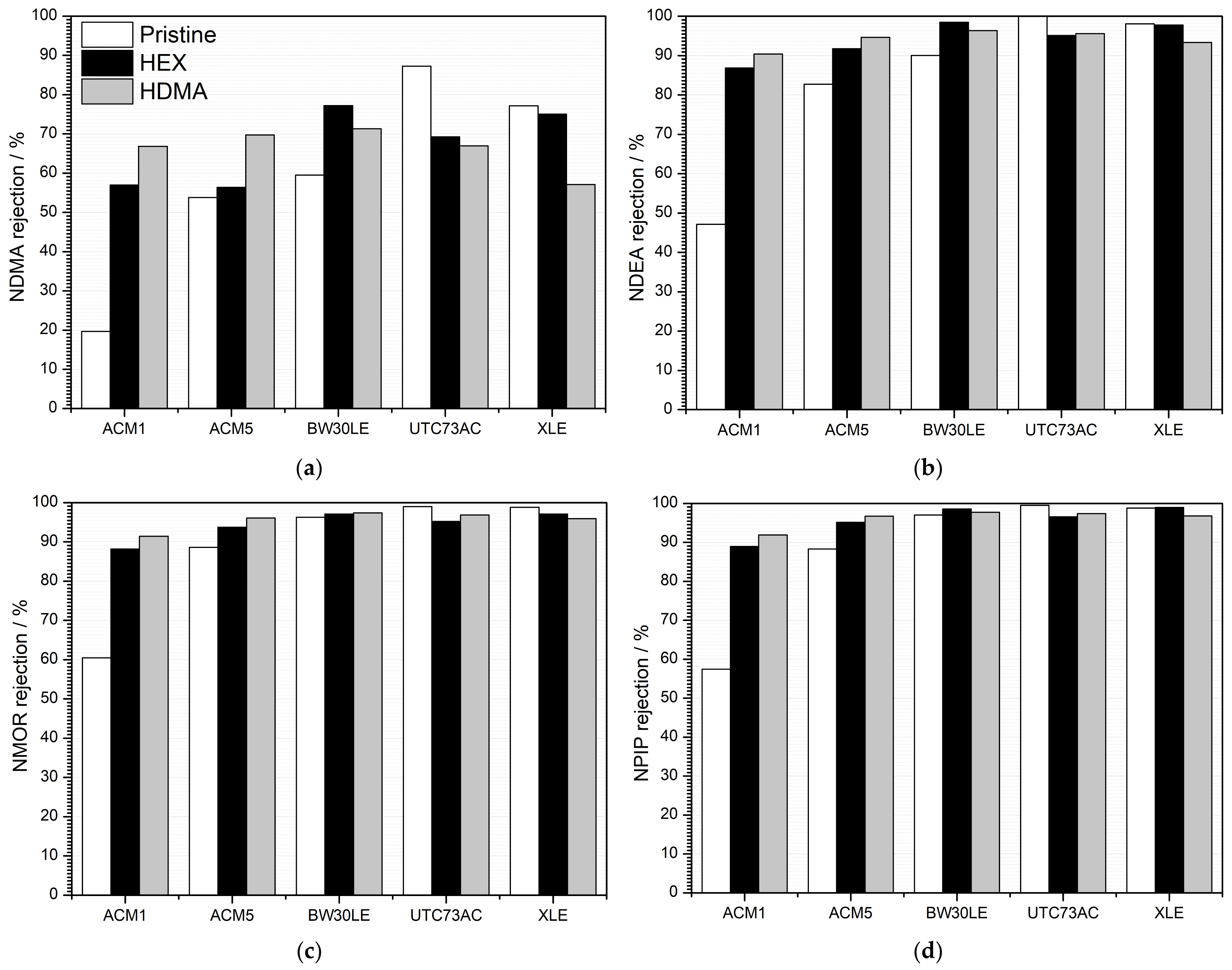
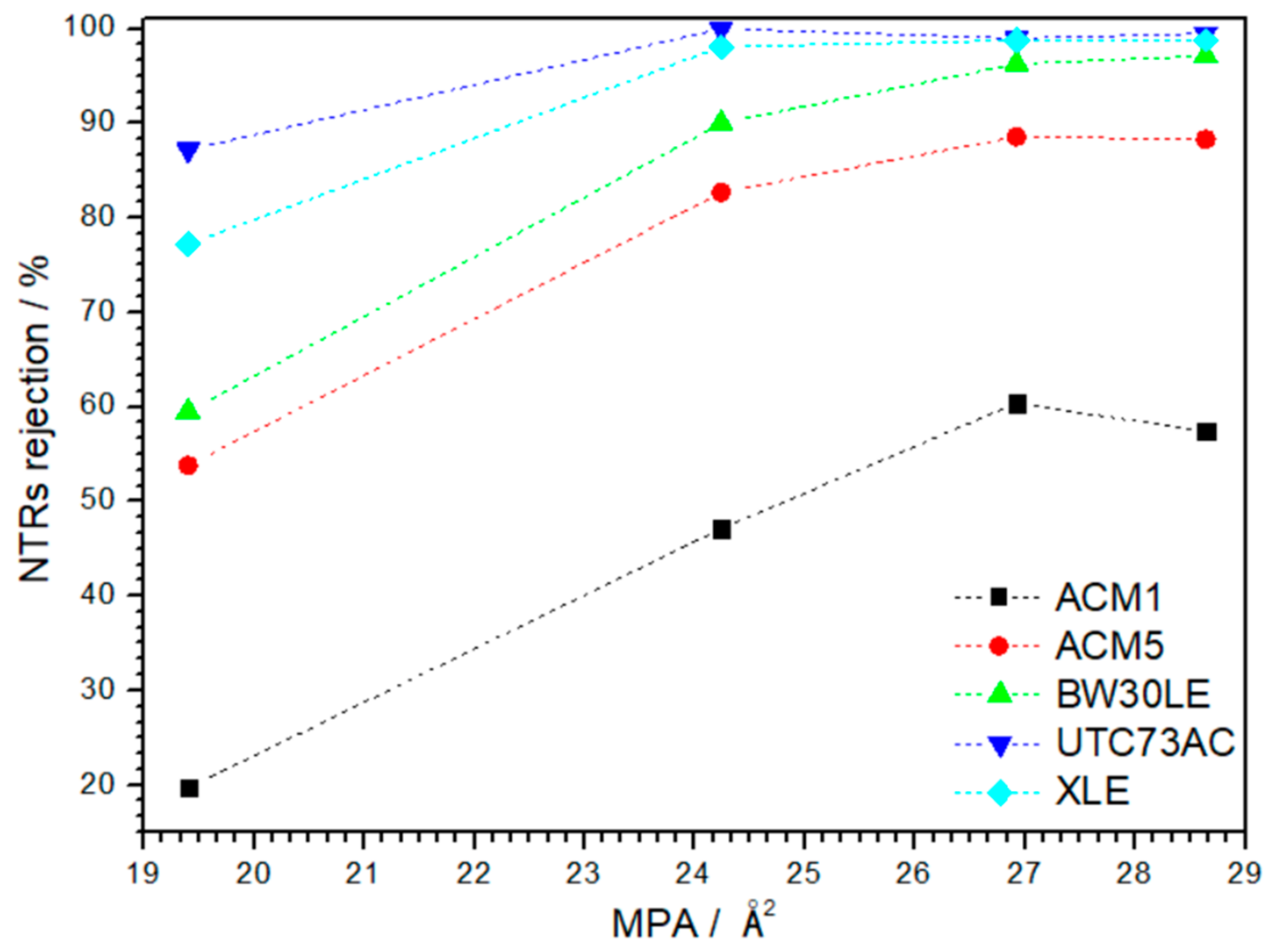
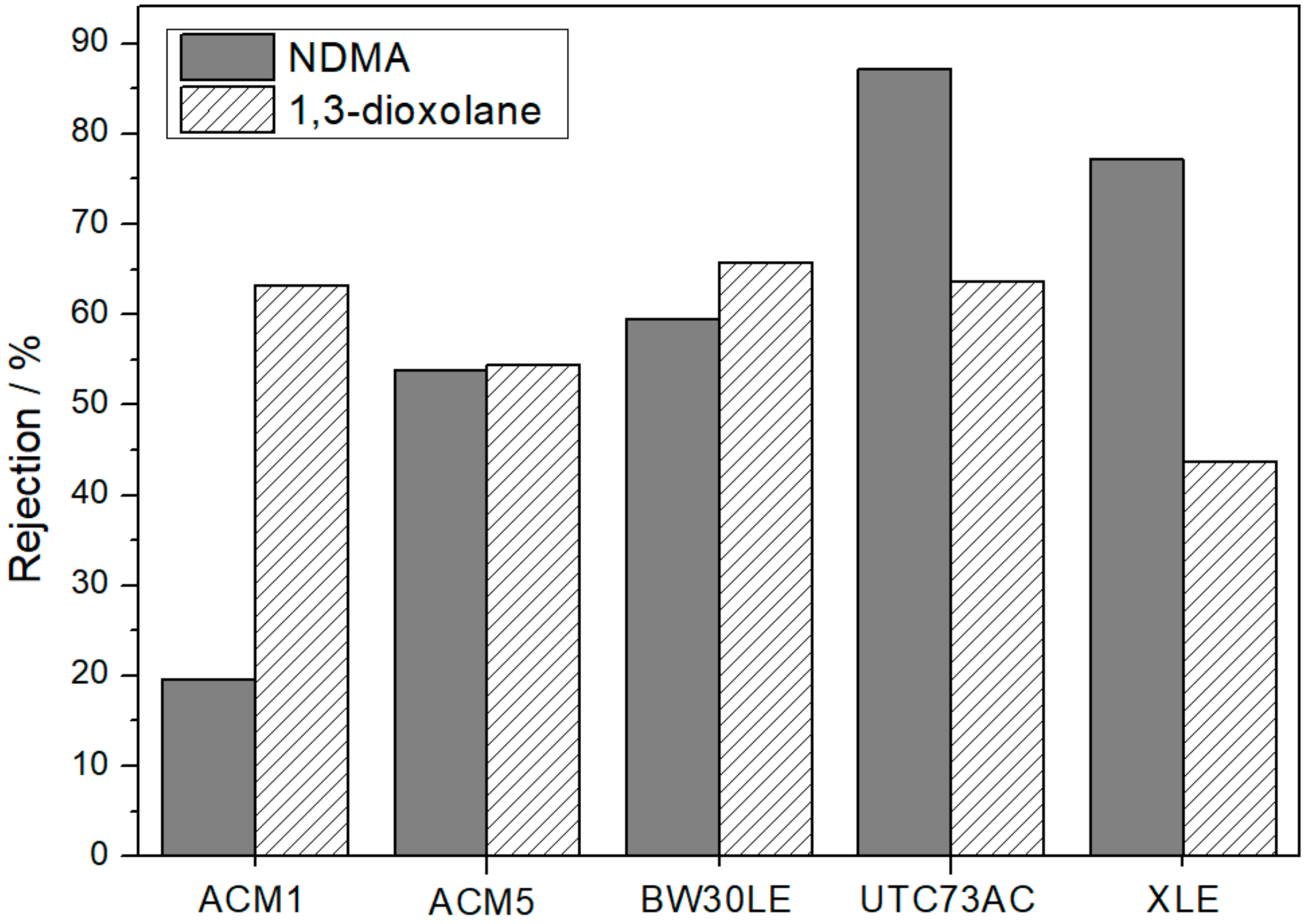
3.2. Rejection of NTRs and NaCl Rejection
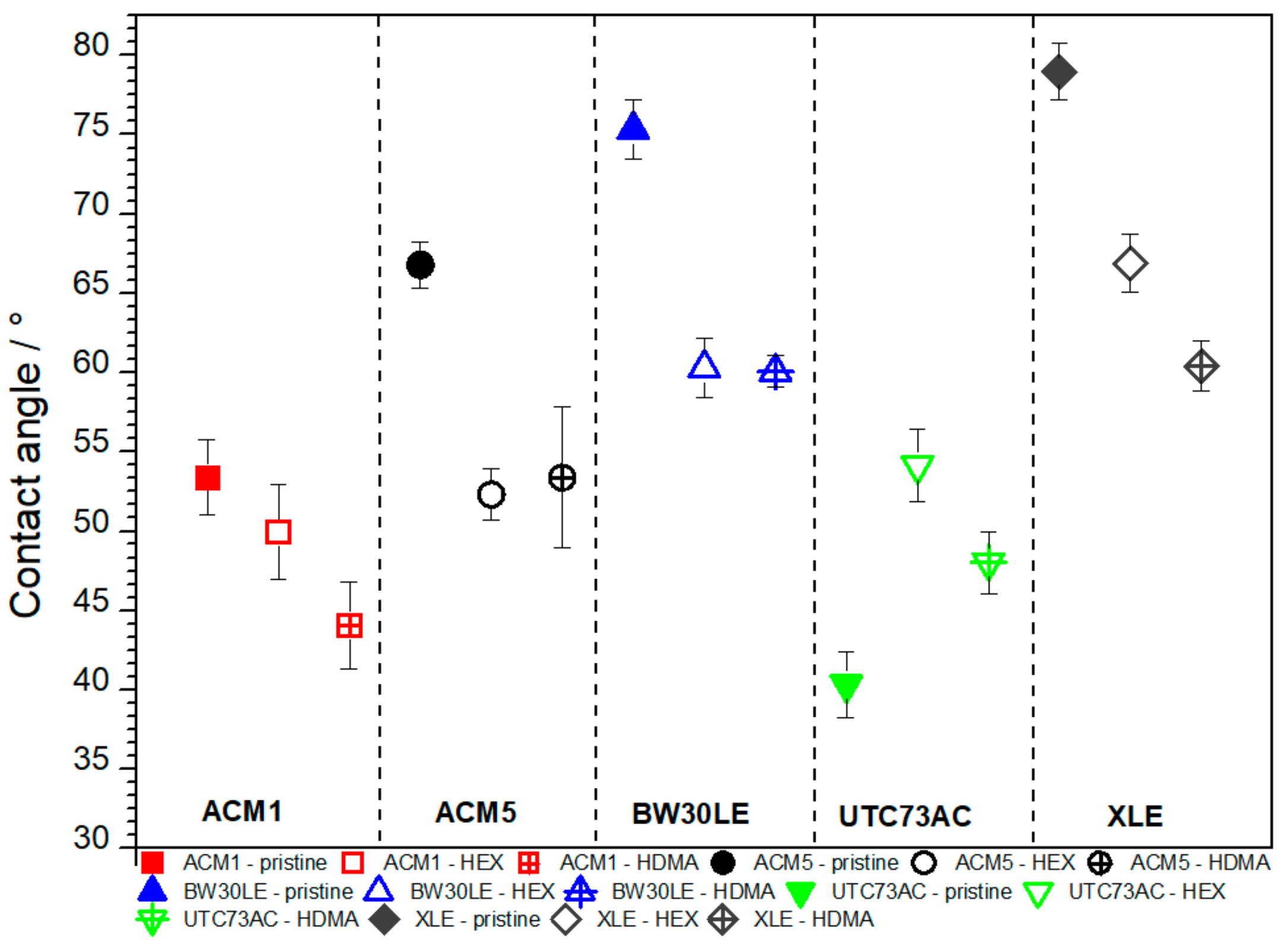
3.3. Contact Angle
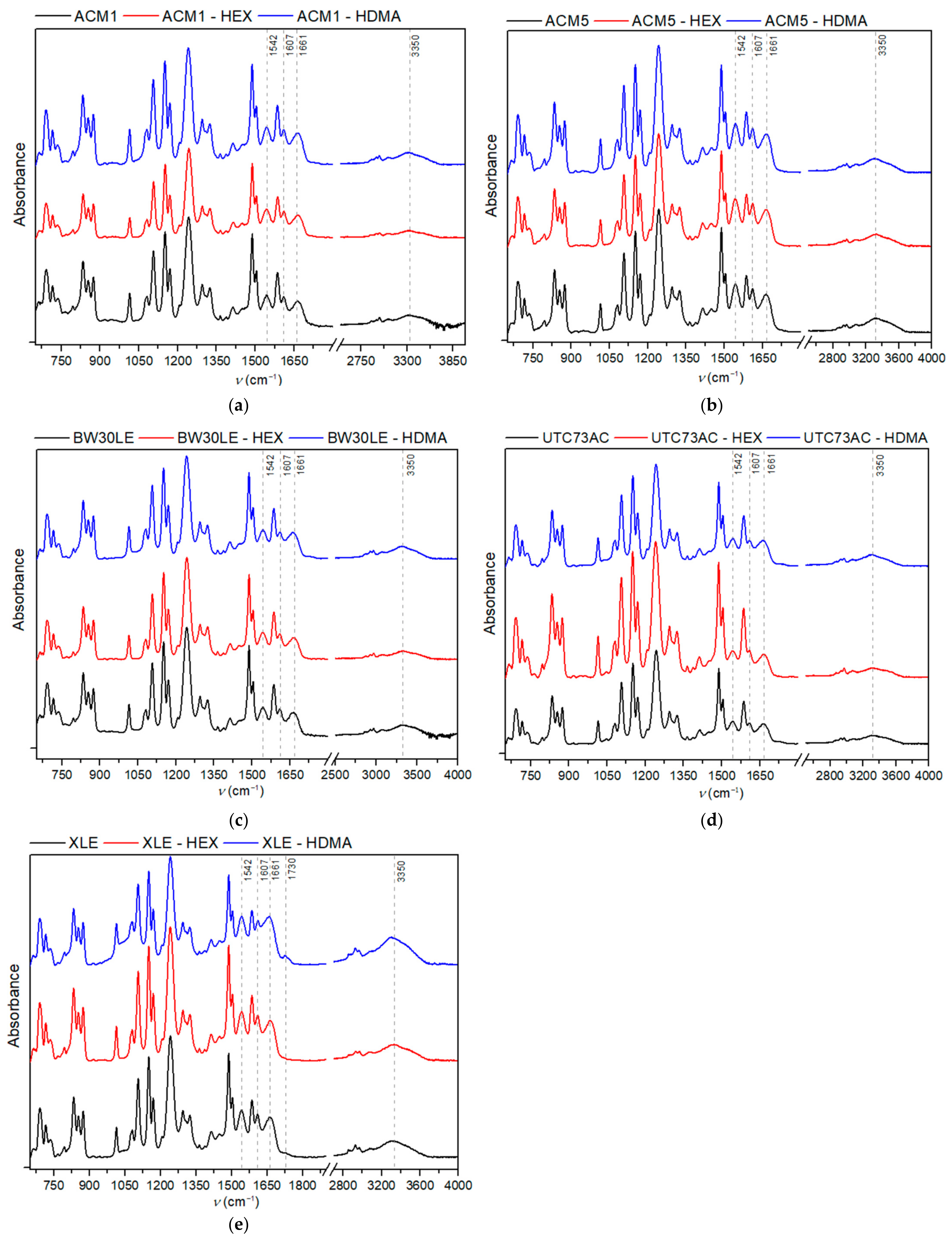
3.4. ATR-IR
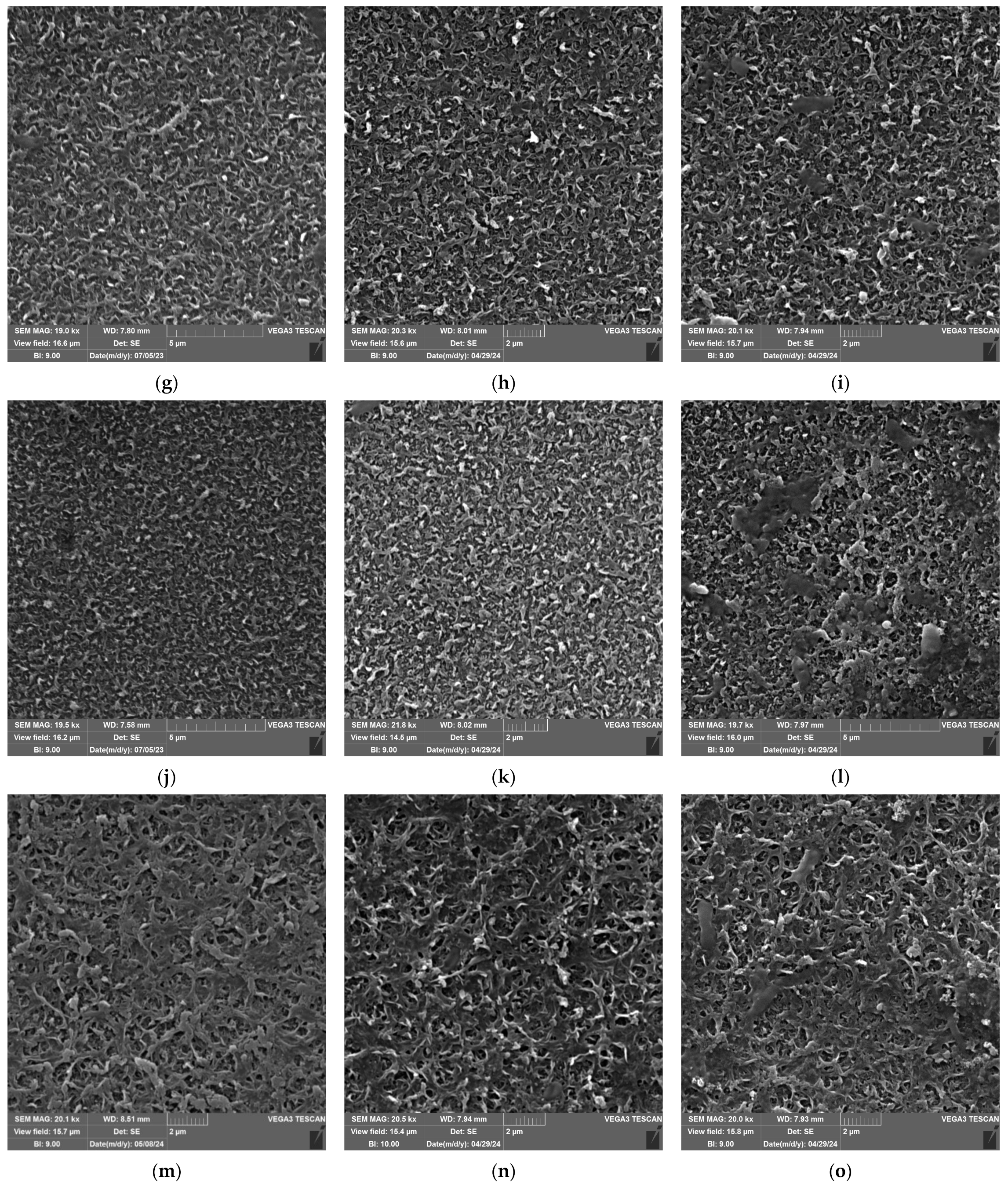
3.5. SEM
3.6. Pore Size Distribution
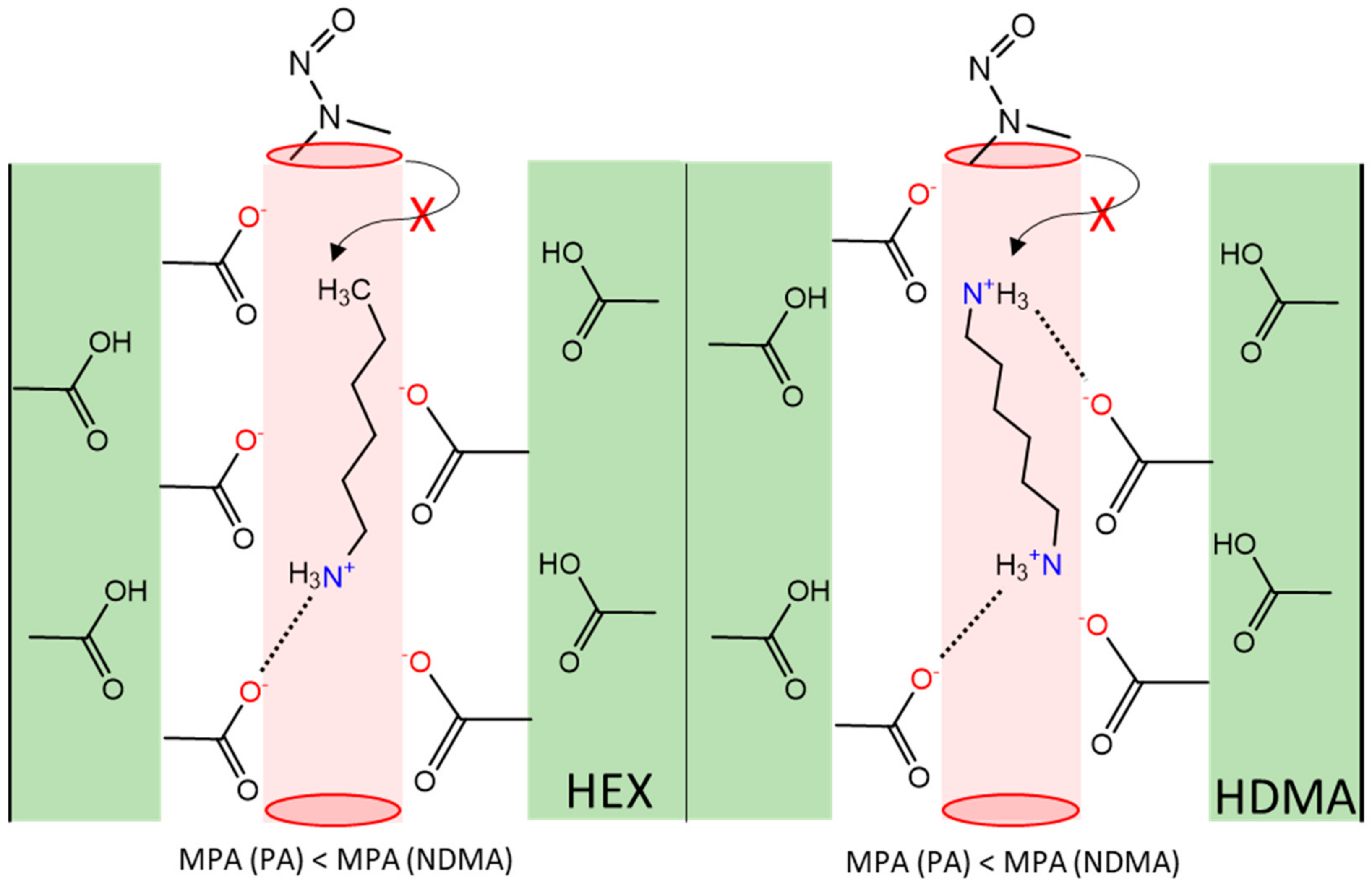
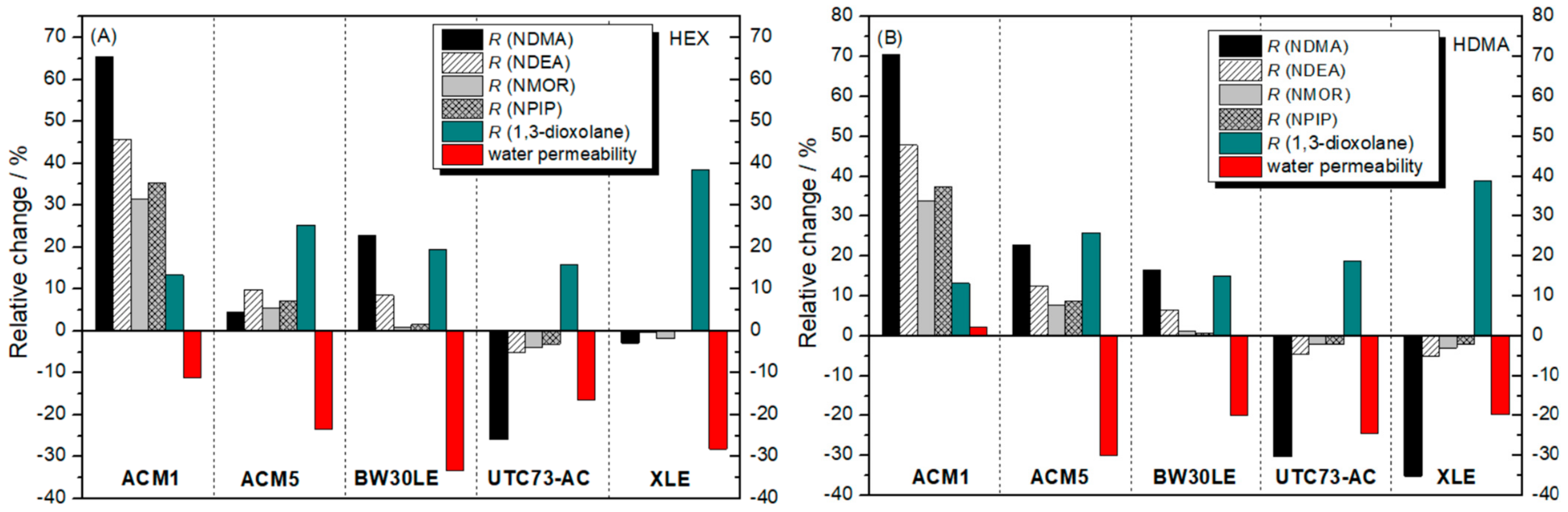
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shemer, H.; Wald, S.; Semiat, R. Challenges and Solutions for Global Water Scarcity. Membranes 2023, 13, 612. [Google Scholar] [CrossRef] [PubMed]
- Water Scarcity. Available online: https://www.unicef.org/wash/water-scarcity (accessed on 16 May 2024).
- Silva, J.A. Wastewater Treatment and Reuse for Sustainable Water Resources Management: A Systematic Literature Review. Sustainability 2023, 15, 10940. [Google Scholar] [CrossRef]
- EEA. Urban Waste Water Treatment Map. Available online: https://www.eea.europa.eu/themes/water/european-waters/water-use-and-environmental-pressures/uwwtd/interactive-maps/urban-waste-water-treatment-maps-3 (accessed on 4 December 2023).
- Eniola, J.O.; Kumar, R.; Barakat, M.A.; Rashid, J. A Review on Conventional and Advanced Hybrid Technologies for Pharmaceutical Wastewater Treatment. J. Clean. Prod. 2022, 356, 131826. [Google Scholar] [CrossRef]
- Phoon, B.L.; Ong, C.C.; Mohamed Saheed, M.S.; Show, P.-L.; Chang, J.-S.; Ling, T.C.; Lam, S.S.; Juan, J.C. Conventional and Emerging Technologies for Removal of Antibiotics from Wastewater. J. Hazard. Mater. 2020, 400, 122961. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, P.; Shukla, P.; Giri, B.S.; Chowdhary, P.; Chandra, R.; Gupta, P.; Pandey, A. Prevalence and Hazardous Impact of Pharmaceutical and Personal Care Products and Antibiotics in Environment: A Review on Emerging Contaminants. Environ. Res. 2021, 194, 110664. [Google Scholar] [CrossRef] [PubMed]
- Farré, M.J.; Insa, S.; Gernjak, W.; Corominas, L.; Čelić, M.; Acuña, V. N-Nitrosamines and Their Precursors in Wastewater Effluents from Selected Industries in Spain. J. Hazard. Mater. 2023, 451, 131159. [Google Scholar] [CrossRef] [PubMed]
- Shen, R.; Andrews, S.A. Demonstration of 20 Pharmaceuticals and Personal Care Products (PPCPs) as Nitrosamine Precursors during Chloramine Disinfection. Water Res. 2011, 45, 944–952. [Google Scholar] [CrossRef]
- Krasner, S.W.; Mitch, W.A.; McCurry, D.L.; Hanigan, D.; Westerhoff, P. Formation, Precursors, Control, and Occurrence of Nitrosamines in Drinking Water: A Review. Water Res. 2013, 47, 4433–4450. [Google Scholar] [CrossRef]
- Sanchís, J.; Gernjak, W.; Munné, A.; Catalán, N.; Petrovic, M.; Farré, M.J. Fate of N-Nitrosodimethylamine and Its Precursors during a Wastewater Reuse Trial in the Llobregat River (Spain). J. Hazard. Mater. 2021, 407, 124346. [Google Scholar] [CrossRef]
- Fujioka, T.; Khan, S.J.; Poussade, Y.; Drewes, J.E.; Nghiem, L.D. N-Nitrosamine Removal by Reverse Osmosis for Indirect Potable Water Reuse—A Critical Review Based on Observations from Laboratory-, Pilot- and Full-Scale Studies. Sep. Purif. Technol. 2012, 98, 503–515. [Google Scholar] [CrossRef]
- Fujioka, T.; Khan, S.J.; McDonald, J.A.; Roux, A.; Poussade, Y.; Drewes, J.E.; Nghiem, L.D. N-Nitrosamine Rejection by Reverse Osmosis Membranes: A Full-Scale Study. Water Res. 2013, 47, 6141–6148. [Google Scholar] [CrossRef]
- Freger, V.; Ramon, G.Z. Polyamide Desalination Membranes: Formation, Structure, and Properties. Prog. Polym. Sci. 2021, 122, 101451. [Google Scholar] [CrossRef]
- Kim, J.-H.; Park, P.-K.; Lee, C.-H.; Kwon, H.-H. Surface Modification of Nanofiltration Membranes to Improve the Removal of Organic Micro-Pollutants (EDCs and PhACs) in Drinking Water Treatment: Graft Polymerization and Cross-Linking Followed by Functional Group Substitution. J. Membr. Sci. 2008, 321, 190–198. [Google Scholar] [CrossRef]
- Park, J.; Yoon, H.W.; Paul, D.R.; Freeman, B.D. Gas Transport Properties of PDMS-Coated Reverse Osmosis Membranes. J. Membr. Sci. 2020, 604, 118009. [Google Scholar] [CrossRef]
- Baransi-Karkaby, K.; Bass, M.; Freger, V. In Situ Modification of Reverse Osmosis Membrane Elements for Enhanced Removal of Multiple Micropollutants. Membranes 2019, 9, 28. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Deng, Y.; Tao, Z.; Yao, Z.; Wang, J.; Lin, C.; Zhang, T.; Zhu, B.; Tang, C.Y. Does Hydrophilic Polydopamine Coating Enhance Membrane Rejection of Hydrophobic Endocrine-Disrupting Compounds? Environ. Sci. Technol. Lett. 2016, 3, 332–338. [Google Scholar] [CrossRef]
- Liu, L.; Xie, X.; Qi, S.; Li, R.; Zhang, X.; Song, X.; Gao, C. Thin Film Nanocomposite Reverse Osmosis Membrane Incorporated with UiO-66 Nanoparticles for Enhanced Boron Removal. J. Membr. Sci. 2019, 580, 101–109. [Google Scholar] [CrossRef]
- Wu, B.; Wang, S.; Wang, J.; Song, X.; Zhou, Y.; Gao, C. Facile Fabrication of High-Performance Thin Film Nanocomposite Desalination Membranes Imbedded with Alkyl Group-Capped Silica Nanoparticles. Polymers 2020, 12, 1415. [Google Scholar] [CrossRef]
- Ng, Z.C.; Chong, C.Y.; Lau, W.J.; Karaman, M.; Ismail, A.F. Boron Removal and Antifouling Properties of Thin-Film Nanocomposite Membrane Incorporating PECVD-Modified Titanate Nanotubes. J. Chem. Technol. Biotechnol. 2019, 94, 2772–2782. [Google Scholar] [CrossRef]
- Fujioka, T.; Osako, M.; Tanabe, S.; Kodamatani, H.; Shintani, T. Plugging Nonporous Polyamide Membranes for Enhanced Rejection of Small Contaminants during Advanced Wastewater Treatment. Sep. Purif. Technol. 2020, 253, 117490. [Google Scholar] [CrossRef]
- Shultz, S.; Bass, M.; Semiat, R.; Freger, V. Modification of Polyamide Membranes by Hydrophobic Molecular Plugs for Improved Boron Rejection. J. Membr. Sci. 2018, 546, 165–172. [Google Scholar] [CrossRef]
- Li, C.; Lai, G.S.; Zhao, Y.; Wang, R. In-Situ Rapid Construction of Aliphatic Amine Combined Polyamide Seawater Reverse Osmosis Membrane for Efficient Boron Removal. J. Membr. Sci. Lett. 2023, 3, 100056. [Google Scholar] [CrossRef]
- Kaštelan-Kunst, L.; Dananić, V.; Kunst, B.; Košutić, K. Preparation and Porosity of Cellulose Triacetate Reverse Osmosis Membranes. J. Membr. Sci. 1996, 109, 223–230. [Google Scholar] [CrossRef]
- Matsuura, T.; Sourirajan, S. Reverse Osmosis Transport through Capillary Pores under the Influence of Surface Forces. Ind. Eng. Chem. Process Des. Dev. 1981, 20, 273–282. [Google Scholar] [CrossRef]
- Dražević, E.; Košutić, K.; Dananić, V.; Pavlović, D.M. Coating Layer Effect on Performance of Thin Film Nanofiltration Membrane in Removal of Organic Solutes. Sep. Purif. Technol. 2013, 118, 530–539. [Google Scholar] [CrossRef]
- Tang, C.Y.; Kwon, Y.-N.; Leckie, J.O. Effect of Membrane Chemistry and Coating Layer on Physiochemical Properties of Thin Film Composite Polyamide RO and NF Membranes: I. FTIR and XPS Characterization of Polyamide and Coating Layer Chemistry. Desalination 2009, 242, 149–167. [Google Scholar] [CrossRef]
- Kim, S.H.; Kwak, S.-Y.; Suzuki, T. Positron Annihilation Spectroscopic Evidence to Demonstrate the Flux-Enhancement Mechanism in Morphology-Controlled Thin-Film-Composite (TFC) Membrane. Environ. Sci. Technol. 2005, 39, 1764–1770. [Google Scholar] [CrossRef]
- Zhao, Y.-Y.; Kong, F.-X.; Wang, Z.; Yang, H.-W.; Wang, X.-M.; Xie, Y.F.; Waite, T.D. Role of Membrane and Compound Properties in Affecting the Rejection of Pharmaceuticals by Different RO/NF Membranes. Front. Environ. Sci. Eng. 2017, 11, 20. [Google Scholar] [CrossRef]
- Bellona, C.; Drewes, J.E.; Xu, P.; Amy, G. Factors Affecting the Rejection of Organic Solutes during NF/RO Treatment—A Literature Review. Water Res. 2004, 38, 2795–2809. [Google Scholar] [CrossRef]
- Nghiem, L.D.; Schäfer, A.I.; Waite, T.D. Adsorptive interactions between membranes and trace contaminants. Desalination 2002, 147, 269–274. [Google Scholar] [CrossRef]
- Otitoju, T.A.; Saari, R.A.; Ahmad, A.L. Progress in the Modification of Reverse Osmosis (RO) Membranes for Enhanced Performance. J. Ind. Eng. Chem. 2018, 67, 52–71. [Google Scholar] [CrossRef]
- Ritt, C.L.; Werber, J.R.; Wang, M.; Yang, Z.; Zhao, Y.; Kulik, H.J.; Elimelech, M. Ionization Behavior of Nanoporous Polyamide Membranes. Proc. Natl. Acad. Sci. USA 2020, 117, 30191–30200. [Google Scholar] [CrossRef] [PubMed]
- Perry, L.A.; Coronell, O. Reliable, Bench-Top Measurements of Charge Density in the Active Layers of Thin-Film Composite and Nanocomposite Membranes Using Quartz Crystal Microbalance Technology. J. Membr. Sci. 2013, 429, 23–33. [Google Scholar] [CrossRef]
- Coronell, O.; Mariñas, B.J.; Cahill, D.G. Depth Heterogeneity of Fully Aromatic Polyamide Active Layers in Reverse Osmosis and Nanofiltration Membranes. Environ. Sci. Technol. 2011, 45, 4513–4520. [Google Scholar] [CrossRef] [PubMed]
- Widjaya, A.; Hoang, T.; Stevens, G.W.; Kentish, S.E. A Comparison of Commercial Reverse Osmosis Membrane Characteristics and Performance under Alginate Fouling Conditions. Sep. Purif. Technol. 2012, 89, 270–281. [Google Scholar] [CrossRef]
- Tang, C.Y.; Kwon, Y.-N.; Leckie, J.O. Probing the Nano- and Micro-Scales of Reverse Osmosis Membranes—A Comprehensive Characterization of Physiochemical Properties of Uncoated and Coated Membranes by XPS, TEM, ATR-FTIR, and Streaming Potential Measurements. J. Membr. Sci. 2007, 287, 146–156. [Google Scholar] [CrossRef]
- Kiso, Y. Factors affecting adsorption of organic solutes on cellulose acetate in an aqueous solution system. Chromatographia 1986, 22, 55–58. [Google Scholar] [CrossRef]
- Zhang, N.; Chen, S.; Yang, B.; Huo, J.; Zhang, X.; Bao, J.; Ruan, X.; He, G. Effect of Hydrogen-Bonding Interaction on the Arrangement and Dynamics of Water Confined in a Polyamide Membrane: A Molecular Dynamics Simulation. J. Phys. Chem. B 2018, 122, 4719–4728. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, T.; Kodamatani, H.; Nghiem, L.D.; Shintani, T. Transport of N-Nitrosamines through a Reverse Osmosis Membrane: Role of Molecular Size and Nitrogen Atoms. Environ. Sci. Technol. Lett. 2019, 6, 44–48. [Google Scholar] [CrossRef]
- Li, Q.; Pan, X.; Qu, Z.; Zhao, X.; Jin, Y.; Dai, H.; Yang, B.; Wang, X. Understanding the Dependence of Contact Angles of Commercially RO Membranes on External Conditions and Surface Features. Desalination 2013, 309, 38–45. [Google Scholar] [CrossRef]



| Compound | Chemical Structure | Molecular Formula | Molecular Weight/g mol−1 | Minimal Projection Area (MPA)/Å2 | pKa | log Kow |
|---|---|---|---|---|---|---|
| NDMA | 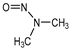 | C2H6N2O | 74.08 | 19.4 | 3.52 | −0.57 |
| NDEA |  | C4H10N2O | 102.14 | 24.2 | 3.32 | 0.34 |
| NMOR |  | C4H8N2O2 | 116.12 | 26.9 | 3.14 | −0.44 |
| NPIP | 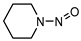 | C5H10N2O | 114.08 | 27.2 | 3.30 | 0.72 |
| 1,3-dioxolane | 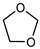 | C3H6O2 | 74.08 | 19.2 | N.I. | −0.37 |
| DOW-FILMTEC™BW30LE | Toray™ UTC-73AC | TriSep™ ACM1 | TriSep™ ACM5 | DOW FILMTEC™ XLE | |
|---|---|---|---|---|---|
| Feed | Brackish Water | Brackish Water | Brackish Water | Brackish Water | Brackish Water |
| Type | Low Energy | High Rejection, Low Energy, Cl Resistant | “Tight” | Low Pressure, High permeability | Extra-Low Energy |
| pH Range (25 °C) | 2–11 | 2–11 | 2–11 | 2–11 | 2–11 |
| Rejection (NaCl)/% | 99.0% | 99.8% | 99.5% | 98.5% | 98.7% |
| Pore size/MWCO | N/A | N/A | N/A | N/A | N/A |
| Polymer | Polyamide-TFC | Polyamide-TFC | Polyamide-TFC | Polyamide-TFC | Polyamide-TFC |
| Water permeability/L m−2 h−1 bar−1 | 4.06–5.05 | 3.37 | 2.74 | 6.7 | 6.5–8.1 |
| Compound | Chemical Structure | Molecular Formula | Molecular Weight/g mol−1 | Min Projection Area (MPA)/Å2 | pKa | Log D at pH 7.4 | Solubility/g L−1 |
|---|---|---|---|---|---|---|---|
| HMDA | 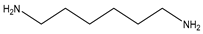 | C6H16N2 | 116.208 | 21.03 | 10.51 | −5.16 | 100 |
| HEX |  | C6H15N | 101.2 | 20.7 | 10.5 | −1.02 | 12 |
| Membranes | MPA/Å2 | Rejection/% | |||||||
|---|---|---|---|---|---|---|---|---|---|
| DIOX | NDMA | NDEA | NMOR | NPIP | |||||
| 19.20 | 19. 40 | 24.24 | 26.92 | 28.64 | |||||
| Pristine | |||||||||
| ACM1 | 27.3 | 63.3 | 19.7 | 47.1 | 60.4 | 57.4 | |||
| ACM5 | 24.3 | 27.7 | 33.0 | 54.5 | 53.8 | 82.7 | 88.6 | 88.3 | |
| BW30LE | 21.2–31.8 | 65.8 | 59.5 | 90.0 | 96.3 | 97.1 | |||
| UTC73AC | 27.9 | 63.7 | 87.2 | 100.0 | 99.0 | 99.5 | |||
| XLE | 24.3 | 34.2 | 43.7 | 77.2 | 98.1 | 98.8 | 98.8 | ||
| HEX | |||||||||
| ACM1 | 28.4 | 73.1 | 57.0 | 86.9 | 88.2 | 89.0 | |||
| ACM5 | 27.9 | 72.9 | 56.4 | 91.8 | 93.7 | 95.2 | |||
| BW30LE | 27.9 | 81.8 | 77.2 | 98.5 | 97.1 | 98.5 | |||
| UTC73AC | 24.3 | 28.1 | 75.6 | 69.3 | 95.1 | 95.2 | 96.5 | ||
| XLE | 28.4 | 70.9 | 75.0 | 97.8 | 97.1 | 99.0 | |||
| HDMA | |||||||||
| ACM1 | 26.6 | 72.9 | 66.8 | 90.4 | 91.4 | 91.9 | |||
| ACM5 | 24.3 | 26.8 | 73.5 | 69.7 | 94.7 | 96.1 | 96.7 | ||
| BW30LE | 27.7 | 77.4 | 71.3 | 96.3 | 97.4 | 97.7 | |||
| UTC73AC | 27.3 | 78.5 | 67.0 | 95.6 | 96.8 | 97.4 | |||
| XLE | 26.6 | 71.5 | 57.1 | 93.4 | 95.9 | 96.8 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morović, S.; Drmić, K.M.; Babić, S.; Košutić, K. Maximizing N-Nitrosamine Rejection via RO Membrane Plugging with Hexylamine and Hexamethylenediamine. Nanomaterials 2024, 14, 1117. https://doi.org/10.3390/nano14131117
Morović S, Drmić KM, Babić S, Košutić K. Maximizing N-Nitrosamine Rejection via RO Membrane Plugging with Hexylamine and Hexamethylenediamine. Nanomaterials. 2024; 14(13):1117. https://doi.org/10.3390/nano14131117
Chicago/Turabian StyleMorović, Silvia, Katarina Marija Drmić, Sandra Babić, and Krešimir Košutić. 2024. "Maximizing N-Nitrosamine Rejection via RO Membrane Plugging with Hexylamine and Hexamethylenediamine" Nanomaterials 14, no. 13: 1117. https://doi.org/10.3390/nano14131117
APA StyleMorović, S., Drmić, K. M., Babić, S., & Košutić, K. (2024). Maximizing N-Nitrosamine Rejection via RO Membrane Plugging with Hexylamine and Hexamethylenediamine. Nanomaterials, 14(13), 1117. https://doi.org/10.3390/nano14131117







