Smartphone-Based Platforms for Clinical Detections in Lung-Cancer-Related Exhaled Breath Biomarkers: A Review
Abstract
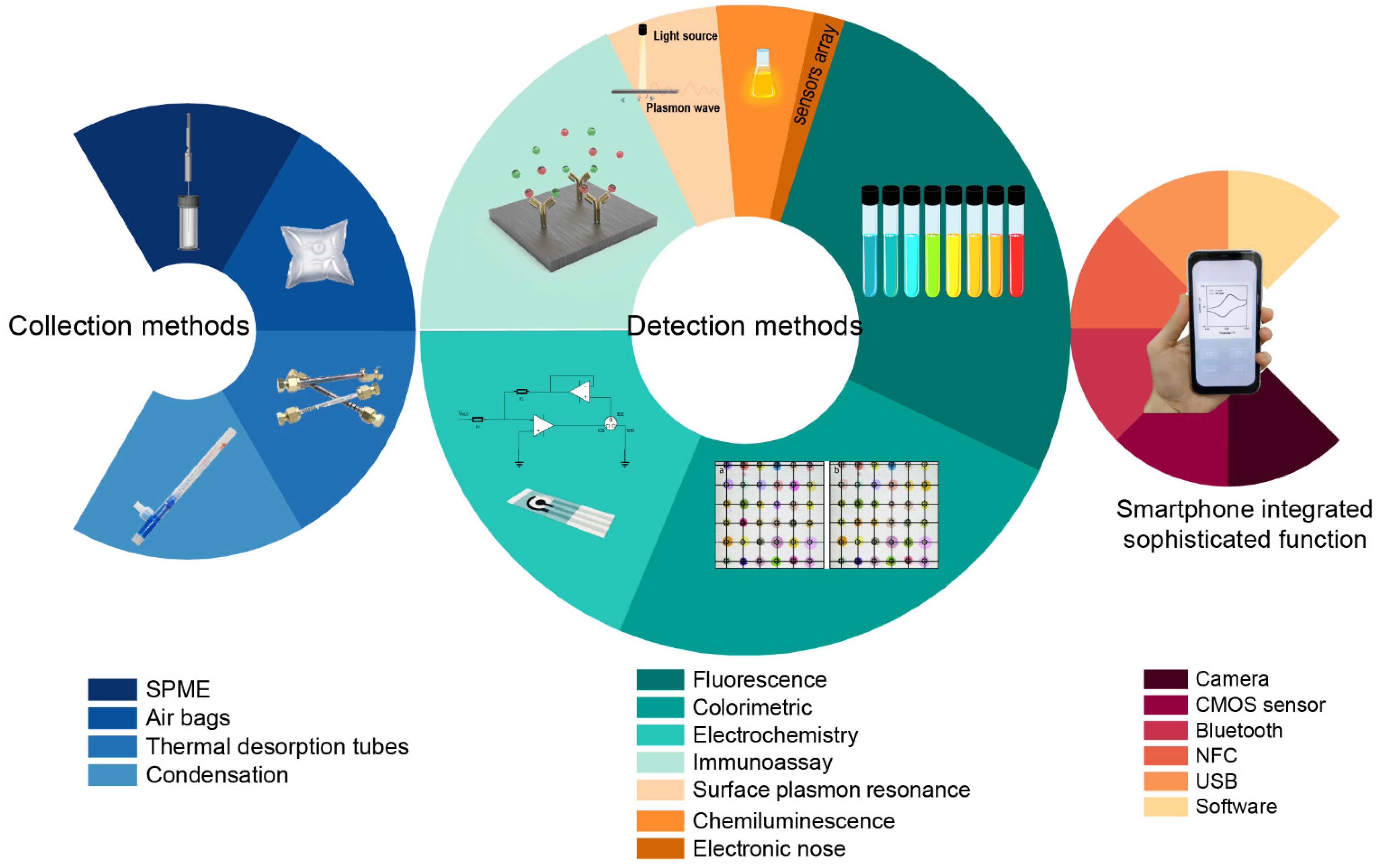
:1. Introduction
2. Smartphone-Based Detection Methods
2.1. Optical Sensors
2.1.1. Colorimetric Sensors
2.1.2. Immunoassay Sensors
2.2. Electrochemical Sensors
2.3. Electronic Noses
3. Biomarkers in VOCs and EBCs
3.1. Breath Sampling and Preconcentration
3.2. Biomarkers in VOCs
3.3. Biomarkers in EBCs
3.3.1. IL-6
3.3.2. Microsatellite DNA
3.3.3. MicroRNAs
4. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA-Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammerschmidt, S.; Wirtz, H. Lung cancer: Current diagnosis and treatment. Dtsch. Arztebl. Int. 2009, 106, 809–818; quiz 819–820. [Google Scholar] [PubMed]
- Phillips, M.; Cataneo, R.N.; Cummin, A.R.; Gagliardi, A.J.; Gleeson, K.; Greenberg, J.; Maxfield, R.A.; Rom, W.N. Detection of lung cancer with volatile markers in the breath. Chest 2003, 123, 2115–2123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dai, X.; Deng, S.; Wang, T.; Qiu, G.; Li, J.; Yang, B.; Feng, W.; He, X.; Deng, Q.; Ye, J.; et al. Associations between 25 Lung Cancer Risk-Related SNPs and Polycyclic Aromatic Hydrocarbon-Induced Genetic Damage in Coke Oven Workers. Cancer Epidemiol. Biomarkers Prev. 2014, 23, 986–996. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Wu, H.; Chen, B.; Hu, G.; Huang, L.; Qin, K.; Chen, Y.; Yuan, X.; Liao, Z. SNPs in the TGF-beta signaling pathway are associated with increased risk of brain metastasis in patients with non-small-cell lung cancer. PLoS ONE 2012, 7, e51713. [Google Scholar] [CrossRef]
- Rosell, R.; Wei, J. Single nucleotide polymorphisms (SNPs) in non-small cell lung cancer (NSCLC) patients. Oncologist 2012, 17, 1484–1485. [Google Scholar] [CrossRef] [Green Version]
- Islam, S.; Walker, R.C. Advanced Imaging (Positron Emission Tomography and Magnetic Resonance Imaging) and Image-Guided Biopsy in Initial Staging and Monitoring of Therapy of Lung Cancer. Cancer J. 2013, 19, 208–216. [Google Scholar] [CrossRef] [Green Version]
- Hunt, J. Exhaled breath condensate: An evolving tool for noninvasive evaluation of lung disease. J. Allergy Clin. Immunol. 2002, 110, 28–34. [Google Scholar] [CrossRef]
- Campanella, A.; De Summa, S.; Tommasi, S. Exhaled breath condensate biomarkers for lung cancer. J. Breath Res. 2019, 13, 044002. [Google Scholar] [CrossRef]
- Rattray, N.J.; Hamrang, Z.; Trivedi, D.K.; Goodacre, R.; Fowler, S.J. Taking your breath away: Metabolomics breathes life in to personalized medicine. Trends Biotechnol. 2014, 32, 538–548. [Google Scholar] [CrossRef]
- Baldini, C.; Billeci, L.; Sansone, F.; Conte, R.; Domenici, C.; Tonacci, A. Electronic Nose as a Novel Method for Diagnosing Cancer: A Systematic Review. Biosensors 2020, 10, 84. [Google Scholar] [CrossRef] [PubMed]
- Spira, A.; Beane, J.E.; Shah, V.; Steiling, K.; Liu, G.; Schembri, F.; Gilman, S.; Dumas, Y.M.; Calner, P.; Sebastiani, P.; et al. Airway epithelial gene expression in the diagnostic evaluation of smokers with suspect lung cancer. Nat. Med. 2007, 13, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Amann, A.; Corradi, M.; Mazzone, P.; Mutti, A. Lung cancer biomarkers in exhaled breath. Expert Rev. Mol. Diagn. 2011, 11, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, P.J.; Hammel, J.; Dweik, R.; Na, J.; Czich, C.; Laskowski, D.; Mekhail, T. Diagnosis of lung cancer by the analysis of exhaled breath with a colorimetric sensor array. Thorax 2007, 62, 565–568. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Rotaru, A.; Robeyns, K.; Garcia, Y. A colorimetric sensor for the highly selective, ultra-sensitive, and rapid detection of volatile organic compounds and hazardous gases. Ind. Eng. Chem. Res. 2021, 60, 8788–8798. [Google Scholar] [CrossRef]
- Cooper, E.H.; Splinter, T.A.; Brown, D.A.; Muers, M.F.; Peake, M.D.; Pearson, S.L. Evaluation of a radioimmunoassay for neuron specific enolase in small cell lung cancer. Br. J. Cancer 1985, 52, 333–338. [Google Scholar] [CrossRef] [Green Version]
- Andersen, L.; Dinesen, B.; Jorgensen, P.N.; Poulsen, F.; Roder, M.E. Enzyme immunoassay for intact human insulin in serum or plasma. Clin. Chem. 1993, 39, 578–582. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Klapperich, C.M. Design and testing of a disposable microfluidic chemiluminescent immunoassay for disease biomarkers in human serum samples. Biomed. Microdevices 2007, 9, 245–251. [Google Scholar] [CrossRef]
- Tang, D.; Wang, Y.; Chang, W.; Ci, Y.; Guo, Z. Time-resolved fluorescence immunoassay of estradiol in serum. Chin. J. Anal. Chem. 1999, 27, 899–903. [Google Scholar]
- Yalow, R.S.; Berson, S.A. Immunoassay of endogenous plasma insulin in man. J Clin Investig. 1960, 39, 1157–1175. [Google Scholar] [CrossRef]
- Engvall, E. Enzyme immunoassay ELISA and EMIT. Methods Enzymol. 1980, 70, 419–439. [Google Scholar] [CrossRef] [PubMed]
- Rubenstein, K.E.; Schneider, R.S.; Ullman, E.F. “Homogeneous” enzyme immunoassay. A new immunochemical technique. Biochem. Biophys. Res. Commun. 1972, 47, 846–851. [Google Scholar] [CrossRef]
- Uddin, M.J.; Bhuiyan, N.H.; Hong, J.H.; Shim, J.S. Smartphone-Based Fully Automated Optofluidic Device with Laser Irradiation-Induced Image Whitening. Anal. Chem. 2021, 93, 6394–6402. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.-B.; He, Y.-F.; Han, S.-Q. Matrix interference in serum total thyroxin (T4) time-resolved fluorescence immunoassay (TRFIA) and its elimination with the use of streptavidin–biotin separation technique. Clin. Chim. Acta 2001, 308, 117–126. [Google Scholar] [CrossRef]
- Liu, Z.; Hua, Q.; Wang, J.; Liang, Z.; Li, J.; Wu, J.; Shen, X.; Lei, H.; Li, X. A smartphone-based dual detection mode device integrated with two lateral flow immunoassays for multiplex mycotoxins in cereals. Biosens. Bioelectron. 2020, 158, 112178. [Google Scholar] [CrossRef]
- Li, S.; Liu, J.; Chen, Z.; Lu, Y.; Low, S.S.; Zhu, L.; Cheng, C.; He, Y.; Chen, Q.; Su, B. Electrogenerated chemiluminescence on smartphone with graphene quantum dots nanocomposites for Escherichia Coli detection. Sens. Actuators, B 2019, 297, 126811. [Google Scholar] [CrossRef]
- Zhu, L.; Li, S.; Liu, W.; Chen, J.; Yu, Q.; Zhang, Z.; Li, Y.; Liu, J.; Chen, X. Real time detection of 3-nitrotyrosine using smartphone-based electrochemiluminescence. Biosens. Bioelectron. 2021, 187, 113284. [Google Scholar] [CrossRef]
- Cui, F.; Zhou, Z.; Zhou, H.S. Measurement and analysis of cancer biomarkers based on electrochemical biosensors. J. Electrochem. Soc. 2019, 167, 037525. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, D.; Zhang, Q.; Chen, X.; Xu, G.; Lu, Y.; Liu, Q. Smartphone-based sensing system using ZnO and graphene modified electrodes for VOCs detection. Biosens. Bioelectron. 2017, 93, 94–101. [Google Scholar] [CrossRef]
- Rock, F.; Barsan, N.; Weimar, U. Electronic nose: Current status and future trends. Chem. Rev. 2008, 108, 705–725. [Google Scholar] [CrossRef]
- Zhao, Y.; Ruan, X.; Song, Y.; Smith, J.N.; Vasylieva, N.; Hammock, B.D.; Lin, Y.; Du, D. Smartphone-Based Dual-Channel Immunochromatographic Test Strip with Polymer Quantum Dot Labels for Simultaneous Detection of Cypermethrin and 3-Phenoxybenzoic Acid. Anal. Chem. 2021, 93, 13658–13666. [Google Scholar] [CrossRef] [PubMed]
- Zohora, S.E.; Khan, A.; Hundewale, N. Chemical Sensors Employed in Electronic Noses: A Review. In Advances in Computing and Information Technology; Springer: Berlin/Heidelberg, Germany, 2013; pp. 177–184. [Google Scholar]
- Wang, D.; Yu, K.; Wang, Y.S.; Hu, Y.J.; Zhao, C.; Wang, L.; Ying, K.J.; Wang, P. A Hybrid Electronic Noses’ System Based on Mos-Saw Detection Units Intended for Lung Cancer Diagnosis. J. Innov. Opt. Health Sci. 2012, 5. [Google Scholar] [CrossRef]
- Guo, X.; Zhang, J.; Chen, Y.; He, Y.; Wang, L.; Zheng, Y. Ultrasensitive Surface Acoustic Wave Gas Sensor for Trace VOCs Detection Based on Sensing Mechanism of Gas-Liquid Phase Transition. In Proceedings of the 2021 21st International Conference on Solid-State Sensors, Actuators and Microsystems (Transducers), Orlando, FL, USA, 20–24 June 2021; pp. 1227–1230. [Google Scholar]
- Kononov, A.; Korotetsky, B.; Jahatspanian, I.; Gubal, A.; Vasiliev, A.; Arsenjev, A.; Nefedov, A.; Barchuk, A.; Gorbunov, I.; Kozyrev, K. Online breath analysis using metal oxide semiconductor sensors (electronic nose) for diagnosis of lung cancer. J. Breath Res. 2019, 14, 016004. [Google Scholar] [CrossRef] [PubMed]
- Lawal, O.; Ahmed, W.M.; Nijsen, T.M.E.; Goodacre, R.; Fowler, S.J. Exhaled breath analysis: A review of ‘breath-taking’ methods for off-line analysis. Metabolomics 2017, 13, 110. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.-E.; Lee, D.-S.; Ban, S.-W.; Oh, J.; Jung, M.Y.; Kim, S.-H.; Park, S.; Persaud, K.; Jheon, S. Analysis of volatile organic compounds in exhaled breath for lung cancer diagnosis using a sensor system. Sens. Actuators, B 2018, 255, 800–807. [Google Scholar] [CrossRef]
- Di Gilio, A.; Catino, A.; Lombardi, A.; Palmisani, J.; Facchini, L.; Mongelli, T.; Varesano, N.; Bellotti, R.; Galetta, D.; de Gennaro, G.; et al. Breath Analysis for Early Detection of Malignant Pleural Mesothelioma: Volatile Organic Compounds (VOCs) Determination and Possible Biochemical Pathways. Cancers 2020, 12, 1262. [Google Scholar] [CrossRef]
- Amann, A.; de Lacy Costello, B.; Miekisch, W.; Schubert, J.; Buszewski, B.; Pleil, J.; Ratcliffe, N.; Risby, T. The human volatilome: Volatile organic compounds (VOCs) in exhaled breath, skin emanations, urine, feces and saliva. J. Breath Res. 2014, 8, 034001. [Google Scholar] [CrossRef]
- Reyes-Garces, N.; Gionfriddo, E.; Gmez-Rios, G.A.; Alam, M.N.; Boyaci, E.; Bojko, B.; Singh, V.; Grandy, J.; Pawliszyn, J. Advances in Solid Phase Microextraction and Perspective on Future Directions. Anal. Chem. 2018, 90, 302–360. [Google Scholar] [CrossRef]
- Harshman, S.W.; Rubenstein, M.H.; Qualley, A.V.; Fan, M.; Geier, B.A.; Pitsch, R.L.; Slusher, G.M.; Hughes, G.T.; Dershem, V.L.; Grigsby, C.C. Evaluation of thermal desorption analysis on a portable GC–MS system. Int. J. Environ. Anal. Chem. 2017, 97, 247–263. [Google Scholar] [CrossRef]
- de Lacy Costello, B.; Amann, A.; Al-Kateb, H.; Flynn, C.; Filipiak, W.; Khalid, T.; Osborne, D.; Ratcliffe, N.M. A review of the volatiles from the healthy human body. J. Breath Res. 2014, 8, 014001. [Google Scholar] [CrossRef]
- Risby, T.H.; Sehnert, S.S. Clinical application of breath biomarkers of oxidative stress status. Free Radic. Biol. Med. 1999, 27, 1182–1192. [Google Scholar] [CrossRef]
- Kneepkens, C.M.; Ferreira, C.; Lepage, G.; Roy, C.C. The hydrocarbon breath test in the study of lipid peroxidation: Principles and practice. Clin. Investig. Med. 1992, 15, 163–186. [Google Scholar]
- Kneepkens, F.; Lepage, G.; Roy, C.C. The potential of the hydrocarbon breath test as a measure of lipid peroxidation. Free Radic. Biol. Med. 1994, 17, 127–160. [Google Scholar] [CrossRef]
- Phillips, M.; Cataneo, R.N.; Greenberg, J.; Gunawardena, R.; Naidu, A.; Rahbari-Oskoui, F. Effect of age on the breath methylated alkane contour, a display of apparent new markers of oxidative stress. J. Lab. Clin. Med. 2000, 136, 243–249. [Google Scholar] [CrossRef] [Green Version]
- Gordon, S.M.; Szidon, J.P.; Krotoszynski, B.K.; Gibbons, R.D.; O’Neill, H.J. Volatile organic compounds in exhaled air from patients with lung cancer. Clin. Chem. 1985, 31, 1278–1282. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.; Gleeson, K.; Hughes, J.M.B.; Greenberg, J.; Cataneo, R.N.; Baker, L.; McVay, W.P. Volatile organic compounds in breath as markers of lung cancer: A cross-sectional study. Lancet 1999, 353, 1930–1933. [Google Scholar] [CrossRef]
- Poli, D.; Carbognani, P.; Corradi, M.; Goldoni, M.; Acampa, O.; Balbi, B.; Bianchi, L.; Rusca, M.; Mutti, A. Exhaled volatile organic compounds in patients with non-small cell lung cancer: Cross sectional and nested short-term follow-up study. Respir. Res. 2005, 6, 71. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Jiang, N.; Wang, L.; Liu, H.; He, R. Chronic obstructive pulmonary disease and risk of lung cancer: A meta-analysis of prospective cohort studies. Oncotarget 2017, 8, 78044. [Google Scholar] [CrossRef] [Green Version]
- Wehinger, A.; Schmid, A.; Mechtcheriakov, S.; Ledochowski, M.; Grabmer, C.; Gastl, G.A.; Amann, A. Lung cancer detection by proton transfer reaction mass-spectrometric analysis of human breath gas. Int. J. Mass Spectrom. 2007, 265, 49–59. [Google Scholar] [CrossRef]
- Bajtarevic, A.; Ager, C.; Pienz, M.; Klieber, M.; Schwarz, K.; Ligor, M.; Ligor, T.; Filipiak, W.; Denz, H.; Fiegl, M. Noninvasive detection of lung cancer by analysis of exhaled breath. BMC Cancer 2009, 9, 348. [Google Scholar] [CrossRef] [Green Version]
- Fuchs, P.; Loeseken, C.; Schubert, J.K.; Miekisch, W. Breath gas aldehydes as biomarkers of lung cancer. Int. J. Cancer 2010, 126, 2663–2670. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Qin, T.; Liu, H.; Xu, G.-B.; Pan, Y.-Y.; Xiong, F.-X.; Gu, K.-S.; Sun, G.-P.; Chen, Z.-D. Quantitative breath analysis of volatile organic compounds of lung cancer patients. Lung Cancer 2010, 67, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Ulanowska, A.; Kowalkowski, T.; Trawińska, E.; Buszewski, B. The application of statistical methods using VOCs to identify patients with lung cancer. J. Breath Res. 2011, 5, 046008. [Google Scholar] [CrossRef] [PubMed]
- Buszewski, B.; Ligor, T.; Jezierski, T.; Wenda-Piesik, A.; Walczak, M.; Rudnicka, J. Identification of volatile lung cancer markers by gas chromatography–mass spectrometry: Comparison with discrimination by canines. Anal. Bioanal. Chem. 2012, 404, 141–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.; Huang, J.; Abbassi-Ghadi, N.; Mackenzie, H.A.; Veselkov, K.A.; Hoare, J.M.; Lovat, L.B.; Španěl, P.; Smith, D.; Hanna, G.B. Mass spectrometric analysis of exhaled breath for the identification of volatile organic compound biomarkers in esophageal and gastric adenocarcinoma. Ann. Surg. 2015, 262, 981–990. [Google Scholar] [CrossRef]
- Schallschmidt, K.; Becker, R.; Jung, C.; Bremser, W.; Walles, T.; Neudecker, J.; Leschber, G.; Frese, S.; Nehls, I. Comparison of volatile organic compounds from lung cancer patients and healthy controls—Challenges and limitations of an observational study. J. Breath Res. 2016, 10, 046007. [Google Scholar] [CrossRef]
- Sakumura, Y.; Koyama, Y.; Tokutake, H.; Hida, T.; Sato, K.; Itoh, T.; Akamatsu, T.; Shin, W. Diagnosis by volatile organic compounds in exhaled breath from lung cancer patients using support vector machine algorithm. Sensors 2017, 17, 287. [Google Scholar] [CrossRef] [Green Version]
- Phillips, M.; Bauer, T.L.; Pass, H.I. A volatile biomarker in breath predicts lung cancer and pulmonary nodules. J. Breath Res. 2019, 13, 036013. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Dai, W.; Liu, M.; Long, Y.; Wang, C.; Xie, S.; Liu, Y.; Zhang, Y.; Shi, Q.; Peng, X.; et al. VOC biomarkers identification and predictive model construction for lung cancer based on exhaled breath analysis: Research protocol for an exploratory study. BMJ Open 2019, 9. [Google Scholar] [CrossRef] [Green Version]
- Salimi, M.; Hosseini, S.M.R.M. Smartphone-based detection of lung cancer-related volatile organic compounds (VOCs) using rapid synthesized ZnO nanosheet. Sens. Actuators, B 2021, 344, 130127. [Google Scholar] [CrossRef]
- Yang, F.; Lin, D.; Pan, L.; Zhu, J.; Shen, J.; Yang, L.; Jiang, C. Portable Smartphone Platform Based on a Single Dual-Emissive Ratiometric Fluorescent Probe for Visual Detection of Isopropanol in Exhaled Breath. Anal. Chem. 2021, 93, 14506–14513. [Google Scholar] [CrossRef] [PubMed]
- Dalaveris, E.; Kerenidi, T.; Katsabeki-Katsafli, A.; Kiropoulos, T.; Tanou, K.; Gourgoulianis, K.I.; Kostikas, K. VEGF, TNF-alpha and 8-isoprostane levels in exhaled breath condensate and serum of patients with lung cancer. Lung Cancer 2009, 64, 219–225. [Google Scholar] [CrossRef]
- Liloglou, T.; Bediaga, N.G.; Brown, B.R.B.; Field, J.K.; Davies, M.P.A. Epigenetic biomarkers in lung cancer. Cancer Lett. 2014, 342, 200–212. [Google Scholar] [CrossRef]
- Duruisseaux, M.; Esteller, M. Lung cancer epigenetics: From knowledge to applications. Semin. Cancer Biol. 2018, 51, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Wang, T.; Reilly, A.A.; Keller, S.M.; Spivack, S.D. Gene promoter methylation assayed in exhaled breath, with differences in smokers and lung cancer patients. Respir. Res. 2009, 10, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahrendt, S.A.; Chow, J.T.; Xu, L.H.; Yang, S.C.; Eisenberger, C.F.; Esteller, M.; Herman, J.G.; Wu, L.; Decker, P.A.; Jen, J.; et al. Molecular detection of tumor cells in bronchoalveolar lavage fluid from patients with early stage lung cancer. J. Natl. Cancer Inst. 1999, 91, 332–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Zou, Y.C.; An, C.; Ying, K.J.; Chen, X.; Wang, P. A miniaturized immunosensor platform for automatic detection of carcinoembryonic antigen in EBC. Sens. Actuators, B 2014, 205, 94–101. [Google Scholar] [CrossRef]
- Dodig, S.; Čepelak, I. Exhaled breath condensate–from an analytical point of view. Biochem. Med. 2013, 23, 281–295. [Google Scholar] [CrossRef]
- Quan, Z.; Purser, C.; Baker, R.C.; Dwyer, T.; Bhagat, R.; Sheng, Y.; Leszczynski, J.R. Determination of derivatized urea in exhaled breath condensate by LC-MS. J. Chromatogr. Sci. 2010, 48, 140–144. [Google Scholar] [CrossRef] [Green Version]
- Pine, S.R.; Mechanic, L.E.; Enewold, L.; Chaturvedi, A.K.; Katki, H.A.; Zheng, Y.-L.; Bowman, E.D.; Engels, E.A.; Caporaso, N.E.; Harris, C.C. Increased levels of circulating interleukin 6, interleukin 8, C-reactive protein, and risk of lung cancer. J. Natl. Cancer Inst. 2011, 103, 1112–1122. [Google Scholar] [CrossRef]
- Adrover-Jaume, C.; Alba-Patiño, A.; Clemente, A.; Santopolo, G.; Vaquer, A.; Russell, S.M.; Barón, E.; Del Campo, M.D.M.G.; Ferrer, J.M.; Berman-Riu, M. Paper biosensors for detecting elevated IL-6 levels in blood and respiratory samples from COVID-19 patients. Sens. Actuators, B 2021, 330, 129333. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.; Luo, H.; Krawczyk, M.; Wei, W.; Wang, W.; Wang, J.; Flagg, K.; Hou, J.; Zhang, H.; Yi, S.; et al. DNA methylation markers for diagnosis and prognosis of common cancers. Proc. Natl. Acad. Sci. USA 2017, 114, 7414–7419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hascher, A.; Haase, A.-K.; Hebestreit, K.; Rohde, C.; Klein, H.-U.; Rius, M.; Jungen, D.; Witten, A.; Stoll, M.; Schulze, I.; et al. DNA Methyltransferase Inhibition Reverses Epigenetically Embedded Phenotypes in Lung Cancer Preferentially Affecting Polycomb Target Genes. Clin. Cancer Res. 2014, 20, 814–826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herman, J.G.; Graff, J.R.; Myohanen, S.; Nelkin, B.D.; Baylin, S.B. Methylation-specific PCR: A novel PCR assay for methylation status of CpG islands. Proc. Natl. Acad. Sci. USA 1996, 93, 9821–9826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalligosfyri, P.M.; Sevastou, A.; Kyriakou, I.K.; Tragoulias, S.S.; Kalogianni, D.P.; Christopoulos, T.K. Smartphone-based chemiluminometric hybridization assays and quantitative competitive polymerase chain reaction. Anal. Chim. Acta 2019, 1088, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.; Thakur, R.K.; Yadav, V.K.; Nanda, R.; Chowdhury, S.; Agrawal, A. Lung cancer biomarkers: State of the art. J Carcinog 2013, 12, 3. [Google Scholar]
- Mallick, R.; Patnaik, S.K.; Yendamuri, S. MicroRNAs and lung cancer: Biology and applications in diagnosis and prognosis. J. Carcinog. 2010, 9, 8. [Google Scholar]
- Mozzoni, P.; Banda, I.; Goldoni, M.; Corradi, M.; Tiseo, M.; Acampa, O.; Balestra, V.; Ampollini, L.; Casalini, A.; Carbognani, P.; et al. Plasma and EBC microRNAs as early biomarkers of non-small-cell lung cancer. Biomarkers 2013, 18, 679–686. [Google Scholar] [CrossRef]
- Lee, J.; Na, H.-K.; Lee, S.; Kim, W.-K. Advanced graphene oxide-based paper sensor for colorimetric detection of miRNA. Microchim. Acta 2022, 189, 1–8. [Google Scholar] [CrossRef]
- Low, S.S.; Pan, Y.; Ji, D.; Li, Y.; Lu, Y.; He, Y.; Chen, Q.; Liu, Q. Smartphone-based portable electrochemical biosensing system for detection of circulating microRNA-21 in saliva as a proof-of-concept. Sens. Actuators B 2020, 308. [Google Scholar]
- Li, Y.; Zhou, L.; Ni, W.; Luo, Q.; Zhu, C.; Wu, Y. Portable and Field-Ready Detection of Circulating MicroRNAs with Paper-Based Bioluminescent Sensing and Isothermal Amplification. Anal. Chem. 2019, 91, 14838–14841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, L.; Zhang, L.; Yang, L.; Ni, W.; Li, Y.; Wu, Y. Tandem reassembly of split luciferase-DNA chimeras for bioluminescent detection of attomolar circulating microRNAs using a smartphone. Biosens. Bioelectron. 2021, 173, 112824. [Google Scholar] [CrossRef]
- Cheng, S.; Hideshima, S.; Kuroiwa, S.; Nakanishi, T.; Osaka, T. Label-free detection of tumor markers using field effect transistor (FET)-based biosensors for lung cancer diagnosis. Sens. Actuators B 2015, 212, 329–334. [Google Scholar] [CrossRef]
- Cheng, S.; Hotani, K.; Hideshima, S.; Kuroiwa, S.; Nakanishi, T.; Hashimoto, M.; Mori, Y.; Osaka, T. Field Effect Transistor Biosensor Using Antigen Binding Fragment for Detecting Tumor Marker in Human Serum. Materials 2014, 7, 2490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Cao, Z.; Zhang, Y.; Lau, C.; Lu, J. Simultaneous detection of two lung cancer biomarkers using dual-color fluorescence quantum dots. Analyst 2011, 136, 1399–1405. [Google Scholar] [CrossRef]
- Wu, Z.; Li, H.; Liu, Z. An aptasensor for carcinoembryonic antigen based on upconversion fluorescence resonance energy transfer. Sens. Actuators B 2015, 206, 531–537. [Google Scholar] [CrossRef]
- Xu, G.; Lu, Y.; Cheng, C.; Li, X.; Xu, J.; Liu, Z.; Liu, J.; Liu, G.; Shi, Z.; Chen, Z. Battery-free and wireless smart wound dressing for wound infection monitoring and electrically controlled on-demand drug delivery. Adv. Funct. Mater. 2021, 31, 2100852. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA-Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef]



| Years | Author | Collection Method | Sample | VOCs |
|---|---|---|---|---|
| 1985 | Gordon [47] | Tenax GC sorbent cartridges | Expired breath | Acetone, 2-butanone, n-propanol |
| 1999 | Phillips [48] | Sorbent trap | Alveolar breath | Styrene, 2,2,4,6,6-pentamethylheptane, 2-methylheptane, decane, n-propylbenzene undecane, methyl cyclopentane, 1-methyl-2-pentylcyclopropane, trichlorofluoromethane, benzene, 1,2,4-trimethylbenzene, isoprene, 3-methyloctane, 1-hexene, 3-methylnonane, 1-heptene, 1,4-dimethylbenzene, 2,4-dimethylheptane, hexanal, cyclohexane, 1-methylethenylbenzene, heptanal |
| 2005 | Poli [49] | Teflon® bulb; SPME | Mixed expiratory samples | Isoprene; methylpentane; pentane; ethylbenzene; xylenes; trimethylbenzene; toluene; benzene; heptane; decane, styrene; octane; pentamethylheptane |
| 2007 | Wehinger [51] | Tedlar® bags | Alveolar breath | Formaldehyde, isopropanol |
| 2009 | Bajtarevic [52] | Tedlar® bags | Mixed expiratory and indoor air | Isoprene, acetone, methanol; 2-butanone, benzaldehyde, 2,3-butanedione, 1-propanol, 2-butanone, 3-hydroxy-, 3-butyn-2-ol, butane, 2-methyl-, 2-butene, 2-methyl-, acetophenone, 1-cyclopentene, methyl propyl sulfide, urea, tetramethyl-, n-pentanal, 1,3-cyclopentadiene, 1-methyl-, 2-butanol, 2,3-dimethyl-, isoquinoline, 1,2,3,4-tetrahydro-, undecane, 3,7-dimethyl-, benzene, cyclobutyl-, butyl acetate, ethylenimine, n-undecane, |
| 2010 | Fuchs [53] | Mylar sampling bag | Alveolar breath | p-Cymene, toluene, dodecane, 3,3-dimethylpentane, 2,3,4-trimethylhexane, (1-phenyl-1-butenyl)benzene 1,3-dimethylbenzene, 1-iodononane, [(1,1-dimethylethyl) thiol]acetic acid, 4-(4-propylcyclohexyl)-4′-cyano [1,1′-biphenyl]4-yl ester benzoic acid, 2-amino-5-isopropyl-8-methyl-1-azulenecarbonitrile, 5-(2-methylpropyl)nonane, 2,3,4-trimethyldecane, 6-ethyl-3-octanyl 2-(trifluoromethyl)benzoate, p-xylene, and 2,2-dimethyldecane |
| 2010 | Song [54] | Tedlar® gas bags; SPME | Mixed expiratory samples | 1-Butanol and 3-hydroxy-2-butanone |
| 2011 | Ulanowska [55] | Tedlar® bags; SPME | Alveolar breath | Ethanol, acetone, butane, dimethyl sulfide, isoprene, propanal, 1-propanol, 2-pentanone, furan, o-xylene, ethylbenzene, pentanal, hexanal, nonane |
| 2012 | Buszewski [56] | Tedlar® bags; SPME | Alveolar breath | Butanal, ethyl acetate, 2-pentanone, ethylbenzene, 1-propanol, 2-propanol |
| 2015 | Kumar [57] | Nalophan bag | Mixed alveolar breath | Pentanoic acid; hexanoic acid; phenol; methyl phenol; ethyl phenol; butanal; pentanal; hexanal; heptanal; octanal; nonanal; decanal |
| 2016 | Schallschmidt [58] | Gas bulbs; SPME | Tidal breath | Propanal, butanal, decanal, butanal, 2-butanone, ethylbenzene |
| 2017 | Sakumura [59] | Analytic Barrier Bag | Alveolar breath | Hydrogen cyanide, methanol, acetonitrile, isoprene, 1-propanol |
| 2019 | Phillips [60] | Carbotrap C and Carbopack C | Alveolar breath | 1,4-Butanediol, 2-pentanamine, 4-methyl-, 2-propanamine, 3-butenamide, 4-penten-2-ol, acetamide, 2-cyanoalanine, n-methylglycine, octodrine |
| 2019 | Li [61] | Tedlar® bags; SPME | End-tidal breath | Isopropanol, n-butanol, n-heptanol, n-hexanal, n-heptanal, n-decanal |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Q.; Chen, J.; Fu, W.; Muhammad, K.G.; Li, Y.; Liu, W.; Xu, L.; Dong, H.; Wang, D.; Liu, J.; et al. Smartphone-Based Platforms for Clinical Detections in Lung-Cancer-Related Exhaled Breath Biomarkers: A Review. Biosensors 2022, 12, 223. https://doi.org/10.3390/bios12040223
Yu Q, Chen J, Fu W, Muhammad KG, Li Y, Liu W, Xu L, Dong H, Wang D, Liu J, et al. Smartphone-Based Platforms for Clinical Detections in Lung-Cancer-Related Exhaled Breath Biomarkers: A Review. Biosensors. 2022; 12(4):223. https://doi.org/10.3390/bios12040223
Chicago/Turabian StyleYu, Qiwen, Jing Chen, Wei Fu, Kanhar Ghulam Muhammad, Yi Li, Wenxin Liu, Linxin Xu, Hao Dong, Di Wang, Jun Liu, and et al. 2022. "Smartphone-Based Platforms for Clinical Detections in Lung-Cancer-Related Exhaled Breath Biomarkers: A Review" Biosensors 12, no. 4: 223. https://doi.org/10.3390/bios12040223
APA StyleYu, Q., Chen, J., Fu, W., Muhammad, K. G., Li, Y., Liu, W., Xu, L., Dong, H., Wang, D., Liu, J., Lu, Y., & Chen, X. (2022). Smartphone-Based Platforms for Clinical Detections in Lung-Cancer-Related Exhaled Breath Biomarkers: A Review. Biosensors, 12(4), 223. https://doi.org/10.3390/bios12040223






