3.1. Antimicrobial Activity and Peptides’ Sequences
Since SJGAP’s C-terminal portion is strongly homologous to the AMGAP sequence, and that it has shown strong antimicrobial activity against Gram-negative bacteria, Gram-positive bacteria and the yeast
Candida albicans [
40], it has been selected as the model peptide for this study. The first thing to note is that, contrary to Seo et al. [
40], we could not demonstrate antibacterial activity associated with native SJGAP, even though we tested three of the same bacterial species (
E. coli,
P. aeruginosa, and
M. luteus). The strains are not the same, but the assumption of strain-specific inhibitory activity of SJGAP is unlikely given that AMPs generally have mechanisms of action that are not directed at a specific molecular target [
2]. In fact, it is widely accepted that most AMPs act on the cytoplasmic membrane of targeted microorganisms, causing pore formation or complete membrane destabilization, leading to loss of intracellular material and cell dysfunction [
4]. This membrane destabilization is generally not linked to specific structures that might be strain-specific and depends, rather, on the chemical interactions between peptides and major microbial membrane components (notably phospholipids), which is in contrast to the mechanism of some particular AMPs that interact with specific molecular targets (such as lipid II for nisin and mutacin), hence explaining the narrower activity spectrum of these two bacteriocins [
50].
A more likely hypothesis is related to the fact that synthetic SJGAP were used, rather than extracted and purified (HPLC) from skipjack tuna skin, to perform the antimicrobial tests. Indeed, despite efficient purification, it is possible that other compounds eluted simultaneously with SJGAP have a synergistic effect on its antimicrobial activity [
51]. Such a synergistic effect has also been proposed to explain the higher activity of purified AMGAP, compared to the synthesized one [
38]. However, our results show that the native SJGAP (analog 1) is active against a yeast (
Z. rouxii) and filamentous fungus (
Paecilomyces sp.), confirming the antifungal activity detected by Seo et al. [
40].
The microbial strains used in this study were selected to thoroughly test the spectrum of activity of the peptides and their potential for therapeutic and food applications.
E. coli ATCC 25922 is a reference strain widely used in the literature and, therefore, allows us to rigorously compare the activity of SJGAP and its analogs with other antimicrobial peptides and compounds.
P. aeruginosa ATCC 27853 and
L. ivanovii ATCC 19119 are two bacterial strains known to have pathogenic potential, as attested by the biosafety level 2, assigned by ATCC. Then, two strains of
Aeromonas salmonicida were selected, as this bacterial species represents a major threat to salmonid fish farming and stimulates the search for alternatives to the use of antibiotics, in this context [
52,
53]. Finally,
M. luteus LMA-272, as well as the three yeasts and three molds tested, are all strains that have been isolated from food products, thus allowing us to study the food application potential of the peptides tested.
Seven peptide analogs have been designed and synthesized, based on the 32-amino acid residues sequence of the native SJGAP. Peptide analogs 2, 3, and 4 were synthesized to assess whether SJGAP’s activity was directly linked to a particular region in the peptide chain. Indeed, while the AMGAP sequence is homologous to the C-terminal end of the SJGAP, it has been shown in another study that a 12-residue, C-terminal fragment of the fish antimicrobial peptide pleurocidin displayed an antimicrobial activity, comparable that of the native pleurocidin [
54]. This indicates that some fragments may possess activity equivalent to that of the native peptide from which they are derived. Results (presented in
Table 2 and
Table 3) show that none of these three fragments displayed antimicrobial activity, which is not surprising, considering the very low antimicrobial activity that could be found for the native SJGAP. Surprisingly, however, the C-terminal fragment of SJGAP (analog 4) showed evident inhibitory activity (MIC = 64 μg/mL) against the yeast
Z. rouxii, even more so than the native SJGAP (MIC = 128 μg/mL). According to this result, it would certainly be interesting to see if the AMGAP, highly homologous to this fragment, has the same activity against this yeast. Given the lack of conclusive antimicrobial activity of these three SJGAP fragments, they were not included in the membrane permeabilization experiments.
Peptide analogs 5 and 6 were designed to evaluate both the impact of negatively charged residues and the increase of the net charge on SJGAP’s activity. As a matter of fact, the importance of net charge for the antimicrobial activity of numerous peptides has been highlighted, whether they come from fish [
9,
32,
36,
37] or other sources [
55,
56]. It can also be noted that anionic residues, such as aspartic acid (D) and glutamic acid (E), are rarely present in the sequences of AMPs [
57].
Analog 7 was conceptualized, in order to investigate the impact of increasing the hydrophobicity of the hydrophobic face of the alpha helix of the SJGAP structure, as obtained by homology modeling and represented by a Schiffer-Edmundson wheel projection [
40]. This also has the direct consequence of increasing the amphipathicity of the analog. The hydrophobicity of the C-terminal end was also slightly increased with the substitution of the A
29 residue with a leucine residue. In fact, hydrophobicity and amphipathicity have been extensively linked with the inhibitory activity of several AMPs [
35,
58,
59,
60,
61].
The C-terminal amidated SJGAP (analog 8) has been synthesized because C-terminal amidation is a very common post-translational modification of AMPs, which can have a major impact on antimicrobial activity [
62]. In fact, C-terminal amidation raises both the net charge of the peptide (+1) and its hydrophobicity, because it suppresses the negative charge bore by the carboxyl group on the free C-terminal end [
63]. Besides, it has been shown that a C-terminal amidation can have a stabilizing effect on the structural conformation of peptides, which has already been correlated with their antimicrobial activity [
64].
The MIC values presented in
Table 2 and
Table 3 show that some of these analogs have considerable antimicrobial activity against several bacterial and fungal strains. These values indicate moderate antimicrobial activity [
65], but the MIC value of 16 μg/mL, shown by analogs 5 and 6 against two bacterial strains and analog 7 against the fungal strain
Paecilomyces sp., is close to the very interesting activity threshold set at 10 μg/mL by Ríos and Recio (2005) [
66]. From the above results, it can be deduced that the net charge of the analogs is strongly correlated with their antibacterial activity. The two most cationic analogs, which are analogs 5 and 6 (net charges of +6 and +8, respectively), proved to be the most active against all the tested bacterial strains. Analog 8, having a charge of +1, compared to the native SJGAP, also showed inhibitory activity against four of the five Gram-negative bacterial strains, but was inactive against the Gram-positive strains. For example, the +1 net charge, caused by the C-terminal amidation, also increased the antibacterial activity of pteroicidin-α, another AMP from fish [
64]. However, analog 7, with a net charge (+4) equal to that of native SJGAP, did not show antibacterial activity.
Several points can be made here. Bacterial membranes are well-known to contain negatively charged phospholipids, and the outer membrane of Gram-negative bacteria contains lipopolysaccharides (LPS), which are also anionic in nature. These strongly electronegative membranes, thus, allow the initial electrostatic interactions with cationic peptides, explaining why it has been observed that an increase in the cationicity of AMPs is often correlated with an increase in their inhibitory activity [
4,
9]. This is true regardless of whether the peptides have the microbial membrane as a direct target or intracellular targets, in that these peptide interactions with the membrane are prerequisites for all these mechanisms [
57]. It can also be noted that the distribution of charges on the peptide sequence can have an impact on its activity [
3,
4]. Thus, it is possible that the reduction of the anionic character of the C-terminal portion of SJGAP, by the substitution of anionic residues E
26 and D
32 with neutral alanine residues (analog 5), cationic lysine residues (analog 6), or by C-terminal amidation (analog 8), is important if this portion is critical in the initial attachment of the peptide to the membrane, which will be studied in subsequent work. Furthermore, it is known that the cationic amino acids lysine and arginine are very common in the sequences of AMPs, as they are essential for electrostatic interactions with the anionic components of microbial membranes [
57].
Regarding the superior inhibitory activity of the peptides against Gram-negative bacteria, it can first be evoked the presence of LPS on the outer membrane, which can favor the first interactions with the cationic peptides. Gram-positive bacteria, on the other hand, have a cell wall consisting of a thick layer of peptidoglycan, in which lipoteichoic acids (LTAs) are present, which are also anionic polymers. The interactions between peptidoglycan and AMPs are still relatively unclear, but it seems that it does not have a negative impact on the penetration of AMPs and their interactions with the phospholipids of the cytoplasmic membrane, given its relatively high porosity. However, LTAs could very well retain cationic peptides and affect their interactions with the cytoplasmic membrane, thus offering a possible explanation for their lower activity against Gram-positive bacteria [
67,
68].
The results obtained with the two
A. salmonicida strains also tend to support this link between the net charge of the analogs, their interactions with membranes, and their inhibitory activity. Indeed, the only difference between the 69 R3 and 69 R5 strains is the presence of the A-proteins that form the A-layer on some strains of this bacterium [
69,
70]. These A proteins are not only quite hydrophobic, but they also have an acidic isoelectric point [
69], which, therefore, translates into their anionic nature. It is, thus, possible that this more anionic surface of the 69 R3 strain of bacteria could explain the greater activity of the cationic SJGAP analogs, compared to the 69 R5 strain of bacteria, which lack this protein layer.
The impact of the positive net charge seems somewhat less direct, with respect to the antifungal activity of SJGAP analogs. Firstly, it is interesting to note that native SJGAP is weakly active against two fungal strains, including a mold (Paecilomyces sp.) and yeast (Z. rouxii), while this activity is slightly enhanced by the C-terminal amidation (analog 8), resulting in a two-fold lower MIC value for the same strains. Analogs 5, 6, and 7 showed the strongest antifungal activity, not only noticeable by the lower MIC values for the same two strains, but also by their inhibitory activity against the yeast R. mucilaginosa. The high antifungal activity of analog 7, with a net charge equal to that of native SJGAP, thus shows that the cationicity of the analogs is not the only factor to be considered in explaining their antifungal activity.
In this respect, the hydrophobicity of the peptides also seems to play an important role. Similarly to what was observed in the present study, such an increase in hydrophobicity by the substitution of residues on the hydrophobic face of the alpha helix of AMPs with more hydrophobic residues has already been shown to be favorable for their antifungal activity [
56]. The differences in composition between bacterial and fungal membranes may offer an interesting avenue for explaining this result. Indeed, even if fungal membranes contain a non-negligible proportion of anionic phospholipids (notably phosphatidylserine and phosphatidylinositol), the surface of eukaryotic cells is generally much less negatively charged than that of prokaryotes, which contain a greater proportion of anionic lipids, such as phosphatidylglycerol and cardiolipin [
4,
71]. In fact, the major phospholipids in fungal membranes, phosphatidylcholine, and phosphatidylethanolamine are zwitterionic in nature [
61]. It is also relevant to mention the presence of ergosterol in the fungal membrane, which is a hydrophobic molecule absent in bacterial membranes. It is, therefore, likely that the less anionic nature of the fungal membrane compared to bacterial membranes may favor hydrophobic interactions with analog 7, which could explain why it has inhibitory activity against fungi but not bacteria.
3.2. Secondary Structure
The antimicrobial activity of many peptides is clearly linked to their secondary structure. In fact, the spatial conformation of peptides has a direct impact on their amphipathicity: certain secondary structure patterns, notably alpha helices and beta strands, promote the amphipathic character of peptides [
4]. These structural motifs are the key to the activity of most AMPs, despite their great structural diversity [
5]. However, many AMPs are unstructured in aqueous conditions or before contact with microbial membranes [
72,
73]. In this respect, alcohols, in particular TFE, are often used to simulate the membrane environment and well-known to induce alpha helix formation, which is why this solvent is frequently used in SAR studies of AMPs [
32,
58].
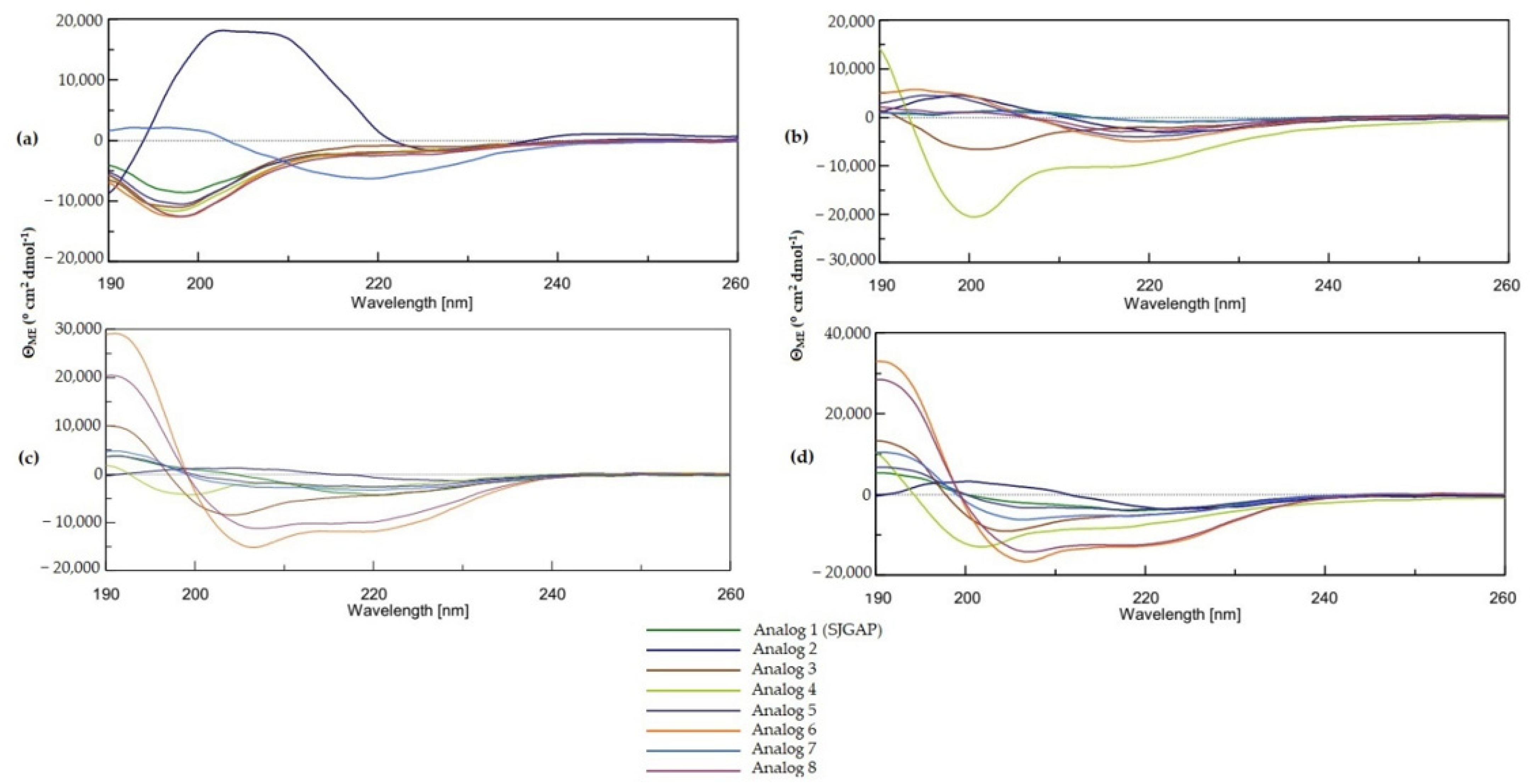
In this study, the secondary structure of SJGAP, and analogs thereof, was investigated by CD with different concentrations of TFE and by a bioinformatics prediction method. As expected, in aqueous media (PB), as well as in the presence of a low concentration of TFE (25%), all peptides showed low helicity, but the structures of analogs 6 and 8 are strongly affected by the increase in TFE concentration, which causes the formation of the central alpha helix, as predicted by PSIPRED. Indeed, it can be seen that the central alpha helix of analogs 1, 5, 6, and 7, as predicted by PSIPRED (see
Figure 2), is formed by 12 residues (11 for peptide 6), which corresponds quite precisely to the alpha helix contents of peptide 6 (41%) and peptide 8 (36%), as determined by CD in 75% TFE.
However, the same helix predicted for analogs 1, 5, and 7 was not observed with CD. This is not necessarily surprising, as PSIPRED is a multiple sequence alignment (MSA)-based method [
74], which, in the present situation, used the sequences of several GAPDHs, given the high homology between SJGAP and this enzyme. This explains why the sequences of the four analogs submitted to PSIPRED have almost exactly the same predicted secondary structures, as the homology modeling was based on the same sequence alignments of different GAPDHs. This shows how useful it is to combine different secondary structure analysis methods for the analysis of peptides that are produced by the cleavage of a protein, whose secondary structure is already well-known.
In contrast to what has been described elsewhere [
9], the present study did not detect a clear correlation between the helical structure of the peptides and their antimicrobial activity. Indeed, analog 5, which is very weakly helical, according to the CD experiments, showed antibacterial and antifungal activities similar to those of analog 6 and superior to those of analog 8, which has a much more helical structure. Similarly, analog 7 showed high antifungal activity, despite its low helicity. It should be noted, however, that the antifungal activity of peptides has been previously observed to not be as dependent on their helicity as for their antibacterial activity [
75]. Moreover, SJGAP is not a predominant alpha-helical peptide, as are other AMPs from fish, such as piscidin-1 [
76], pleurocidin [
77], or myxinidin [
32]. It is, therefore, very possible that both beta strands of SJGAP and its analogs are strongly involved in interactions with microbial membranes, thus explaining why the central alpha helix is not essential for their activity.
3.3. Membrane Permeabilization
To obtain information on the mode of action of SJGAP and its analogs, two experiments to assess their membrane permeabilization capacity were conducted. The LIVE/DEAD BacLight
TM Bacterial Viability kit consists of two fluorescent dyes, namely SYTO 9 and PI, and is commonly used to evaluate AMPs’ ability to disrupt microbial membranes [
78]. These two dyes, when bound to nucleic acids, emit a high fluorescence (green for SYTO 9 and red for PI), but do not have the same membrane permeability properties. Indeed, while SYTO 9 can penetrate intact membranes, as well as membranes with compromised integrity, PI cannot pass through intact membranes, but only through disrupted membranes. Furthermore, when both dyes cross the membrane, SYTO 9 is dislodged by PI, due to the higher affinity of the latter for nucleic acids. It is then possible to evaluate the membrane integrity of microorganisms by measuring the relative intensity of these two fluorescent dyes [
79,
80]. We also wanted to evaluate the degree of membrane permeabilization caused by the peptides by measuring whether they produce a loss of intracellular material absorbing at 260 nm, mainly DNA and RNA.
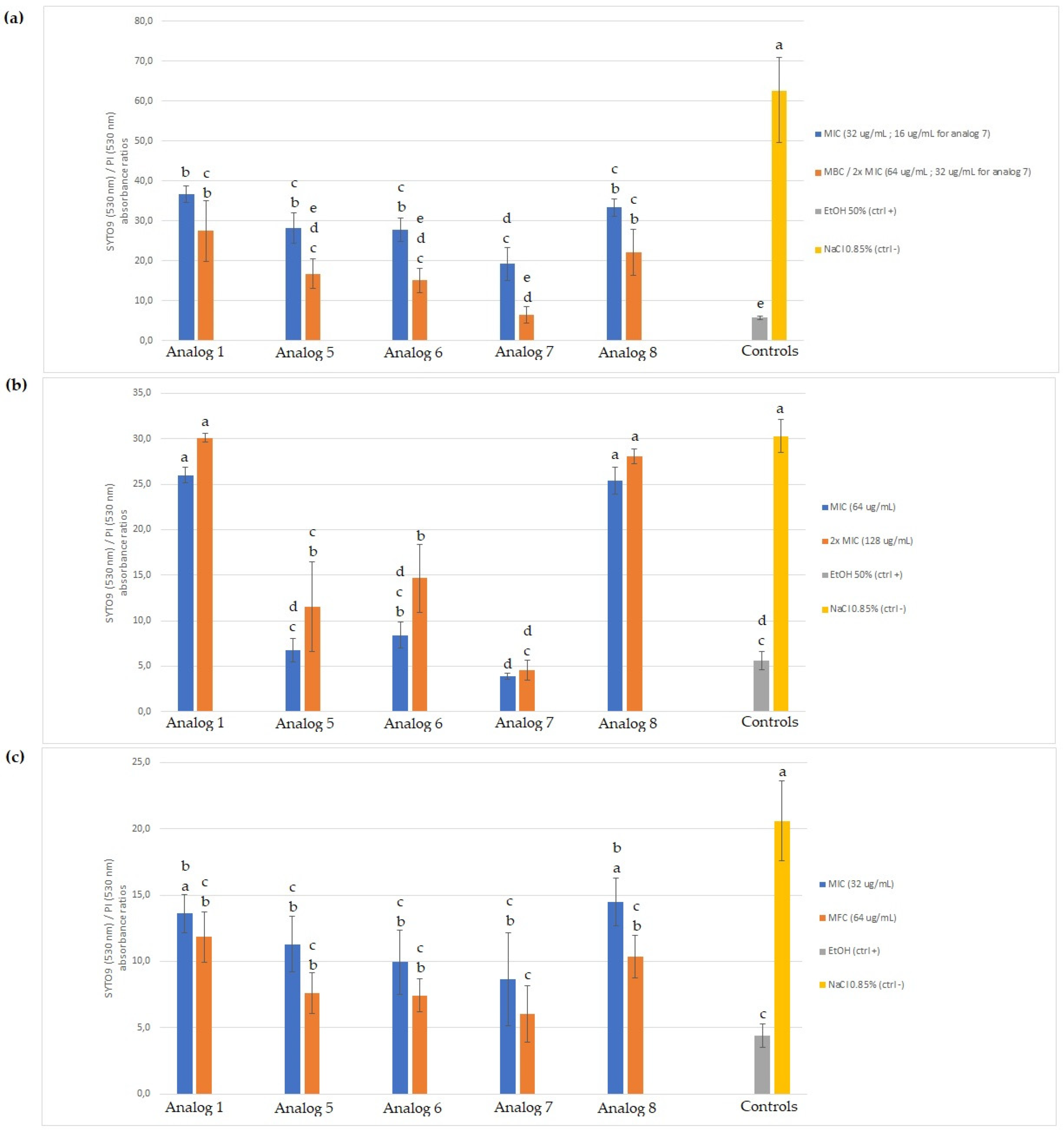
The fact that all tested peptides caused a significant level of membrane permeabilization on
E. coli, as assessed by the BacLight test, but that native SJGAP and analog 7 did not show any inhibitory activity against this microorganism, requires some consideration. Likewise, analog 7 caused significant membrane permeabilization of
M. luteus cells, without having any inhibitory activity. Firstly, it is worth noting that these results are somewhat contradictory to those obtained by Seo et al. [
40], who, using a different methodology, concluded that SJGAP did not really induce membrane permeabilization in
E. coli. Secondly, the fact that SJGAP and analog 7 showed membrane permeabilizing activity against one or both bacteria, without revealing quantifiable inhibitory activity in microplates is surprising, but some hypotheses can be put forward. It is generally accepted that PI-stained cells have their cytoplasmic membrane permeabilized and are, therefore, non-viable [
81]. However, some cases in which bacteria stained red by PI without being non-viable have been described in the literature. Indeed, it has been reported that extracellular nucleic acids can induce PI fluorescence, thus causing an underestimation of the viability of the cells studied, but this was observed in the case of biofilms [
82]. Moreover, cases of permeabilized membrane repair have been reported in both bacteria and yeast under stressful conditions, highlighting the possibility of PI staining, without signifying cell non-viability [
83,
84].
Another avenue of explanation concerns the nature of the permeabilization, induced by SJGAP and its analogs. Membrane permeabilization caused by AMPs is generally classified into three mechanisms of action: barrel-stave pores, toroidal pores (wormholes), and the carpet model. However, other alternatives are possible, in the case of non-lytic mechanisms of action, i.e., not directly based on membrane disruption. Indeed, some peptides can permeabilize the cytoplasmic membrane, without damaging, it by forming transient pores, which can then allow peptides to reach the cytoplasm and target intracellular components [
57]. In fact, some peptides are even specifically dependent on pore disintegration to cross the membrane [
3].
In this regard, it is relevant to mention that Branco et al. found that other AMPs (saccharomycin and synthetic analogs) homologous to GAPDH not only induce permeabilization of yeast cytoplasmic membranes, as evidenced by PI uptake, but that these peptides are also internalized in the cytoplasm of these yeasts [
85]. These cells also showed specific markers related to apoptosis, supporting the hypothesis of intracellular mechanisms. However, the mechanism of action of the peptides, studied by Branco et al., should not be linked too directly with those of SJGAP and its analogs, because the latter are homologous to the N-terminal portion of GAPDH, whereas the former are homologous to the C-terminal segment of this enzyme [
41]. Still, this clearly shows the possibility of membrane permeabilization that allows intracellular action of AMPs, rather than direct lethal membrane disruption. Thus, although analogs 1 and 7 can induce membrane permeabilization and possibly enter the cytoplasm of bacteria, they may not be able to interact with intracellular targets, as analogs 5, 6, and 8 do, which would explain their membrane permeabilizing activity, without showing inhibitory activity.
The latter hypothesis would also be consistent with the results obtained with leakage assays of intracellular absorbent material at 260 nm. Indeed, no significant leakage was detected for the three microorganisms, in contact with SJGAP and analogs thereof. This clearly shows that these peptides do not cause a complete disintegration of the membranes, thus showing that a carpet model mechanism of action is not possible. Such a result is, however, very compatible with the possibility of small temporary pores caused by the peptides. DNA and RNA polymers, which are large molecules, would not have the possibility to leave the cytoplasm of cells through these small, short-lived pores. These results also confirm the findings of Seo et al., who showed that SJGAP did not cause calcein to leak out of phospholipid liposomes [
40]. The hypothesis of mixed mechanisms cannot be ruled out either: it is possible that the antimicrobial activity of SJGAP analogs is due to both membrane permeabilization (leakage of ions and other small molecules) and their action on intracellular targets. Such a mixed mode of action would also allow us to account for the bactericidal (against Gram-negatives) and fungicidal action of the peptides, as well as for their bacteriostatic action against Gram-positives, because it is known that membrane mechanisms normally induce cell death, while some intracellular targets, notably DNA and RNA, are, rather, associated with a bacteriostatic action [
4,
86].
However, it should also be noted that for the Gram-positive
M. luteus, analogs 1 (native SJGAP) and 8, which caused no significant membrane permeabilization, also showed no inhibitory activity. Moreover, analogs 5 and 6, having shown strong inhibitory activity, also caused membrane permeabilization in this microorganism. The same expected relationship between membrane permeabilization and inhibitory activity can be observed for
R. mucilaginosa, but to an even greater degree: for this yeast, analogs 1 and 8, which showed only weak inhibitory activity, induced membrane permeabilization when used at their MFC, but not at their MIC, while analogs 5, 6, and 7 caused both strong membrane permeabilization (MICs and MFCs) and growth inhibition in microplates. This positive correlation between the degree of membrane permeabilization and inhibitory activity highlights the fact that peptide insertion into membranes and pore formation, transient or not, are important for antimicrobial activity, whether due to direct disrupting action on the membrane or non-lytic mechanisms targeting intracellular structures [
57].