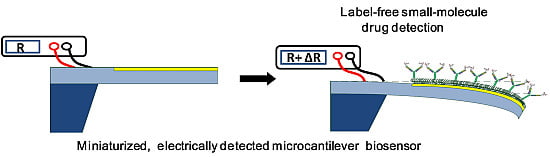
Direct Determination of a Small-Molecule Drug, Valproic Acid, by an Electrically-Detected Microcantilever Biosensor for Personalized Diagnostics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fabrication

2.2. Surface Modification
3. Results and Discussion
3.1. Detection of Valproic Acid


3.2. Drug-Antibody Binding Capability and Measured Reproducibility

3.3. Detection of Valproic Acid in Serum

3.4. Comparison Results

4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Buchapudi, K.; Huang, X.; Yang, X.; Ji, H.; Thundat, T. Microcantilever biosensors for chemicals and bioorganisms. Analyst 2011, 136, 1539–1556. [Google Scholar] [CrossRef] [PubMed]
- Fritz, J.; Baller, M.; Lang, H.; Rothuizen, H.; Vettiger, P.; Meyer, E.; Guntherodt, H.; Gerber, C.; Gimzewski, J. Translating biomolecularrecognition into nanomechanics. Science 2000, 288, 316–318. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, R.; Lorentzen, M.; Kjems, J.; Besenbacher, F. Nanomechanicalsensing of DNA sequences using piezoresistivecantilevers. Langmuir 2005, 21, 8400–8408. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Datar, R.; Hansen, K.; Thundat, T.; Cote, R.; Majumdar, A. Bioassay of prostate-specific antigen (PSA) using microcantilevers. Nat. Biotechnol. 2001, 19, 856–860. [Google Scholar] [CrossRef] [PubMed]
- Wee, K.; Kang, G.; Park, J.; Kang, J.; Yoon, D.; Park, J.; Kim, T. Novel electrical detection of label-free disease marker proteins using piezoresistive self-sensing micro-cantilevers. Biosen. Bioelectron. 2005, 20, 1932–1938. [Google Scholar] [CrossRef]
- Pei, J.; Tian, F.; Thundat, T. Glucose biosensor based on the microcantilever. Anal. Chem. 2003, 76, 292–297. [Google Scholar] [CrossRef]
- Zhang, Y.; Venkatachalan, S.; Xu, H.; Xu, X.; Joshi, P.; Ji, H.; Schulte, M. Micromechanical measurement of membrane receptor binding for label-free drug discovery. Biosen. Bioelectron. 2004, 19, 1473–1478. [Google Scholar] [CrossRef]
- Huang, L.; Pheanpanitporn, Y.; Yen, Y.; Chang, K.; Lin, L.; Lai, D. Detection of the antiepileptic drug phenytoin using a single free-standing piezoresistivemicrocantilever for therapeutic drug monitoring. Biosen. Bioelectron. 2014, 19, 233–238. [Google Scholar] [CrossRef]
- Cooper, M.; Singleton, V. A survey of the 2001 to 2005 quartz crystal microbalance biosensor literature: Applications of acoustic physics to the analysis of biomolecular interaction. J. Mol. Recognit. 2007, 20, 154–184. [Google Scholar] [CrossRef] [PubMed]
- Arce, A.; Zougagh, M.; Arce, C.; Moreno, A.; Ríos, A.; Valcárcel, M. Self-assembled monolayer-based piezoelectric flow immunosensor for the determination of canine immunoglobulin. Biosen. Bioelectron. 2007, 22, 3217–3223. [Google Scholar] [CrossRef]
- Mitchell, J. Smallmolecules immunosensingusing surface plasmonresonance. Sensors 2010, 10, 7323–7346. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Chen, C.; Lin, K.; Fang, Y.; Lieber, C. Label-free detection of small-molecule-protein interactions by using nanowire nanosensors. Proc. Natl. Acad. Sci. USA 2005, 102, 3208–3212. [Google Scholar] [CrossRef] [PubMed]
- Roopa, B.; Narayan, S.; Sharma, G.; Rodrigues, R.; Kulkarni, C. Pattern of adverse drug reactions to anti-epiletpic drugs: A cross-sectional one-year survey at a tertiary care hospital. Pharmacoepidemiol. Drug Saf. 2008, 17, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Perucca, E. Is there a role for therapeutic drug monitoring of new anticonvulsants? Clin. Pharmacokinet. 2000, 38, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Külpmann, W.; Gey, S.; Beneking, M.; Kohl, B.; Oellerich, M. Determination of total and free phenytoin in serum by non-isotopic immunoassays and gas chromatography. J. Clin. Chem. Clin. Biochem. 1984, 22, 773–779. [Google Scholar] [PubMed]
- Watari, M.; Galbraith, J.; Lang, H.; Sousa, M.; Hegner, M.; Gerber, C.; Horton, M.; McKendry, R. Investigating the molecular mechanisms of in-plane mechanochemistry on cantilever arrays. J. Am. Chem. Soc. 2006, 129, 601–609. [Google Scholar] [CrossRef]
- Ji, H.; Hansen, K.; Hu, Z.; Thundat, T. Detection of pH variation using modified microcantilever sensors. Sens. Actuator B Chem. 2001, 72, 233–238. [Google Scholar] [CrossRef]
- Yen, Y.; Lai, Y.; Hong, W.; Pheanpanitporn, Y.; Chen, C.; Huang, L. Electrical detection of C-reactive protein using a single free-standing, thermally controlled piezoresistive microcantilever for highly reproducible and accurate measurements. Sensors 2013, 13, 9653–9668. [Google Scholar] [CrossRef] [PubMed]
- Godin, M.; Tabard-Cossa, V.; Miyahara, Y.; Monga, T.; Williams, P.; Beaulieu, L.; Bruce Lennox, R.; Grutter, P. Cantilever-based sensing: The origin of surface stress and optimization strategies. Nanotechnology 2010. [Google Scholar] [CrossRef]
- Shih, Y.; Chen, C.; Wu, K. Chain length effect on surface stress of alkanethiolates adsorbed onto Au (111) surface: A van der Waals density functional study. J. Mech. 2014, 30, 241–246. [Google Scholar] [CrossRef]
- Pei, Z.; Anderson, H.; Myrskog, A.; Duner, G.; Ingemarsson, B.; Aastrup, T. Optimizing immobilization on two-dimensional carboxyl surface: pH dependence of antibody orientation and antigen binding capacity. Anal. Biochem. 2010, 398, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Yen, Y.; Huang, C.; Chen, C.; Hung, C.; Wu, K.; Lee, C.; Chang, J.; Lin, S.; Huang, L. A novel, electrically protein-manipulated microcantilever biosensor for enhancement of capture antibody immobilization. Sens. Actuator B Chem. 2009, 141, 498–505. [Google Scholar] [CrossRef]
- Manallack, D. The pKa distribution of drugs: Application to drug discovery. Perspect. Med. Chem. 2008, 1, 25–38. [Google Scholar]
- Zhao, Q.; Duan, R.; Yuan, J.; Quan, Y.; Yang, H.; Xi, M. A reusable localized surface plasmon resonance biosensor for quantitative detection of serum squamous cell carcinoma antigen in cervical cancer patients based on silver nanoparticles array. Int. J. Nanomed. 2014, 9, 1097–1104. [Google Scholar]
- Ndieyira, J.; Watari, M.; Barrera, A.; Zhou, D.; Vogtli, M.; Batchelor, M.; Cooper, M.; Strunz, T.; Horton, M.; Abell, C.; et al. Nanomechanical detection of antibiotic-mucopeptide binding in a model for superbug drug resistance. Nat. Nanotechnol. 2008, 3, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Kuan, S.; Chi, S.; Cheng, Y.; Chia, T.; Huang, L. Binding kinetics of grouper nervous necrosis viruses with functionalized antimicrobial peptides by nanomechanical detection. Biosen. Bioelectron. 2012, 31, 116–123. [Google Scholar] [CrossRef]
- Kodama, Y.; Kodama, H.; Kuranari, M.; Tsutsumi, K.; Ono, S.; Yamaguchi, T.; Fujimura, A. Protein binding of valproic acid in Japanese pediatric and adult patients with epilepsy. Am. J. Health Syst. Pharm. 2002, 59, 835–840. [Google Scholar] [PubMed]
- Gómez Bellver, M.; García Sánchez, M.; Alonso González, A.; Santos Buelga, D.; Domínguez-Gil, A. Plasma protein binding kinetics of valproic acid over a broad dosage range: Therapeutic implications. J. Clin. Pharm. Ther. 1993, 18, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, A. Usefulness of monitoring free (unbound) concentrations of therapeutic drugs in patient management. Clin. Chim. Acta 2007, 377, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Adkins, J.; Varnum, S.; Auberry, K.; Moore, R.; Angell, N.; Smith, R.; Springer, D.; Pounds, J. Toward a human blood serum proteome—Analysis by multidimensional separation coupled with mass spectrometry. Mol. Cell. Proteomics 2002, 1, 947–955. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, L.-S.; Gunawan, C.; Yen, Y.-K.; Chang, K.-F. Direct Determination of a Small-Molecule Drug, Valproic Acid, by an Electrically-Detected Microcantilever Biosensor for Personalized Diagnostics. Biosensors 2015, 5, 37-50. https://doi.org/10.3390/bios5010037
Huang L-S, Gunawan C, Yen Y-K, Chang K-F. Direct Determination of a Small-Molecule Drug, Valproic Acid, by an Electrically-Detected Microcantilever Biosensor for Personalized Diagnostics. Biosensors. 2015; 5(1):37-50. https://doi.org/10.3390/bios5010037
Chicago/Turabian StyleHuang, Long-Sun, Christian Gunawan, Yi-Kuang Yen, and Kai-Fung Chang. 2015. "Direct Determination of a Small-Molecule Drug, Valproic Acid, by an Electrically-Detected Microcantilever Biosensor for Personalized Diagnostics" Biosensors 5, no. 1: 37-50. https://doi.org/10.3390/bios5010037
APA StyleHuang, L.-S., Gunawan, C., Yen, Y.-K., & Chang, K.-F. (2015). Direct Determination of a Small-Molecule Drug, Valproic Acid, by an Electrically-Detected Microcantilever Biosensor for Personalized Diagnostics. Biosensors, 5(1), 37-50. https://doi.org/10.3390/bios5010037