1. Introduction
The rise of antibiotic-resistant bacteria is considered a crucial health threat globally [
1]. The World Health Organization (WHO) has reported annual mortality of more than half a million people from antimicrobial-resistant infections [
1]. Therefore, it is essential to develop novel approaches to overcome such a menace; one possible solution is to advance new antimicrobial drugs against multidrug-resistant (MDR) pathogens. This approach has encouraged the scientific community to engage and address the threat through various drug development methods. The process of discovering new applications for existing drugs [
2] and using computational approaches [
3], such as artificial intelligence (AI), have been utilized to find new antimicrobial agents. The latter approach generates and exploits algorithms that can link different genes and compositions by recognizing active reagents and observing their inhibition to microorganisms [
4].
A team of scientists at MIT has recently reported using artificial intelligence to repurpose a new compound with antibacterial activities against broad spectrum bacteria, including MDR [
5,
6]. The team used a machine-learning algorithm AI from the ZINC15 database, an online collection of nearly 1.5 billion chemical compounds, to discover a previously developed anti-diabetic ‘Halicin’ (formerly known as SU-3327) with remarkable antibacterial activity against MDR bacteria [
4]. Halicin is a c-Jun N-terminal protein kinase (JNK) inhibitor, which displayed unusual antibacterial mechanisms involving the disruption of the transmembrane electrochemical gradient and the upregulation of bacterial genes responsible for iron homeostasis, triggering the disturbance of the pH regulation across the bacterial cell membrane, which will halt bacterial growth. Hence, the bacteria may not be able to acquire resistance to this mechanism of action [
7]. This discovery has opened the path to using AI in repurposing drugs that the drug authorities have approved to treat various infectious diseases, especially MDR bacteria.
As a result of a recent discovery, a couple of studies have evaluated the antibacterial activity of the drug. Unfortunately, the standard guidelines reference lacks the antibacterial activity of halicin against pathogenic bacteria, making it difficult to validate its antibacterial efficacy. Here, halicin MIC was validated and determined. The results of the antibacterial activity were correlated with the disc diffusion assay against gram-positive and gram-negative bacteria, including multidrug-resistant bacterium, to construct linear regression graphs of the halicin antibacterial drug.
2. Materials and Methods
2.1. Materials
Halicin was purchased from Carbosynth (Carbosynth Ltd., Compton, UK), while Mueller-Hinton broth was bought from Scharlab (Scharlab, S.L., Barcelona, Spain). Distilled water was generated through Milli Q (Millipore Corporation, Bedford, MA, USA) and had been used throughout this study. The measurement was made with CytationTM 3 Cell Imaging Multi-Mode Reader (BioTek Instruments, Winooski, VT, USA).
2.2. Preparation of Bacterial Suspensions
The American Type Culture Collection (ATCC) bacteria were used in this study and MDR clinical isolate. Muller-Hinton broth was used to revive the bacterial strains,
Staphylococcus aureus (
S. aureus; ATCC BAA-977),
Escherichia coli (
E. coli; ATCC 25922), and
Acinetobacter baumannii (
A. baumannii; ATCC BAA-747 as a control, and 3086 as an MDR clinical isolate from the Carbapenem-resistant patients in Saudi Arabia—resistant against beta-lactams, aminoglycosides, quinolones, tetracycline, and trimethoprim/sulfonamide). Bacterial suspensions were adjusted to a turbidity equivalent to that of a 0.5 McFarland Standard and incubated overnight under aerobic conditions at 37 °C. The media were prepared according to the manufacturer Scharlab’s recommendations [
8].
2.3. Minimum Inhibitory Concentration (MIC) and Zone of Inhibition Assay
The MIC of halicin was determined by preparing a serial twofold dilution of the drug, ranging from 256 to 0.125 μg/mL, in Mueller–Hinton broth, and filtered by a 0.22 μm filter and added into 96-well plates containing bacterial suspensions. The total culture volume was 200 μL. A final bacterial inoculum of 1 × 10
6 colony-forming-unit/mL (CFU/mL) was used, and incubated overnight at 37 °C with a continuous shaking speed of 120 RPM [
8]. In addition, a similar serial dilution of halicin was prepared and stored at 4 °C for almost a week to assess the antibacterial efficacy upon reserving. The minimum concentration at which there was no bacterial growth (i.e., lack of turbidity) was considered the MIC; the plate was measured at 600 nm absorbance using CytationTM. This test was performed in independent duplicates.
The zone of inhibition assay was performed with discs impregnated with halicin solutions, at a twofold concentration range of 128 to 16 μg/mL against S. aureus (ATCC BAA-977) and E. coli (ATCC 25922), while the concentration range was 256 to 16 μg/mL against A. baumannii (ATCC BAA-747 and 3086 MDR isolate), according to the MIC results. A final concentration of 1 × 106 CFU/mL inoculum was equally distributed on the Mueller-Hinton agar surface. The diameters of the clear areas of no growth were recorded in millimeters (mm). This test was performed in independent triplicates.
2.4. Statistical Analysis
The mean, standard deviation (SD), regression equation, correlation coefficient, and (R) coefficient of determination (R2) were calculated using Microsoft Excel 2016 software (Microsoft Corporation, Redmond, MA, USA). A t-test was used to compare the results’ variance, in which p < 0.05 was considered as statistically significant.
3. Results and Discussion
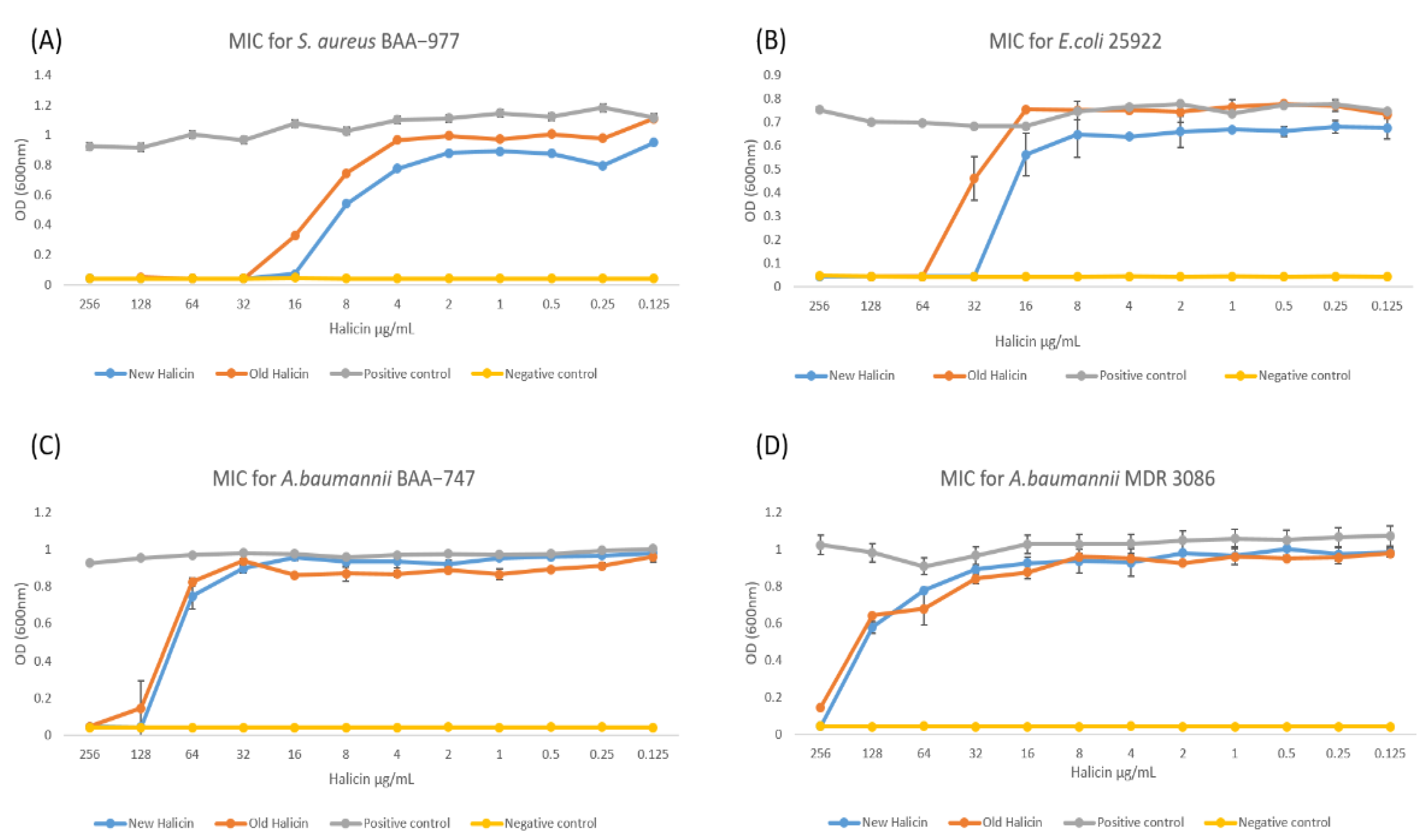
Drug repurposing is crucial in drug discovery, and it implicates comprehensive testing of drug activities pertinent to the disease occurrence. We aimed to demonstrate the antibacterial effect of halicin as an active ingredient against gram-positive and gram-negative, including MDR bacteria, by the zone of inhibition and MIC assays. These assays showed that the bacterial growth was suppressed or inhibited by the action of the tested substrate ‘halicin’. Simulating through the Clinical and Laboratory Standards Institute (CLSI) helped clarify the MIC with a sensitive inhibition zone diameter. Furthermore, these assays are characterized by their simplicity; they are fast and cost-effective compared to other microbiological tests. All of these factors are essential for the reproducibility of the data, mainly when testing different phenotypic bacteria.
Consistent with previous studies, halicin was able to inhibit the growth of gram-negative and gram-positive bacteria, including MDR strains [
8]. The MICs of halicin were measured at 16 μg/mL for
S. aureus (ATCC BAA-977), 32 μg/mL for
E. coli (ATCC 25922), 128 μg/mL for
A. baumannii (ATCC BAA-747), and 256 μg/mL for MDR
A. baumannii (MDR 3086), as shown in
Figure 1. These values were consistent with our previous study [
8]. This variation in the MIC values is due to the different bacterial susceptibilities against the tested drug. Upon storage at 4 °C, the MICs were significantly increased to be 32 μg/mL for
S. aureus, significantly increased to be 64 μg/mL for
E. coli, insignificantly changed from 128 μg/mL for
A. baumannii, and significantly increased to be ≥256 μg/mL for MDR
A. baumannii (
Figure 1). This result indicates that the efficacy of the halicin solution can be affected during its storage, as the
p < 0.05 value presented. Hence, a stability evaluation is further required.
Due to the ease of its application and its low cost, the inhibition zone test is usually used to determine the sensitivity of the microbe that affects clinical decision-making. The result can be linked either to the ATCC strain samples, or compared with the CSLI or the European Committee on Antimicrobial Susceptibility Testing (EUCAST) reference platforms. On the other hand, the MIC test is relatively expensive since a higher amount of a drug is required to be measured. There is no reference evidence for halicin effectiveness against bacteria; hence, standard strains were used in this study to estimate the MIC and determine the region of inhibition on agar plates [
9].
The inhibition zones demonstrated that halicin could inhibit all tested gram-positive and gram-negative bacterial strains, but with variable efficacy. The diameter of the zone of inhibition at a halicin concentration of 128 μg/mL was recorded as 29 mm for strains of
S. aureus BAA-977, 20 mm for
E. coli ATCC 25922, and 20 mm for
A. baumannii ATCC BAA-747. However, the MDR
A. baumannii was only sensitive at >128 μg/mL, with a diameter of 22 mm for a halicin concentration of 256 μg/mL.
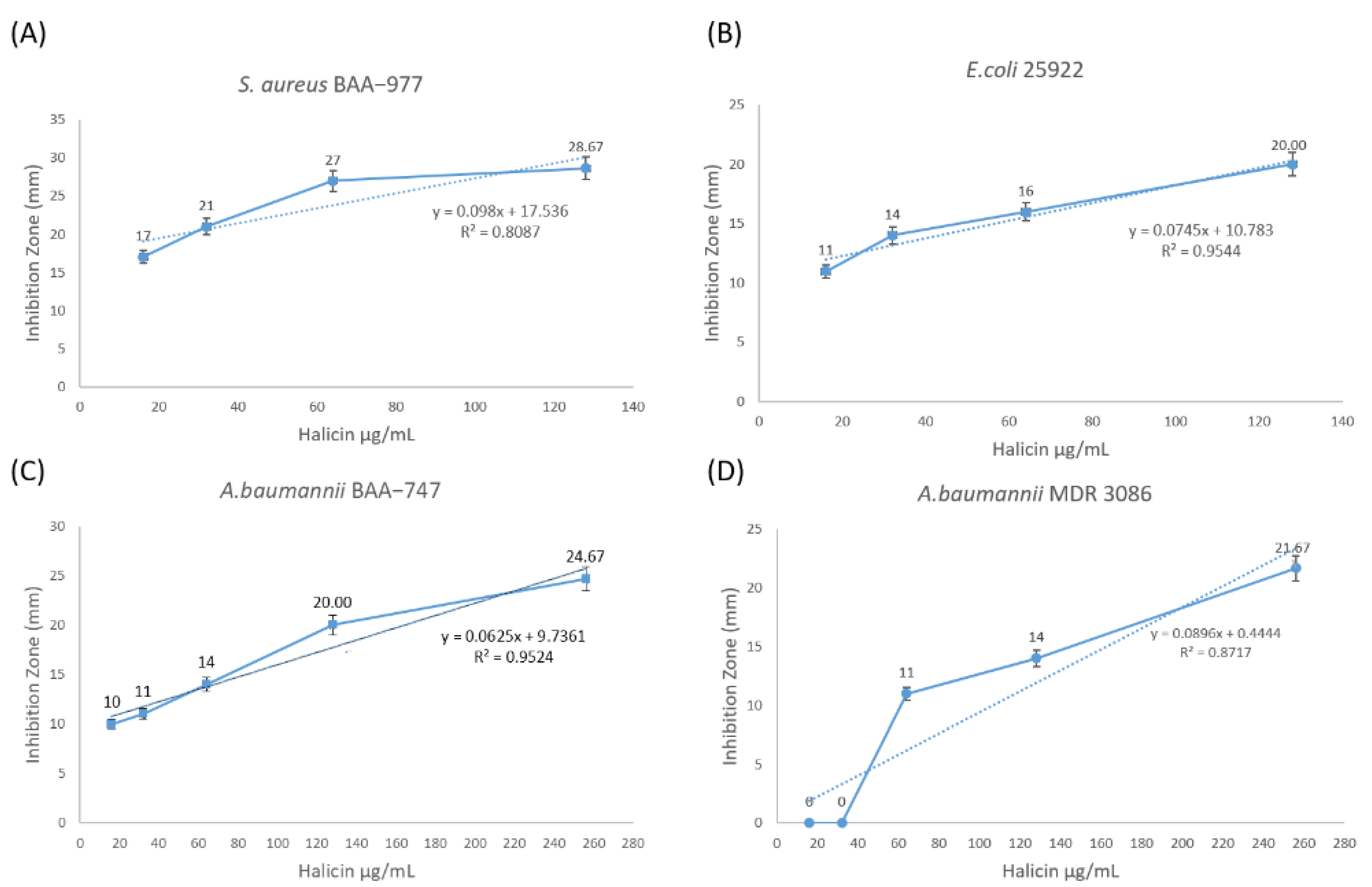
Table 1 summarizes the result of this assay. The correlation coefficient (R) for the zone of inhibition against all tested bacterial strains was determined, as shown in
Table 1, to compare the diameters of the zones with dilution values of halicin, in which a value of ≥0.9 is considered a strong correlation.
The statistical coefficient of determination (R
2) between halicin MIC and its bacterial inhibitory effect was also shown in
Figure 2 [
10]. This statistical model is based on the proportion of the total variation of outcomes explained by the model, and it showed the relation between halicin concentration and its bacterial inhibitory effect, which is considered strong (R
2 ≥ 0.95) against
E. coli ATCC 25922 and
A. baumannii ATCC BAA-747, and fairly strong (R
2 ≥ 0.8 and <0.9) against
S. aureus ATCC BAA-977 and MDR
A. baumannii MDR 3086.
Many factors might affect the reproducibility, susceptibility, and accuracy of the agar diffusion assay. Therefore, the interpretations consider all the variable factors [
11]. The correlation of results from both assays can improve the accuracy and reproducibility of the data, and compensate for the breakpoint variations in the susceptibility testing that might occur due to handling error [
12]. However, to compare between the halicin MIC and the antibacterial effect using zone of inhibition,
Table 2 shows that
S. aureus was sensitive to halicin at an inhibition diameter of ≥17 mm compared with the MIC (16 μg/mL), while
E. coli was sensitive at a diameter of ≥14 mm at MIC 32 μg/mL. The
A. baumannii ATCC strain was also sensitive at an inhibition diameter of ≥20 mm at MIC 128 μg/mL. In contrast, the MDR strain was resistant at the same concentration but sensitive at a higher concentration (265 μg/mL), which gave an inhibition diameter of >20 mm. These variable data suggest that the pharmacokinetics of the halicin drug may behave differently with sensitive and drug-resistant bacteria.