Multicenter Surveillance of Antimicrobial Resistance among Gram-Negative Bacteria Isolated from Bloodstream Infections in Ghana
Abstract
1. Introduction
2. Results
2.1. Characteristics of the Study Samples
2.2. Comparison between the Bacteria Isolated at the Sentinel and Reference Laboratories
2.3. Antimicrobial Drug Susceptibility of the Gram-Negative Blood Isolates
3. Discussion
4. Materials and Methods
4.1. Study Design, Sites, and Sampling
4.2. Laboratory Analysis
4.3. Data Analysis
4.4. Ethical Approval
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Laupland, K.B. Incidence of bloodstream infection: A review of population-based studies. Clin. Microbiol. Infect. 2013, 19, 492–500. [Google Scholar] [CrossRef] [PubMed]
- Obeng-Nkrumah, N.; Labi, A.-K.; Acquah, M.E.; Donkor, E.S. Bloodstream infections in patients with malignancies: Implications for antibiotic treatment in a Ghanaian tertiary setting. BMC Res. Notes 2015, 8, 742. [Google Scholar] [CrossRef] [PubMed]
- Amanati, A.; Sajedianfard, S.; Khajeh, S.; Ghasempour, S.; Mehrangiz, S.; Nematolahi, S.; Shahhosein, Z. Bloodstream infections in adult patients with malignancy, epidemiology, microbiology, and risk factors associated with mortality and multi-drug resistance. BMC Infect. Dis. 2021, 21, 636. [Google Scholar] [CrossRef] [PubMed]
- Ginzburg, A.; Goldstein, G.; Raviv, D.; Cohen, H.; Weinreb, S.; Harlev, D.; Nitsan-Luques, A.; Saoud, M.Y.A.; Strahilevitz, J.; Averbuch, D. Bacteremia in children with solid tumors: Etiology, antimicrobial susceptibility, factors associated with multidrug resistance, and mortality. Microb. Drug Resist. 2022, 28, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Pai, S.; Enoch, D.A.; Aliyu, S.H. Bacteremia in children: Epidemiology, clinical diagnosis and antibiotic treatment. Expert Rev. Anti Infect. Ther. 2015, 13, 1073–1088. [Google Scholar] [CrossRef]
- Obeng-Nkrumah, N.; Labi, A.-K.; Addison, N.O.; Labi, J.E.M.; Awuah-Mensah, G. Trends in paediatric and adult bloodstream infections at a Ghanaian referral hospital: A retrospective study. Ann. Clin. Microbiol. Antimicrob. 2016, 15, 49. [Google Scholar] [CrossRef]
- Hattori, H.; Maeda, M.; Nagatomo, Y.; Takuma, T.; Niki, Y.; Naito, Y.; Sasaki, T.; Ishino, K. Epidemiology and risk factors for mortality in bloodstream infections: A single-center retrospective study in Japan. Am. J. Infect. Control 2018, 46, e75–e79. [Google Scholar] [CrossRef]
- Tsuzuki, S.; Matsunaga, N.; Yahara, K.; Shibayama, K.; Sugai, M.; Ohmagari, N. Disease burden of bloodstream infections caused by antimicrobial-resistant bacteria: A population-level study, Japan, 2015–2018. Int. J. Infect. Dis. 2021, 108, 119–124. [Google Scholar] [CrossRef]
- De Kraker, M.E.; Davey, P.G.; Grundmann, H.; BURDEN Study Group. Mortality and hospital stay associated with resistant Staphylococcus aureus and Escherichia coli bacteremia: Estimating the burden of antibiotic resistance in Europe. PLoS Med. 2011, 8, e1001104. [Google Scholar] [CrossRef] [PubMed]
- Stewardson, A.J.; Allignol, A.; Beyersmann, J.; Graves, N.; Schumacher, M.; Meyer, R.; Tacconelli, E.; De Angelis, G.; Farina, C.; Pezzoli, F.; et al. The health and economic burden of bloodstream infections caused by antimicrobial-susceptible and non-susceptible Enterobacteriaceae and Staphylococcus aureus in European hospitals, 2010 and 2011: A multicentre retrospective cohort study. Eurosurveillance 2016, 21, 30319. [Google Scholar] [CrossRef]
- Schechner, V.; Wulffhart, L.; Temkin, E.; Feldman, S.F.; Nutman, A.; Shitrit, P.; Schwaber, M.J.; Carmeli, Y. One-year mortality and years of potential life lost following bloodstream infection among adults: A nationwide population based study. Lancet Reg. Health Eur. 2022, 23, 100511. [Google Scholar] [CrossRef] [PubMed]
- Marchello, C.S.; Dale, A.P.; Pisharody, S.; Rubach, M.P.; Crump, J.A. A systematic review and meta-analysis of the prevalence of community-onset bloodstream infections among hospitalized patients in Africa and Asia. Antimicrob. Agents Chemother. 2019, 64, e01974-19. [Google Scholar] [CrossRef] [PubMed]
- Iroh Tam, P.Y.; Musicha, P.; Kawaza, K.; Cornick, J.; Denis, B.; Freyne, B.; Everett, D.; Dube, Q.; French, N.; Feasey, N.; et al. Emerging resistance to empiric antimicrobial regimens for pediatric bloodstream infections in Malawi (1998–2017). Clin. Infect. Dis. 2019, 69, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Musicha, P.; Cornick, J.E.; Bar-Zeev, N.; French, N.; Masesa, C.; Denis, B.; Kennedy, N.; Mallewa, J.; Gordon, M.A.; Msefula, C.L.; et al. Trends in antimicrobial resistance in bloodstream infection isolates at a large urban hospital in Malawi (1998–2016): A surveillance study. Lancet Infect. Dis. 2017, 17, 1042–1052. [Google Scholar] [CrossRef] [PubMed]
- Cross, A.; Levine, M.M. Patterns of bacteraemia aetiology. Lancet Infect. Dis. 2017, 17, 1005–1006. [Google Scholar] [CrossRef] [PubMed]
- Labi, A.-K.; Obeng-Nkrumah, N.; Addison, N.O.; Donkor, E.S. Salmonella blood stream infections in a tertiary care setting in Ghana. BMC Infect. Dis. 2014, 14, 3857. [Google Scholar] [CrossRef]
- Marks, F.; von Kalckreuth, V.; Aaby, P.; Adu-Sarkodie, Y.; El Tayeb, M.A.; Ali, M.; Aseffa, A.; Baker, S.; Biggs, H.M.; Bjerregaard-Andersen, M.; et al. Incidence of invasive salmonella disease in sub-Saharan Africa: A multicentre population-based surveillance study. Lancet Glob. Health 2017, 5, e310–e323. [Google Scholar] [CrossRef]
- Sader, H.S.; Castanheira, M.; Streit, J.M.; Flamm, R.K. Frequency of occurrence and antimicrobial susceptibility of bacteria isolated from patients hospitalized with bloodstream infections in United States medical centers (2015–2017). Diagn. Microbiol. Infect. Dis. 2019, 95, 114850. [Google Scholar] [CrossRef]
- Weiner-Lastinger, L.M.; Abner, S.; Edwards, J.R.; Kallen, A.J.; Karlsson, M.; Magill, S.S.; Pollock, D.; See, I.; Soe, M.M.; Walters, M.S.; et al. Antimicrobial-resistant pathogens associated with adult healthcare-associated infections: Summary of data reported to the National Healthcare Safety Network, 2015–2017. Infect. Control Hosp. Epidemiol. 2020, 41, 1–18. [Google Scholar] [CrossRef]
- Weiner-Lastinger, L.M.; Abner, S.; Benin, A.L.; Edwards, J.R.; Kallen, A.J.; Karlsson, M.; Magill, S.S.; Pollock, D.; See, I.; Soe, M.M.; et al. Antimicrobial-resistant pathogens associated with pediatric healthcare-associated infections: Summary of data reported to the National Healthcare Safety Network, 2015–2017. Infect. Control Hosp. Epidemiol. 2020, 41, 19–30. [Google Scholar] [CrossRef]
- GBD 2017 Non-Typhoidal Salmonella Invasive Disease Collaborators. The global burden of non-typhoidal salmonella invasive disease: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Infect. Dis. 2019, 19, 1312–1324. [Google Scholar] [CrossRef] [PubMed]
- Still, W.L.; Tapia, M.D.; Tennant, S.M.; Sylla, M.; Touré, A.; Badji, H.; Keita, A.M.; Sow, S.O.; Levine, M.M.; Kotloff, K.L. Surveillance for invasive salmonella disease in Bamako, Mali, from 2002 to 2018. Clin. Infect. Dis. 2020, 71 (Suppl. 2), S130–S140. [Google Scholar] [CrossRef]
- Mandomando, I.; Macete, E.; Sigaúque, B.; Morais, L.; Quintó, L.; Sacarlal, J.; Espasa, M.; Vallès, X.; Bassat, Q.; Aide, P.; et al. Invasive non-typhoidal Salmonella in Mozambican children. Trop. Med. Int. Health 2009, 14, 1467–1474. [Google Scholar] [CrossRef] [PubMed]
- Feasey, N.A.; Dougan, G.; Kingsley, R.A.; Heyderman, R.S.; Gordon, M.A. Invasive non-typhoidal salmonella disease: An emerging and neglected tropical disease in Africa. Lancet 2012, 379, 2489–2499. [Google Scholar] [CrossRef] [PubMed]
- Russo, T.A.; Johnson, J.R. Diseases Caused by Gram-Negative Enteric Bacilli. In Harrison’s Principles of Internal Medicine, 20th ed.; Jameson, J.L., Fauci, A.S., Kasper, D.L., Hauser, S.L., Longo, D.L., Loscalzo, J., Eds.; McGraw-Hill Education/Medical: New York, NY, USA, 2018. [Google Scholar]
- Lee, C.-R.; Lee, J.H.; Park, K.S.; Jeon, J.H.; Kim, Y.B.; Cha, C.-J.; Jeong, B.C.; Lee, S.H. Antimicrobial resistance of hypervirulent Klebsiella pneumoniae: Epidemiology, hypervirulence-associated determinants, and resistance mechanisms. Front. Cell. Infect. Microbiol. 2017, 7, 483. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Guo, J. Hypervirulent Klebsiella pneumoniae (hypermucoviscous and aerobactin positive) infection over 6 years in the elderly in China: Antimicrobial resistance patterns, molecular epidemiology and risk factor. Ann. Clin. Microbiol. Antimicrob. 2019, 18, 4. [Google Scholar] [CrossRef] [PubMed]
- Seale, A.C.; Gordon, N.; Islam, J.; Peacock, S.J.; Scott, J.A.G. AMR Surveillance in low and middle-income settings—A roadmap for participation in the Global Antimicrobial Surveillance System (GLASS). Wellcome Open Res. 2017, 2, 92. [Google Scholar] [CrossRef]
- Collaborators, A.R. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar]
- Murray, C.J.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. 2016. Available online: https://amr-review.org/sites/default/files/160518_Final%20paper_with%20cover.pdf (accessed on 1 May 2020).
- O’Neill, J. “Review on Antimicrobial Resistance.” Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations. 2014. Available online: https://amr-review.org/sites/default/files/AMR%20Review%20Paper%20-%20Tackling%20a%20crisis%20for%20the%20health%20and%20wealth%20of%20nations_1.pdf (accessed on 16 November 2021).
- Simonsen, G.S.; Tapsall, J.W.; Allegranzi, B.; Talbot, E.A.; Lazzari, S. The antimicrobial resistance containment and surveillance approach--a public health tool. Bull. World Health Organ. 2004, 82, 928–934. [Google Scholar]
- Dar, O.A.; Hasan, R.; Schlundt, J.; Harbarth, S.; Caleo, G.; Dar, F.K.; Littmann, J.; Rweyemamu, M.; Buckley, E.J.; Shahid, M.; et al. Exploring the evidence base for national and regional policy interventions to combat resistance. Lancet 2016, 387, 285–295. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.M.; Wertheim, H.F.L.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H.; et al. Antibiotic resistance – the need for global solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- Opintan, J.A.; Newman, M.J.; Arhin, R.E.; Donkor, E.S.; Gyansa-Lutterodt, M.; Mills-Pappoe, W. Laboratory-based nationwide surveillance of antimicrobial resistance in Ghana. Infect. Drug Resist. 2015, 8, 379–389. [Google Scholar] [CrossRef]
- Opintan, J.A.; Newman, M.J. Prevalence of antimicrobial resistant pathogens from blood cultures: Results from a laboratory based nationwide surveillance in Ghana. Antimicrob. Resist. Infect. Control 2017, 6, 64. [Google Scholar] [CrossRef]
- Kohli, R.; Omuse, G.; Revathi, G. Antibacterial susceptibility patterns of blood stream isolates in patients investigated at the Aga Khan University Hospital, Nairobi. East Afr. Med. J. 2010, 87, 74–80. [Google Scholar] [CrossRef]
- Toy, T.; Pak, G.D.; Duc, T.P.; Campbell, J.I.; El Tayeb, M.A.; Von Kalckreuth, V.; Im, J.; Panzner, U.; Cruz Espinoza, L.M.; Eibach, D.; et al. Multicountry distribution and characterization of extended-spectrum beta-lactamase-associated Gram-negative bacteria from bloodstream infections in sub-Saharan Africa. Clin. Infect. Dis. 2019, 69 (Suppl. 6), S449–S458. [Google Scholar] [CrossRef]
- WHO. WHO Antibiotic Categorization. Available online: https://aware.essentialmeds.org/groups (accessed on 18 February 2021).
- Lester, R.; Musicha, P.; van Ginneken, N.; Dramowski, A.; Hamer, D.H.; Garner, P.; Feasey, N.A. Prevalence and outcome of bloodstream infections due to third-generation cephalosporin-resistant Enterobacteriaceae in sub-Saharan Africa: A systematic review. J. Antimicrob. Chemother. 2020, 75, 492–507. [Google Scholar] [CrossRef]
- Tack, B.; Vanaenrode, J.; Verbakel, J.Y.; Toelen, J.; Jacobs, J. Invasive non-typhoidal Salmonella infections in sub-Saharan Africa: A systematic review on antimicrobial resistance and treatment. BMC Med. 2020, 18, 212. [Google Scholar] [CrossRef]
- WHO. Critically Important Antimicrobials for Human Medicine, 6th ed.; World Health Organization: Geneva, Switzerland, 2019.
- Papp-Wallace, K.M.; Endimiani, A.; Taracila, M.A.; Bonomo, R.A. Carbapenems: Past, present, and future. Antimicrob. Agents Chemother. 2011, 55, 4943–4960. [Google Scholar] [CrossRef]
- Legese, M.H.; Asrat, D.; Swedberg, G.; Hasan, B.; Mekasha, A.; Getahun, T.; Worku, M.; Shimber, E.T.; Getahun, S.; Ayalew, T.; et al. Sepsis: Emerging pathogens and antimicrobial resistance in Ethiopian referral hospitals. Antimicrob. Resist. Infect. Control 2022, 11, 83. [Google Scholar] [CrossRef]
- Labi, A.K.; Enweronu-Laryea, C.C.; Nartey, E.T.; Bjerrum, S.; Ayibor, P.K.; Andersen, L.P.; Newman, M.J.; Kurtzhals, J.A. Bloodstream Infections at two neonatal intensive care units in Ghana: Multidrug resistant Enterobacterales undermine the usefulness of standard antibiotic regimes. Pediatr. Infect. Dis. J. 2021, 40, 1115–1121. [Google Scholar] [CrossRef] [PubMed]
- Mhondoro, M.; Ndlovu, N.; Bangure, D.; Juru, T.; Gombe, N.T.; Shambira, G.; Nsubuga, P.; Tshimanga, M. Trends in antimicrobial resistance of bacterial pathogens in Harare, Zimbabwe, 2012–2017: A secondary dataset analysis. BMC Infect. Dis. 2019, 19, 746. [Google Scholar] [CrossRef] [PubMed]
- Brink, A.J. Epidemiology of carbapenem-resistant Gram-negative infections globally. Curr. Opin. Infect. Dis. 2019, 32, 609–616. [Google Scholar] [CrossRef] [PubMed]
- M100; Performance Standard for Antimicrobial Susceptibility Testing. 31st ed. Clinical and Laboratory Standards Institute (CLSI): Wayne, PA, USA, 2021.
| Number | % | ||
|---|---|---|---|
| Sentinel Site | 37 Military Hospital | 11 | 3.3 |
| Accra Regional Hospital | 56 | 16.8 | |
| Eastern Regional Hospital | 41 | 12.3 | |
| Effia Nkwanta Regional Hospital | 22 | 6.6 | |
| Komfo Anokye Teaching Hospital | 34 | 10.2 | |
| Korle Bu Teaching Hospital | 93 | 27.8 | |
| Princess Marie Louise Children’s Hospital | 30 | 9 | |
| Private Labs | 31 | 9.3 | |
| St. Patrick’s Hospital | 9 | 2.7 | |
| Volta Regional Hospital | 7 | 2.1 | |
| City | Accra | 221 | 66.2 |
| Effia Nkwanta | 22 | 6.6 | |
| Ho | 7 | 2.1 | |
| Koforidua | 41 | 12.3 | |
| Kumasi | 34 | 10.2 | |
| Maase-Offinso | 9 | 2.7 | |
| Region | Ashanti | 43 | 12.9 |
| Eastern | 41 | 12.3 | |
| Greater Accra | 221 | 66.2 | |
| Volta | 7 | 2.1 | |
| Western | 22 | 6.6 | |
| Gender | Female | 78 | 23.4 |
| Male | 76 | 22.8 | |
| Unknown | 180 | 53.9 | |
| Age groups | under 5 years | 85 | 25.0 |
| 5–19 years | 22 | 6.5 | |
| 20–59 years | 55 | 16.2 | |
| 60+ years | 18 | 5.3 | |
| Unknown | 160 | 47.1 | |
| Diagnosis | Bacteremia | 33 | 9.9 |
| Sepsis | 296 | 88.6 | |
| Otitis media | 1 | 0.3 | |
| Severe anemia & malaria | 1 | 0.3 | |
| Urosepsis | 2 | 0.6 | |
| Not indicated | 1 | 0.3 | |
| Sentinel Laboratory | Reference Laboratory | |||||
|---|---|---|---|---|---|---|
| Number | Percent | Number | Percent | Kappa Coefficient | p Value | |
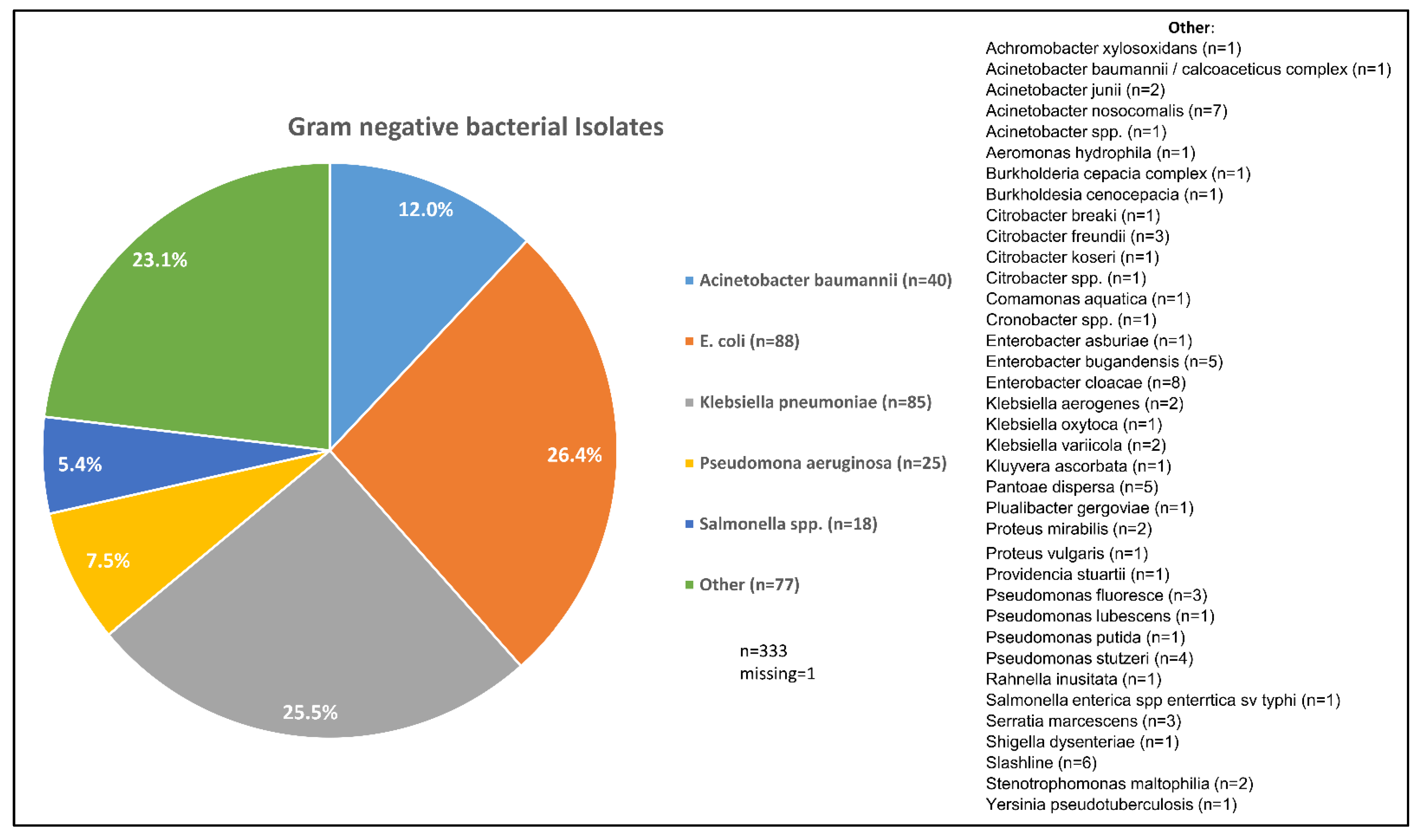
| Acinetobacter baumannii | 23 | 6.9 | 40 | 12.0 | 0.669 | <0.001 |
| Escherichia coli | 85 | 25.4 | 88 | 26.3 | 0.805 | <0.001 |
| Klebsiella pneumoniae | 67 | 20.1 | 85 | 25.4 | 0.796 | <0.001 |
| Pseudomonas aeruginosa | 22 | 6.6 | 25 | 7.5 | 0.840 | <0.001 |
| Non-typhoidal Salmonella | 21 | 6.3 | 18 | 5.4 | 0.864 | <0.001 |
| Other | 115 | 34.4 | 77 | 23.1 | ||
| Missing | 1 | 0.3 | 1 | 0.3 | ||
| Total | 334 | 100.0 | 334 | 100.0 | ||
| All Gram-Negative Isolates | Klebsiella pneumoniae | Non-typhoidal Salmonella | Acinetobacter baumannii | Escherichia coli | Pseudomonas aeruginosai | |
|---|---|---|---|---|---|---|
| Total | 330 | 84 | 18 | 40 | 88 | 25 |
| Meropenem | 92 a (28.0%) | 20 (23.8%) | 3 (16.7%) | 12 g (30.8%) | 23 (26.1%) | 13 (52.0%) |
| Tetracycline | 251 (76.1%) | 75 (89.3%) | 6 (33.3%) | 22 (55.0%) | 71 (80.7%) | 23 (92.0%) |
| Chloramphenicol | 197 (59.7%) | 59 (70.2%) | 5 (27.8%) | 35 (87.5%) | 36 (40.9%) | 19 (76.0%) |
| Gentamicin | 157 b (53.8%) | 63 e (82.9%) | 4 f (23.5%) | 14 g (45.2%) | 34 e (44.7%) | 6 (24.0%) |
| Cefotaxime | 256 a (77.8%) | 72 (85.7%) | 7 (38.9%) | 36 (90.0%) | 64 h (73.6%) | 21 (84.0%) |
| Azithromycin | 138 c (42.1%) | 39 (46.4%) | 4 f (23.5%) | 17 (42.5%) | 41 (46.6%) | 9 (36.0%) |
| Ciprofloxacin | 177 a (55.3%) | 62 (73.8%) | 5 (27.8%) | 15 g (44.1%) | 54 (61.4%) | 8 (32.0%) |
| Ceftriaxone | 227 d (73.7%) | 70 e (84.3%) | 4 (22.2%) | 24 g (88.9%) | 67 h (77.9%) | 20 (80.0%) |
| Ampicillin | 293 a (89.3%) | 83 e (100.0%) | 6 (33.3%) | 37 g (94.9%) | 77 (87.5%) | 24 (96.0%) |
| Ceftazidime | 178 (56.3%) | 58 (69.0%) | 5 (27.8%) | 19 g (57.6%) | 59 (67.0%) | 10 (40.0%) |
| Ertapenem | 146 a (44.4%) | 25 (29.8%) | 3 (16.7%) | 36 (90.0%) | 26 (29.5%) | 21 (84.0%) |
| Nalidixic acid | 209 (63.3%) | 49 (58.3%) | 3 (16.7%) | 20 (50.0%) | 68 (77.3%) | 23 (92.0%) |
| Trimethoprim-sulfamethoxazole | 236 (71.5%) | 72 (85.7%) | 6 (33.3%) | 23 (57.5%) | 63 (71.6%) | 23 (92.0%) |
| Multidrug resistant | 290 (88.0%) | 81 (96.4%) | 7 (38.9%) | 39 (97.5%) | 83 (94.3%) | 25 (100.0%) |
| Number Tested | Multidrug Resistant N (%) | p-Value | ||
|---|---|---|---|---|
| Sentinel Site | 37 Military Hospital | 11 | 9 (81.8) | 0.315 |
| Accra Regional Hospital | 56 | 45 (80.4) | ||
| Eastern Regional Hospital | 41 | 36 (90.0) | ||
| Effia Nkwanta Regional Hospital | 22 | 17 (81.0) | ||
| Komfo Anokye Teaching Hospital | 34 | 33 (97.1) | ||
| Korle Bu Teaching Hospital | 93 | 83 (91.2) | ||
| Princess Marie Louise Children’s Hospital | 30 | 25 (83.3) | ||
| Private labs | 31 | 26 (83.8) | ||
| St. Patrick’s Hospital | 9 | 7 (100.0) | ||
| Volta Regional Hospital | 7 | 7 (100.0) | ||
| City | Accra | 219 | 188 (85.8) | 0.231 |
| Effia Nkwanta | 21 | 17 (81.0) | ||
| Ho | 7 | 7 (100.0) | ||
| Koforidua | 40 | 36 (90.0) | ||
| Kumasi | 34 | 33 (97.1) | ||
| Maase-Offinso | 9 | 9 (100.0) | ||
| Region | Ashanti | 43 | 42 (97.7) | 0.318 |
| Eastern | 40 | 36 (90.0) | ||
| Greater Accra | 219 | 188 (85.8) | ||
| Volta | 7 | 7 (100.0) | ||
| Western | 21 | 17 (81.0) | ||
| Gender | Female | 78 | 70 (89.7) | 0.534 |
| Male | 76 | 64 (86.5) | ||
| Age | <5 years | 85 | 74 (87.1) | 0.962 |
| 5–19 years | 22 | 19 (86.4) | ||
| 20–59 years | 51 | 45 (88.2) | ||
| 60+ years | 18 | 15 (83.3) | ||
| Sentinel Sites | Reference Laboratory and Coordinating Centre |
|---|---|
| Greater Accra Regional Hospital | Department of Medical Microbiology, University of Ghana Medical School *. |
| Princess Marie Louise Children’s Hospital | |
| Korle Bu Teaching Hospital * | |
| 37 Military Hospital | |
| Eastern Regional Hospital, Koforidua * | |
| Komfo Anokye Teaching Hospital | |
| Two private laboratories | |
| Bolgatanga Regional Hospital | |
| Volta Regional Hospital * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donkor, E.S.; Muhsen, K.; Johnson, S.A.M.; Kotey, F.C.N.; Dayie, N.T.K.D.; Tetteh-Quarcoo, P.B.; Tette, E.M.A.; Osei, M.-M.; Egyir, B.; Nii-Trebi, N.I.; et al. Multicenter Surveillance of Antimicrobial Resistance among Gram-Negative Bacteria Isolated from Bloodstream Infections in Ghana. Antibiotics 2023, 12, 255. https://doi.org/10.3390/antibiotics12020255
Donkor ES, Muhsen K, Johnson SAM, Kotey FCN, Dayie NTKD, Tetteh-Quarcoo PB, Tette EMA, Osei M-M, Egyir B, Nii-Trebi NI, et al. Multicenter Surveillance of Antimicrobial Resistance among Gram-Negative Bacteria Isolated from Bloodstream Infections in Ghana. Antibiotics. 2023; 12(2):255. https://doi.org/10.3390/antibiotics12020255
Chicago/Turabian StyleDonkor, Eric S., Khitam Muhsen, Sherry A. M. Johnson, Fleischer C. N. Kotey, Nicholas T. K. D. Dayie, Patience B. Tetteh-Quarcoo, Edem M. A. Tette, Mary-Magdalene Osei, Beverly Egyir, Nicholas I. Nii-Trebi, and et al. 2023. "Multicenter Surveillance of Antimicrobial Resistance among Gram-Negative Bacteria Isolated from Bloodstream Infections in Ghana" Antibiotics 12, no. 2: 255. https://doi.org/10.3390/antibiotics12020255
APA StyleDonkor, E. S., Muhsen, K., Johnson, S. A. M., Kotey, F. C. N., Dayie, N. T. K. D., Tetteh-Quarcoo, P. B., Tette, E. M. A., Osei, M.-M., Egyir, B., Nii-Trebi, N. I., Owusu-Okyere, G., Owusu-Ofori, A., Amir, Y., Perlman, S., Lopes, P. H., Mfodwo, A., Gordon, N. C., Gresham, L., Smolinski, M., & Cohen, D. (2023). Multicenter Surveillance of Antimicrobial Resistance among Gram-Negative Bacteria Isolated from Bloodstream Infections in Ghana. Antibiotics, 12(2), 255. https://doi.org/10.3390/antibiotics12020255








