Abstract
Antibiotic resistance in Salmonella is a global public health problem. Salmonella enterica serovar 1,4,[5],12:i:- (S. 1,4,[5],12:i:-), a monophasic variant of Salmonella Typhmurium, is one of the leading Salmonella serovars in several countries. This study aimed to assess the prevalence of antibiotic resistance to this serovar in China through a systematic review and meta-analysis. Nineteen eligible studies during 2011–2021 were included. A total of 4514 isolates from humans, animals, foods, and the environment were reported, which mainly concerned isolates found in Guangdong, Guangxi, Jiangsu, and Shanghai. A random-effects model was used to estimate the pooled resistance rate of S. 1,4,[5],12:i:-. Rates were found to be very high (values ≥ 75%) for tetracycline, ampicillin, sulfisoxazole, and streptomycin; high (50–75%) for nalidixic acid, amoxicillin–clavulanic acid, and chloramphenicol; and moderate (25–50%) for trimethoprim–sulfamethoxazole, kanamycin, trimethoprim, and gentamicin. The rates of resistance to ciprofloxacin, cefotaxime, ceftriaxone, cefepime, ceftazidime, and colistin were low (values ≤ 25%), but of great concern in terms of their current clinical importance. Furthermore, a high multidrug resistance rate (86%, 95% CI: 78–92%) was present in S. 1,4,[5],12:i:-, with the ASSuT pattern largely dominating. Subgroup analysis results showed that the high heterogeneity of resistance rates was not entirely dependent on isolated sources. Taken together, the severity of antibiotic resistance in S. 1,4,[5],12:i:- urgently requires the rational use of antibiotics in future infection control and antibiotic stewardship programs.
1. Introduction
Salmonella is an important foodborne pathogen that has been estimated to cause 115 million human infections and 370,000 deaths per year globally [1]. Salmonella has been classified into 2659 serovars based on the unique combination of their surface antigens [2]. Among them, Salmonella enterica serovar Enteritidis and serovar Typhimurium are the most common causative agents of human non-typhoidal salmonellosis [3,4]. Notably, Salmonella enterica serovar 1,4,[5],12:i:- (S. 1,4,[5],12:i:-) is a monophasic variant of Salmonella Typhimurium that lacks the fljB-encoded second-phase H antigen. Its rapid spread across the world has notably affected human health in the last two decades [5,6,7,8,9]. It has been reported to be predominant in S. 1,4,[5],12:i:- isolates of various origins, for instance in USA, Italy, and Spain [10,11,12].
Since having been confirmed as an emerging pathogen, this particular serovar is now considered one of the most frequent Salmonella serotypes responsible for human infections. It has been recognized as causing human salmonellosis, particularly in countries and regions that are associated with the prominence of the swine-related food production chain [8,13,14,15,16,17]. Several foodborne outbreaks around the world caused by this monophasic serovar have since been reported due to contamination of pigs, and consequently pork and dried pork sausage products [5,15,16,17,18]. In Europe, it is the most common serovar among human, pig, and pork isolates [19,20,21], while in the United States it is in the top five serovars associated with human salmonellosis [15,22]. The serovar was ranked as the fourth most frequent serovar responsible for human salmonellosis in Guangdong province, China, since 2009 [23], and was found to be the fifth most frequent epidemic serovar in retail meat and meat production from 2011 to 2016 in China [16].
Next to the prevalence of S. 1,4,[5],12:i:-, its resistance to particular antibiotics [24,25,26,27,28] and its multidrug resistance profile [11,29,30,31] have been reported in several countries and regions globally. The majority of its isolates showed three prominent lineages at different times and in different regions. One of its clonal lines, identified as the “Spanish clone”, was specifically associated with pigs and pork products and phenotypically showed the multidrug resistance pattern ACSuGSTTm (i.e., ampicillin, chloramphenicol, sulfonamides, gentamicin, streptomycin, tetracycline, and trimethoprim) [32]. A clonal line referred to as the “USA clone” was associated with pigs. Notably, multidrug resistance of S. 1,4,[5],12:i:- strains was rare in the USA clones, which were different from Spanish clone isolates [12]. At present, the most prominent clonal line, designated as the “European clone” prevalence in pigs, has spread in several countries across the EU, for which ampicillin, streptomycin, sulfonamides, and tetracyclines (ASSuT) is the more prominent pattern [33]. In several provinces of China, such as Guangdong, Guangxi, and Jiangsu, the drug resistance phenotype of this serovar has been explored and it has been shown to be multidrug-resistant [34,35,36].
Concerning the genetic makeup of S. 1,4,[5],12:i:-, the genes underlying the ASSuT phenotype are present on a chromosomal resistance island that frequently includes blaTEM, strA-strB, sul2, and tetB [33,37]. The fluoroquinolone resistance-conferring genes qnrS1 and aac(6′)lb-cr have been detected in MDR isolate S. 1,4,[5],12:i:- in Australia [38]. Extended-spectrum β-lactamase-encoding resistance genes (i.e., blaCTX-M-1, blaCTX-M-14) in S. 1,4,[5],12:i:- have been reported for isolates found in Germany and China [39,40], whereas plasmid-mediated colistin resistance genes, i.e., mcr-1 and mcr-3, have been reported for isolates found in Switzerland, Australia, and China [38,41,42]. The aspects of the prevalence of antibiotic-resistant phenotypes in isolates from different sources, levels of antibiotic resistance, or MDR profiles in isolates S. 1,4,[5],12:i:- have been individually studied and documented in many different countries; however, there is a paucity in the literature of combinations of the various aspects mentioned in the context of China.
Therefore, this study aimed to quantify the prevalence of antibiotic resistance to S. 1,4,[5],12:i:- from humans, animals, foods, and environment sources in China based on relevant peer-reviewed publications available in the public domain, using the meta-analysis approach. A better understanding of antibiotic resistance prevalence and rates for individual antibiotic agents, as well as of MDR features, was sought to provide concrete information for relevant risk managers and policymakers deciding on preventive and therapeutic human clinical strategies or regulatory measures concerning food and food production regarding public health safety, including the utility of antibiotics in the food chain.
2. Results
2.1. Characteristics of Eligible Studies and Datasets
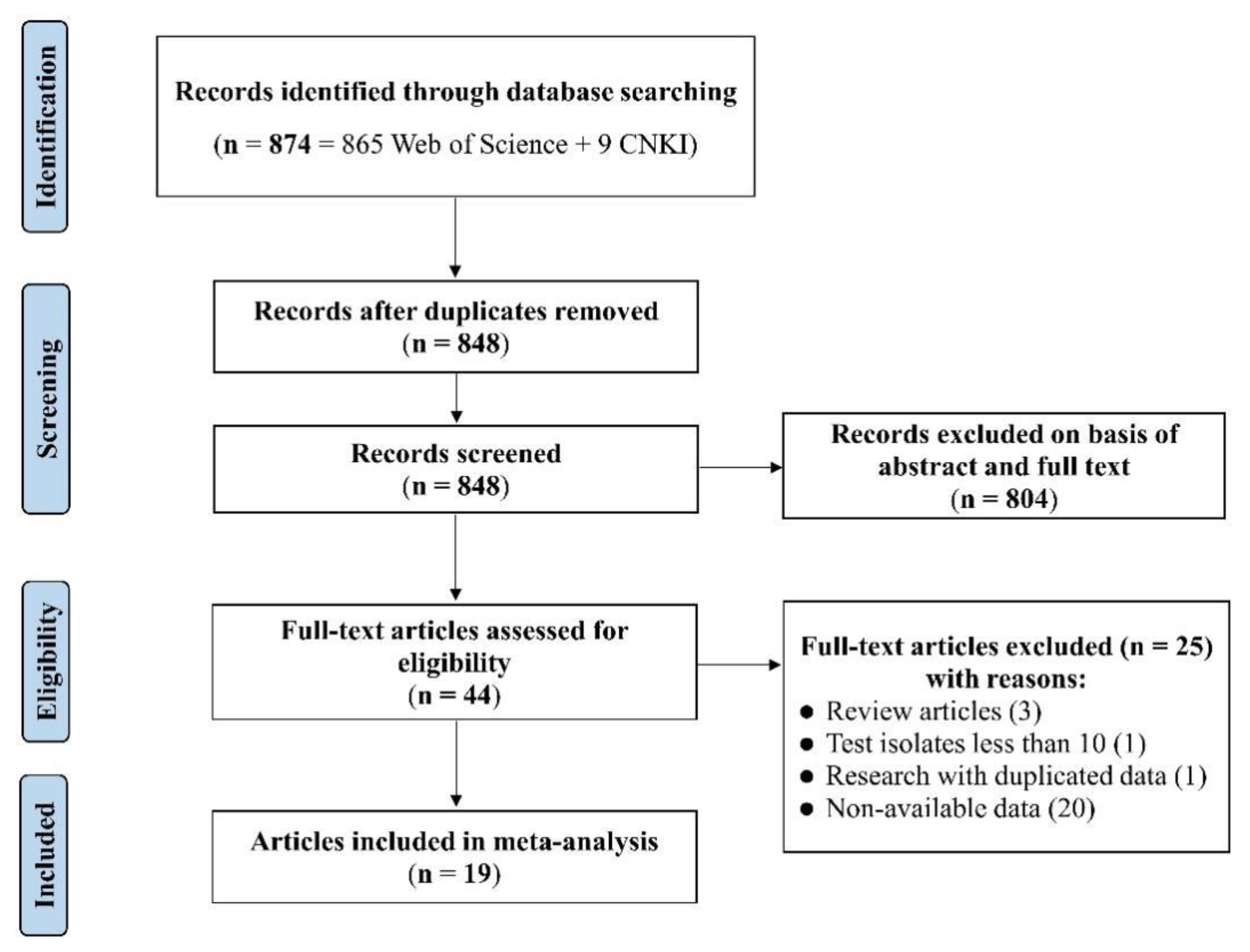
The detailed flowchart of the literature meta-analysis is shown in Figure 1. A total of 874 potentially relevant S. 1,4,[5],12:i:- publications were identified in the online databases considered based on the keywords used, from which 26 duplicated articles were removed. The remaining 848 articles were screened for relevant information on antibiotic resistance, which removed 804 articles. The remaining 44 articles were evaluated against the pre-established eligibility criteria. As a result, 25 articles were excluded due to low numbers of isolates (<10), reporting duplicated data, or non-availability data on resistance rates in the literature. Finally, 19 articles (i.e., 12 in English and 7 in Chinese) were included in the outcome of the meta-analysis, reporting on antibiotic resistance of S. 1,4,[5],12:i:- isolates from different sources in China in the period 2011–2021 (Table S1). Collectively, they are referred to as eligible studies. It should be noted that these eligible studies may not represent the insights into the antibiotic resistance traits of isolates of this serovar across all of China that a systematic and targeted investigation would have achieved, but rather provide an indication of the situation and the potential relevance for more targeted investigations and surveillance.
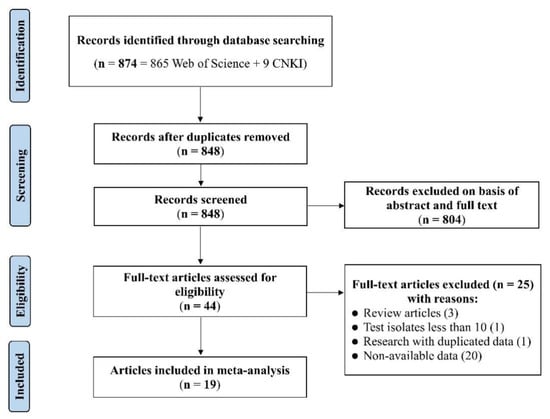
Figure 1.
PRISMA flowchart diagram for the selection process of eligible studies.
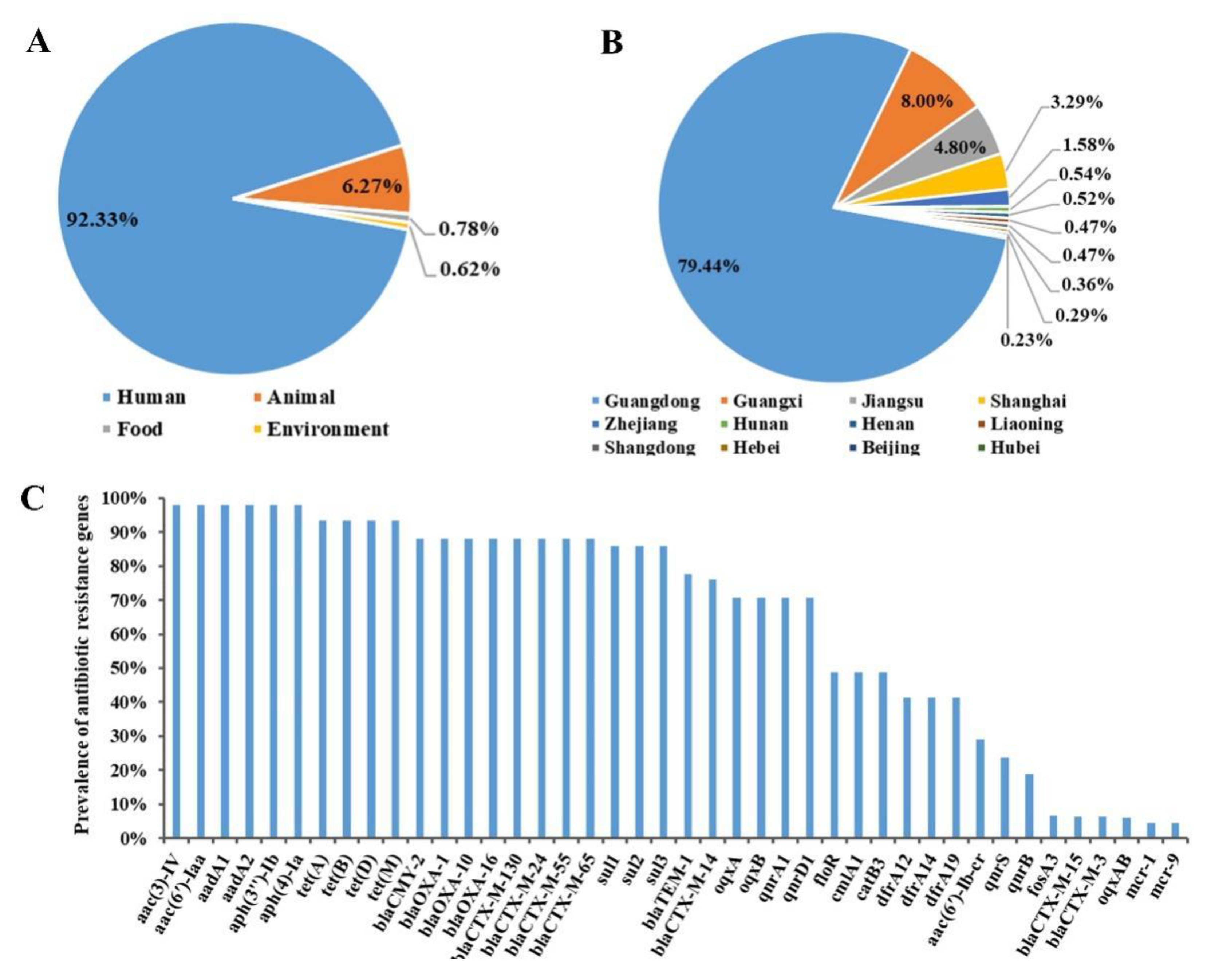
Following full-text quality checking, 186 sets of antibiotic resistance and MDR data for S. 1,4,[5],12:i:- were retrieved from the 19 eligible reports, encompassing a total of 4514 isolates derived from humans, animals, foods, and environment sources. Human-derived isolates accounted for the highest proportion retrieved from the eligible studies, followed by animal-derived isolates, then smaller numbers of isolates from foods and environment sources (Figure 2A). Of these, notably 74% of the human-derived isolates were from infants younger than 3 years old, while 98% of the animal-derived isolates were from pigs; food-derived isolates were mainly from pork and beef products, whereas all environment-derived isolates were from a single pig farm. As for the geographic origin of the isolates reported in eligible studies, these were mainly from provinces and municipalities in the eastern part of China, such as Guangdong, Guangxi, Jiangsu, Shanghai, Zhejiang, Liaoning, Shandong, Hunan, Henan, Beijing, Hebei, and Hubei (Figure 2B). Among them, by far the highest proportion of isolates was from Guangdong, followed by Guangxi, Jiangsu, and Shanghai.
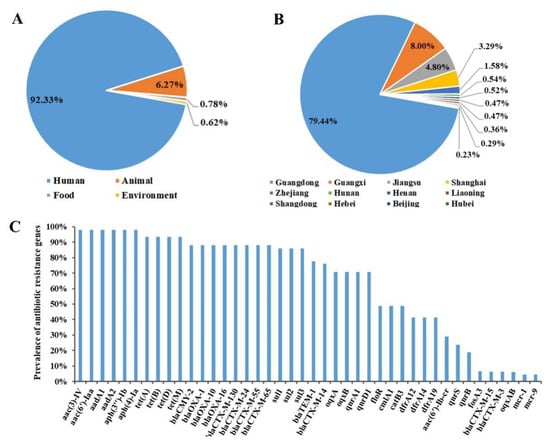
Figure 2.
Data on isolates of S. 1,4,[5],12:i:- extracted from eligible studies concerning (A) sources, (B) geographical locations in China, and (C) detection rates of antibiotic resistance genes.
Information concerning antibiotic resistance genes of the serovar was included in four eligible studies (three in English and one in Chinese). The rate of occurrence of antibiotic resistance genes was determined by dividing the number of resistant S. 1,4,[5],12:i:- isolates by the number of total S. 1,4,[5],12:i:- isolates, as presented in Figure 2C. A total of 42 genes conferring resistance against antibiotics were identified for S. 1,4,[5],12:i:- isolates reported in the eligible studies. The most prominent resistance genes were those conferring resistance against aminoglycosides (aac(3)-IV, aac(6′)-Iaa, aadA1, and aadA2), tetracyclines (tetA, tetB, tetD, and tetM), β-lactamases (blaTEM, blaCTX-M, blaOXA, and blaCMY-2), sulfonamides (sul1, sul2, sul3, dfrA1, and dfrA12), quinolones (oqxAB, qnrB, and qnrS), chloramphenicol (cmlA, catB, and floR), and colistin (mcr-1, and mcr-9).
2.2. Pooled Prevalence of Antibiotic Resistance in S. 1,4,[5],12:i:-
In all eligible studies, the antibiotic susceptibility of S. 1,4,[5],12:i:- was interpreted according to Clinical and Laboratory Standards Institute CLSI guidelines. Data on seven major types of antibiotics, including 17 individual antibiotic agents, were covered in the studies resulting from the meta-analysis, allowing us to calculate antibiotic resistance rates for β-lactams (i.e., ampicillin, amoxicillin–clavulanic acid, cefotaxime, ceftazidime, cefepime, and ceftriaxone), aminoglycosides (i.e., gentamicin, streptomycin, and kanamycin), sulfonamides (i.e., trimethoprim–sulfamethoxazole, sulfisoxazole, and trimethoprim), phenicols (i.e., chloramphenicol), polymyxin (i.e., colistin), quinolones (i.e., ciprofloxacin and nalidixic acid), and tetracyclines (i.e., tetracycline). Eight of these antibiotic agents (i.e., ampicillin, ceftazidime, gentamicin, streptomycin, trimethoprim–sulfamethoxazole, chloramphenicol, ciprofloxacin, and tetracycline) were reported in at least 70% of the eligible studies.
The meta-analysis results on the prevalence and heterogeneity of antibiotic resistance in S. 1,4,[5],12:i:- are shown in Table 1. Based on an arbitrary categorization of resistance rates into very high (≥75%), high (50–75%), moderate (25–50%), and low (≤25%), resistance rates of S. 1,4,[5],12:i:- isolates to different antibiotic agents were found to vary from very high to low as follows (with their 95% confidence interval in parentheses): tetracycline, 93% (90–95%); ampicillin, 91% (83–94%); sulfisoxazole, 86% (76–93%); streptomycin, 76% (66–83%); nalidixic acid, 73% (69–76%); amoxicillin–clavulanic acid, 70% (26–94%); chloramphenicol, 54% (47–60%); trimethoprim–sulfamethoxazole, 48% (33–63%); kanamycin, 45% (40–50%); trimethoprim, 45% (39–52%); gentamicin, 44% (29–59%); ciprofloxacin, 22% (15–32%); ceftriaxone, 21% (10–38%); cefotaxime, 19% (12–29%); cefepime, 12% (9–15%); ceftazidime, 10% (7–15%); colistin, 2% (1–5%).

Table 1.
Meta-analysis results concerning antibiotic resistance in S. 1,4,[5],12:i:- isolates in China.
A significant low heterogeneity was observed for tetracycline (46%; p < 0.1) among the eligible studies (Table 1), indicating a relatively low variance in data for this antibiotic agent. Rather high levels of heterogeneity and wide CI ranges for resistance rates derived from the literature were observed for the other antibiotic agents. For instance, as shown in Table 1, heterogeneities were significant (p < 0.1) and high for amoxicillin–clavulanic acid (96%), gentamicin (91%), ciprofloxacin (90%), cefotaxime (89%,), chloramphenicol (88%), trimethoprim–sulfamethoxazole (86%), sulfisoxazole (83%), trimethoprim (83%), streptomycin (79%), and ceftazidime (74%). A significant moderate level was derived for ampicillin (63%). There were lower, non-significant heterogeneity values regarding the antibiotic resistance of S. 1,4,[5],12:i:- to cefepime, ceftriaxone, kanamycin, and nalidixic acid, which may be due to the small number of relevant studies.
2.3. Pooled Prevalence of Multiple-Drug Resistance in S. 1,4,[5],12:i:-
The results of the meta-analysis on the prevalence and heterogeneity of S. 1,4,[5],12:i:- MDR are presented in Table 2. The prevalence of MDR in isolates of the serovar was 86% (95% CI: 78–92%) out of 2251 isolates for which resistance was reported, indicating that the majority of the S. 1,4,[5],12:i:- strains showed resistance to three or more antibiotics. Notably, the prevalence rates in S. 1,4,[5],12:i:- isolates of ASSuT, ACSSuT, and ACSuGSTTm MDR profiles were 70% (95% CI: 66–73%), 29% (95% CI: 18–43%), and 27% (95% CI: 24–31%), respectively. The heterogeneity in data across the eligible literature was high at a significant level.

Table 2.
Meta-analysis results concerning MDR rates in S. 1,4,[5],12:i:- isolates in China.
2.4. Prevalence of Antibiotic-Resistant S. 1,4,[5],12:i:- from Different Isolated Sources
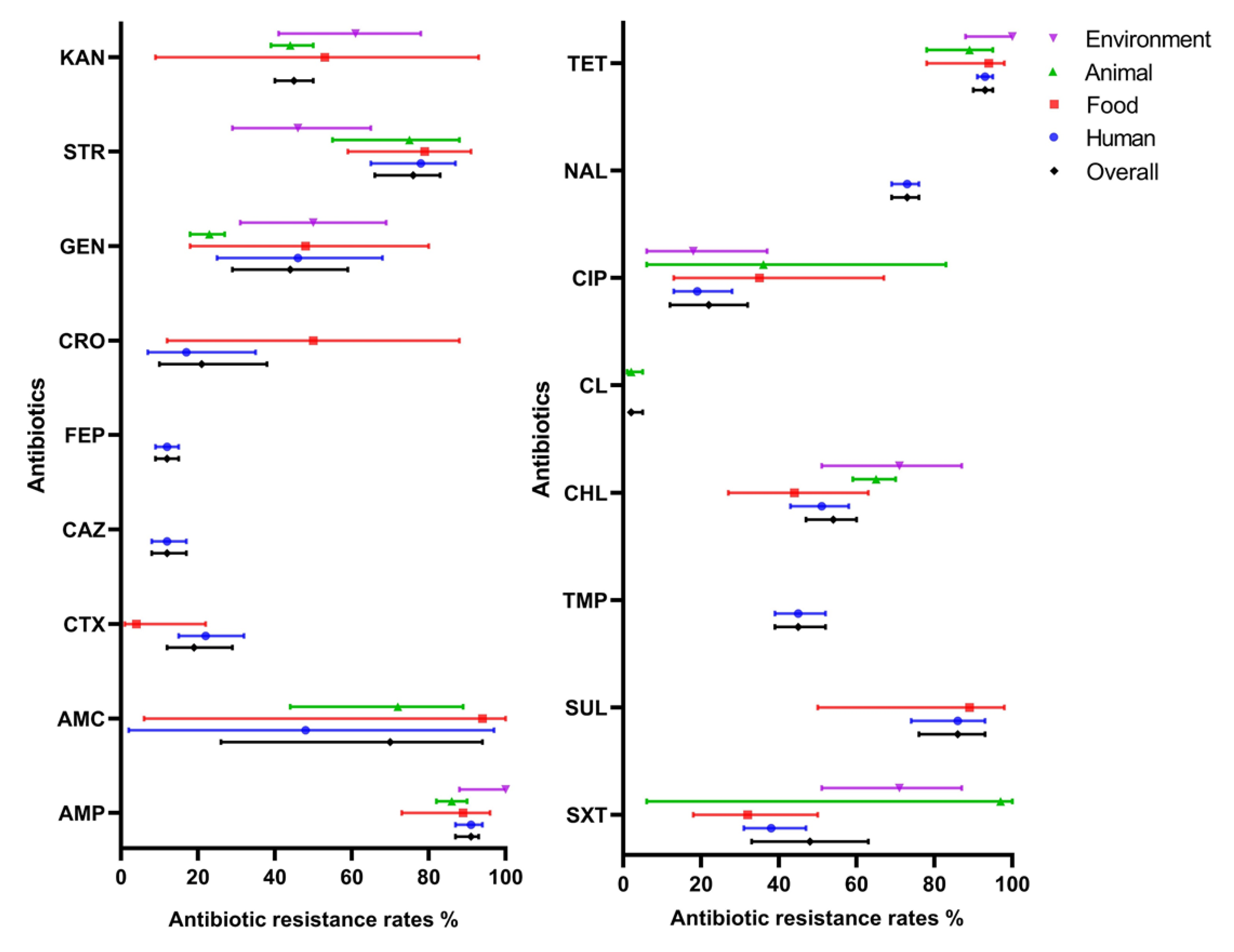
Given that there was considerable heterogeneity among the eligible studies retrieved and with an aim to analyze possible reasons for this, stratified analyses were used to assess heterogeneity across subgroups defined by the various source that isolates were obtained from humans, animals, foods, and the environment. The results are shown in Figure 3. In many cases, quite broad ranges of antibiotic resistance were deduced from the available data and there was substantial variation in occurrence across sources. For instance, some antibiotic agents were only reported from human sources (i.e., ceftazidime, cefepime, trimethoprim, and nalidixic acid), while others were not reported from animal sources (cefotaxime, ceftriaxone, and sulfisoxazole) or were reported from all sources (i.e., ampicillin, chloramphenicol, ciprofloxacin, gentamicin, streptomycin, tetracycline and trimethoprim–sulfamethoxazole). Of the agents reported from food and animal sources, relatively high resistance rates were observed in the case of ampicillin, streptomycin, sulfisoxazole, and tetracycline.
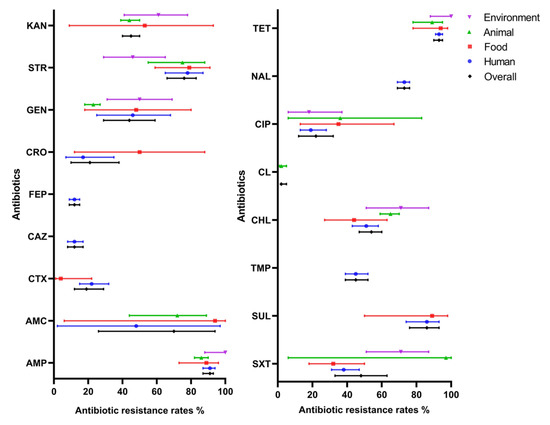
Figure 3.
Resistance rate of S. 1,4,[5],12:i:- to antibiotics by sampling sources. Antibiotics in the left picture: AMP, ampicillin; AMC, amoxicillin–clavulanic acid; CTX, cefotaxime; CAZ, ceftazidime; FEP, cefepime; CRO, ceftriaxone; GEN, gentamicin; STR, streptomycin; KAN, kanamycin. Antibiotics in the right picture: SXT, trimethoprim–sulfamethoxazole; SUL, sulfisoxazole; TMP, trimethoprim; CHL, chloramphenicol; CL, colistin; CIP, ciprofloxacin; NAL, nalidixic acid; TET, tetracycline. Lines indicate 95% confidence intervals.
2.5. Prevalence of Multiple-Resistant S. 1,4,[5],12:i:- from Different Geographical Regions in China
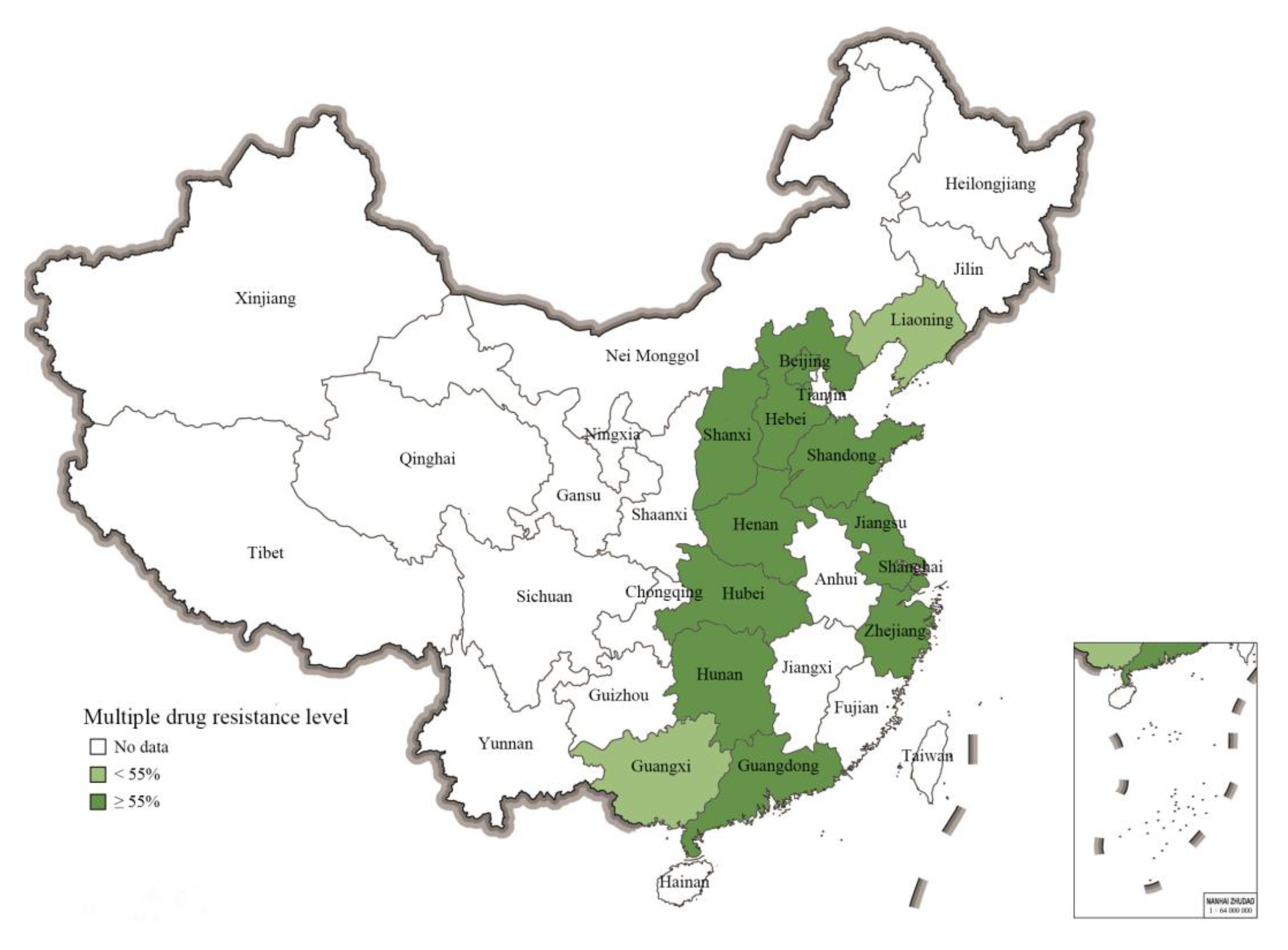
Albeit the limitations of a meta-analysis study, the MDR prevalence data on S. 1,4,[5],12:i:- covered an important number of provinces (n = 11) and municipalities (n = 2) in the eastern part of China. As shown in Figure 4, the mean pooled prevalence of MDR-level values for S. 1,4,[5],12:i:- isolates (calculated with a CI of 95% in all cases) in different regions were as follows: Shaanxi, 100% (40–100%); Hubei, 100%, (69–100%); Shandong, 96% (31–100%); Hunan, 96% (76–99%); Hebei, 94% (66–99%); Zhejiang, 93% (42–100%); Shanghai, 92% (71–98%); Jiangsu, 91% (86–94%); Henan, 91% (71–98%); Beijing, 85% (55–96%); Guangdong, 81% (73–87%); Guangxi, 54% (49–59%); Liaoning, 31% (14–57%). Notably, the actual numbers of isolates from different parts of China reported on in the eligible studies varied strongly. Consequently, the numbers of isolates displaying MDR also varied between provinces and municipalities, including Guangdong (n = 1340), Guangxi (n = 355), Hunan (n = 24), Henan (n = 23), Shandong (n = 21), Hebei (n = 16), Hubei (n = 10), and Shaanxi (n = 4).
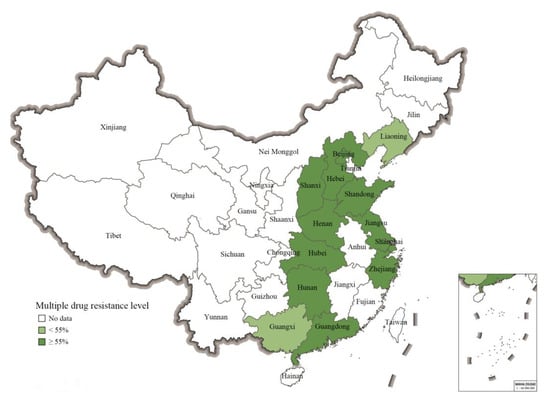
Figure 4.
The pooled prevalence rates of MDR S. 1,4,[5],12:i:- in 13 Chinese provincial regions based on the eligible studies.
3. Discussion
A meta-analysis approach was used to retrieve relevant published data concerning the prevalence and antibiotic resistance traits of isolates of S. 1,4,[5],12:i:- in China, considering that this serovar has become prevalent in Asia, Europe, Africa, North America, and South America and has been implicated in several outbreaks of salmonellosis [8,9,19,43]. It should be noted that the data retrieved by this meta-analysis study do not provide a complete and balanced picture of the S. 1,4,[5],12:i:- situation across China, such as would be obtained through systematic surveillance and targeted research. The study may rather reflect the interests of researchers in this particular serovar and their choices concerning the study scope, data generation, and region. Despite this caveat, the results of this study may still provide useful insights for government decisionmakers and others on the relevance and urgency to consider the impacts of the spread of S. 1,4,[5],12:i:- in China and the antibiotic resistance properties of isolates of this serovar.
Of the 4514 S. 1,4,[5],12:i:- isolates in China reported on in the studies retrieved from the literature, about 92% was from humans. Of these isolates from human sources, 74% were from infants and young children under 3 years old, most probably indicating that this specific serovar is predominantly prevalent in these hosts because of its high infection rate in them. High infections rate in infants and young children have been reported before [26,35]. Such a host infection rate may be quite universal. Concerning the reported prevalence of the serovar in non-human sources, about 6% of isolates were from animal sources, while the remainder was from food sources and a pig farm (less than 1% of the isolates in each case). Of the animal associated isolates, 98% was from pigs, which is in line with earlier publications noting pigs, pork, and pork products as the main vehicle for S. 1,4,[5],12:i:- [17,28,44,45]. A relatively high prevalence of the serovar in pig sources has also been reported for the USA (18.7%) and EU (48%) [19,46]. However, compared to these countries, the proportion of the serovar in non-human sources in China is relatively low, which may be due to the lack of consistent surveillance of and research on this serovar across the country, and the limitations of the detection method.
In terms of antibiotic resistance, this study identified very high or high levels of resistance of S. 1,4,[5],12:i:- isolates from various sources to tetracycline (93%), ampicillin (91%), sulfisoxazole (86%), streptomycin (76%), nalidixic acid (73%), amoxicillin–clavulanic acid (70%), and chloramphenicol (54%), which is consistent with previous results [36,47,48]. S. 1,4,[5],12:i:- resistance rates to other antibiotics (e.g., ceftriaxone, cefotaxime, kanamycin, and ciprofloxacin) in China were low to moderately high. Notably, high levels of heterogeneity were observed in the resistance rates calculated for most antibiotics, which may be related to the fact that the overwhelming amount of isolates in the eligible studies were from human sources, leaving a limited number of isolates from other sources in the pooled data for the resistance rate calculation. Nevertheless, high antibiotic resistance rates were noted for multiple key antibiotics, which may be the result of improper use or misuse of these antibiotics in human clinical treatment and animal food production. The risk is that improper use and misuse of antibiotics may select for antibiotic-resistant bacteria, since such uses can result in sublethal exposure of bacteria to antibiotics, possibly generating antibiotic-resistant bacteria. Inadequate antibiotic concentrations may occur in patients and livestock during antibiotic therapy, resulting in resistance bacteria that are widely distributed in the environment such as sewage water, rivers, lakes, and even drinking water [49]. Importantly, the increased antibiotic resistance levels of S. 1,4,[5],12:i:- isolates may contribute to further spread of the serovar, suggesting the importance of establishing targeted nation-wide surveillance to obtain a better view of the current spread of S. 1,4,[5],12:i:- and its prevalence in different sources, such that suitable risk mitigation measures can be established.
Although a limited number of different sources have been reported in the studies retrieved through the meta-analysis here, the occurrence of antibiotic-resistant S. 1,4,[5],12:i:- isolates in humans, animals, foods, and the environment suggests that resistant isolates of the serovar are probably widespread in China. Of the four antibiotic resistances noted in S. 1,4,[5],12:i:- isolates exclusively obtained from humans, rates against ceftazidime and cefepime were low (10 and 12%, respectively), while rates against nalidixic acid and trimethoprim were higher (73 and 45%, respectively). The latter antibiotics may have been used for longer or more intensively in human clinical treatment than the other antibiotic agents, leading to increased resistance rates. Extensive and imprudent usage of antimicrobials in veterinary practices and animal husbandry may lead to increased antibiotic resistance rates [50,51], and in this study high resistance rates were for instance found against ampicillin, chloramphenicol, and tetracycline, which are often used in animal treatments. In addition, differences in antibiotic resistance among isolates from different sources may be mainly related to the limited number of isolates from animals, foods, and the environment.
Previous studies have already shown that S. 1,4,[5],12:i:- isolates may display multiple drug resistance, which may be an important reason for their rapid spread since it emerged some decades ago [52,53]. Genes conferring resistance against quinolones, tetracyclines, aminoglycosides, β-lactam, and chloramphenicol are often located in transferable genetic elements, such as integrons and plasmids [54,55]. In the investigations retrieved in this study, numerous antibiotic-resistant genes located in chromosome and mobile genetic elements were detected in S. 1,4,[5],12:i:- isolates (i.e., aac(3)-IV, aac(6′)-Iaa, blaTEM, blaCTX-M, sul, tet, and mcr). Additionally, several plasmid-mediated quinolone resistance genes (i.e., oqxAB, qnrS, and qnrB) were also identified. The presence of the above-mentioned resistance genes is consistent with the antibiotic resistance phenotypes of the S. 1,4,[5],12:i:- isolates. Previous studies also reported these resistance genes in the serovar, next to others. For instance, genes encoding β-lactamase (blaTEM, blaCTX-M), aminoglycosides (aac(3)-IV, aac(6′)-Iaa, aadA1, and aadA2), tetracyclines (tetA, tetB), sulfonamides (sul, dfrA1, and dfrA12), quinolones (oqxAB, qnrB, and qnrS), chloramphenicol (cmlA, catB), and colistin (mcr-1, and mcr-3) have been observed in isolates from different sources in Germany [37], Spain [32], Portugal [56], Canada [29], and Thailand [17].
Given that MDR is a factor that may significantly contribute to the potential of S. 1,4,[5],12:i:- to spread in and across countries, it has been identified as a particular concern globally. In this study, 86% of 2251 S. 1,4,[5],12:i:- isolates were found to be multidrug-resistant strains. This rate is in the same ballpark as some MDR rates reported elsewhere, such as the 92% rate in S. 1,4,[5],12:i:- isolates from different origins and regions in Portugal [57], the 81.4% rate reported for human isolates in the EU [4], the 84.7% rate in swine isolates [36], and the 76.9% rate in food isolates [58]. For the S. 1,4,[5],12:i:- isolates from China involved in this study, three MDR patterns were noted, namely ASSuT, ACSSuT, and ACSuGSTTm, with the ASSuT pattern largely dominating in terms of prevalence (70%). In the EU this particular pattern also dominates [33], although isolates with an ACSuGSTTm pattern typically are associated with pigs and pork products in Spain [32]. The highest absolute numbers of isolates showing MDR were from Guangdong (n = 1340) and Guangxi (n = 355). The MDR rates determined for these provinces (mean values of 81% and 54%, respectively) may be more representative of the magnitude of the spread of multidrug-resistant isolates of S. 1,4,[5],12:i:- than the often higher prevalence rates derived for various other provinces and municipalities, for which only very limited numbers of isolates were available in the eligible studies retrieved. Guangdong and Guangxi are provinces with considerable levels of pig production. Considering the high prevalence of isolates of S. 1,4,[5],12:i:- in pigs and pork products and especially the potential exacerbating effect of multidrug-resistant strains, as well as the importance of pork production in China as a whole, it is of concern that systematic monitoring of this pathogen in pigs and pork products is lacking in China.
The isolates of S. 1,4,[5],12:i:- in this study were mainly from Eastern China according to the eligible studies. Except for Guangdong, Guangxi, Jiangsu, and Shanghai, the numbers of strains from other provinces are very limited, so the results of this study cannot fully reflect the overall drug resistance of S. 1,4,[5],12:i:- in China. Most of the isolates were derived from clinical patients, while the numbers of isolates from animals, foods, and the environment were relatively limited, so more surveillance data are needed to accurately reflect the antibiotic resistance of isolates from these sources.
4. Materials and Methods
4.1. Search Strategy
Two databases were systematically searched to collect potentially relevant publications, namely Web of Science and China National Knowledge Infrastructure (CNKI). The following keywords and their combinations were used in Web of Science: “Salmonella enterica Serotype 4,5,12:i:-” OR “Salmonella 4,5,12:i:-” OR “Salmonella 1, 4,[5],12:i:-” OR “Salmonella Typhimurium-like” OR “Monophasic S. Typhimurium” OR “Monophasic variants of Salmonella Typhimurium-like” OR “S. 4,12:i:-” OR “S. 4,5,12:i:-” OR “Monophasic Salmonella” OR “Salmonella 1,4,[5],12:i:-” OR “1,4,[5],12:i:-” OR “1,4,5,12:-:-” OR “1:4,[5],12:i:-”. Search terms (in Chinese) used for searching in CNKI were: “Salmonella enterica Serotype 4,5,12:i:-” OR “Salmonella 1,4,[5],12:i:-” OR “1,4,[5],12:i:-” OR “Monophasic Salmonella Typhimurium” OR “1,4,[5],12:i:- Salmonella” OR “Salmonella 4,[5],12:i:-” OR “Salmonella Typhimurium Monophasic variant” OR “Salmonella 1,4,[5],12:i:-”. Subsequently, the cited references of retrieved articles were manually searched to identify any additional relevant studies. Searches were performed between XJQ and MZY.
4.2. Selection Criteria
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement approach was employed for reporting the literature screening and retrieval process and outcomes [59]. After removing duplicate records, all publications were checked against the following set of selection criteria. A study was considered eligible if it contained information about specific antibiotic agent resistance or MDR patterns of isolates of S. 1,4,[5],12:i:-, as well as the percentage of specific antibiotic agent resistance or MDR of isolates. A study was excluded if (1) it was a duplicate report; (2) it was published as a conference abstract or as a review; (3) it was not relevant to antibiotic prevalence, resistance, or MDR levels, such as studies rather focusing on plasmids, comparative genomes, or virulence; or (4) the total number of isolates was lower than 10. In this study, only original research articles were included.
4.3. Data Extraction
The following specific data concerning various aspects of antibiotic resistance of S. 1,4,[5],12:i:- in China mentioned in articles retrieved on the basis of the selection criteria were extracted: author(s), publication year, sampling location(s), the total number of tested isolates, the total number of resistant isolates, antibiotic agents tested, antibiotic resistance levels, and MDR rates, the origin of isolates, method for drug susceptibility test, and antibiotic resistance gene information.
4.4. Meta-Analysis and Statistical Analysis
This meta-analysis was performed using R-software (meta-package, version 4.9.2; The University of Auckland). Rates of antibiotic resistance were estimated by the number of antibiotic resistance isolates divided by the total number of isolates expressed as percentages. A random-effects model was applied to analyze the pooled resistance rate of S. 1,4,[5],12:i:-. In all instances, rates were calculated using a 95% confidence interval (CI). Heterogeneity among studies was assessed using Cochran’s Q-test (significance level of p < 0.1) and the I2 statistic [60]. I2 values with percentages of 25%, 50%, and 75% were considered low, moderate, and high heterogeneity levels, respectively [61]. A subgroup analysis was carried out to explore the possible reasons for heterogeneity based on the source of isolates (i.e., humans, animals, foods, environment). All statistical tests with a Q-test value (p) < 0.05 were considered significant.
5. Conclusions
The data retrieved in this study indicate that S. 1,4,[5],12:i:- is already highly prevalent in some provinces in China, and it poses a significant risk to humans due to further spread of the serovar. Factors exacerbating the risk posed by the serovar are its resistance to antibiotics and its multidrug resistance properties. Resistance of S. 1,4,[5],12:i:- isolates to antibiotics was found to be reported as very high (i.e., tetracycline, ampicillin, sulfisoxazole, streptomycin) or high (i.e., nalidixic acid, amoxicillin–clavulanic acid, and chloramphenicol), while the resistance rate for clinically important antibiotics, such as ciprofloxacin and cefotaxime, is low at present according to this study. Furthermore, MDR may contribute to the rapid spread of the serovar, and a high level of MDR was evident in the S. 1,4,[5],12:i:- isolates from China, in which ASSuT was the most predominant MDR pattern and numerous antibiotic resistance genes were detected. More systematic, targeted surveillance and research into the prevalence rates of antibiotic resistance and MDR of S. 1,4,[5],12:i:- in humans, animals, foods, and the environment are needed to provide the necessary data on the levels of resistance to key antibiotics to inform management decisions related to the use of antibiotics. Future research should further explore the paths of the evolution and transmission of this relatively recently emerged serovar, particularly in food and food-producing environments, in order to better understand its occurrence and spread valuable information to mitigate its further spread in China and to address forthcoming emerging serotypes.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/antibiotics11040532/s1, Table S1: List and characteristics of the 19 eligible studies [13,16,30,34,35,36,40,48,58,62,63,64,65,66,67,68,69,70,71].
Author Contributions
Conceptualization, X.Q., M.Y. and H.C.; methodology, Y.L., K.J. and T.S.; validation, X.Q. and M.Y.; formal analysis, X.Q. and M.Y.; investigation, X.Q. and M.Y.; data curation, X.Q. and M.Y.; writing—original draft preparation, X.Q. and M.Y.; writing—review and editing, X.Q., L.G., M.Z.A., X.W. and Q.D.; visualization, X.Q. and M.Y.; supervision, Q.D.; project administration, Q.D.; funding acquisition, Q.D. and X.Q. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by Shanghai Agriculture Applied Technology Development Program, China (Grant No. X2021–02-08-00-12-F00782), and the Three-Year Initiative Plan for Strengthening Public Health System Construction in Shanghai (2020–2022, GWV-1.1).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data related to the research are presented in the article.
Acknowledgments
We would like to thank Taisong Fang for advice on R code and Jiaming Zheng for his help in drawing the graphics.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Seif, Y.; Kavvas, E.; Lachance, J.C.; Yurkovich, J.T.; Nuccio, S.P.; Fang, X.; Catoiu, E.; Raffatellu, M.; Palsson, B.O.; Monk, J.M. Genome-scale metabolic reconstructions of multiple Salmonella strains reveal serovar-specific metabolic traits. Nat. Commun. 2018, 9, 3771. [Google Scholar] [CrossRef] [PubMed]
- Sholpan, A.; Lamas, A.; Cepeda, A.; Franco, C.M. Salmonella spp. quorum sensing: An overview from environmental persistence to host cell invasion. AIMS Microbiol. 2021, 7, 238–256. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, R.G.; Rosario, D.K.A.; Cunha-Neto, A.; Mano, S.B.; Figueiredo, E.E.S.; Conte-Junior, C.A. Worldwide epidemiology of Salmonella serovars in animal-based foods: A meta-analysis. Appl. Environ. Microb. 2019, 85, e00591-19. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority; European Center for Disease Prevention and Control. The European Union one health 2018 zoonoses report. EFSA J. 2019, 17, 5926. [Google Scholar] [CrossRef]
- Switt, A.I.M.; Soyer, Y.; Warnick, L.D.; Wiedmann, M. Emergence, distribution, and molecular and phenotypic characteristics of Salmonella enterica serotype 4,5,12: I:-. Foodborne Pathog. Dis. 2009, 6, 407–415. [Google Scholar] [CrossRef]
- Ido, N.; Lee, K.; Iwabuchi, K.; Izumiya, H.; Uchida, I.; Kusumoto, M.; Iwata, T.; Ohnishi, M.; Akiba, M. Characteristics of Salmonella enterica Serovar 4,[5],12: I:- as a monophasic variant of serovar Typhimurium. PLoS ONE 2014, 9, e104380. [Google Scholar] [CrossRef]
- Crayford, G.; Coombes, J.L.; Humphrey, T.J.; Wigley, P. Monophasic expression of FliC by Salmonella 4,[5],12:i:- DT193 does not alter its pathogenicity during infection of porcine intestinal epithelial cells. Microbiology 2014, 160, 2507–2516. [Google Scholar] [CrossRef]
- Andreoli, G.; Merla, C.; Valle, C.D.; Corpus, F.; Morganti, M.; D’incau, M.; Colmegna, S.; Marone, P.; Fabbi, M.; Barco, L.; et al. Foodborne salmonellosis in Italy: Characterization of Salmonella enterica serovar Typhimurium and monophasic variant 4,[5],12:i- isolated from salami and human patients. J. Food Prot. 2017, 80, 632–639. [Google Scholar] [CrossRef]
- Sun, H.H.; Wan, Y.P.; Du, P.C.; Bai, L. The epidemiology of monophasic Salmonella Typhimurium. Foodborne Pathog. Dis. 2020, 17, 87–97. [Google Scholar] [CrossRef]
- Molina-lópez, R.A.; Vidal, A.; Obón, E.; Martín, M.; Darwich, L. Multidrug-resistant Salmonella enterica serovar Typhimurium monophasic variant 4,12:i:- isolated from asymptomatic wildlife in a Catalonian Wildlife Rehabilitation Center, Spain. J. Wildl. Dis. 2015, 51, 759–763. [Google Scholar] [CrossRef]
- Proroga, Y.T.R.; Mancusi, A.; Peruzy, M.F.; Carullo, M.R.; Montone, A.M.I.; Fulgione, A.; Capuano, F. Characterization of Salmonella Typhimurium and its monophasic variant 1,4,[5],12:i:- isolated from different sources. Folia. Microbiol. 2019, 64, 711–718. [Google Scholar] [CrossRef]
- Elnekave, E.; Hong, S.; Taylor, A.; Boxrud, D.; Rovira, A.; Alvarez, J. Tracing the evolutionary history of an emerging Salmonella 4,[5],12:i:- clone in the United States. Virus Evol. 2019, 5, 19–20. [Google Scholar] [CrossRef]
- Tian, Y.Q.; Gu, D.; Wang, F.; Liu, B.W.; Li, J.W.; Kang, X.L.; Meng, C.; Jiao, X.A.; Pan, Z.M. Prevalence and characteristics of Salmonella spp. from a pig farm in Shanghai, China. Foodborne Pathog. Dis. 2021, 18, 477–488. [Google Scholar] [CrossRef]
- Barco, L.; Barrucci, F.; Cortini, E.; Ramon, E.; Olsen, J.E.; Luzzi, I.; Lettini, A.A.; Ricci, A. Ascertaining the relationship between Salmonella Typhimurium and Salmonella 4,[5],12:i:- by MLVA and inferring the sources of human salmonellosis due to the two serovars in Italy. Front Microbiol. 2015, 6, 301. [Google Scholar] [CrossRef]
- Marder, E.P.; Griffin, P.M.; Cieslak, P.R.; Dunn, J.; Hurd, S.; Jervis, R.; Lathrop, S.; Muse, A.; Ryan, P.; Smith, K.; et al. Preliminary incidence and trends of infections with pathogens transmitted commonly through food—foodborne diseases active surveillance network, 10 U.S. Sites, 2006–2017. Mmwr. Morb. Mortal. Wkly. Rep. 2018, 67, 324–328. [Google Scholar] [CrossRef]
- Yang, X.J.; Wu, Q.P.; Zhang, J.M.; Huang, J.H.; Chen, L.; Wu, S.; Zeng, H.Y.; Wang, J.; Chen, M.T.; Wu, H.M.; et al. Prevalence, bacterial load, and antimicrobial resistance of Salmonella serovars isolated from retail meat and meat products in China. Front. Microbiol. 2019, 10, 2121. [Google Scholar] [CrossRef]
- Poonchareon, K.; Pulsrikarn, C.; Nuanmuang, N.; Khamai, P. Effectiveness of BOX-PCR in differentiating genetic relatedness among Salmonella enterica serotype 4,[5],12:i:- isolated from hospitalized patients and minced pork samples in northern Thailand. Int. J. Microbiol. 2019, 2019, 5086240. [Google Scholar] [CrossRef]
- Mossong, J.; Margues, P.; Raqimbeau, C.; Huberty-Krau, P.; Losch, S.; Meyer, G.; Moris, G.; Strottner, C.; Rabsch, W.; Schneider, F. Outbreaks of monophasic Salmonella enterica serovar 4,[5],12:i:- in Luxembourg, 2006. Eurosurveillance 2007, 12, 156–158. [Google Scholar] [CrossRef]
- Hauser, E.; Tietze, E.; Helmuth, R.; Junker, E.; Blank, K.; Prager, R.; Rabsch, W.; Appel, B.; Fruth, A.; Malorny, B. Pork contaminated with Salmonella enterica serovar 4,[5],12:i:-, an emerging health risk for humans. Appl. Environ. Microb. 2010, 76, 4601–4610. [Google Scholar] [CrossRef]
- Clemente, L.; Manageiro, V.; Ferreira, E.; Jonesdias, D.; Correia, I.; Themudo, P.; Albuquerque, T.; Canica, M. Occurrence of extended-spectrum β-lactamases among isolates of Salmonella enterica subsp. enterica from food-producing animals and food products, in Portugal. Int. J. Microbiol. 2013, 167, 221–228. [Google Scholar] [CrossRef]
- Fois, F.; Piras, F.; Torpdahl, M.; Mazza, R.; Consolati, S.G.; Spanu, C.; Scarano, C.; De Santis, E.P.L. Occurrence, characterization, and antimicrobial susceptibility of Salmonella enterica in slaughtered pigs in Sardinia. J. Food Sci. 2017, 82, 969–976. [Google Scholar] [CrossRef]
- Centers for Diseases Control and Prevention. National Enteric Disease Surveillance (NEDS): Salmonella Annual Report, 2016; CDC: Atlanta, GA, USA, 2018. Available online: https://www.cdc.gov/nationalsurveillance/pdfs/2016-Salmonella-report-508.pdf (accessed on 17 July 2021).
- Deng, X.L.; Ran, L.; Wu, S.Y.; Ke, B.X.; He, D.M.; Yang, X.F.; Zhang, Y.H.; Ke, C.W.; Klena, J.D.; Yan, M.Y.; et al. Laboratory-based surveillance of non-typhoidal Salmonella infections in Guangdong province, China. Foodborne Pathog. Dis. 2012, 9, 305–312. [Google Scholar] [CrossRef]
- Arguello, H.; Sørensen, G.; Carvajal, A.; Baggesen, D.L.; Rubio, P.; Pedersen, K. Characterization of the emerging Salmonella 4,[5],12:i:- in Danish animal production. Foodborne Pathog. Dis. 2014, 11, 366–372. [Google Scholar] [CrossRef]
- Alessiani, A.; Sacchini, L.; Pontieri, E.; Gavini, J.; Giannatale, E.D. Molecular typing of Salmonella enterica subspecies enterica serovar Typhimurium isolated in Abruzzo region (Italy) from 2008 to 2010. Vet. Ital. 2014, 50, 31–39. [Google Scholar] [CrossRef]
- Seixas, R.; Santos, T.R.; Machado, J.; Tavares, L.; Bernardo, F.; Semedo-Lemsaddek, T.; Oliveira, M. Phenotypic and molecular characterization of Salmonella 1,4,[5],12:i:- R-Type ASSuT isolates from humans, animals, and environment in Portugal, 2006-2011. Foodborne Pathog. Dis. 2016, 13, 633–641. [Google Scholar] [CrossRef]
- Possebon, F.S.; Casas, M.R.T.; Nero, L.A.; Yamatogi, R.S.; Araújo, J.P.; Pinto, J. Prevalence, antibiotic resistance, PFGE and MLST characterization of Salmonella in swine mesenteric lymph nodes. Prev. Vet. Med. 2020, 179, 105024. [Google Scholar] [CrossRef]
- Bone, A.; Noel, H.; Hello, S.L.; Pihier, N.; Jourdan-da Silva, N. Nationwide outbreak of Salmonella enterica serotype 4,12:i:- infections in France, linked to dried pork sausage, March-May 2010. Eurosurveillance 2010, 15, 19592. [Google Scholar] [CrossRef]
- Mulvey, M.R.; Finley, R.; Allen, V.; Ang, L.; Bekal, S.; Bailey, S.E.; Haldane, D.; Hoang, L.; Horsman, G.; Louie, M.; et al. Emergence of multidrug-resistant Salmonella enterica serotype 4,[5],12:i:- involving human cases in Canada: Results from the Canadian Integrated Program on Antibiotic Resistance Surveillance (CIPARS), 2003–2010. J. Antimicrob. Chemoth. 2013, 9, 1982–1986. [Google Scholar] [CrossRef] [PubMed]
- Qu, M.; Lv, B.; Zhang, X.; Yan, H.Q.; Huang, Y.; Qian, H.K.; Pang, B.; Jia, L.; Kan, B.; Wang, Q.Y. Prevalence and antibiotic resistance of bacterial pathogens isolated from childhood diarrhea in Beijing, China (2010–2014). Gut Pathog. 2016, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Mechesso, A.F.; Moon, D.C.; Kim, S.J.; Song, H.J.; Kang, H.Y.; Na, S.H.; Choi, J.H.; Kim, H.Y.; Yoon, S.S.; Lim, S.K. Nationwide surveillance on serotype distribution and antimicrobial resistance profiles of non-typhoidal Salmonella serovars isolated from food-producing animals in South Korea. Int. J. Food Microbiol. 2020, 355, 108893. [Google Scholar] [CrossRef] [PubMed]
- García, P.; Guerra, B.; Bances, M.; Mendoza, M.C.; Rodicio, M.R. IncA/C plasmids mediate antimicrobial resistance linked to virulence genes in the Spanish clone of the emerging Salmonella enterica serotype 4,[5],12:i:-. J. Antimicrob. Chemoth. 2011, 66, 543–549. [Google Scholar] [CrossRef]
- Hopkins, K.L.; Kirchner, M.; Guerra, B.; Granier, S.A.; Lucarelli, C.; Porrero, M.C.; Jakubczak, A.; Threlfall, E.J.; Mevius, D.J. Multiresistant Salmonella enterica serovar 4,[5],12:i:- in Europe: A new pandemic strain? Eurosurveillance 2010, 15, 19580. [Google Scholar] [CrossRef]
- Su, J.H.; Zhu, Y.H.; Ren, T.Y.; Guo, L.; Yang, G.Y.; Jiao, L.G.; Wang, J.F. Distribution and antimicrobial resistance of Salmonella isolated from pigs with diarrhea in China. Microorganisms 2018, 6, 117. [Google Scholar] [CrossRef]
- Zheng, D.Y.; Ma, K.; Du, J.L.; Zhou, Y.J.; Wu, G.L.; Qiao, X.; Wang, Y.M.; Ni, Y.L.; Fu, J.J.; Huo, X. Characterization of human origin Salmonella serovar 1,4,[5],12:i:- in Eastern China, 2014 to 2018. Foodborne Pathog. Dis. 2021, 18, 790–797. [Google Scholar] [CrossRef]
- Zeng, X.Y.; Lv, S.L.; Qu, C.; Lan, L.; Tan, D.M.; Li, X.G.; Bai, L. Serotypes, antibiotic resistance, and molecular characterization of non-typhoidal salmonella isolated from diarrheic patients in Guangxi Zhuang Autonomous Region, China, 2014-2017. Food Control. 2021, 120, 107478. [Google Scholar] [CrossRef]
- Lucarelli, C.; Dionisi, A.M.; Torpdahl, M.; Villa, L.; Graziani, C.; Hopkins, K.; Threlfall, J.; Caprioli, A.; Luzzi, I. Evidence for a second genomic island conferring multidrug resistance in a clonal group of strains of Salmonella enterica serovar Typhimurium and its monophasic variant circulating in Italy, Denmark, and the United Kingdom. J. Clin. Microbiol. 2010, 48, 2103–2109. [Google Scholar] [CrossRef]
- Arnott, A.; Wang, Q.N.; Bachmann, N.; Sadsad, R.; Biswas, C.; Sotomayor, C.; Howard, P.; Rockett, R.; Wiklendt, A.; Iredell, J.R.; et al. Multidrug-resistant Salmonella enterica 4,[5],12:i:- sequence type 34, new south wales, Australia, 2016-2017. Emerg Infect. Dis. 2018, 24, 751–753. [Google Scholar] [CrossRef]
- Rodríguez, I.; Jahn, S.; Schroeter, A.; Malorny, B.; Helmuth, R.; Guerra, B. Extended-spectrum β-lactamases in German isolates belonging to the emerging monophasic Salmonella enterica subsp. Enterica serovar Typhimurium 4,[5],12:i:- European clone. J. Antimicrob. Chemoth. 2012, 67, 505–508. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Wang, X.N.; Li, R.H.; Xue, W.C.; Zhai, R.B.; Liu, L.W.; Liu, Y.; Jin, H. Prevalence, resistance, and extended-spectrum beta-lactamases among Salmonella from patients in northeast China. J. Infect. Dev. Countr. 2017, 11, 470–478. [Google Scholar] [CrossRef]
- Carroll, L.M.; Zurfluh, K.; Jang, H.; Gopinath, G.; Nuesch-Inderbinen, M.; Poirel, L.; Nordmann, P.; Stephan, R.; Guldimann, C. First report of an mcr-1 harboring Salmonella enterica subsp. Enterica serotype 4,5,12:i:- strain isolated from blood of a patient in Switzerland. Int. J. Antimicrob. Agents 2018, 52, 740–741. [Google Scholar] [CrossRef]
- Portes, A.B.; Rodrigues, G.; Leitão, M.P.; Ferrari, R.; Junior, C.A.C.; Panzenhagen, P. Global distribution of plasmid-mediated colistin resistance mcr gene in Salmonella: A systematic review. J. Appl. Microbiol. 2022, 132, 872–889. [Google Scholar] [CrossRef]
- Naberhaus, S.A.; Krull, A.C.; Bradner, L.K.; Harmon, K.M.; Arruda, P.; Arruda, B.L.; Sahin, O.; Burrough, E.R.; Schwartz, K.J.; Kreuder, A.J. Emergence of Salmonella enterica serovar 4,[5],12:i:- as the primary serovar identified from swine clinical samples and development of a multiplex real-time PCR for improved Salmonella serovar-level identification. J. Vet. Diagn. Investig. 2019, 31, 818–827. [Google Scholar] [CrossRef]
- Kawakami, V.M.; Bottichio, L.; Angelo, K.; Linton, N.; Kissler, B.; Basler, C.; Lloyd, J.; Inouye, W.; Gonzales, E.; Rietberg, K.; et al. Outbreak of multidrug-resistant Salmonella infections linked to pork-Washington, 2015. Mmwr. Morb. Mortal. Wkly. 2016, 65, 379–381. [Google Scholar] [CrossRef]
- Xie, X.L.; Wang, Z.Y.; Zhang, K.; Li, Y.; Hu, Y.C.; Pan, Z.M.; Chen, X.; Li, Q.C.; Jiao, X.A. Pig as a reservoir of CRISPR type TST4 Salmonella enterica serovar Typhimurium monophasic variant during 2009–2017 in China. Emerg. Microbes Infec. 2020, 1, 1–4. [Google Scholar] [CrossRef]
- Bonardi, S.; Alpigiani, I.; Bruini, I.; Barilli, E.; Brindani, F.; Morganti, M.; Cavallini, P.; Bolzoni, L.; Pongolini, S. Detection of Salmonella enterica in pigs at slaughter and comparison with human isolates in Italy. Int. J. Food Microbiol. 2016, 218, 44–50. [Google Scholar] [CrossRef]
- Thai, T.H.; Hirai, T.; Lan, N.T.; Yamaguchi, R. Antibiotic resistance profiles of Salmonella serovars isolated from retail pork and chicken meat in North Vietnam. Int. J. Food Microbiol. 2012, 156, 147–151. [Google Scholar] [CrossRef]
- He, J.J.; Sun, F.; Sun, D.W.; Wang, Z.Y.; Jin, S.S.; Pan, Z.M.; Xu, Z.Z.; Chen, X.; Jiao, X.A. Multidrug resistance and prevalence of quinolone resistance genes of Salmonella enterica serotypes 4,[5],12:i:- in China. Int. J. Food Microbiol. 2020, 330, 108692. [Google Scholar] [CrossRef]
- Andersson, D.I.; Hughes, D. Microbiological effects of sublethal levels of antibiotics. Nat. Rev. Microbiol. 2014, 12, 465–478. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.L.; Caffrey, N.P.; Diego, B. Restricting the use of antibiotics in food-producing animals and its associations with antibiotic resistance in food-producing animals and human beings: A systematic review and meta-analysis. Lancet Planet. Health 2017, 1, 316–327. [Google Scholar] [CrossRef]
- Campos, J.; Mourão, J.; Peixe, L.; Antunes, P. Non-typhoidal Salmonella in the pig production chain: A comprehensive analysis of its impact on human health. Pathogens 2019, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Doyle, M.E. Multidrug-resistant pathogens in the food supply. Foodborne Pathog. Dis. 2015, 12, 261–279. [Google Scholar] [CrossRef]
- Varela, M.F.; Stephen, J.; Lekshmi, M.; Ojha, M.; Wezel, N.; Sanford, L.M.; Hernandez, A.J.; Parvathi, A.; Kumar, S.H. Bacterial resistance to antimicrobial agents. Antibiotics 2021, 10, 593. [Google Scholar] [CrossRef]
- Hao, H.; Sander, P.; Iqbal, Z.; Wang, Y.; Cheng, G.; Yuan, Z. The risk of some veterinary antimicrobial agents on public health associated with antimicrobial resistance and their molecular basis. Front. Microbiol. 2016, 7, 1–11. [Google Scholar] [CrossRef]
- Vtnair, D.; Venkitanarayanan, K.; Kollanoor Johny, A. Antibiotic-resistance Salmonella in the food supply and the potential role of antibiotic alternatives for control. Foods 2018, 7, 167. [Google Scholar] [CrossRef]
- Fernandes, L.; Centeno, M.M.; Couto, N.; Nunes, T.; Almeida, V.; Alban, L.; Pomba, C. Longitudinal characterization of monophasic Salmonella Typhimurium throughout the pig’s life cycle. Vet. Microbiol. 2016, 192, 231–237. [Google Scholar] [CrossRef]
- Mourão, J.; Machado, J.; Novais, C.; Antunes, P.; Peixe, L. Characterization of the emerging clinically-relevant multidrug-resistant Salmonella enterica serotype 4,[5],12:i:- (monophasic variant of S. Typhimurium) clones. Eur. J. Clin. Microbiol. 2014, 33, 2249–2257. [Google Scholar] [CrossRef]
- Yang, X.; Wu, Q.; Zhang, J.; Huang, J.; Guo, W.; Cai, S. Prevalence and characterization of monophasic Salmonella Serovar 1,4,[5],12:i:- of food origin in China. PLoS ONE 2015, 10, e0137967. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J. Clin. Epidemiol 2009, 62, 1006–1012. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. Brit. Med. J. 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Zheng, Z.B.; Zheng, W.; Wang, H.Q.; Yu, H.; Pan, J.C.; Chen, Q.; Liu, X.D. Characteristics and molecular subtyping of multidrug resistant Salmonella in Hangzhou. Microbiol. China 2021, 48, 536–544. (In Chinese) [Google Scholar]
- Huang, J.M.; Ke, B.X.; Li, B.S.; He, D.M.; Liu, Z.; Li, Z.C.; Deng, X.L. Molecular epidemiological characteristics and antibiotic resistance of multi-drug resistant Salmonella 1,4,[5],12:i:- in Guangdong. Dis. Surveill. 2021, 36, 501–508. (In Chinese) [Google Scholar]
- Zeng, X.Y.; Lv, S.L.; Qu, C.; Lan, L.; Tan, D.M.; Li, X.G. The drug resistance and molecular characteristics of Salmonella typhimurium 1,4,[5],12:i:- in Guangxi. Chin. J. Food Hygiene 2021, 33, 558–565. (In Chinese) [Google Scholar]
- Yuan, Z.H.; Zheng, Y.K.; Wu, C.Q.; Liu, Q.M.; Qiu, Q.L.; Yang, S.H. Serotype distribution and drug resistance analysis of 540 Salmonella spp. isolated from diarrhea cases from Zhongshan City. Chin. J. Food Hygiene 2020, 32, 134–138. (In Chinese) [Google Scholar]
- Huang, S.F.; Peng, S.P.; Xu, M.Q.; Liao, G.D. Epidemiological characteristics and drug resistance analysis of Salmonella 1,4,[5],12:i:- in Maoming City from 2017 to 2019. Chin. J. Food Hygiene 2020, 32, 681–685. (In Chinese) [Google Scholar]
- Wu, W.; Tu, L.H.; Zhang, W.X.; Zhang, X.; Chen, M. Identification and antimicrobial susceptibility of Salmonella typhimurium monophasic variant in Shanghai. Lab. Med. 2018, 33, 798–802. (In Chinese) [Google Scholar]
- Ke, B.X.; Zeng, H.H.; He, D.M.; Tan, H.L.; Li, B.S.; Liang, Y.H.; Ke, C.W. Circulation and etiological characterization of Salmonella enterica serotype in human in Guangdong province, 2007–2016. Chin. J. Epidemiol. 2018, 39, 64–66. (In Chinese) [Google Scholar]
- He, X.H.; Xu, X.B.; Li, K.; Liu, B.; Yue, T.L. Identification of Salmonella enterica Typhimurium and variants using a novel multiplex PCR assay. Food Control 2016, 65, 152–159. [Google Scholar] [CrossRef]
- Liang, Z.M.; Ke, B.X.; Deng, X.L.; Liang, J.H.; Ran, L.; Lu, L.L.; He, D.M.; Huang, Q.; Ke, C.W.; Li, Z.J.; et al. Serotypes, seasonal trends, and antibiotic resistance of non-typhoidal Salmonella from human patients in Guangdong Province, China, 2009–2012. BMC Infect. Dis. 2015, 15, 53. [Google Scholar] [CrossRef] [PubMed]
- Ke, B.X.; Sun, J.F.; He, D.M.; Li, X.C.; Liang, Z.M.; Ke, C. Serovar distribution, antimicrobial resistance profiles, and PFGE typing of Salmonella enterica strains isolated from 2007–2012 in Guangdong, China. BMC Infect. Dis. 2014, 14, 338. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).