Occult Vancomycin-Resistant Enterococcus faecium ST117 Displaying a Highly Mutated vanB2 Operon
Abstract
1. Introduction
2. Results
2.1. Phenotypic and Genotypic (PCR) Characterization of E. faecium Isolates
2.2. Multi Locus Sequence Typing (MLST)
2.3. WGS In Silico Analysis
2.4. CgMLST Phylogenetic Analysis
3. Discussion
4. Materials and Methods
4.1. E. faecium Isolates, Antimicrobial Susceptibility
4.2. Molecular Characterization
4.2.1. Screening for vanA, -B, -C Genes and MLST
4.2.2. WGS and In Silico Analysis
4.3. Phylogenetic Analysis by cgMLST
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arias, C.A.; Murray, B.E. The rise of the Enterococcus: Beyond vancomycin resistance. Nat. Rev. Microbiol. 2012, 10, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Cheah, A.L.Y.; Spelman, T.; Liew, D.; Peel, T.; Howden, B.P.; Spelman, D.; Graysonca, M.L.; Nationa, R.L.; Konga, D.D.M. Enterococcal bacteraemia: Factors influencing mortality, length of stay and costs of hospitalization. Clin. Microbiol. Infect. 2013, 19, 384. [Google Scholar] [CrossRef] [PubMed]
- Cattoir, V.; Leclercq, R. Twenty-five years of shared life with vancomycin-resistant enterococci: Is it time to divorce. J. Antimicrob. Chemother. 2013, 68, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Brinkwirth, S.; Ayobami, O.; Eckmanns, T.; Markwart, R. Hospital-acquired infections caused by enterococci: A systematic review and meta-analysis, WHO European Region, 1 january 2010 to 4 february 2020. Eurosurveillance 2021, 26, 2001628. [Google Scholar] [CrossRef] [PubMed]
- Correa-Martínez, C.L.; Jurke, A.; Schmitz, J.; Schaumburg, F.; Kampmeier, S.; Mellmann, A. Molecular Epidemiology of Vancomycin-Resistant Enterococci Bloodstream Infections in Germany: A Population-Based Prospective Longitudinal Study. Microorganisms 2022, 10, 130. [Google Scholar] [CrossRef]
- Leavis, H.L.; Willems, R.J.L.; van Wamel, W.J.B.; Schuren, F.H.; Caspers, M.P.M.; Bonten, M.J.M. Insertion sequence-driven diversification creates a globally dispersed emerging multiresistant subspecies of E. faecium. PLoS Pathog. 2007, 3, e7. [Google Scholar] [CrossRef]
- Hegstad, K.; Mikalsen, T.; Coque, T.M.; Werner, G.; Sundsfjord, A. Mobile genetic elements and their contribution to the emergence of antimicrobial resistant Enterococcus faecalis and Enterococcus faecium. Clin. Microbiol. Infect. 2010, 16, 541–554. [Google Scholar] [CrossRef]
- Lee, T.; Pang, S.; Abraham, S.; Coombs, G.W. Antimicrobial-resistant CC17 Enterococcus faecium: The past, the present and the future. J. Glob. Antimicrob. Resist. 2019, 16, 36–47. [Google Scholar] [CrossRef]
- Walker, S.V.; Wolke, M.; Plum, G.; Weber, R.E.; Werner, G.; Hamprecht, A. Failure of Vitek2 to reliably detect vanB-mediated vancomycin resistance in Enterococcus faecium. J. Antimicrob. Chemother. 2021, 76, 1698–1702. [Google Scholar] [CrossRef] [PubMed]
- Hammerum, A.M.; Justesen, U.S.; Pinholt, M.; Roer, L.; Kaya, H.; Worning, P.; Nygaard, S.; Kemp, M.; Clausen, M.E.; Nielsen, K.L.; et al. Surveillance of vancomycin-resistant enterococci reveals shift in dominating clones and national spread of a vancomycin-variable vanA Enterococcus faecium ST1421-CT1134 clone, Denmark, 2015 to March 2019. Eurosurveillance 2019, 24, 1900503. [Google Scholar] [CrossRef]
- Gagnon, S.; Lévesque, S.; Lefebvre, B.; Bourgault, A.-M.; Labbé, A.-C.; Roger, M. vanA-containing Enterococcus faecium susceptible to vancomycin and teicoplanin because of major nucleotide deletions in Tn1546. J. Antimicrob. Chemother. 2011, 66, 2758–2762. [Google Scholar] [CrossRef]
- Sivertsen, A.; Pedersen, T.; Larssen, K.W.; Bergh, K.; Rønning, T.G.; Radtke, A.; Hegstada, K. A silenced vanA gene cluster on a transferable plasmid caused an outbreak of vancomycin-variable enterococci. Antimicrob. Agents Chemother. 2016, 60, 4119–4127. [Google Scholar] [CrossRef]
- Wagner, T.M.; Janice, J.; Sivertsen, A.; Sjögren, I.; Sundsfjord, A.; Hegstad, K. Alternative vanHAX promoters and increased vanA-plasmid copy number resurrect silenced glycopeptide resistance in Enterococcus faecium. J. Antimicrob. Chemother. 2021, 76, 876–882. [Google Scholar] [CrossRef] [PubMed]
- Thaker, M.N.; Kalan, L.; Waglechner, N.; Eshaghi, A.; Patel, S.N.; Poutanen, S.; Willey, B.; Coburn, B.; McGeer, A.; Low, D.E.; et al. Vancomycin-variable enterococci can give rise to constitutive resistance during antibiotic therapy. Antimicrob. Agents Chemother. 2015, 59, 1405–1410. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Kurushima, J.; Nomura, T.; Tanimoto, K.; Tamai, K.; Yanagisawa, H.; Shirabe, K.; Iket, Y.; Tomita, H. Dissemination and genetic analysis of the stealthy vanB gene clusters of Enterococcus faecium clinical isolates in Japan. BMC Microbiol. 2018, 18, 213. [Google Scholar] [CrossRef] [PubMed]
- De Been, M.; Pinholt, M.; Top, J.; Bletz, S.; Mellmann, A.; van Schaik, W.; Brouwer, E.; Rogers, M.; Kraat, Y.; Bonten, M.; et al. Core genome multilocus sequence typing scheme for high-resolution typing of Enterococcus faecium. J. Clin. Microbiol. 2015, 53, 3788–3797. [Google Scholar] [CrossRef]
- Guzman, P.A.M.; van Schaik, W.; Rogers, M.R.C.; Coque, T.M.; Baquero, F.; Corander, J.; Willems, R.J.L. Global Emergence and Dissemination of Enterococci as Nosocomial Pathogens: Attack of the Clones? Front. Microbiol. 2016, 7, 788. [Google Scholar] [CrossRef]
- Merlino, J.; Gray, T. Vancomycin variable Enterococcus (VVE), E. faecium, harbouring the vanA gene complex. Pathology 2021, 53, 680–682. [Google Scholar] [CrossRef]
- Lu, J.J.; Perng, C.L.; Ho, M.F.; Chiueh, T.S.; Lee, W.H. High prevalence of VanB2 vancomycin-resistant Enterococcus faecium in Taiwan. J. Clin. Microbiol. 2001, 39, 2140–2145. [Google Scholar] [CrossRef] [PubMed]
- Guffey, A.A.; Loll, P.J. Regulation of Resistance in Vancomycin-Resistant Enterococci: The VanRS Two-Component System. Microorganisms 2021, 9, 2026. [Google Scholar] [CrossRef]
- Stogios, P.J.; Savchenko, A. Molecular mechanisms of vancomycin resistance. Protein Sci. 2020, 29, 654–669. [Google Scholar] [CrossRef] [PubMed]
- Freitas, A.R.; Pereira, A.P.; Novais, C.; Peixe, L. Multidrug-resistant high-risk Enterococcus faecium clones: Can we really define them? Int. J. Antimicrob. Agents 2021, 57, 106227. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.; Maechler, F.; Schwab, F.; Gastmeier, P.; Kola, A. Increase of vancomycin-resistant Enterococcus faecium strain type ST117 CT71 at Charité—Universitätsmedizin Berlin, 2008 to 2018. Antimicrob. Resist. Infect. Control 2020, 9, 109. [Google Scholar] [CrossRef]
- Rodríguez-Lucas, C.; Fernández, J.; Raya, C.; Bahamonde, A.; Quiroga, A.; Muñoz, R.; Rodicio, M.R. Establishment and Persistence of Glycopeptide-Resistant Enterococcus faecium ST80 and ST117 Clones within a Health Care Facility Located in a Low-Prevalence Geographical Region. Microb. Drug Resist. 2022, 28, 217–221. [Google Scholar] [CrossRef]
- Lisotto, P.; Couto, N.; Rosema, S.; Lokate, M.; Zhou, X.; Bathoorn, E.; Harmsen, H.J.M.; Friedrich, A.W.; Rossen, J.W.A.; Chlebowicz-Fliss, M.A. Molecular Characterisation of Vancomycin-Resistant Enterococcus faecium Isolates Belonging to the Lineage ST117/CT24 Causing Hospital Outbreaks. Front. Microbiol. 2021, 12, 728356. [Google Scholar] [CrossRef] [PubMed]
- Fioriti, S.; Simoni, S.; Caucci, S.; Morroni, G.; Ponzio, E.; Coccitto, S.N.; Brescini, L.; Cirioni, O.; Menzo, S.; Biavasco, F.; et al. Trend of clinical vancomycin-resistant enterococci isolated in a regional Italian hospital from 2001 to 2018. Braz. J. Microbiol. 2020, 51, 1607–1613. [Google Scholar] [CrossRef]
- The European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for Interpretation of MICs and Zone Diameters. Version 12.0. 2022. Available online: http://www.eucast.org (accessed on 30 March 2022).
- Santona, A.; Taviani, E.; Hoang, H.M.; Fiamma, M.; Deligios, M.; Ngo, T.V.Q.; Van, A.L.; Cappuccinelli, P.; Rubino, S.; Paglietti, B. Emergence of unusual vanA/vanB2 genotype in a highly mutated vanB2 -vancomycin-resistant hospital-associated E. faecium background in Vietnam. Int. J. Antimicrob. Agents 2018, 52, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Homan, W.L.; Tribe, D.; Poznanski, S.; Li, M.; Hogg, G.; Spalburg, E.; Van Embden, J.D.; Willems, R.J. Multilocus sequence typing scheme for Enterococcus faecium. J. Clin. Microbiol. 2002, 40, 1963–1971. [Google Scholar] [CrossRef]
- Zerbino, D.R.; Birney, E. Velvet: Algorithms for de novo short read assembly using de Bruijn graphs. Genome Res. 2008, 18, 821–829. [Google Scholar] [CrossRef]
- Jünemann, S.; Sedlazeck, F.J.; Prior, K.; Albersmeier, A.; John, U.; Kalinowski, J.; Mellmann, A.; Goesmann, A.; von Haeseler, A.; Stoye, J. Updating benchtop sequencing performance comparison. Nat. Biotechnol. 2013, 31, 294–296. [Google Scholar] [CrossRef] [PubMed]
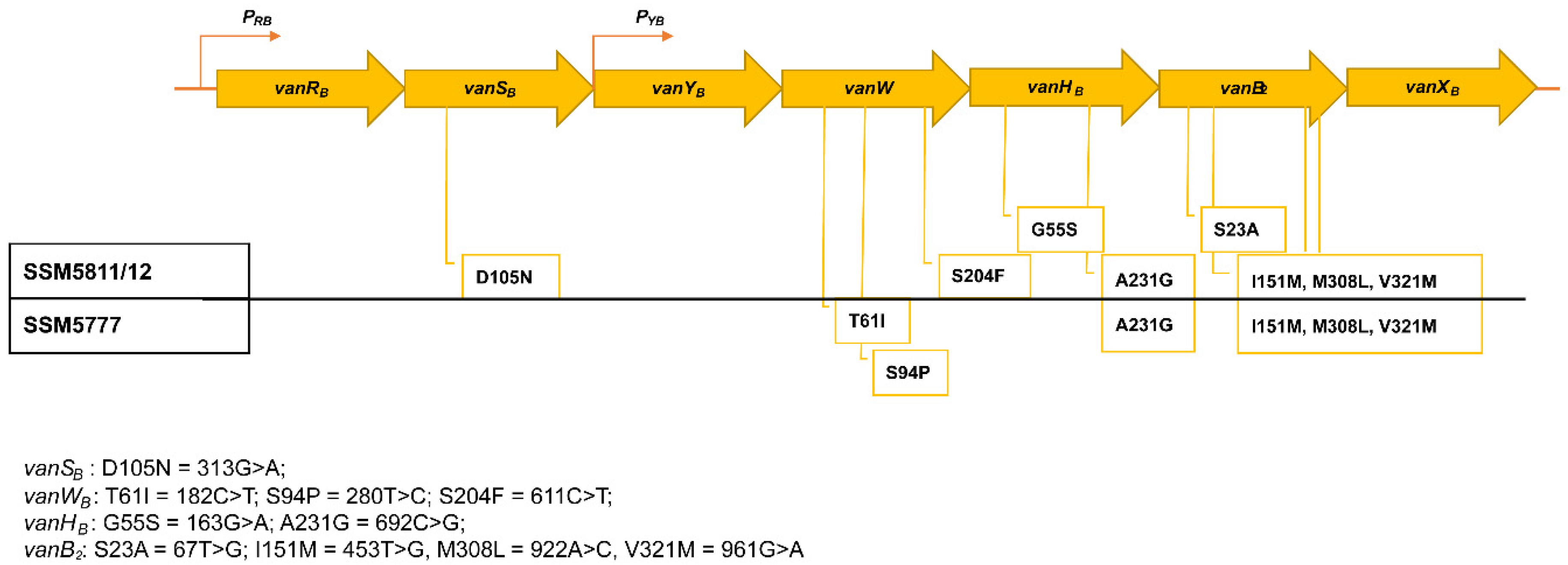
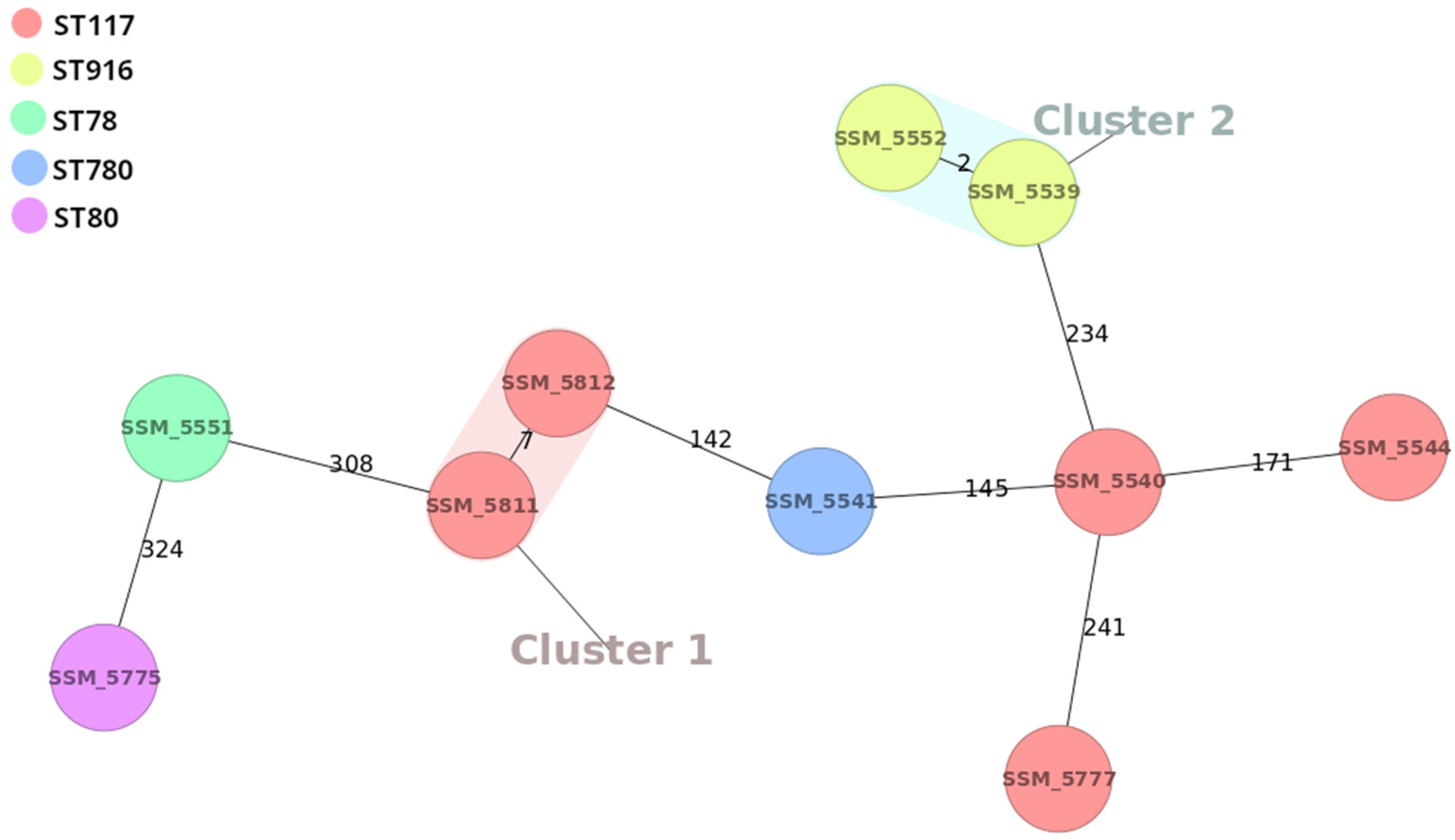
| SSM Strain | Date | Source | Ward | AMP | GEN | STR | ERY | CIP | IMI | LNZ | TGC | VAN mg/L | TEC mg/L | Van Genes | ST |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5538 | 4 February 2013 | B. aspirate | Medicine | R | R | R | R | R | R | S | S | 1 | ≤0.5 | - | 117 |
| 5550 | 6 March 2013 | Urine | Medicine | R | S | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 117 |
| 5563 | 4 April 2013 | Pus | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 78 |
| 5556 | 7 April 2013 | F. fluid | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 78 |
| 5542 | 17April 2013 | Sputum | Surgery | S | R | R | R | R | S | S | S | 1 | ≤0.5 | - | 78 |
| 5540 * | 22 April 2013 | CVC | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 117 |
| 5548 | 1 June 2013 | Wound | ICU | R | R | R | R | R | R | S | S | 1 | ≤0.5 | - | 78 |
| 5549 | 3 June 2013 | Bile | Surgery | R | R | R | R | R | R | S | S | 1 | ≤0.5 | - | 78 |
| 5551 * | 6 June 2013 | A. fluid | Surgery | R | R | R | R | R | R | S | S | 1 | ≤0.5 | - | 78 |
| 5562 | 8 June 2013 | Urine | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | 1 | - | 117 |
| 5555 | 1 July 2013 | Blood | Medicine | R | R | R | R | R | R | S | S | 1 | ≤0.5 | - | 78 |
| 5646 | 12 July 2013 | Urine | Surgery | R | R | R | R | R | R | S | S | 1 | 1 | - | 117 |
| 5560 | 19 July 2013 | B. aspirate | Medicine | S | S | S | R | R | S | S | S | ≤0.5 | ≤0.5 | - | 54 |
| 5539 * | 4 August 2013 | Bile | Surgery | R | S | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 916 |
| 5587 | 10 August 2013 | D. fluid | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 78 |
| 5588 | 10 August 2013 | B. aspirate | Medicine | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 916 |
| 5583 | 28 September 2013 | Biopsy | Medicine | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 117 |
| 5552 * | 6 October 2013 | Blood | Surgery | R | R | S | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 916 |
| 5591 | 2 November 2013 | Bile | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 78 |
| 5544 * | 4 November 2013 | B. aspirate | Surgery | R | R | R | R | R | R | S | S | ≤0.25 | ≤0.25 | - | 117 |
| 5592 | 4 November 2013 | Pus | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 78 |
| 5541 * | 4 December 2013 | F. fluid | Surgery | R | R | R | R | R | R | S | S | 1 | ≤0.12 | - | 780 |
| 5574 | 9 December 2013 | Wound | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 78 |
| 5652 | 28 December 2013 | Blood | Medicine | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 780 |
| 5662 | 21 February 2014 | A. fluid | Surgery | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 117 |
| 5670 | 26 February 2014 | B. aspirate | ICU | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 117 |
| 5673 | 20 March 014 | Blood | ICU | R | S | R | R | R | R | S | S | 1 | 2 | - | 117 |
| 5671 | 27 March 2014 | Blood | ICU | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 117 |
| 5653 | 2 April 2014 | Blood | ICU | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 117 |
| 5655 | 1 May 2014 | Bile | Surgery | R | R | R | R | R | R | S | S | 1 | ≤0.5 | - | 78 |
| 5654 | 1 May 2014 | Urine | Medicine | R | R | R | R | R | R | S | S | ≤0.5 | 1 | - | 117 |
| 5658 | 2 May 2014 | Urine | Medicine | R | R | R | R | R | R | S | S | ≤0.5 | 2 | - | 117 |
| 5656 | 2 May 2014 | Pus | ICU | S | S | S | R | R | S | S | S | ≤0.5 | ≤0.5 | - | 74 |
| 5775 * | 20 January 2017 | D. fluid | Surgery | R | S | R | R | R | R | S | S | ≤0.5 | 1 | - | 80 |
| 5777 * | 25 January 2017 | A. fluid | ICU | R | R | R | R | R | R | S | S | ≤0.5 | ≤0.5 | vanB | 117 |
| 5779 | 9 February 2017 | Bile | Surgery | R | S | R | R | R | R | S | S | ≤0.5 | 2 | - | 117 |
| 5780 | 9 February 2017 | Bile | Surgery | R | S | R | R | R | R | S | S | ≤0.5 | ≤0.5 | - | 80 |
| 5811 * | 2 May 2018 | Wound | ICU | R | R | R | R | R | R | S | S | 8 | ≤0.5 | vanB | 117 |
| 5812 * | 3 May 2018 | CVC | ICU | R | R | R | R | R | R | S | S | ≥32 | ≤0.5 | vanB | 117 |
| SSM Strain | Date | Source | Ward | A Vitek2 | B MicroScan | C Vitek2 | |||
|---|---|---|---|---|---|---|---|---|---|
| VAN mg/L | TEC mg/L | VAN mg/L | TEC mg/L | VAN mg/L | TEC mg/L | ||||
| 5777 | 25/01/2017 | A. fluid | ICU | ≤1 | ≤0.5 | 8 | ≤2 | ≥32 | ≤0.5 |
| 5811 * | 02/05/2018 | Wound | ICU | 8 | ≤0.5 | >8 | ≤2 | ≥32 | ≤0.5 |
| 5812 * | 03/05/2018 | CVC | ICU | ≥32 | ≤0.5 | >8 | ≤2 | ≥32 | ≤0.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santona, A.; Taviani, E.; Fiamma, M.; Deligios, M.; Hoang, H.M.; Sanna, S.; Rubino, S.; Paglietti, B. Occult Vancomycin-Resistant Enterococcus faecium ST117 Displaying a Highly Mutated vanB2 Operon. Antibiotics 2023, 12, 476. https://doi.org/10.3390/antibiotics12030476
Santona A, Taviani E, Fiamma M, Deligios M, Hoang HM, Sanna S, Rubino S, Paglietti B. Occult Vancomycin-Resistant Enterococcus faecium ST117 Displaying a Highly Mutated vanB2 Operon. Antibiotics. 2023; 12(3):476. https://doi.org/10.3390/antibiotics12030476
Chicago/Turabian StyleSantona, Antonella, Elisa Taviani, Maura Fiamma, Massimo Deligios, Hoa M. Hoang, Silvana Sanna, Salvatore Rubino, and Bianca Paglietti. 2023. "Occult Vancomycin-Resistant Enterococcus faecium ST117 Displaying a Highly Mutated vanB2 Operon" Antibiotics 12, no. 3: 476. https://doi.org/10.3390/antibiotics12030476
APA StyleSantona, A., Taviani, E., Fiamma, M., Deligios, M., Hoang, H. M., Sanna, S., Rubino, S., & Paglietti, B. (2023). Occult Vancomycin-Resistant Enterococcus faecium ST117 Displaying a Highly Mutated vanB2 Operon. Antibiotics, 12(3), 476. https://doi.org/10.3390/antibiotics12030476






