Abstract
Background: COVID-19 patients are typically prescribed antibiotics empirically despite concerns. There is a need to evaluate antibiotic use among hospitalized COVID-19 patients during successive pandemic waves in Pakistan alongside co-infection rates. Methods: A retrospective review of patient records among five tertiary care hospitals during successive waves was conducted. Data were collected from confirmed COVID-19 patients during the first five waves. Results: 3221 patients were included. The majority were male (51.53%), residents from urban areas (56.35%) and aged >50 years (52.06%). Cough, fever and a sore throat were the clinical symptoms in 20.39%, 12.97% and 9.50% of patients, respectively. A total of 23.62% of COVID-19 patients presented with typically mild disease and 45.48% presented with moderate disease. A high prevalence of antibiotic prescribing (89.69%), averaging 1.66 antibiotics per patient despite there only being 1.14% bacterial co-infections and 3.14% secondary infections, was found. Antibiotic use significantly increased with increasing severity, elevated WBCs and CRP levels, a need for oxygen and admittance to the ICU; however, this decreased significantly after the second wave (p < 0.001). Commonly prescribed antibiotics were piperacillin plus an enzyme inhibitor (20.66%), azithromycin (17.37%) and meropenem (15.45%). Common pathogens were Staphylococcus aureus (24.19%) and Streptococcus pneumoniae (20.96%). The majority of the prescribed antibiotics (93.35%) were from the WHO’s “Watch” category. Conclusions: Excessive prescribing of antibiotics is still occurring among COVID-19 patients in Pakistan; however, rates are reducing. Urgent measures are needed for further reductions.
1. Introduction
In Pakistan, the first laboratory-confirmed case of a novel coronavirus, subsequently referred to as COVID-19, was reported on 26 February 2020. Following this, an appreciable number of positive cases were reported throughout the country in successive waves [1,2], which is similar to other countries [3,4]. The first wave of COVID-19 in Pakistan peaked on 14 June 2020, infecting more than 300,000 people [5,6]. However, due to strict lockdown activities, travel restrictions, enhanced capacity testing for COVID-19 as well as improved contact tracing and isolation, cases started to decline substantially, mirroring other countries [5,7,8,9]. The second wave of COVID-19 was declared by the Government of Pakistan on 28 October 2020, with greater infection, positivity and mortality rates [10,11]. The third COVID-19 wave impacted mainly the Punjab and Khyber Pakhtunkhwa provinces of Pakistan, with the peak of cases in this wave occurring in April 2021 [12,13]. The fourth wave of COVID-19 was declared in Pakistan in July 2021, with the highest number of positive cases reported in August 2021, and a positivity rate of 6.78% [14]. According to the official statement from the National Command and Operation Center (NCOC), Government of Pakistan, the omicron-driven fifth COVID-19 wave peaked on 23 January 2022 with 7586 positive cases reported in 24 h [15]. More than 1,520,817 confirmed cases and 30,326 deaths have been reported in Pakistan as of 21 March 2022 [16]. On 21 March 2022, in the last 24 h, 1,476,120 patients had recovered, with 361 positive cases reported [17].
While most patients with COVID-19 in Pakistan manifest with mild to moderate disease, severe cases still typically required hospitalization as well as mechanical ventilation [18,19]. Since the start of the pandemic, numerous pharmacological options have been proposed to treat hospitalized COVID-19 patients. These include antipyretics, antihistamines, anticoagulants, corticosteroids, immunomodulatory agents, hydroxychloroquine, and antivirals, including remdesivir, and ivermectin, with the most evidence for corticosteroids [20,21,22,23]. There were concerns about the safety of hydroxychloroquine with deaths reported [24,25]. Subsequent studies, including systematic reviews, failed to find clinical benefits from the use of hydroxychloroquine, lopinavir/ritonavir, remdesivir and ivermectin, which led to their removal from the World Health Organization’s (WHO) and other guidelines [26,27,28,29,30,31,32]. Antibiotics are usually not recommended prophylactically in patients with COVID-19 as this is a viral disease, and should ideally only be prescribed following confirmation of any bacterial co-infection or secondary infections [20,32]. However, a number of studies have documented appreciable prescribing of antibiotics among hospitalized COVID-19 patients, including children, despite only 1.2–14.3% of hospitalized patients being identified as having bacterial co-infections or secondary infections [33,34,35,36,37,38].
The excessive and unnecessary use of antibiotics in patients with COVID-19 is a crucial driver of antimicrobial resistance (AMR), compromising global health and food security [39,40]. As a result, there have been concerns about the worsening of AMR during the current pandemic, especially in hospital settings, which urgently need to be addressed [41,42,43,44]. The lack of infection prevention and control measures along with antimicrobial stewardship programs (ASPs), combined with the disruption of surveillance activities in hospitals during the current pandemic, could have a disastrous impact on increasing AMR, especially among low-middle income countries (LMICs), if not addressed [45,46,47]. The antimicrobial resistance collaborators (2022) calculated that 1.27 million deaths had occurred in 2019 due to bacterial AMR, with 4.95 million deaths associated with bacterial AMR globally in 2019 [48], making AMR the next pandemic if not addressed. This is because the impact of AMR will worsen unless addressed with more than 10 million people likely to die annually from AMR by 2050, along with economic losses estimated at $US100 trillion per year, which is equivalent to a 3.8% reduction in annual gross domestic product per country [49,50].
The morbidity, mortality and costs associated with AMR resulted in multiple global, regional and national activities in an attempt to improve future antibiotic use, including those from the WHO [39]. The WHO, in 2015, instigated the Global Action Plan (GAP) against AMR, highlighting the need for countries to develop their own national action plans (NAP) to tackle rising rates [51]. More recently, the WHO developed the AWaRe (Access, Watch and Reserve) classification of antibiotics as a monitoring tool for antimicrobial stewardship (AMS) activities across sectors and countries. Antibiotics in the ‘Access’ group should be used against commonly encountered infections with a lower resistance rate. Antibiotics in the ‘Watch’ group should only be used in critical conditions, as they have a greater chance of resistance development, while those in the ‘Reserve’ group should only be prescribed in multi-drug resistance cases [52,53]. The aim is to curb rising AMR rates, especially surrounding ‘Watch’ and ‘Reserve’ antibiotics [52,53,54,55], with ongoing activities across countries to monitor antibiotic prescribing using these criteria as part of the national or local quality improvement programmes [56,57,58].
Pakistan developed its national action plan against AMR in 2017. Strategic priorities included the establishment of robust antimicrobial surveillance, regulations of appropriate antimicrobial utilization and estimations of the costs of AMR [59,60]. However, there are ongoing challenges with the implementation of the NAP in Pakistan that need addressing going forward [61].
Currently, though, surveillance data on antimicrobial use among hospitalized patients, particularly among those with COVID-19 is scarce in Pakistan, with typically paper-based systems and limited resources in terms of personnel and available finances [61]. There have been a few studies, including point prevalence survey (PPS) studies, that have reported antimicrobial use in the early phases of the pandemic [37,62]. Consequently, there is a need to address this to provide future guidance, given the concerns with rising AMR rates in Pakistan. There is also a need to track antimicrobial consumption during successive waves of COVID-19 to monitor trends in their prescribing among hospitalized patients with COVID-19 [63]. In view of this, the objective of this study was to evaluate antimicrobial use among hospitalized COVID-19 patients during the first five successive waves of the pandemic in Pakistan. These findings can be used to guide future activities to reduce unnecessary antimicrobial prescribing among patients in Pakistan who are hospitalized with COVID-19. This includes potential quality improvement programmes, including the instigation of ASPs.
2. Results
2.1. Demographic Characteristics of COVID-19 Patients
A total of 3221 patients hospitalized with COVID-19 across the first five COVID-19 waves were included in the study (Table 1). The majority of patients were male (51.53%), residents from an urban area (56.35%) and belonged to the age groups >50 years (52.06%) followed by 30–50 years (32.25%). Nearly two-thirds of the COVID-19 patients (63.89%) did not have any comorbidity conditions. The frequent comorbidities that were identified and documented in patients’ records included hypertension (14.25%), diabetes mellitus (10.92%), and heart disease (4.43%).

Table 1.
Demographic characteristics of the hospitalized COVID-19 patients during different waves.
2.2. Clinical Characteristics of COVID-19 Patients
The common clinical symptoms that were identified included coughing (20.39%), fever (12.97), a sore throat (9.50%), headache (10.49%) and dyspnea (6.92%). Overall, 13.78% of surveyed patients manifested multiple symptoms (Table 2). More than one-third of COVID-19 patients (34.11%) had white blood cell (WBCs) values out of range, with abnormal X-ray findings evident in 30.12% of the surveyed patients. Less than a quarter of COVID-19 patients (24.18%) were on oxygen therapy and two-thirds of patients (66.02%) stayed in a hospital for 7 to 14 days. The majority of patients (81.83%) were admitted to the medical unit of hospitals, followed by the intensive care unit (18.16%). Most of the COVID-19 patients (45.48%) manifested moderate symptoms while 23.62% presented with mild symptoms. The vast majority of patients were discharged (96.98%) with 3.01% dying from the virus (Table 2), with similar rates across successive waves.

Table 2.
Clinical characteristics of hospitalized COVID-19 patients in different waves.
2.3. Antimicrobials Prescribed to COVID-19 Patients during Different Waves
An appreciable number of COVID-19 patients (89.69%) were prescribed antibiotics during their hospital stay, even though only a few of those prescribed antibiotics had documented bacterial co-infections (1.14%) or bacterial secondary infections (3.14%) (Table 3). Out of the total number of patients surveyed, 2889 patients hospitalized with COVID-19 were prescribed 5565 antibiotics, with an average of 1.66 ± 0.85 antibiotics per patient.

Table 3.
Detail of prescribed antimicrobials during different waves of COVID-19.
In both the first and second waves, there was a high prevalence of prescribed antibiotics at an average of 2.03 ± 1.01 and 2.07 ± 0.87 antibiotics/patient, compared with the fourth and fifth waves at an average of 1.35 ± 0.64 and 1.23 ± 0.81 antibiotics/patient, respectively. Most of the antibiotics (84.43%) were prescribed at the time of hospital admission. However, among the patients identified with a bacterial co-infection or secondary infections, antibiotics were typically prescribed after confirmation from the culture results. Overall, 36.62% of surveyed patients hospitalized with COVID-19 were prescribed one antibiotic, 34.12% two antibiotics and 29.24% prescribed three or more antibiotics. The majority (64.81%) of COVID-19 patients were prescribed antibiotics for 6–10 days while 26.63% of patients were prescribed antibiotics for between 1–5 days. Other antimicrobials prescribed included antivirals (remdesivir, 10.28%), antifungals (2.44%) and antiprotozoals (0.66%).
2.4. Association between Antibiotic Use and Demographics as Well as Clinical Characteristics
The differences in antibiotic use among patients based on their demographic and clinical characteristics are shown in Table 4. The utilization of antibiotics (average number of antibiotics per patient) was significantly higher among male patients with COVID-19 (p = 0.043) compared to females. There was no significant difference in antibiotics utilization between patients hospitalized during the first and second waves of COVID-19. However, antibiotic utilization was significantly reduced in the three later waves (p < 0.001). As expected, antibiotic prescribing increased significantly with increasing severity of COVID-19 (p < 0.001), elevated WBCs (p < 0.001) and CRP levels (p < 0.001) as well as the need for oxygen therapy (p < 0.001). Furthermore, antibiotic utilization was also significantly higher among patients initially admitted to the ICU (p < 0.001).

Table 4.
Differences in antibiotic usage patterns among selected demographic and clinical variables.
2.5. Bacterial Agents Identified as Co-Infection and Secondary Infection
Amongst patients with bacterial co-infections along with COVID-19, common pathogens included Staphylococcus aureus (42.42%), Streptococcus pneumoniae (27.27%) and Haemophilus influenzae (24.24%) (Table 5). Pseudomonas aeruginosa (24.19%), Streptococcus pneumoniae (20.96%) and Staphylococcus aureus (19.35%) were frequent among those with secondary bacterial infections.

Table 5.
Bacterial agents identified as bacterial co-infection and bacterial secondary infection.
2.6. Prescribed Antibiotics during the Different Waves according to the ATC Classification
Third-generation cephalosporins were the most frequently prescribed antibiotics in 24.1% of patients hospitalized with COVID-19 who were prescribed antibiotics. Other frequently prescribed antibiotics or groups included piperacillin plus enzyme inhibitors (20.7%), macrolides (17.37%), carbapenems (15.45%) and fluoroquinolones (14.32%) (Table 6).

Table 6.
Prescribed antibiotics during different waves of COVID-19 according to their ATC classification.
2.7. Antibiotics Prescribed during the Different Waves according to WHO AWaRe Classification
The majority of the prescribed antibiotics (93.4%) during the successive waves belonged to the ‘Watch’ category. Overall, 2.3% of prescribed antibiotics were from the ‘Access’ category while 4.3% were from the ‘Reserve’ category (Figure 1).
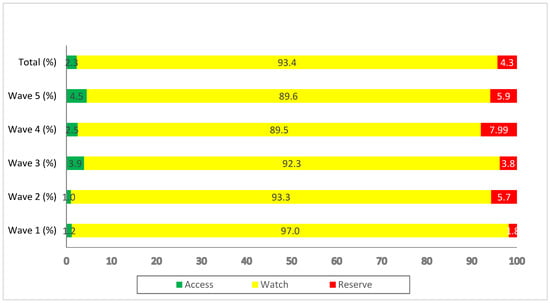
Figure 1.
Antibiotics prescribed during the different waves of COVID-19, categorized according to the WHO AWaRe classification.
3. Discussion
We believe this is one of the first studies in Pakistan, and potentially among other LMICs, to document the characteristics and management of patients hospitalized with COVID-19 among the same set of tertiary hospitals during successive waves. There have been studies documenting the profile of patients during early waves in LMICs, but not necessarily documenting their characteristics and management during successive waves [64,65,66]. The most common comorbidities reported include hypertension, diabetes mellitus, heart diseases and respiratory diseases, similar to previous studies reported in Pakistan, China, Germany, France and the USA [19,67,68,69,70].
The common symptoms of admitted patients, including coughing, fever, sore throats and headaches, or a combination of these, are also similar to previous studies from Pakistan as well as those from Bangladesh and Iran [19,71,72,73,74]. Our findings, that approximately one-third of patients hospitalized with COVID-19 had abnormal X-ray findings and WBCs counts, with 17.1% manifesting out of range of CRP levels, is similar to a study from Jordan regarding abnormal X-ray findings [75] and China regarding WBCs [76]. This contrasts with previous studies in Pakistan where there was a higher percentage of X-ray abnormalities among hospitalized patients [77,78], as well as studies from India and the USA [79,80]. In addition, in a published study in the USA, almost all hospitalized patients with COVID-19 had raised CRP levels [81]. We are not sure of the reasons behind these differences within and between these countries. This may just reflect the current situation at the time across countries; however, further research is needed before we can say anything with certainty
Our findings that less than a quarter of the total hospitalized COVID-19 patients had mild disease, whilst moderate and severe disease were reported in more than 45 and 16% of patients, respectively, contrasts with a previous study from three hospitals in Punjab where more than one-third of COVID-19 patients had severe disease [82]. This may again reflect the changing nature of the virus, especially with ongoing vaccination programmes, which have shown to be effective in reducing the extent and severity of COVID-19 [83,84,85]. However, further research is again needed in this area, including the impact of the vaccination programmes on the severity of admitted patients, before we can say anything with certainty.
Similar to a previous study conducted in the UK, more than two-thirds of patients stayed in hospitals for 7–14 days with more than 20% of patients staying for ≥ 15 days [86]. This was lower than a systematic review published in 2020 [87], which may again reflect greater knowledge in successfully treating patients with COVID-19 in recent years as well as reduced severity with the increasing availability of vaccines. Our findings that 18% of hospitalized patients with COVID-19 required an ICU stay is similar to studies from the USA [88,89]. This contrasts though with a systematic review and meta-analysis published in 2020, where approximately one-third of patients with COVID-19 were admitted to the ICU during hospitalization [90].
Encouragingly, the vast majority of patients (~97%) in our study were discharged from hospital. This contrasts with previous studies from Pakistan as well as from China, India, Iran, Poland and the USA, with lower rates [82,91,92,93,94,95,96]. These combined findings may again reflect greater knowledge in successfully treating patients with COVID-19 in successive waves.
Of concern, however, was the high prevalence (~90%) of antibiotic prescribing in our study, particularly broad-spectrum antibiotics, despite a very low prevalence of bacterial co-infections and secondary infections. This, though, is in line with previous studies from Pakistan [37,38], a study from China during the first COVID-19 wave [96], and patients with COVID-19 in an ICU in Kosovo [97]. However, studies from the USA and other countries have reported lower figures, with only two-thirds of hospitalized COVID-19 patients prescribed antibiotics [98,99,100]. Similar to studies from Bangladesh and India, more than one-third of patients in our study received one antibiotic, whilst two-thirds of patients were prescribed multiple antibiotics during their hospitalization [101,102]. This high rate of prescribing is a concern for the future, as this will continue to increase AMR rates in the country. Alongside this, piperacillin plus an enzyme inhibitor, azithromycin, meropenem, ceftriaxone and moxifloxacin were the top five frequently prescribed antibiotics in our study, similar to previous studies from Pakistan [37,38]. Other countries have also documented high prescribing rates of broad-spectrum antibiotics among patients hospitalized with COVID-19 [102,103]. In contrast, a study conducted in Scotland reported amoxicillin, doxycycline and co-amoxiclav were the top three antibiotics prescribed among patients hospitalized with COVID-19 [104]. Of equal concern, is that most of the antibiotics prescribed in our study were from the ‘Watch’ group followed by the ‘Reserve’ group’, although this was similar to studies conducted in Africa, Bangladesh and India [102,105,106].
Mirroring the findings from a study in the US, most of the antibiotics prescribed in our study were administered at admission [107]. This contrasts with a study from Scotland where only two-thirds of the antibiotics prescribed were on the first day of hospital admission [104].
The over-prescribing of antibiotics in our study, especially on admission, is emphasized by very few patients actually having bacterial co-infections or secondary bacterial infections at 1.14% and 3.14%, respectively, with Staphylococcus aureus and Streptococcus pneumoniae, similar to other studies [102,108]. Having said this, the extent of antibiotics prescribed per patient significantly decreased after the second wave. This again may reflect increasing knowledge about managing patients hospitalized with COVID-19; however, there are still concerns with the extent to which COVID-19 patients are being prescribed even one antibiotic in our study.
ASPs and other measures are urgently needed among hospitals in Pakistan to address the unnecessary antimicrobial prescribing, especially among patients admitted with COVID-19 [40,109,110,111,112]. There have been concerns with the instigation of ASPs in LMICs with resources and other issues; however, this is beginning to change [109,110,113,114,115,116]. This is important for Pakistan, given the rising AMR rates and ongoing challenges with the implementation of their NAP [59,60,61].
Consequently, a multi-sectorial engagement of clinicians, pharmacists and other key groups within hospitals, including infection, prevention and control groups, is essential to formulate hospital-based ASPs. This can start with the instigation and monitoring of guidelines among initially tertiary hospitals in the region regarding the appropriate use of antibiotics on admission based on patient diagnosis, local resistance patterns and the WHO’s AWaRe list [40,110,111,117,118,119]. Such activities should reduce inappropriate empiric prescribing of antibiotics on admission. Alongside this, there should be enhanced microbiological testing capacities, starting with tertiary hospitals. This builds on increased testing of patients during the current pandemic, with the findings documented in patients’ notes to guide future antimicrobial prescribing. In addition, measures should be instigated, including education and monitoring, to enhance the early transition from intravenous to oral therapy where appropriate, alongside their discontinuation if no bacterial infections are detected among COVID-19 patients [111]. Patient review dates, as well as start and stop dates, should also be documented in patients’ notes to assess their continued appropriateness as more microbiological data becomes available. These quality improvement initiatives can be part of future ASPs.
Alongside this, there should be repeated surveillance on antibiotic use generally among patients hospitalized with COVID-19 in the surveyed hospitals in Pakistan to document key changes, thus, building on our initial findings. We will also research key factors associated with bacterial co-infections and secondary infections in patients hospitalized with COVID-19 among the surveyed hospitals to further improve appropriate management and discourage inappropriate antimicrobial prescribing.
We are aware of a number of limitations of our study. Firstly, we only included five tertiary care hospitals within the Punjab Province of Pakistan. Secondly, we were unable to record other patient and clinical characteristics apart from those recorded in the study, since there was no proper documentation in the available medical records. However, this is similar to the drawbacks of point prevalence studies, generally. We also did not research the key factors associated with bacterial co-infections and secondary infections, as this was outside the scope of the study. However, this will be a consideration for the future. Similarly, we did not record the AMR patterns among participating hospitals prior to the pandemic, as this was outside the objectives of the study. Despite these concerns, we believe this is one of the first studies conducted in Pakistan to evaluate antibiotics use among patients hospitalized with COVID-19 during the first five waves of the pandemic with robust findings. Consequently, we believe our findings provide insight to improve future antimicrobial prescribing among clinicians, public health experts and policymakers during future waves of the pandemic.
4. Materials and Methods
4.1. Study Settings and Design
This retrospective medical record review study was conducted among the COVID-19 wards of five tertiary care/teaching hospitals in the Province of Punjab. Punjab was selected for the purpose of this study, as it is the most populous province in Pakistan [58]. Tertiary hospitals were specifically included as they are likely to provide guidance to other hospitals, including secondary and primary hospitals. The medical records of patients admitted over a period of one month during each of the five COVID-19 waves, i.e., June 2020, November 2020, April 2021, August 2021, and January 2022, were reviewed retrospectively.
4.2. Study Variables
Based on previous studies [32,52,55,58,70,71,72,78,82,108,120], data on the following variables were recorded from the individual medical records of all patients, hospitalized for COVID-19:
- Demographic characteristics include the patients’ ages, their sex, residence and presence or absence of any comorbidities, including diabetes mellitus, hypertension and other respiratory diseases. The age distribution categories, i.e., 10–30 years, 31–50 and >50 years, were based on previous studies by the co-authors.
- Clinical symptoms include a fever, cough, sore throat or headache.
- Laboratory findings, including X-rays, white blood cell counts (WBC) and C-reactive protein (CRP) were documented. The X-ray findings were reviewed by medical doctors and the treating physician was consulted in case of any confusion. Normal ranges of WBCs and CRP were taken from the reference mentioned on the testing kits.
- Whether hospitalized COVID-19 patients were on oxygen therapy or not.
- Ward subspecialty, including medical wards or intensive care units (ICU) on admission.
- Duration of hospital stay in days.
- Status of COVID-19 severity, categorized as asymptomatic, mild, moderate, severe or critical. These were categorized as per the guidelines issued by the Ministry of National Health Services, Regulation and Coordination, Government of Pakistan.
- Outcomes include whether patients were discharged from a hospital or died.
- Details about the antibiotics prescribed. This includes how many hospitalized COVID-19 patients were prescribed antibiotics during their stay in hospitals, as well as the presence of bacterial co-infection and bacterial secondary infections. Antibiotics were further classified according to the ATC classification as well as the WHO AWaRe classification.
- Bacterial co-infection was identified as those bacterial infections identified in ≤2 days after hospital admission due to COVID-19, and bacterial secondary infection as bacterial infections identified in >2 days after admission, microbiologically.
- The total number of antibiotics, the average number of antibiotics per patient, the duration of antibiotic therapy and the consumption of other antimicrobials, including antivirals, antifungal and antiprotozoal antimicrobials.
4.3. Data Collection Procedures
A team of investigators retrospectively collected the data from the COVID-19 wards of the five participating hospitals between January and February 2022. The medical records of inpatients during the months of the five waves of the COVID-19 pandemic (June 2020, November 2020, April 2021, August 2021, and January 2022) were reviewed retrospectively to obtain the required information. Data collection was facilitated and overseen by the principal investigator (ZUM) after all investigators were thoroughly trained on the data collection procedures. All data were recorded on a specifically designed data collection form. Clinical staff were only approached where clarity was needed in terms of accessing records or any anomalies in the patient records. This included any confusion regarding X-ray findings.
4.4. Inclusion and Exclusion Criteria
All patients who were admitted into the COVID-19 wards of the selected tertiary hospitals during the first five different waves of the pandemic were included in the study. Patients who were not admitted, isolated at home, or admitted at a time other than previously mentioned were excluded from the study.
4.5. Statistical Analysis
All data were entered in Microsoft Excel and, after the appropriate coding, were imported into SPSS, version 22, for analysis. Continuous data were expressed as means and standard deviations, whereas categorical data were presented as frequency and percentages. The Student’s t-test was used to evaluate the difference between the two groups, whereas one-way ANOVA was performed for ≥3 groups. To determine the significance between the intergroup variables, Tukey’s honestly significant difference test and Games–Howell post hoc tests were conducted, where applicable. An alpha (p) value of less than 0.05 was considered statistically significant.
4.6. Ethical Considerations
Ethical approval for the study was obtained from the Human Research Ethics Committee, Department of Pharmacy Practice, The University of Lahore (REC/DPP/FOP/46). Permission to conduct the study in the different hospitals was obtained from the administrators of each hospital prior to data collection.
Patients were not approached to provide informed consent, since this was a retrospective study based on data collected from patients’ medical records with no direct contact with them. This is in line with previous PPS studies undertaken by the co-authors [36,58,121,122,123,124,125].
No personal patient information was collected, and all patient data was kept confidential. All patients had an anonymized study identifier, which was kept confidential and separate from the data, for the purpose of verifying the accuracy of recorded data, where concerns were identified during data cleaning.
5. Conclusions and Next Steps
There was excessive prescribing of antibiotics among COVID-19 patients admitted to tertiary hospitals in Pakistan during the first five waves of COVID-19. This was despite a very low prevalence of bacterial co-infections and secondary infections. Whilst the average number of antibiotics prescribed per patient decreased in later waves, there were still high rates of inappropriate prescribing, which needs to be addressed moving forward. Future activities include the instigation of institutional guidelines with the help of key stakeholder groups alongside monitoring their adherence. This can be achieved with the instigation of ASPs, starting in tertiary hospitals where these do not currently exist. Key targets for ASPs also include encouraging greater prescribing of the WHO ‘Access’ group of antibiotics as well as encouraging greater documentation of start and stop dates for antibiotic prescribing in patients’ notes alongside their rationale. Encouraging a de-escalation from IV to oral antibiotics where appropriate is another key target. We will be monitoring these activities to improve future antibiotic prescribing among tertiary hospitals in Pakistan during future waves of this and other pandemics.
Author Contributions
Conceptualization, K.R., S.S., I.R., Z.U.M. and B.G.; methodology, Z.U.M., B.G. and J.C.M.; software, Z.U.M.; validation, K.R., S.S., I.R., Z.U.M., M.S. and A.H.K.; formal analysis, K.R., S.S., I.R., Z.U.M., M.S., A.H.K. and J.C.M.; investigation, K.R., S.S., I.R., Z.U.M., M.S. and A.H.K.; resources, Z.U.M.; data curation, K.R., S.S., I.R. and Z.U.M.; writing—original draft preparation, Z.U.M. and B.G.; writing—review and editing, K.R., S.S., I.R., Z.U.M., M.S., A.H.K., J.C.M. and B.G.; visualization, K.R., S.S., I.R. and Z.U.M.; supervision, Z.U.M. and B.G.; project administration, Z.U.M.; funding acquisition, not applicable. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The current study was conducted after approval from the ethics committee of The Department of Pharmacy Practice, Faculty of Pharmacy, The University of Lahore (REC/DPP/FOP/46). Patient informed consent was not required, with medical records of patients who were discharged from the hospital reviewed retrospectively.
Informed Consent Statement
As mentioned, patients were not approached for consent since this was a retrospective study based on data collected from medical records only, with no direct contact with patients. This is in line with previous point prevalence studies undertaken by the co-authors.
Data Availability Statement
The datasets used during the current study are available from the corresponding author on reasonable request.
Acknowledgments
We are thankful to the staff and administration of the participating hospitals for their support with this research.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Abid, K.; Bari, Y.A.; Younas, M.; Javaid, S.T.; Imran, A. Progress of COVID-19 Epidemic in Pakistan. Asia Pac. J. Public Health 2020, 32, 154–156. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, H.; Afridi, M.; Akhtar, S.; Ahmad, H.; Ali, S.; Khalid, S.; Awan, S.M.; Jahangiri, S.; Khader, Y.S. Pakistan’s Response to COVID-19: Overcoming National and International Hypes to Fight the Pandemic. JMIR Public Health Surveill. 2021, 7, e28517. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, S.; Bakhshwin, D.; Shirbeeny, W.; Bakhshwin, A.; Bahabri, F.; Bakhshwin, A.; Alsaggaf, S.M. Successive waves of COVID 19: Confinement effects on virus-prevalence with a mathematical model. Eur. J. Med. Res. 2021, 26, 128. [Google Scholar] [CrossRef] [PubMed]
- Hale, T.; Angrist, N.; Hale, A.J.; Kira, B.; Majumdar, S.; Petherick, A.; Phillips, T.; Sridhar, D.; Thompson, R.N.; Webster, S.; et al. Government responses and COVID-19 deaths: Global evidence across multiple pandemic waves. PLoS ONE 2021, 16, e0253116. [Google Scholar] [CrossRef] [PubMed]
- Rahim, S.; Dhrolia, M.; Qureshi, R.; Nasir, K.; Ahmad, K. A Comparative Study of the First and Second Waves of COVID-19 in Hemodialysis Patients From Pakistan. Cureus 2022, 14, e21512. [Google Scholar] [CrossRef]
- Imran, M.; Khan, S.; Khan, S.; Uddin, A.; Khan, M.S.; Ambade, A. COVID-19 situation in Pakistan: A broad overview. Respirology 2021, 26, 891–892. [Google Scholar] [CrossRef]
- Godman, B.; Haque, M.; Islam, S.; Iqbal, S.; Urmi, U.L.; Kamal, Z.M.; Shuvo, S.A.; Rahman, A.; Kamal, M.; Haque, M.; et al. Rapid assessment of price instability and paucity of medicines and protection for COVID-19 across Asia: Findings and public health implications for the future. Public Health Front. 2020, 8, 585832. [Google Scholar] [CrossRef]
- Ayouni, I.; Maatoug, J.; Dhouib, W.; Zammit, N.; Fredj, S.B.; Ghammam, R.; Ghannem, H. Effective public health measures to mitigate the spread of COVID-19: A systematic review. BMC Public Health 2021, 21, 1015. [Google Scholar] [CrossRef]
- Talic, S.; Shah, S.; Wild, H.; Gasevic, D.; Maharaj, A.; Ademi, Z.; Li, X.; Xu, W.; Mesa-Eguiagaray, I.; Rostron, J.; et al. Effectiveness of public health measures in reducing the incidence of covid-19, SARS-CoV-2 transmission, and covid-19 mortality: Systematic review and meta-analysis. BMJ 2021, 375, e068302. [Google Scholar]
- Looi, M.K. Covid-19: Is a second wave hitting Europe? BMJ 2020, 371, m4113. [Google Scholar] [CrossRef]
- Shahid, R.; Zeb, S. Second Wave of COVID-19 Pandemic: Its deleterious and mortal repercussion in Pakistan. J. Rawalpindi Med. Coll. 2020, 24, 288–289. [Google Scholar] [CrossRef]
- Basheer, A.; Zahoor, I. Genomic epidemiology of SARS-CoV-2 divulge B. 1, B. 1.36, and B. 1.1. 7 as the most dominant lineages in first, second, and third wave of SARS-CoV-2 infections in Pakistan. Microorganisms 2021, 9, 2609. [Google Scholar] [CrossRef] [PubMed]
- Kamran, K.; Ali, A. Challenges and Strategies for Pakistan in the Third Wave of COVID-19: A Mini Review. Public Health Front. 2021, 9, 690820. [Google Scholar] [CrossRef] [PubMed]
- The United Nations Children’s Fund (UNICEF). Pakistan Humanitarian. Situation Report No. 28. 2021. Available online: https://www.unicef.org/media/107031/file/%20Pakistan-Humanitarian-sitRep-No28-31-August-2021.pdf (accessed on 21 March 2022).
- Geo News. COVID-19 Situation Continues to Worsen in Pakistan Amid Fifth Wave. 2021. Available online: https://www.geo.tv/latest/394761-covid-19-situation-continues-to-worsen-in-pakistan-amid-fifth-wavepdf (accessed on 21 March 2022).
- National Disaster Management Authority (NDMA). 2022. Available online: http://cms.ndma.gov.pk/ (accessed on 12 March 2022).
- National Command and Operation Center (NCOC); Government of Islamic Government of Pakistan. 2022. Available online: https://ncoc.gov.pk/ (accessed on 21 March 2022).
- Sarfaraz, S.; Shaikh, Q.; Saleem, S.G.; Rahim, A.; Herekar, F.F.; Junejo, S.; Hussain, A. Determinants of in-hospital mortality in COVID-19; a prospective cohort study from Pakistan. PLoS ONE 2021, 16, e0251754. [Google Scholar] [CrossRef]
- Kamran, S.H.; Ul Mustafa, Z.; Rao, A.Z.; Hasan, S.S.; Zahoor, F.; Sarwar, M.U.; Khan, S.; Butt, S.; Rameez, M.; Abbas, M.A. SARS-CoV-2 infection pattern, transmission and treatment: Multicenter study in low to middle-income districts hospitals in Punjab, Pakistan. Pak. J. Pharm. Sci. 2021, 34, 1135–1142. [Google Scholar]
- World Health Organization (WHO). Clinical Management of COVID-19: Interim Guidance. 27 May 2020. Available online: https://apps.who.int/iris/handle/10665/332196 (accessed on 21 March 2022).
- Sanders, J.M.; Monogue, M.L.; Jodlowski, T.Z.; Cutrell, J.B. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): A review. JAMA 2020, 323, 1824–1836. [Google Scholar] [CrossRef]
- Scavone, C.; Brusco, S.; Bertini, M.; Sportiello, L.; Rafaniello, C.; Zoccoli, A.; Berrino, L.; Racagni, G.; Rossi, F.; Capuano, A. Current pharmacological treatments for COVID-19: What’s next? Br. J. Pharmacol. 2020, 177, 4813–4824. [Google Scholar] [CrossRef]
- RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef]
- Abena, P.M.; Decloedt, E.H.; Bottieau, E.; Suleman, F.; Adejumo, P.; Sam-Agudu, N.A.; TamFum, J.-J.M.; Seydi, M.; Eholie, S.P.; Edward J Mills, E.J.; et al. Chloroquine and Hydroxychloroquine for the Prevention or Treatment of COVID-19 in Africa: Caution for Inappropriate Off-label Use in Healthcare Settings. Am. J. Trop. Med. Hyg. 2020, 102, 1184–1188. [Google Scholar] [CrossRef]
- Khadka, S.; Shrestha, D.B.; Budhathoki, P.; Rawal, E. Hydroxychloroquine in COVID-19: The Study Points to Premature Decisions on Efficacy While Bells Ringing for Safety. Clin. Pharmacol. 2020, 12, 115–121. [Google Scholar] [CrossRef]
- World Health Organization (WHO). WHO Discontinues Hydroxychloroquine and Lopinavir/Ritonavir Treatment Arms for COVID-19. 2020. Available online: https://www.who.int/news/item/04-07-2020-who-discontinues-hydroxychloroquine-and-lopinavir-ritonavir-treatment-arms-for-covid-19 (accessed on 20 March 2022).
- Horby, P.; Mafham, M.; Linsell, L.; Bell, J.L.; Staplin, N.; Emberson, J.R.; Wiselka, M.; Ustianowski, A.; Elmahi, E.; Prudon, B.; et al. Effect of Hydroxychloroquine in Hospitalized Patients with Covid-19. N. Engl. J. Med. 2020, 383, 2030–2040. [Google Scholar] [PubMed]
- Lopinavir-ritonavir in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial. Lancet 2020, 396, 1345–1352. [CrossRef]
- Dyer, O. Covid-19: Remdesivir has little or no impact on survival, WHO trial shows. BMJ 2020, 371, m4057. [Google Scholar] [CrossRef] [PubMed]
- Kow, C.S.; Merchant, H.A.; Mustafa, Z.U.; Hasan, S.S. The association between the use of ivermectin and mortality in patients with COVID-19: A meta-analysis. Pharmacol. Rep. 2021, 73, 1473–1479. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Zhou, F.; Ali, S.; Heybati, K.; Hou, W.; Huang, E.; Wonh, C.Y. Efficacy and safety of ivermectin for the treatment of COVID-19: A systematic review and meta-analysis. QJM 2021, 114, 721–732. [Google Scholar] [CrossRef]
- Ministry Of National Health Services. Clinical Management Guidelines for COVID-19 Infections. Regulations and Coordination, Government of Pakistan; 2020. Available online: https://storage.covid.gov.pk/new_guidelines/11December2020_20201211_Clinical_Management_Guidelines_for_COVID-19_infection_1204.pdf (accessed on 19 March 2020).
- Langford, B.J.; So, M.; Raybardhan, S.; Leung, V.; Soucy, J.-P.R.; Westwood, D.; Daneman, N.; MacFadden, D.R. Antibiotic prescribing in patients with COVID-19: Rapid review and meta-analysis. Clin. Microbiol. Infect. 2021, 27, 520–531. [Google Scholar] [CrossRef]
- Langford, B.J.; So, M.; Raybardhan, S.; Leung, V.; Westwood, D.; MacFadden, D.R.; Soucy, J.-P.R.; Daneman, N. Bacterial co-infection and secondary infection in patients with COVID-19: A living rapid review and meta-analysis. Clin. Microbiol. Infect. 2020, 26, 1622–1629. [Google Scholar] [CrossRef]
- Vaughn, V.M.; Gandhi, T.N.; Petty, L.A.; Patel, P.K.; Prescott, H.C.; Malani, A.N.; Ratz, D.; McLaughlin, E.; Chopra, V.; Flanders, S.A. Empiric antibacterial therapy and community-onset bacterial coinfection in patients hospitalized with coronavirus disease 2019 (COVID-19): A multi-hospital cohort study. Clin. Infect. Diseases. 2021, 72, e533–e541. [Google Scholar] [CrossRef]
- Chowdhury, K.; Haque, M.; Nusrat, N.; Adnan, N.; Islam, S.; Lutfor, A.B.; Begum, D.; Rabbany, A.; Karim, E.; Malek, A.; et al. Management of Children Admitted to Hospitals across Bangladesh with Suspected or Confirmed COVID-19 and the Implications for the Future: A Nationwide Cross-Sectional Study. Antibiotics 2022, 11, 105. [Google Scholar] [CrossRef]
- Mustafa, Z.U.; Saleem, M.S.; Ikram, M.N.; Salman, M.; Butt, S.A.; Khan, S.; Godman, B.; Seaton, R.A. Co-infections and antimicrobial use among hospitalized COVID-19 patients in Punjab, Pakistan: Findings from a multicenter, point prevalence survey. Pathog. Glob. Health 2021, 1–7. [Google Scholar] [CrossRef]
- Akhtar, H.; Akhtar, S.; Rahman, F.-U.; Afridi, M.; Khalid, S.; Ali, S.; Akhtar, N.; Khader, Y.S.; Ahmad, H.; Khan, M.M. An Overview of the Treatment Options Used for the Management of COVID-19 in Pakistan: Retrospective Observational Study. JMIR Public Health Surveill. 2021, 7, e28594. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Antimicrobial Resistance. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 13 March 2022).
- Nathwani, D.; Varghese, D.; Stephens, J.; Ansari, W.; Martin, S.; Charbonneau, C. Value of hospital antimicrobial stewardship programs [ASPs]: A systematic review. Antimicrob. Resist. Infect. Control 2019, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Lucien, M.A.; Canarie, M.F.; Kilgore, P.E.; Jean-Denis, G.; Fénélon, N.; Pierre, M.; Cerpa, M.; Joseph, G.A.; Maki, G. Antibiotics and antimicrobial resistance in the COVID-19 era: Perspective from resource-limited settings. Int. J. Infect. Dis. 2021, 104, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Centers for Diseases Control and Prevention (CDC). COVID-19 & Antibiotic Resistance. Available online: https://www.cdc.gov/drugresistance/covid19.html (accessed on 13 March 2022).
- Founou, R.C.; Blocker, A.J.; Noubom, M.; Tsayem, C.; Choukem, S.P.; Dongen, M.V.; Founou, L.L. The COVID-19 pandemic: A threat to antimicrobial resistance containment. Future Sci. OA 2021, 7, FSO736. [Google Scholar] [CrossRef]
- Hsu, J. How covid-19 is accelerating the threat of antimicrobial resistance. BMJ 2020, 369, m1983. [Google Scholar] [CrossRef]
- Tomczyk, S.; Taylor, A.; Brown, A.; de Kraker, M.E.A.; El-Saed, A.; Alshamrani, M.; Hendriksen, R.S.; Jacob, M.; Löfmark, S.; Perovic, O.; et al. Impact of the COVID-19 pandemic on the surveillance, prevention and control of antimicrobial resistance: A global survey. J. Antimicrob. Chemother. 2021, 76, 3045–3058. [Google Scholar] [CrossRef]
- Knight, G.M.; Glover, R.E.; McQuaid, C.F.; Olaru, I.D.; Gallandat, K.; Leclerc, Q.J.; Fuller, N.M.; Willcocks, S.J.; Hasan, R.; van Kleef, E.; et al. Antimicrobial resistance and COVID-19: Intersections and implications. Elife 2021, 10, e64139. [Google Scholar] [CrossRef]
- Nieuwlaat, R.; Mbuagbaw, L.; Mertz, D.; Burrows, L.L.; Bowdish, D.M.E.; Moja, L.; Wright, G.D.; Schünemann, H.J. Coronavirus disease 2019 and antimicrobial resistance: Parallel and interacting health emergencies. Clin. Infect. Dis. 2021, 72, 1657–1659. [Google Scholar] [CrossRef]
- Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- World Bank Group. Pulling Together to Beat Superbugs—Knowledge and Implementation Gaps in Addressing Antimicrobial Resistance. 2019. Available online: https://documents1.worldbank.org/curated/en/430051570735014540/pdf/Pulling-Together-to-Beat-Superbugs-Knowledge-and-Implementation-Gaps-in-Addressing-Antimicrobial-Resistance.pdf (accessed on 18 March 2022).
- The World Bank. Antimicrobial Resistance (AMR). 2021. Available online: https://www.worldbank.org/en/topic/health/brief/antimicrobial-resistance-amr (accessed on 17 March 2022).
- World Health Organization (WHO). Global Action Plan on Antimicrobial Resistance. 2016. Available online: https://www.who.int/publications/i/item/9789241509763 (accessed on 17 March 2022).
- Sharland, M.; Pulcini, C.; Harbarth, S.; Zeng, M.; Gandra, S.; Mathur, S.; Magrini, N. Classifying antibiotics in the WHO Essential Medicines List for optimal use—Be AWaRe. Lancet Infect. Dis. 2018, 18, 18–20. [Google Scholar] [CrossRef] [Green Version]
- Sharland, M.; Gandra, S.; Huttner, B.; Moja, L.; Pulcini, C.; Zeng, M.; Mendelson, M.; Cappello, B.; Cooke, G.; Magrini, N.; et al. Encouraging AwaRe-ness and discouraging inappropriate antibiotic use-the new 2019 Essential Medicines List becomes a global antibiotic stewardship tool. Lancet Infect. Dis. 2019, 19, 1278–1280. [Google Scholar] [CrossRef]
- Hsia, Y.; Lee, B.R.; Versporten, A.; Yang, Y.; Bielicki, J.; Jackson, C.; Newland, J.; Goossens, H.; Magrini, N.; Sharland, M.; et al. Use of the WHO Access, Watch, and Reserve classification to define patterns of hospital antibiotic use (AwaRe): An analysis of paediatric survey data from 56 countries. Lancet Glob. Health 2019, 7, e861–e871. [Google Scholar] [CrossRef] [Green Version]
- WHO Access, Watch, Reserve, Classification of Antibiotics for Evaluation and Monitoring of Use. 2021. Available online: https://www.who.int/publications/i/item/2021-aware-classification (accessed on 17 March 2022).
- Hsia, Y.; Sharland, M.; Jackson, C.; Wong, I.C.K.; Magrini, N.; Bielicki, J.A. Consumption of oral antibiotic formulations for young children according to the WHO Access, Watch, Reserve (AWaRe) antibiotic groups: An analysis of sales data from 70 middle-income and high-income countries. Lancet Infect. Dis. 2019, 19, 67–75. [Google Scholar] [CrossRef]
- Klein, E.Y.; Milkowska-Shibata, M.; Sharland, M.; Gabdra, S.; Pulcini, C.; Laxminarayan, R. Assessment of WHO antibiotic consumption and access targets in 76 countries, 2000–2015: An analysis of pharmaceutical sales data. Lancet Infect. Dis. 2021, 21, 107–115. [Google Scholar] [CrossRef]
- Mustafa, Z.U.; Salman, M.; Yasir, M.; Godman, B.; Majeed, H.A.; Kanwal, M.; Iqbal, M.; Riaz, M.B.; Hayat, K.; Hasan, S.S. Antibiotic consumption among hospitalized neonates and children in Punjab province, Pakistan. Expert Rev. Anti-Infect. Ther. 2022, 20, 931–939. [Google Scholar] [CrossRef]
- Antimicrobial Resistance. National Action Plan Pakistan. 2017. Available online: https://www.nih.org.pk/wp-content/uploads/2018/08/AMR-National-Action-Plan-Pakistan.pdf (accessed on 20 March 2022).
- Saleem, Z.; Hassali, M.A.; Hashmi, F.K. Pakistan’s national action plan for antimicrobial resistance: Translating ideas into reality. Lancet Infect. Dis. 2018, 18, 1066–1067. [Google Scholar] [CrossRef]
- Saleem, Z.; Godman, B.; Azhar, F.; Kalungia, A.C.; Fadare, J.; Opanga, S.; Markovic-Pekovic, V.; Hoxha, I.; Saeed, A.; Al-Gethamy, M.; et al. Progress on the national action plan of Pakistan on antimicrobial resistance (AMR): A narrative review and the implications. Expert Rev. Anti-Infect. Ther. 2022, 20, 71–93. [Google Scholar] [CrossRef]
- Ul Mustafa, Z.; Salman, M.; Aldeyab, M.; Kow, C.S.; Hasan, S.S. Antimicrobial consumption among hospitalized patients with COVID-19 in Pakistan. SN Compr. Clin. Med. 2021, 3, 1691–1695. [Google Scholar] [CrossRef]
- Zeshan, B.; Karobari, M.I.; Afzal, N.; Siddiq, A.; Basha, S.; Basheer, S.N.; Peeran, S.W.; Mustafa, M.; Daud, N.H.A.; Ahmed, N.; et al. The Usage of Antibiotics by COVID-19 Patients with Comorbidities: The Risk of Increased Antimicrobial Resistance. Antibiotics 2022, 11, 35. [Google Scholar] [CrossRef]
- Kumar, G.; Mukherjee, A.; Sharma, R.K.; Menon, G.R.; Sahu, D.; Wig, N.; Panda, S.; Rao, V.V.; Singh, S.; Guleria, R.; et al. Clinical profile of hospitalized COVID-19 patients in first & second wave of the pandemic: Insights from an Indian registry based observational study. Indian J. Med. Res. 2021, 153, 619–628. [Google Scholar]
- Krishnamurthy, S.; Kar, S.S.; Dhodapkar, R.; Parameswaran, N. Comparison of COVID-19 Infection in Children During the First and Second Wave. Indian J. Pediatr. 2022, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Hashim, J.H.; Adman, M.A.; Hashim, Z.; Radi, M.F.M.; Kwan, S.C. COVID-19 Epidemic in Malaysia: Epidemic Progression, Challenges, and Response. Front. Public Health 2021, 9, 560592. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.-J.; Liang, W.-H.; Zhao, Y.; Liang, H.-R.; Chen, Z.-S.; Li, Y.-M.; Liu, X.-Q.; Chen, R.-C.; Tang, C.-L.; Wang, T.; et al. Comorbidity and its impact on 1590 patients with COVID-19 in China: A nationwide analysis. Eur. Respir. J. 2020, 55, 200054. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elezkurtaj, S.; Greuel, S.; Ihlow, J.; Michaelis, E.G.; Philip Bischoff, P.; Kunze, C.A.; Sinn, B.V.; Gerhold, M.; Hauptmann, K.; Ingold-Heppner, B.; et al. Causes of death and comorbidities in hospitalized patients with COVID-19. Sci. Rep. 2021, 11, 4263. [Google Scholar] [CrossRef]
- Semenzato, L.; Botton, J.; Drouin, J.; Cuenot, F.; Dray-Spira, R.; Weill, A.; Zureik, M. Chronic diseases, health conditions and risk of COVID-19-related hospitalization and in-hospital mortality during the first wave of the epidemic in France: A cohort study of 66 million people. Lancet Reg. Health Eur. 2021, 8, 100158. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; the Northwell COVID-19 Research Consortium. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef]
- Asghar, M.S.; Kazmi, S.J.H.; Akram, M.; Khan, S.A.; Rasheed, U.; Hassan, M.; Memon, M.M. Clinical profiles, characteristics, and outcomes of the first 100 admitted COVID-19 patients in Pakistan: A single-center retrospective study in a tertiary care hospital of Karachi. Cureus 2020, 12, e8712. [Google Scholar] [CrossRef]
- Ahmad, M.; Beg, B.M.; Majeed, A.; Areej, S.; Riffat, S.; Rasheed, M.A.; Mahmood, S.; Mushtaq, R.M.Z.; Hafeez, M.A. Epidemiological and clinical characteristics of covid-19: A retrospective multi-center study in pakistan. Front. Public Health 2021, 9, 342. [Google Scholar] [CrossRef]
- Mowla, S.G.M.; Azad, K.A.K.; Kabir, A.; Biswas, S.; Islam, M.R.; Banik, G.C.; Khan, M.M.H.; Rohan, K.I.; Alam, A. Clinical profile of 100 confirmed COVID-19 patients admitted in Dhaka medical college Hospital, Dhaka, Bangladesh. J. Bangladesh Coll. Physicians Surg. 2020, 38, 29–36. [Google Scholar] [CrossRef]
- Akbariqomi, M.; Hosseini, M.S.; Rashidiani, J.; Sedighian, H.; Biganeh, H.; Heidari, R.; Moghaddam, M.M.; Farnoosh, G.; Kooshki, H. Clinical characteristics and outcome of hospitalized COVID-19 patients with diabetes: A single-center, retrospective study in Iran. Diabetes Res. Clin. Pract. 2020, 169, 108467. [Google Scholar] [CrossRef]
- Rousan, L.A.; Elobeid, E.; Karrar, M.; Khader, Y. Chest x-ray findings and temporal lung changes in patients with COVID-19 pneumonia. BMC Pulm. Med. 2020, 20, 245. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Chen, L.; Liu, Z.; Pan, J.; Zhou, D.; Wang, H.; Gong, H.; Fu, Z.; Song, Q.; Min, Q.; et al. Clinical features and short-term outcomes of elderly patients with COVID-19. Int. J. Infect. Dis. 2020, 97, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Nishtar, T.; Noor, N.; Khan, S.L. X-ray patterns of COVID-19 in patients presenting to Lady Reading Hospital, Peshawar, Pakistan. Pak. J. Med. Sci. 2021, 37, 28. [Google Scholar] [CrossRef] [PubMed]
- Durrani, M.; Inam ul Haq, U.K.; Yousaf, A. Chest X-rays findings in COVID 19 patients at a University Teaching Hospital-A descriptive study. Pak. J. Med. Sci. 2020, 36, S22. [Google Scholar] [CrossRef] [PubMed]
- Sathi, S.; Tiwari, R.; Verma, S.; Kumar Garg, A.; Singh Saini, V.; Kumar Singh, M.; Mittal, A.; Vohra, D. Role of Chest X-Ray in Coronavirus Disease and Correlation of Radiological Features with Clinical Outcomes in Indian Patients. Can. J. Infect. Dis. Med. Microbiol. 2021, 2021, 6326947. [Google Scholar] [CrossRef]
- Weinstock, M.B.; Echenique, A.; Russell, J.W.; Leib, A.; Miller, J.A.; Cohen, D.J.; Waite, S.; Frye, A.; Illuzzi, F.A. Chest x-ray findings in 636 ambulatory patients with COVID-19 presenting to an urgent care center: A normal chest x-ray is no guarantee. J. Urgent Care Med. 2020, 14, 13–18. [Google Scholar]
- Smilowitz, N.R.; Kunichoff, D.; Garshick, M.; Shah, B.; Pillinger, M.; Hochman, J.S.; Berger, J.S. C-reactive protein and clinical outcomes in patients with COVID-19. Eur. Heart J. 2021, 42, 2270–2279. [Google Scholar] [CrossRef]
- Mustafa, Z.U.; Kow, C.S.; Salman, M.; Kanwal, M.; Riaz, M.B.; Parveen, S.; Hasan, S.S. Pattern of medication utilization in hospitalized patients with COVID-19 in three District Headquarters Hospitals in the Punjab province of Pakistan. Res. Soc. Adm. Pharm. 2022, 5, 100101. [Google Scholar] [CrossRef]
- Khandker, S.S.; Godman, B.; Jawad, M.I.; Meghla, B.A.; Tisha, T.A.; Khondoker, M.U.; Haq, A.; Charan, J.; Talukder, A.A.; Azmuda, N.; et al. A Systematic Review on COVID-19 Vaccine Strategies, Their Effectiveness, and Issues. Vaccines 2021, 9, 1387. [Google Scholar] [CrossRef]
- Mohammed, I.; Nauman, A.; Paul, P.; Ganesan, S.; Chen, K.H.; Jalil, S.M.S.; Jaouni, S.H.; Kawas, H.; Khan, W.A.; Vattoth, A.L.; et al. The efficacy and effectiveness of the COVID-19 vaccines in reducing infection, severity, hospitalization, and mortality: A systematic review. Hum. Vaccin. Immunother. 2022, 18, 2027160. [Google Scholar] [CrossRef]
- Zeng, B.; Gao, L.; Zhou, Q.; Yu, K.; Sun, F. Effectiveness of COVID-19 vaccines against SARS-CoV-2 variants of concern: A systematic review and meta-analysis. BMC Med. 2022, 20, 200. [Google Scholar] [CrossRef] [PubMed]
- Vekaria, B.; Overton, C.; Wiśniowski, A.; Ahmad, S.; Aparicio-Castro, A.; Curran-Sebastian, J.; Eddleston, J.; Hanley, N.A.; House, T.; Kim, J.; et al. Hospital length of stay for COVID-19 patients: Data-driven methods for forward planning. BMC Infect. Dis. 2021, 21, 700. [Google Scholar] [CrossRef] [PubMed]
- Rees, E.M.; Nightingale, E.S.; Jafari, Y.; Waterlow, N.R.; Clifford, S.; Pearson, C.A.B.; Cmmid Working Group; Jombart, T.; Procter, S.R.; Knight, G.M. COVID-19 length of hospital stay: A systematic review and data synthesis. BMC Med. 2020, 18, 270. [Google Scholar] [CrossRef] [PubMed]
- Di Fusco, M.; Shea, K.M.; Lin, J.; Nguyen, J.L.; Angulo, F.J.; Benigno, M.; Malhotra, D.; Emir, B.; Sung, A.H.; Hammond, J.L.; et al. Health outcomes and economic burden of hospitalized COVID-19 patients in the United States. J. Med. Econ. 2021, 24, 308–317. [Google Scholar] [CrossRef]
- Garg, S.; Patel, K.; Pham, H.; Whitaker, M.; O’Halloran, A.; Milucky, J.; Anglin, O.; Kirley, P.D.; Reingold, A.; Kawasaki, B.; et al. Clinical trends among US Adults hospitalized with COVID-19, March to December 2020: A cross-sectional study. Ann. Intern. Med. 2021, 174, 1409–1419. [Google Scholar] [CrossRef]
- Abate, S.M.; Ahmed Ali, S.; Mantfardo, B.; Basu, B. Rate of Intensive Care Unit admission and outcomes among patients with coronavirus: A systematic review and Meta-analysis. PLoS ONE 2020, 15, e0235653. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, L.; Yan, Y.; Zhou, Y.; Yin, P.; Qi, J.; Wang, L.; Pan, J.; You, J.; Yang, J.; et al. Excess mortality in Wuhan city and other parts of China during the three months of the covid-19 outbreak: Findings from nationwide mortality registries. BMJ 2021, 372, n415. [Google Scholar] [CrossRef]
- Malhotra, V.; Basu, S.; Sharma, N.; Kumar, S.; Garg, S.; Dushyant, K.; Borle, A. Outcomes among 10,314 hospitalized COVID-19 patients at a tertiary care government hospital in Delhi, India. J. Med. Virol. 2021, 93, 4553–4558. [Google Scholar] [CrossRef]
- Gujski, M.; Jankowski, M.; Rabczenko, D.; Goryński, P.; Juszczyk, G. Characteristics and Clinical Outcomes of 116,539 Patients Hospitalized with COVID-19—Poland, March–December 2020. Viruses 2021, 13, 1458. [Google Scholar] [CrossRef]
- Ghafari, M.; Kadivar, A.; Katzourakis, A. Excess deaths associated with the Iranian COVID-19 epidemic: A province-level analysis. Int. J. Infect. Dis. 2021, 107, 101–115. [Google Scholar] [CrossRef]
- Finelli, L.; Gupta, V.; Petigara, T.; Yu, K.; Bauer, K.A.; Puzniak, L.A. Mortality among US patients hospitalized with SARS-CoV-2 infection in 2020. JAMA Netw. Open 2021, 4, e216556. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Mustafa, L.; Tolaj, I.; Baftiu, N.; Fejza, H. Use of antibiotics in COVID-19 ICU patients. J. Infect. Dev. Ctries. 2021, 15, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Goncalves Mendes Neto, A.; Lo, K.B.; Wattoo, A.; Salacup, G.; Pelayo, J.; DeJoy, R., 3rd; Bhargav, R.; Gul, F.; Peterson, E.; Albano, J.; et al. Bacterial infections and patterns of antibiotic use in patients with COVID-19. J. Med. Virol. 2021, 93, 1489–1495. [Google Scholar] [CrossRef] [PubMed]
- Karaba, S.M.; Jones, G.; Helsel, T.; Leigh Smith, L.; Avery, R.; Dzintars, K.; Salinas, A.B.; Sara C Keller, S.C.; Townsend, J.L.; Klein, E.; et al. Prevalence of co-infection at the time of hospital admission in COVID-19 patients, a multicenter study. Open Forum Infect. Dis. 2020, 8, ofaa578. [Google Scholar] [CrossRef]
- Chedid, M.; Waked, R.; Haddad, E.; Chetata, N.; Saliba, G.; Choucair, J. Antibiotics in treatment of COVID-19 complications: A review of frequency, indications, and efficacy. J. Infect. Public Health 2021, 14, 570–576. [Google Scholar] [CrossRef]
- Molla, M.M.; Yeasmin, M.; Islam, M.K.; Sharif, M.; Amin, R.; Nafisa, T.; Ghosh, A.K.; Parveen, M.; Arif, M.H.; Alam, J.A.J.; et al. Antibiotic prescribing patterns at COVID-19 dedicated wards in Bangladesh: Findings from a single center study. Infect. Prev. Pract. 2021, 3, 100134. [Google Scholar] [CrossRef]
- Vijay, S.; Bansal, N.; Rao, B.K.; Veeraraghavan, B.; Rodrigues, C.; Wattal, C.; Goyal, J.P.; Tadepalli, K.; Mathur, P.; Venkateswaran, R.; et al. Secondary infections in hospitalized COVID-19 patients: Indian experience. Infect. Drug Resist. 2021, 14, 1893. [Google Scholar] [CrossRef]
- Cong, W.; Poudel, A.N.; Alhusein, N.; Wang, H.; Yao, G.; Lambert, H. Antimicrobial use in COVID-19 patients in the first phase of the SARS-CoV-2 pandemic: A scoping review. Antibiotics 2021, 10, 745. [Google Scholar] [CrossRef]
- Seaton, R.A.; Gibbons, C.L.; Cooper, L.; Malcolm, W.; McKinney, R.; Dundas, S.; Griffith, D.; Jeffreys, D.; Hamilton, K.; Choo-Kang, B.; et al. Survey of antibiotic and antifungal prescribing in patients with suspected and confirmed COVID-19 in Scottish hospitals. J. Infect. 2020, 81, 952–960. [Google Scholar] [CrossRef]
- Adebisi, Y.A.; Jimoh, N.D.; Ogunkola, I.O.; Uwizeyimana, T.; Olayemi, A.H.; Ukor, N.A.; Lucero-Prisno, D.E., 3rd. The use of antibiotics in COVID-19 management: A rapid review of national treatment guidelines in 10 African countries. Trop. Med. Health 2021, 49, 51. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.; Biswas, M.; Al Jubayer, A.; Rahman, F.; Akhtar, Z.; Das, P.; Islam, A.; Chowdhury, F. Use of antimicrobials among suspected COVID-19 patients at selected hospitals, Bangladesh: Findings from the first wave of COVID-19 pandemic. Antibiotics 2021, 10, 738. [Google Scholar]
- Rose, A.N.; Baggs, J.; Wolford, H.; Neuhauser, M.M.; Srinivasan, A.; Gundlapalli, A.V.; Reddy, S.; Kompaniyets, L.; Pennington, A.F.; Grigg, C.; et al. Trends in antibiotic use in United States hospitals during the coronavirus disease 2019 pandemic. Open Forum Infect. Dis. 2021, 8, ofab236. [Google Scholar] [CrossRef] [PubMed]
- Russell, C.D.; Fairfield, C.J.; Drake, T.M.; Turtle, L.; Seaton, R.A.; Wootton, D.G.; Sigfrid, L.; Harrison, E.M.; Docherty, A.B.; de Silva, T.I.; et al. Co-infections, secondary infections, and antimicrobial use in patients hospitalised with COVID-19 during the first pandemic wave from the ISARIC WHO CCP-UK study: A multicentre, prospective cohort study. Lancet Microbe. 2021, 2, e354–e365. [Google Scholar] [CrossRef]
- Kumar, S.; Tadepalli, K.; Joshi, R.; Shrivastava, M.; Malik, R.; Saxena, P.; Saigal, S.; Jhaj, R.; Khadanga, S. Practice of antimicrobial stewardship in a government hospital of India and its impact on extended point prevalence of antibiotic usage. J. Fam. Med. Prim. Care 2021, 10, 991–997. [Google Scholar]
- Arulappen, A.L.; Danial, M.; Haron, N.; Hau, L.C.; Khan, A.H. The Impact of Antimicrobial Stewardship Program on Injudicious Use of Cefuroxime. Front. Pharmacol. 2020, 11, 565818. [Google Scholar] [CrossRef]
- Godman, B.; Egwuenu, A.; Haque, M.; Malande, O.O.; Schellack, N.; Kumar, S.; Saleem, Z.; Sneddon, J.; Hoxha, I.; Islam, S.; et al. Strategies to improve antimicrobial utilization with a special focus on developing countries. Life 2021, 11, 528. [Google Scholar] [CrossRef]
- Uddin, T.M.; Chakraborty, A.J.; Khusro, A.; Zidan, B.R.M.; Mitra, S.; Emran, T.B.; Dhama, K.; Ripon, K.H.; Gajdács, M.; Sahibzada, M.U.K.; et al. Antibiotic resistance in microbes: History, mechanisms, therapeutic strategies and future prospects. J. Infect. Public Health 2021, 14, 1750–1766. [Google Scholar] [CrossRef]
- Cox, J.A.; Vlieghe, E.; Mendelson, M.; Wertheim, H.; Ndegwa, L.; Villegas, M.V.; Gould, I.; Hara, G.L. Antibiotic stewardship in low- and middle-income countries: The same but different? Clin. Microbiol. Infect. 2017, 23, 812–818. [Google Scholar] [CrossRef] [Green Version]
- Pierce, J.; Apisarnthanarak, A.; Schellack, N.; Cornistein, W.; Maani, A.A.; Adnan, S.; Stevens, M.P. Global Antimicrobial Stewardship with a Focus on Low- and Middle-Income Countries. Int. J. Infect. Dis. 2020, 96, 621–629. [Google Scholar] [CrossRef]
- Akpan, M.R.; Isemin, N.U.; Udoh, A.E.; Ashiru-Oredope, D. Implementation of antimicrobial stewardship programmes in African countries: A systematic literature review. J. Glob. Antimicrob. Resist. 2020, 22, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Sartelli, M.; Hardcastle, C.T.; Catena, F.; Chichom-Mefire, A.; Coccolini, F.; Dhingra, S.; Haque, M.; Hodonou, A.; Iskandar, K.; Francesco M Labricciosa, F.M.; et al. Antibiotic Use in Low and Middle-Income Countries and the Challenges of Antimicrobial Resistance in Surgery. Antibiotics 2020, 9, 497. [Google Scholar] [CrossRef] [PubMed]
- Al-Omari, A.; Al Mutair, A.; Alhumaid, S.; Salih, S.; Alanazi, A.; Albarsan, H.; Abourayan, M.; Al Subaie, M. The impact of antimicrobial stewardship program implementation at four tertiary private hospitals: Results of a five-years pre-post analysis. Antimicrob. Resist. Infect. Control 2020, 9, 95. [Google Scholar] [CrossRef] [PubMed]
- Liew, Y.; Lee, W.H.L.; Tan, L.; Kwa, A.L.H.; Thien, S.Y.; Cherng, B.P.Z.; Chung, S.J. Antimicrobial stewardship programme: A vital resource for hospitals during the global outbreak of coronavirus disease 2019 (COVID-19). Int. J. Antimicrob. Agents 2020, 56, 106145. [Google Scholar] [CrossRef]
- Antimicrobial stewardship programmes in health-care facilities in low- and middle-income countries: A WHO practical toolkit. JAC Antimicrob. Resist. 2019, 1, dlz072. [CrossRef] [PubMed]
- WHO. Anatomical Therapeutic Chemical (ATC) Classification. 2021. Available online: https://www.who.int/tools/atc-ddd-toolkit/atc-classification (accessed on 15 March 2022).
- Dlamini, N.N.; Meyer, J.C.; Kruger, D.; Kurdi, A.; Godman, B.; Schellack, N. Feasibility of using point prevalence surveys to assess antimicrobial utilisation in public hospitals in South Africa: A pilot study and implications. Hosp. Pract. 2019, 47, 88–95. [Google Scholar] [CrossRef] [Green Version]
- Skosana, P.P.; Schellack, N.; Godman, B.; Kurdi, A.; Bennie, M.; Kruger, D.; Meyer, J.C. A point prevalence survey of antimicrobial utilisation patterns and quality indices amongst hospitals in South Africa; findings and implications. Expert Rev. Anti-Infect. Ther. 2021, 19, 1353–1366. [Google Scholar] [CrossRef]
- Saleem, Z.; Hassali, M.A.; Versporten, A.; Godman, B.; Hashmi, F.K.; Goossens, H.; Saleem, F. A multicenter point prevalence survey of antibiotic use in Punjab, Pakistan: Findings and implications. Expert Rev. Anti-Infect. Ther. 2019, 17, 285–293. [Google Scholar] [CrossRef] [Green Version]
- Kurdi, A.; Hasan, A.J.; Baker, K.I.; Seaton, R.A.; Ramzi, Z.S.; Sneddon, J.; Godman, B. A multicentre point prevalence survey of hospital antibiotic prescribing and quality indices in the Kurdistan regional government of Northern Iraq: The need for urgent action. Expert Rev. Anti-Infect. Ther. 2021, 19, 805–814. [Google Scholar] [CrossRef]
- Gwebu, P.C.; Meyer, J.C.; Schellack, N.; Matsebula-Myeni, Z.C.; Godman, B. A web-based point prevalence survey of antimicrobial use and quality indicators at Raleigh Fitkin Memorial Hospital in the Kingdom of Eswatini and the implications. Hosp. Pract. 2022, 1–8. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).