Tri-Channel Electrochemical Immunobiosensor for Combined Detections of Multiple Exosome Biomarkers of Lung Cancer
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Reagents
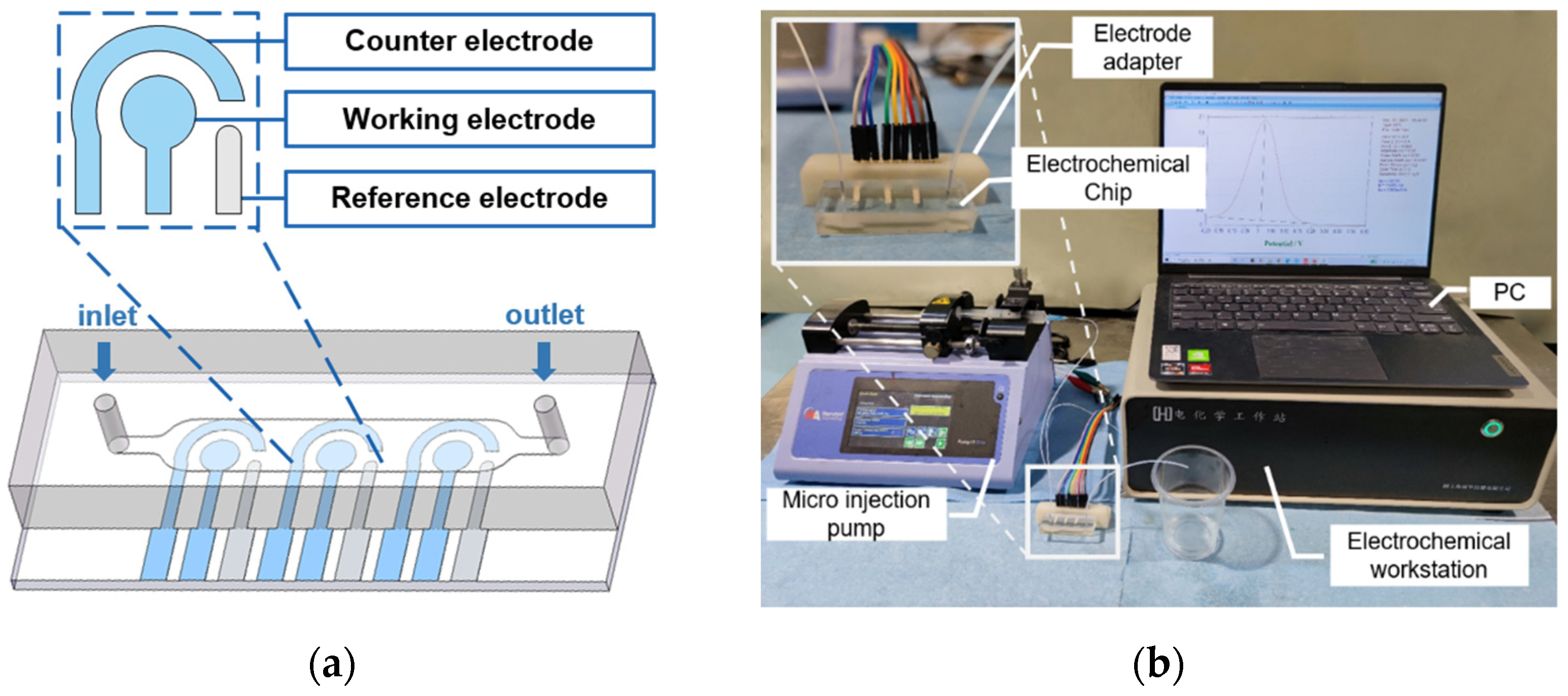
2.2. Fabrication of Tri-Channel Electrodes
2.3. Functionalization of Immunosensor
2.4. Cell Culture and Isolation of Exosomes
2.4.1. Cell Culture
2.4.2. Extraction of Exosome
2.4.3. Characterization of Exosomes
2.5. Electrochemical Detection
3. Results and Discussion
3.1. Electrochemical Chip
3.2. Characterization of Immunosensor Fabrication Steps
3.3. Experimental Parameters Optimization
3.4. Analytical Performance of the Designed Biosensor
3.4.1. Sensitivity
3.4.2. Selectivity
3.4.3. Storage Stability
3.5. Characterization of Exosome
3.6. Multiplexed Detection of Exosome Biomarkers of Lung Cancer
4. Conclusions
- The utilization of ITO glass instead of precious metals such as gold offers an inexpensive, simple, and sensitive system, which makes it easy to realize small sample amounts and integrated detection when combined with microchannels.
- The proposed immunosensor with optimized parameters can detect exosome markers in a range from 10−3 to 10 ng/mL for CEA, 10−4 to 102 ng/mL for NSE, and 10−3 to 102 ng/mL for Cyfra21-1, with a detection limit below 10−4 ng/mL, which was lower than the conventional ELISA method. The immunosensor is suitable for exosome detection in real samples for practical applications.
- The combined detection of multiple exosome markers has a higher efficiency than a single biomarker of ELISA. The difference in expression level may guide the typing of lung cancer; nevertheless, clinical trials with adequate sample volume are still needed for further validation.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Valihrach, L.; Androvic, P.; Kubista, M. Circulating miRNA analysis for cancer diagnostics and therapy. Mol. Asp. Med. 2020, 72, 100825. [Google Scholar] [CrossRef] [PubMed]
- Sateia, H.F.; Choi, Y.; Stewart, R.W.; Peairs, K.S. Screening for lung cancer. Semin. Oncol. 2017, 44, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Xiao, Z.; Tang, C.; Deng, Y.; Huang, H.; He, Z. Recent advances in biosensor for detection of lung cancer biomarkers. Biosens. Bioelectron. 2019, 141, 111416. [Google Scholar] [CrossRef] [PubMed]
- Shao, H.; Im, H.; Castro, C.M.; Breakefield, X.; Weissleder, R.; Lee, H. New Technologies for Analysis of Extracellular Vesicles. Chem. Rev. 2018, 118, 1917–1950. [Google Scholar] [CrossRef] [PubMed]
- Soung, Y.H.; Ford, S.; Zhang, V.; Chung, J. Exosomes in Cancer Diagnostics. Cancers 2017, 9, 8. [Google Scholar] [CrossRef] [Green Version]
- Yokoyama, S.; Takeuchi, A.; Yamaguchi, S.; Mitani, Y.; Watanabe, T.; Matsuda, K.; Hotta, T.; Shively, J.E.; Yamaue, H. Clinical implications of carcinoembryonic antigen distribution in serum exosomal fraction—Measurement by ELISA. PLoS ONE 2017, 12, e0183337. [Google Scholar] [CrossRef]
- Cao, Y.; Li, L.; Han, B.; Wang, Y.; Dai, Y.; Zhao, J. A catalytic molecule machine-driven biosensing method for amplified electrochemical detection of exosomes. Biosens. Bioelectron. 2019, 141, 111397. [Google Scholar] [CrossRef]
- Zong, S.; Wang, L.; Chen, C.; Lu, J.; Zhu, D.; Zhang, Y.; Wang, Z.; Cui, Y. Facile detection of tumor-derived exosomes using magnetic nanobeads and SERS nanoprobes. Anal. Methods 2016, 8, 5001–5008. [Google Scholar] [CrossRef]
- Suthar, J.; Parsons, E.S.; Hoogenboom, B.W.; Williams, G.R.; Guldin, S. Acoustic Immunosensing of Exosomes Using a Quartz Crystal Microbalance with Dissipation Monitoring. Anal. Chem. 2020, 92, 4082–4093. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Yang, Y.; Liu, Y.; Ning, L.; Xiang, Y.; Li, G. Bridging exosome and liposome through zirconium–phosphate coordination chemistry: A new method for exosome detection. Chem. Commun. 2019, 55, 2708–2711. [Google Scholar] [CrossRef]
- Zhao, W.; Hu, J.; Liu, J.; Li, X.; Sun, S.; Luan, X.; Zhao, Y.; Wei, S.; Li, M.; Zhang, Q.; et al. Si nanowire Bio-FET for electrical and label-free detection of cancer cell-derived exosomes. Microsyst. Nanoeng. 2022, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Kwizera, E.A.; O’Connor, R.; Vinduska, V.; Williams, M.; Butch, E.R.; Snyder, S.E.; Chen, X.; Huang, X. Molecular Detection and Analysis of Exosomes Using Surface-Enhanced Raman Scattering Gold Nanorods and a Miniaturized Device. Theranostics 2018, 8, 2722–2738. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Crow, J.; Roth, M.; Zeng, Y.; Godwin, A.K. Integrated immunoisolation and protein analysis of circulating exosomes using microfluidic technology. Lab Chip 2014, 14, 3773–3780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yadav, S.; Boriachek, K.; Islam, M.N.; Lobb, R.; Möller, A.; Hill, M.M.; Hossain, M.S.A.; Nguyen, N.T.; Shiddiky, M.J. An Electrochemical Method for the Detection of Disease-Specific Exosomes. ChemElectroChem 2017, 4, 967–971. [Google Scholar] [CrossRef]
- Doldán, X.; Fagúndez, P.; Cayota, A.; Laíz, J.; Tosar, J.P. Electrochemical Sandwich Immunosensor for Determination of Exosomes Based on Surface Marker-Mediated Signal Amplification. Anal. Chem. 2016, 88, 10466–10473. [Google Scholar] [CrossRef]
- Huang, J.; Yan, H.; Ma, M.; Tian, N.; Ni, H.; Xia, M.; Pei, K.; Ye, P. Hydroxylated Graphene Porous Membrane-Based Biosensor for Exosome Isolation and Detection. ACS Appl. Nano Mater. 2022, 5, 6115–6124. [Google Scholar] [CrossRef]
- Zhu, L.; Wang, K.; Cui, J.; Liu, H.; Bu, X.; Ma, H.; Wang, W.; Gong, H.; Lausted, C.; Hood, L.; et al. Label-Free Quantitative Detection of Tumor-Derived Exosomes through Surface Plasmon Resonance Imaging. Anal. Chem. 2014, 86, 8857–8864. [Google Scholar] [CrossRef] [Green Version]
- Jeong, S.; Park, J.; Pathania, D.; Castro, C.M.; Weissleder, R.; Lee, H. Integrated Magneto–Electrochemical Sensor for Exosome Analysis. ACS Nano 2016, 10, 1802–1809. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Wang, C.; Jin, D.; Yu, Y.; Yang, F.; Zhang, Y.; Yao, Q.; Zhang, G.J. AuNP-Amplified Surface Acoustic Wave Sensor for the Quantification of Exosomes. ACS Sens. 2020, 5, 362–369. [Google Scholar] [CrossRef]
- Zhao, Z.; Yang, Y.; Zeng, Y.; He, M. A microfluidic ExoSearch chip for multiplexed exosome detection towards blood-based ovarian cancer diagnosis. Lab Chip 2016, 16, 489–496. [Google Scholar] [CrossRef] [Green Version]
- Hashkavayi, A.B.; Cha, B.S.; Lee, E.S.; Park, K.S. Dual rolling circle amplification-enabled ultrasensitive multiplex detection of exosome biomarkers using electrochemical aptasensors. Anal. Chim. Acta 2022, 1205, 339762. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Lu, Y.; Wang, K.; Cheng, Z.; Qu, Y.; Qiu, S.; Zhou, L.; Wu, Z.; Liu, H.; Zhao, J.; et al. Rapid Isolation and Multiplexed Detection of Exosome Tumor Markers Via Queued Beads Combined with Quantum Dots in a Microarray. Nano-Micro Lett. 2019, 11, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dal Bello, M.G.; Filiberti, R.A.; Alama, A.; Orengo, A.M.; Mussap, M.; Coco, S.; Vanni, I.; Boccardo, S.; Rijavec, E.; Genova, C.; et al. The role of CEA, CYFRA21-1 and NSE in monitoring tumor response to Nivolumab in advanced non-small cell lung cancer (NSCLC) patients. J. Transl. Med. 2019, 17, 74. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Zhang, G.; Lian, Y.; Yan, L.; Gao, H. Exploration and analysis of the value of tumor-marker joint detection in the pathological type of lung cancer. Cell. Mol. Biol. 2020, 66, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Chia, B.S.; Low, Y.P.; Wang, Q.; Li, P.; Gao, Z. Advances in exosome quantification techniques. TrAC Trends Anal. Chem. 2017, 86, 93–106. [Google Scholar] [CrossRef]
- Lorencova, L.; Bertok, T.; Bertokova, A.; Gajdosova, V.; Hroncekova, S.; Vikartovska, A.; Kasak, P.; Tkac, J. Exosomes as a Source of Cancer Biomarkers: Advances in Electrochemical Biosensing of Exosomes. ChemElectroChem 2020, 7, 1956–1973. [Google Scholar] [CrossRef]
- Reverté, L.; Prieto-Simón, B.; Campàs, M. New advances in electrochemical biosensors for the detection of toxins: Nanomaterials, magnetic beads and microfluidics systems. A review. Anal. Chim. Acta 2016, 908, 8–21. [Google Scholar] [CrossRef]
- Hashkavayi, A.B.; Cha, B.S.; Lee, E.S.; Kim, S.; Park, K.S. Advances in Exosome Analysis Methods with an Emphasis on Electrochemistry. Anal. Chem. 2020, 92, 12733–12740. [Google Scholar] [CrossRef]
- Su, J.; Chen, S.; Dou, Y.; Zhao, Z.; Jia, X.; Ding, X.; Song, S. Smartphone-Based Electrochemical Biosensors for Directly Detecting Serum-Derived Exosomes and Monitoring Their Secretion. Anal. Chem. 2022, 94, 3235–3244. [Google Scholar] [CrossRef]
- Dong, H.; Chen, H.; Jiang, J.; Zhang, H.; Cai, C.; Shen, Q. Highly Sensitive Electrochemical Detection of Tumor Exosomes Based on Aptamer Recognition-Induced Multi-DNA Release and Cyclic Enzymatic Amplification. Anal. Chem. 2018, 90, 4507–4513. [Google Scholar] [CrossRef]
- Jia, X.; Liu, Z.; Liu, N.; Ma, Z. A label-free immunosensor based on graphene nanocomposites for simultaneous multiplexed electrochemical determination of tumor markers. Biosens. Bioelectron. 2014, 53, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Zuo, X.; Li, Z.; Deng, W.; Shi, J.; Zhang, G.; Huang, Q.; Song, S.; Fan, C. A Bubble-Mediated Intelligent Microscale Electrochemical Device for Single-Step Quantitative Bioassays. Adv. Mater. 2014, 26, 4671–4676. [Google Scholar] [CrossRef] [PubMed]
- Lan, Q.; Shen, H.; Li, J.; Ren, C.; Hu, X.; Yang, Z. Facile synthesis of novel reduced graphene oxide@polystyrene nanospheres for sensitive label-free electrochemical immunoassay. Chem. Commun. 2020, 56, 699–702. [Google Scholar] [CrossRef]
- Boriachek, K.; Islam, N.; Gopalan, V.; Lam, A.K.; Nguyen, N.-T.; Shiddiky, M.J.A. Quantum dot-based sensitive detection of disease specific exosome in serum. Analyst 2017, 142, 2211–2219. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Rahimian, A.; Son, K.; Shin, D.-S.; Patel, T.; Revzin, A. Development of an aptasensor for electrochemical detection of exosomes. Methods 2016, 97, 88–93. [Google Scholar] [CrossRef] [Green Version]
- Boriachek, K.; Umer, M.; Islam, N.; Gopalan, V.; Lam, A.K.; Nguyen, N.-T.; Shiddiky, M.J.A. An amplification-free electrochemical detection of exosomal miRNA-21 in serum samples. Analyst 2018, 143, 1662–1669. [Google Scholar] [CrossRef]
- Yin, X.; Hou, T.; Huang, B.; Yang, L.; Li, F. Aptamer recognition-trigged label-free homogeneous electrochemical strategy for an ultrasensitive cancer-derived exosome assay. Chem. Commun. 2019, 55, 13705–13708. [Google Scholar] [CrossRef]
- Phelps, R.M.; Johnson, B.E.; Ihde, D.C.; Gazdar, A.F.; Carbone, D.P.; McClintock, P.R.; Linnoila, R.I.; Matthews, M.J.; Bunn, P.A.; Carney, D.; et al. NCI-navy medical oncology branch cell line data base. J. Cell. Biochem. 1996, 63, 32–91. [Google Scholar] [CrossRef]
- Molazemhosseini, A.; Magagnin, L.; Vena, P.; Liu, C.-C. Single-Use Disposable Electrochemical Label-Free Immunosensor for Detection of Glycated Hemoglobin (HbA1c) Using Differential Pulse Voltammetry (DPV). Sensors 2016, 16, 1024. [Google Scholar] [CrossRef] [Green Version]
- Wei, S.-J.; Wang, L.-P.; Wang, J.-Y.; Ma, J.-X.; Chuan, F.-B.; Zhang, Y.-D. Diagnostic Value of Imaging Combined with Tumor Markers in Early Detection of Lung Cancer. Front. Surg. 2021, 8, 694210. [Google Scholar] [CrossRef]
- Roointan, A.; Mir, T.A.; Wani, S.I.; Rehman, M.U.; Hussain, K.K.; Ahmed, B.; Abrahim, S.; Savardashtaki, A.; Gandomani, G.; Gandomani, M.; et al. Early detection of lung cancer biomarkers through biosensor technology: A review. J. Pharm. Biomed. Anal. 2019, 164, 93–103. [Google Scholar] [CrossRef] [PubMed]











Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, C.; Jiang, B.; Shi, W.; Chen, D.; Zhou, M. Tri-Channel Electrochemical Immunobiosensor for Combined Detections of Multiple Exosome Biomarkers of Lung Cancer. Biosensors 2022, 12, 435. https://doi.org/10.3390/bios12070435
Fan C, Jiang B, Shi W, Chen D, Zhou M. Tri-Channel Electrochemical Immunobiosensor for Combined Detections of Multiple Exosome Biomarkers of Lung Cancer. Biosensors. 2022; 12(7):435. https://doi.org/10.3390/bios12070435
Chicago/Turabian StyleFan, Cui, Bingyan Jiang, Wenjia Shi, Dan Chen, and Mingyong Zhou. 2022. "Tri-Channel Electrochemical Immunobiosensor for Combined Detections of Multiple Exosome Biomarkers of Lung Cancer" Biosensors 12, no. 7: 435. https://doi.org/10.3390/bios12070435
APA StyleFan, C., Jiang, B., Shi, W., Chen, D., & Zhou, M. (2022). Tri-Channel Electrochemical Immunobiosensor for Combined Detections of Multiple Exosome Biomarkers of Lung Cancer. Biosensors, 12(7), 435. https://doi.org/10.3390/bios12070435





